Abstract
Amino acid sequences of human collagen alpha 1(VI) and alpha 2(VI) chains were completed by cDNA sequencing and Edman degradation demonstrating that the mature polypeptides contain 1009 and 998 amino acid residues respectively. In addition, they contain small signal peptide sequences. Both chains show 31% identity in the N-terminal (approximately 235 residues) and C-terminal (approximately 430 residues) globular domains which are connected by a triple helical segment (335-336 residues). Internal alignment of the globular sequences indicates a repetitive 200-residue structure (15-23% identity) occurring three times (N1, C1, C2) in each chain. These repeating subdomains are connected to each other and to the triple helix by short (15-30 residues) cysteine-rich segments. The globular domains possess several N-glycosylation sites but no cell-binding RGD sequences, which are exclusively found in the triple helical segment. Sequencing of alpha 2(VI) cDNA clones revealed two variant chains with a distinct C2 subdomain and 3' non-coding region. The repetitive segments C1, C2 and, to a lesser extent, N1 show significant identity (15-18%) to the collagen-binding A domains of von Willebrand factor (vWF) and they are also similar to some integrin receptors, complement components and a cartilage matrix protein. Since the globular domains of collagen VI come into close contact with triple helical segments during the formation of tissue microfibrils it suggests that the globular domains bind to collagenous structures in a manner similar to the binding of vWF to collagen I.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allore R., O'Hanlon D., Price R., Neilson K., Willard H. F., Cox D. R., Marks A., Dunn R. J. Gene encoding the beta subunit of S100 protein is on chromosome 21: implications for Down syndrome. Science. 1988 Mar 11;239(4845):1311–1313. doi: 10.1126/science.2964086. [DOI] [PubMed] [Google Scholar]
- Argraves W. S., Deák F., Sparks K. J., Kiss I., Goetinck P. F. Structural features of cartilage matrix protein deduced from cDNA. Proc Natl Acad Sci U S A. 1987 Jan;84(2):464–468. doi: 10.1073/pnas.84.2.464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aumailley M., Mann K., von der Mark H., Timpl R. Cell attachment properties of collagen type VI and Arg-Gly-Asp dependent binding to its alpha 2(VI) and alpha 3(VI) chains. Exp Cell Res. 1989 Apr;181(2):463–474. doi: 10.1016/0014-4827(89)90103-1. [DOI] [PubMed] [Google Scholar]
- Brandt A., Glanville R. W., Hörlein D., Bruckner P., Timpl R., Fietzek P. P., Kühn K. Complete amino acid sequence of the N-terminal extension of calf skin type III procollagen. Biochem J. 1984 Apr 15;219(2):625–634. doi: 10.1042/bj2190625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitbart R. E., Andreadis A., Nadal-Ginard B. Alternative splicing: a ubiquitous mechanism for the generation of multiple protein isoforms from single genes. Annu Rev Biochem. 1987;56:467–495. doi: 10.1146/annurev.bi.56.070187.002343. [DOI] [PubMed] [Google Scholar]
- Bruns R. R. Beaded filaments and long-spacing fibrils: relation to type VI collagen. J Ultrastruct Res. 1984 Nov;89(2):136–145. doi: 10.1016/s0022-5320(84)80010-6. [DOI] [PubMed] [Google Scholar]
- Bruns R. R., Press W., Engvall E., Timpl R., Gross J. Type VI collagen in extracellular, 100-nm periodic filaments and fibrils: identification by immunoelectron microscopy. J Cell Biol. 1986 Aug;103(2):393–404. doi: 10.1083/jcb.103.2.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgeson R. E. New collagens, new concepts. Annu Rev Cell Biol. 1988;4:551–577. doi: 10.1146/annurev.cb.04.110188.003003. [DOI] [PubMed] [Google Scholar]
- Carter W. G. The cooperative role of the transformation-sensitive glycoproteins, GP140 and fibronectin, in cell attachment and spreading. J Biol Chem. 1982 Mar 25;257(6):3249–3257. [PubMed] [Google Scholar]
- Chu M. L., Conway D., Pan T. C., Baldwin C., Mann K., Deutzmann R., Timpl R. Amino acid sequence of the triple-helical domain of human collagen type VI. J Biol Chem. 1988 Dec 15;263(35):18601–18606. [PubMed] [Google Scholar]
- Chu M. L., Mann K., Deutzmann R., Pribula-Conway D., Hsu-Chen C. C., Bernard M. P., Timpl R. Characterization of three constituent chains of collagen type VI by peptide sequences and cDNA clones. Eur J Biochem. 1987 Oct 15;168(2):309–317. doi: 10.1111/j.1432-1033.1987.tb13422.x. [DOI] [PubMed] [Google Scholar]
- Colombatti A., Bonaldo P., Ainger K., Bressan G. M., Volpin D. Biosynthesis of chick type VI collagen. I. Intracellular assembly and molecular structure. J Biol Chem. 1987 Oct 25;262(30):14454–14460. [PubMed] [Google Scholar]
- Colombatti A., Bonaldo P. Biosynthesis of chick type VI collagen. II. Processing and secretion in fibroblasts and smooth muscle cells. J Biol Chem. 1987 Oct 25;262(30):14461–14466. [PubMed] [Google Scholar]
- Corbi A. L., Miller L. J., O'Connor K., Larson R. S., Springer T. A. cDNA cloning and complete primary structure of the alpha subunit of a leukocyte adhesion glycoprotein, p150,95. EMBO J. 1987 Dec 20;6(13):4023–4028. doi: 10.1002/j.1460-2075.1987.tb02746.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dayhoff M. O., Barker W. C., Hunt L. T. Establishing homologies in protein sequences. Methods Enzymol. 1983;91:524–545. doi: 10.1016/s0076-6879(83)91049-2. [DOI] [PubMed] [Google Scholar]
- Engel J., Furthmayr H., Odermatt E., von der Mark H., Aumailley M., Fleischmajer R., Timpl R. Structure and macromolecular organization of type VI collagen. Ann N Y Acad Sci. 1985;460:25–37. doi: 10.1111/j.1749-6632.1985.tb51154.x. [DOI] [PubMed] [Google Scholar]
- Engvall E., Hessle H., Klier G. Molecular assembly, secretion, and matrix deposition of type VI collagen. J Cell Biol. 1986 Mar;102(3):703–710. doi: 10.1083/jcb.102.3.703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimura Y., Titani K., Holland L. Z., Roberts J. R., Kostel P., Ruggeri Z. M., Zimmerman T. S. A heparin-binding domain of human von Willebrand factor. Characterization and localization to a tryptic fragment extending from amino acid residue Val-449 to Lys-728. J Biol Chem. 1987 Feb 5;262(4):1734–1739. [PubMed] [Google Scholar]
- Fujimura Y., Titani K., Holland L. Z., Russell S. R., Roberts J. R., Elder J. H., Ruggeri Z. M., Zimmerman T. S. von Willebrand factor. A reduced and alkylated 52/48-kDa fragment beginning at amino acid residue 449 contains the domain interacting with platelet glycoprotein Ib. J Biol Chem. 1986 Jan 5;261(1):381–385. [PubMed] [Google Scholar]
- Furthmayr H., Wiedemann H., Timpl R., Odermatt E., Engel J. Electron-microscopical approach to a structural model of intima collagen. Biochem J. 1983 May 1;211(2):303–311. doi: 10.1042/bj2110303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson M. A., Cleary E. G. CL glycoprotein is the tissue form of type VI collagen. J Biol Chem. 1985 Sep 15;260(20):11149–11159. [PubMed] [Google Scholar]
- Hatamochi A., Aumailley M., Mauch C., Chu M. L., Timpl R., Krieg T. Regulation of collagen VI expression in fibroblasts. Effects of cell density, cell-matrix interactions, and chemical transformation. J Biol Chem. 1989 Feb 25;264(6):3494–3499. [PubMed] [Google Scholar]
- Hynes R. O. Integrins: a family of cell surface receptors. Cell. 1987 Feb 27;48(4):549–554. doi: 10.1016/0092-8674(87)90233-9. [DOI] [PubMed] [Google Scholar]
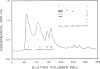
- Jander R., Rauterberg J., Glanville R. W. Further characterization of the three polypeptide chains of bovine and human short-chain collagen (intima collagen). Eur J Biochem. 1983 Jun 1;133(1):39–46. doi: 10.1111/j.1432-1033.1983.tb07427.x. [DOI] [PubMed] [Google Scholar]
- Jander R., Troyer D., Rauterberg J. A collagen-like glycoprotein of the extracellular matrix is the undegraded form of type VI collagen. Biochemistry. 1984 Jul 31;23(16):3675–3681. doi: 10.1021/bi00311a016. [DOI] [PubMed] [Google Scholar]
- Keene D. R., Engvall E., Glanville R. W. Ultrastructure of type VI collagen in human skin and cartilage suggests an anchoring function for this filamentous network. J Cell Biol. 1988 Nov;107(5):1995–2006. doi: 10.1083/jcb.107.5.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kligman D., Hilt D. C. The S100 protein family. Trends Biochem Sci. 1988 Nov;13(11):437–443. doi: 10.1016/0968-0004(88)90218-6. [DOI] [PubMed] [Google Scholar]
- Kozak M. An analysis of 5'-noncoding sequences from 699 vertebrate messenger RNAs. Nucleic Acids Res. 1987 Oct 26;15(20):8125–8148. doi: 10.1093/nar/15.20.8125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo H. J., Keene D. R., Glanville R. W. Orientation of type VI collagen monomers in molecular aggregates. Biochemistry. 1989 May 2;28(9):3757–3762. doi: 10.1021/bi00435a020. [DOI] [PubMed] [Google Scholar]
- Kühn K. Structural and functional domains of collagen: a comparison of the protein with its gene. Coll Relat Res. 1984 Aug;4(4):309–322. doi: 10.1016/s0174-173x(84)80038-2. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lipman D. J., Pearson W. R. Rapid and sensitive protein similarity searches. Science. 1985 Mar 22;227(4693):1435–1441. doi: 10.1126/science.2983426. [DOI] [PubMed] [Google Scholar]
- Oberbäumer I., Laurent M., Schwarz U., Sakurai Y., Yamada Y., Vogeli G., Voss T., Siebold B., Glanville R. W., Kühn K. Amino acid sequence of the non-collagenous globular domain (NC1) of the alpha 1(IV) chain of basement membrane collagen as derived from complementary DNA. Eur J Biochem. 1985 Mar 1;147(2):217–224. doi: 10.1111/j.1432-1033.1985.tb08739.x. [DOI] [PubMed] [Google Scholar]
- Odermatt E., Risteli J., van Delden V., Timpl R. Structural diversity and domain composition of a unique collagenous fragment (intima collagen) obtained from human placenta. Biochem J. 1983 May 1;211(2):295–302. doi: 10.1042/bj2110295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pareti F. I., Fujimura Y., Dent J. A., Holland L. Z., Zimmerman T. S., Ruggeri Z. M. Isolation and characterization of a collagen binding domain in human von Willebrand factor. J Biol Chem. 1986 Nov 15;261(32):15310–15315. [PubMed] [Google Scholar]
- Pytela R. Amino acid sequence of the murine Mac-1 alpha chain reveals homology with the integrin family and an additional domain related to von Willebrand factor. EMBO J. 1988 May;7(5):1371–1378. doi: 10.1002/j.1460-2075.1988.tb02953.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohde H., Wachter E., Richter W. J., Bruckner P., Helles O., Timpl R. Amino acid sequence of the N-terminal non-collagenous segment of dermatosparactic sheep procollagen type I. Biochem J. 1979 Jun 1;179(3):631–642. doi: 10.1042/bj1790631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E., Pierschbacher M. D. New perspectives in cell adhesion: RGD and integrins. Science. 1987 Oct 23;238(4826):491–497. doi: 10.1126/science.2821619. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz-Magdolen U., Oberbäumer I., Kühn K. cDNA and protein sequence of the NC1 domain of the alpha 2-chain of collagen IV and its comparison with alpha 1(IV). FEBS Lett. 1986 Nov 24;208(2):203–207. doi: 10.1016/0014-5793(86)81018-3. [DOI] [PubMed] [Google Scholar]
- Staunton D. E., Marlin S. D., Stratowa C., Dustin M. L., Springer T. A. Primary structure of ICAM-1 demonstrates interaction between members of the immunoglobulin and integrin supergene families. Cell. 1988 Mar 25;52(6):925–933. doi: 10.1016/0092-8674(88)90434-5. [DOI] [PubMed] [Google Scholar]
- Svoboda K. K., Nishimura I., Sugrue S. P., Ninomiya Y., Olsen B. R. Embryonic chicken cornea and cartilage synthesize type IX collagen molecules with different amino-terminal domains. Proc Natl Acad Sci U S A. 1988 Oct;85(20):7496–7500. doi: 10.1073/pnas.85.20.7496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikka L., Pihlajaniemi T., Henttu P., Prockop D. J., Tryggvason K. Gene structure for the alpha 1 chain of a human short-chain collagen (type XIII) with alternatively spliced transcripts and translation termination codon at the 5' end of the last exon. Proc Natl Acad Sci U S A. 1988 Oct;85(20):7491–7495. doi: 10.1073/pnas.85.20.7491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Titani K., Kumar S., Takio K., Ericsson L. H., Wade R. D., Ashida K., Walsh K. A., Chopek M. W., Sadler J. E., Fujikawa K. Amino acid sequence of human von Willebrand factor. Biochemistry. 1986 Jun 3;25(11):3171–3184. doi: 10.1021/bi00359a015. [DOI] [PubMed] [Google Scholar]
- Titani K., Walsh K. A. Human von Willebrand factor: the molecular glue of platelet plugs. Trends Biochem Sci. 1988 Mar;13(3):94–97. doi: 10.1016/0968-0004(88)90048-5. [DOI] [PubMed] [Google Scholar]
- Trüeb B., Schaeren-Wiemers N., Schreier T., Winterhalter K. H. Molecular cloning of chicken type VI collagen. Primary structure of the subunit alpha 2(VI)-pepsin. J Biol Chem. 1989 Jan 5;264(1):136–140. [PubMed] [Google Scholar]
- Trüeb B., Winterhalter K. H. Type VI collagen is composed of a 200 kd subunit and two 140 kd subunits. EMBO J. 1986 Nov;5(11):2815–2819. doi: 10.1002/j.1460-2075.1986.tb04573.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wayner E. A., Carter W. G. Identification of multiple cell adhesion receptors for collagen and fibronectin in human fibrosarcoma cells possessing unique alpha and common beta subunits. J Cell Biol. 1987 Oct;105(4):1873–1884. doi: 10.1083/jcb.105.4.1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber S., Engel J., Wiedemann H., Glanville R. W., Timpl R. Subunit structure and assembly of the globular domain of basement-membrane collagen type IV. Eur J Biochem. 1984 Mar 1;139(2):401–410. doi: 10.1111/j.1432-1033.1984.tb08019.x. [DOI] [PubMed] [Google Scholar]
- Weil D., Mattei M. G., Passage E., N'Guyen V. C., Pribula-Conway D., Mann K., Deutzmann R., Timpl R., Chu M. L. Cloning and chromosomal localization of human genes encoding the three chains of type VI collagen. Am J Hum Genet. 1988 Mar;42(3):435–445. [PMC free article] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Heijne G. Signal sequences. The limits of variation. J Mol Biol. 1985 Jul 5;184(1):99–105. doi: 10.1016/0022-2836(85)90046-4. [DOI] [PubMed] [Google Scholar]
- von der Mark H., Aumailley M., Wick G., Fleischmajer R., Timpl R. Immunochemistry, genuine size and tissue localization of collagen VI. Eur J Biochem. 1984 Aug 1;142(3):493–502. doi: 10.1111/j.1432-1033.1984.tb08313.x. [DOI] [PubMed] [Google Scholar]