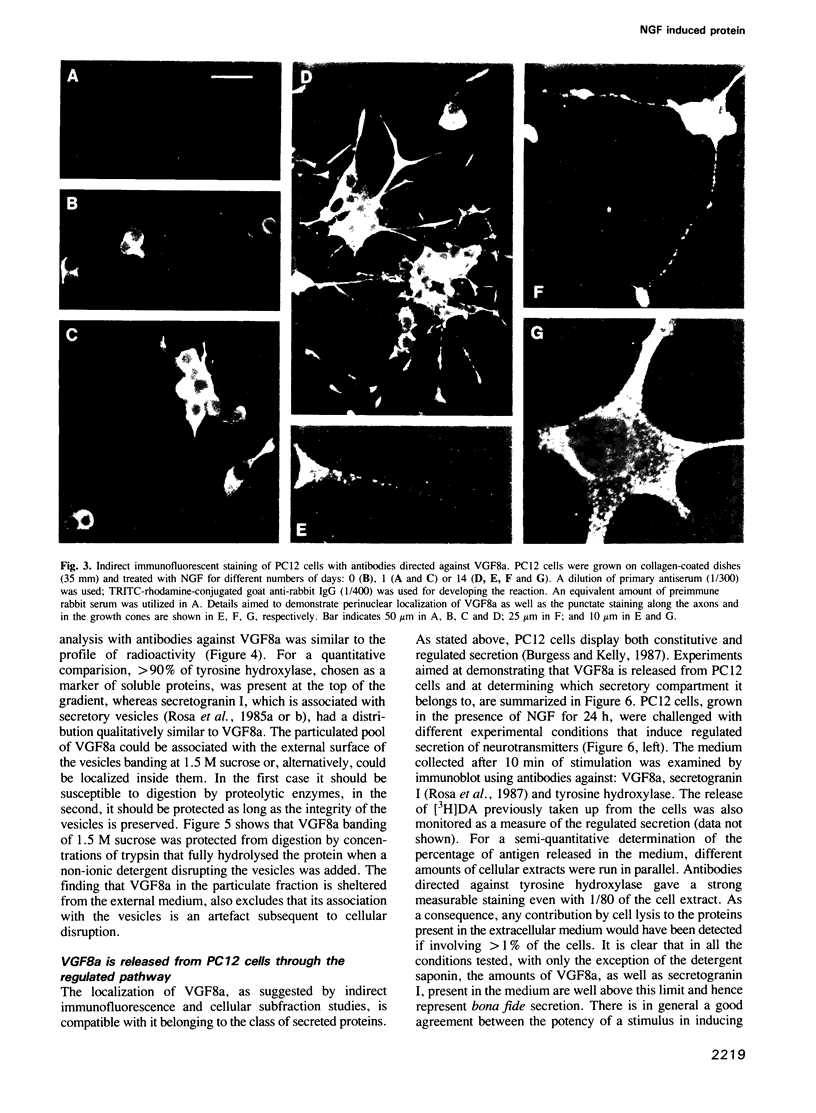
Abstract
We have previously described the isolation of a cDNA clone corresponding to an mRNA rapidly induced to high levels in PC12 cells by treatment with NGF. We report here the complete amino acid sequence of the protein (named VGF8a) as deduced by nucleotide sequencing of overlapping cDNA clones. VGF8a is particularly rich in proline residues and has a conspicuous number of short stretches of basic amino acid residues which may represent potential targets for proteolytic cleavage. Antibodies directed against recombinant VGF8a-beta-galactosidase fusion proteins were used for immunofluorescent staining of the protein in PC12 cells as well as for its localization, by Western blot analysis, in subfractions of cell homogenates. We demonstrate that in PC12 cells, VGF8a protein is stored in secretory vesicles and is released in response to a variety of stimuli that are known to induce the regulated secretion of neurotransmitters.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahn T. G., Cohn D. V., Gorr S. U., Ornstein D. L., Kashdan M. A., Levine M. A. Primary structure of bovine pituitary secretory protein I (chromogranin A) deduced from the cDNA sequence. Proc Natl Acad Sci U S A. 1987 Jul;84(14):5043–5047. doi: 10.1073/pnas.84.14.5043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson D. J., Axel R. Molecular probes for the development and plasticity of neural crest derivatives. Cell. 1985 Sep;42(2):649–662. doi: 10.1016/0092-8674(85)90122-9. [DOI] [PubMed] [Google Scholar]
- Benedum U. M., Baeuerle P. A., Konecki D. S., Frank R., Powell J., Mallet J., Huttner W. B. The primary structure of bovine chromogranin A: a representative of a class of acidic secretory proteins common to a variety of peptidergic cells. EMBO J. 1986 Jul;5(7):1495–1502. doi: 10.1002/j.1460-2075.1986.tb04388.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benedum U. M., Lamouroux A., Konecki D. S., Rosa P., Hille A., Baeuerle P. A., Frank R., Lottspeich F., Mallet J., Huttner W. B. The primary structure of human secretogranin I (chromogranin B): comparison with chromogranin A reveals homologous terminal domains and a large intervening variable region. EMBO J. 1987 May;6(5):1203–1211. doi: 10.1002/j.1460-2075.1987.tb02355.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgess T. L., Kelly R. B. Constitutive and regulated secretion of proteins. Annu Rev Cell Biol. 1987;3:243–293. doi: 10.1146/annurev.cb.03.110187.001331. [DOI] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Dickson G., Prentice H., Julien J. P., Ferrari G., Leon A., Walsh F. S. Nerve growth factor activates Thy-1 and neurofilament gene transcription in rat PC12 cells. EMBO J. 1986 Dec 20;5(13):3449–3453. doi: 10.1002/j.1460-2075.1986.tb04668.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eiden L. E. Is chromogranin a prohormone? Nature. 1987 Jan 22;325(6102):301–301. doi: 10.1038/325301a0. [DOI] [PubMed] [Google Scholar]
- Grasso A., Alemà S., Rufini S., Senni M. I. Black widow spider toxin-induced calcium fluxes and transmitter release in a neurosecretory cell line. Nature. 1980 Feb 21;283(5749):774–776. doi: 10.1038/283774a0. [DOI] [PubMed] [Google Scholar]
- Grasso A. Preparation and properties of a neurotoxin purified from the venom of black widow spider (Latrodectus mactans tredecimguttatus). Biochim Biophys Acta. 1976 Aug 9;439(2):406–412. doi: 10.1016/0005-2795(76)90077-5. [DOI] [PubMed] [Google Scholar]
- Greenberg M. E., Greene L. A., Ziff E. B. Nerve growth factor and epidermal growth factor induce rapid transient changes in proto-oncogene transcription in PC12 cells. J Biol Chem. 1985 Nov 15;260(26):14101–14110. [PubMed] [Google Scholar]
- Greene L. A., Rein G. Release, storage and uptake of catecholamines by a clonal cell line of nerve growth factor (NGF) responsive pheo-chromocytoma cells. Brain Res. 1977 Jul 1;129(2):247–263. doi: 10.1016/0006-8993(77)90005-1. [DOI] [PubMed] [Google Scholar]
- Greene L. A., Tischler A. S. Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc Natl Acad Sci U S A. 1976 Jul;73(7):2424–2428. doi: 10.1073/pnas.73.7.2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huttner W. B., Benedum U. M. Chromogranin A and pancreastatin. Nature. 1987 Jan 22;325(6102):305–305. doi: 10.1038/325305b0. [DOI] [PubMed] [Google Scholar]
- Iacangelo A., Affolter H. U., Eiden L. E., Herbert E., Grimes M. Bovine chromogranin A sequence and distribution of its messenger RNA in endocrine tissues. Nature. 1986 Sep 4;323(6083):82–86. doi: 10.1038/323082a0. [DOI] [PubMed] [Google Scholar]
- Korsching S., Auburger G., Heumann R., Scott J., Thoenen H. Levels of nerve growth factor and its mRNA in the central nervous system of the rat correlate with cholinergic innervation. EMBO J. 1985 Jun;4(6):1389–1393. doi: 10.1002/j.1460-2075.1985.tb03791.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kujubu D. A., Lim R. W., Varnum B. C., Herschman H. R. Induction of transiently expressed genes in PC-12 pheochromocytoma cells. Oncogene. 1987;1(3):257–262. [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leonard D. G., Ziff E. B., Greene L. A. Identification and characterization of mRNAs regulated by nerve growth factor in PC12 cells. Mol Cell Biol. 1987 Sep;7(9):3156–3167. doi: 10.1128/mcb.7.9.3156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levi-Montalcini R. The nerve growth factor 35 years later. Science. 1987 Sep 4;237(4819):1154–1162. doi: 10.1126/science.3306916. [DOI] [PubMed] [Google Scholar]
- Levi A., Eldridge J. D., Paterson B. M. Molecular cloning of a gene sequence regulated by nerve growth factor. Science. 1985 Jul 26;229(4711):393–395. doi: 10.1126/science.3839317. [DOI] [PubMed] [Google Scholar]
- Masiakowski P., Shooter E. M. Nerve growth factor induces the genes for two proteins related to a family of calcium-binding proteins in PC12 cells. Proc Natl Acad Sci U S A. 1988 Feb;85(4):1277–1281. doi: 10.1073/pnas.85.4.1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milbrandt J. A nerve growth factor-induced gene encodes a possible transcriptional regulatory factor. Science. 1987 Nov 6;238(4828):797–799. doi: 10.1126/science.3672127. [DOI] [PubMed] [Google Scholar]
- Rosa P., Fumagalli G., Zanini A., Huttner W. B. The major tyrosine-sulfated protein of the bovine anterior pituitary is a secretory protein present in gonadotrophs, thyrotrophs, mammotrophs, and corticotrophs. J Cell Biol. 1985 Mar;100(3):928–937. doi: 10.1083/jcb.100.3.928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosa P., Hille A., Lee R. W., Zanini A., De Camilli P., Huttner W. B. Secretogranins I and II: two tyrosine-sulfated secretory proteins common to a variety of cells secreting peptides by the regulated pathway. J Cell Biol. 1985 Nov;101(5 Pt 1):1999–2011. doi: 10.1083/jcb.101.5.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schweitzer E. S., Kelly R. B. Selective packaging of human growth hormone into synaptic vesicles in a rat neuronal (PC12) cell line. J Cell Biol. 1985 Aug;101(2):667–676. doi: 10.1083/jcb.101.2.667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon J. P., Bader M. F., Aunis D. Secretion from chromaffin cells is controlled by chromogranin A-derived peptides. Proc Natl Acad Sci U S A. 1988 Mar;85(5):1712–1716. doi: 10.1073/pnas.85.5.1712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sisk W. P., Chirikjian J. G., Lautenberger J., Jorcyk C., Papas T. S., Berman M. L., Zagursky R., Court D. L. A plasmid vector for cloning and expression of gene segments: expression of an HTLV-I envelope gene segment. Gene. 1986;48(2-3):183–193. doi: 10.1016/0378-1119(86)90076-4. [DOI] [PubMed] [Google Scholar]
- Walter P., Lingappa V. R. Mechanism of protein translocation across the endoplasmic reticulum membrane. Annu Rev Cell Biol. 1986;2:499–516. doi: 10.1146/annurev.cb.02.110186.002435. [DOI] [PubMed] [Google Scholar]
- Wieland F. T., Gleason M. L., Serafini T. A., Rothman J. E. The rate of bulk flow from the endoplasmic reticulum to the cell surface. Cell. 1987 Jul 17;50(2):289–300. doi: 10.1016/0092-8674(87)90224-8. [DOI] [PubMed] [Google Scholar]
- von Heijne G. Patterns of amino acids near signal-sequence cleavage sites. Eur J Biochem. 1983 Jun 1;133(1):17–21. doi: 10.1111/j.1432-1033.1983.tb07424.x. [DOI] [PubMed] [Google Scholar]