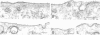Abstract
The human asialoglycoprotein receptor is composed of two homologous subunits, H1 and H2. By expressing the two subunits in transfected fibroblast cell lines, it has been shown previously that the formation of a hetero-oligomeric complex is necessary for the transport of H2 to the plasma membrane and for high-affinity ligand binding. Here we show that subunit H1, when expressed in the absence of H2, is capable of internalization through coated pits and recycling. The kinetics of these processes are very similar to those of the H1-H2 complex. To study endocytosis in the absence of ligand binding, the cell surface was labeled at 4 degrees C with the 125I-iodinated impermeant reagent sulfosuccinimidyl-3-(4-hydroxyphenyl) propionate, the cells were incubated at 37 degrees C for different times and the amount of internalized receptor was determined by protease digestion of surface proteins and immunoprecipitation. Similarly, recycling of surface-labeled and then internalized receptor protein was studied by monitoring its reappearance on the surface in the presence of exogenous protease. Our results show that subunit H1 contains all the signals necessary for receptor endocytosis and recycling independent of ligand binding.
Full text
PDF

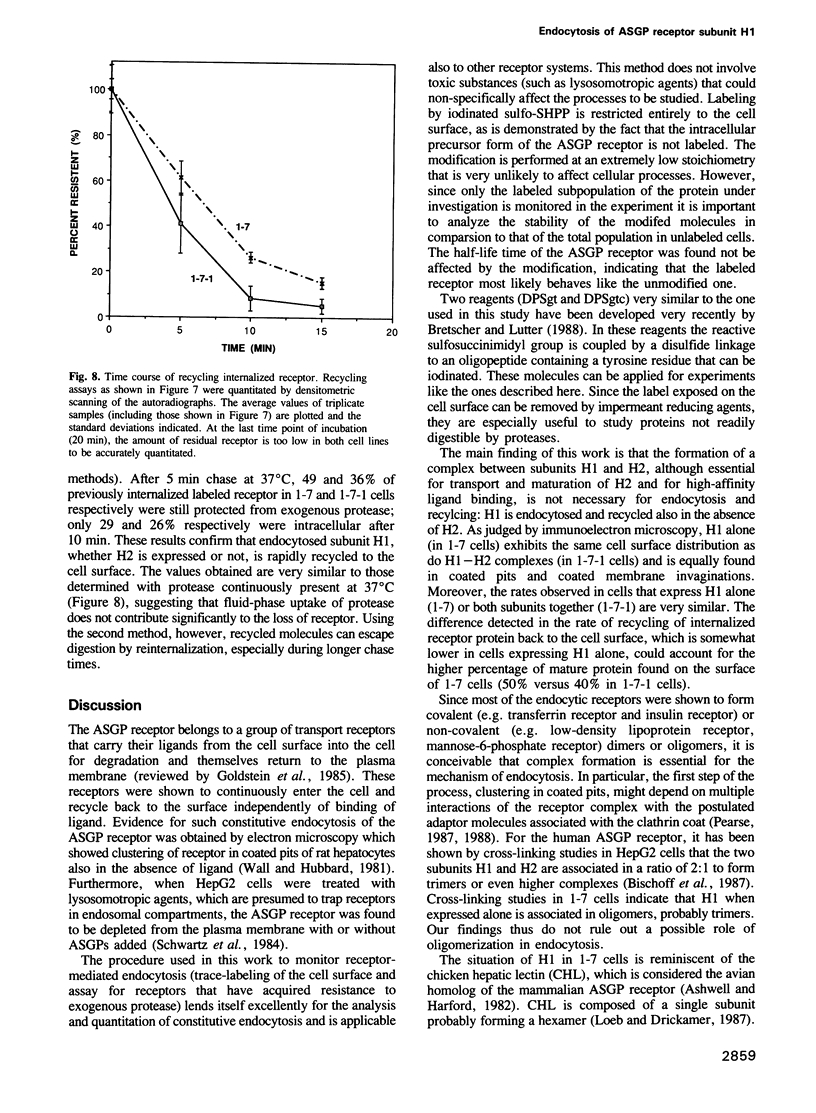


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ashwell G., Harford J. Carbohydrate-specific receptors of the liver. Annu Rev Biochem. 1982;51:531–554. doi: 10.1146/annurev.bi.51.070182.002531. [DOI] [PubMed] [Google Scholar]
- Bischoff J., Libresco S., Shia M. A., Lodish H. F. The H1 and H2 polypeptides associate to form the asialoglycoprotein receptor in human hepatoma cells. J Cell Biol. 1988 Apr;106(4):1067–1074. doi: 10.1083/jcb.106.4.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bischoff J., Lodish H. F. Two asialoglycoprotein receptor polypeptides in human hepatoma cells. J Biol Chem. 1987 Aug 25;262(24):11825–11832. [PubMed] [Google Scholar]
- Bolton A. E., Hunter W. M. The labelling of proteins to high specific radioactivities by conjugation to a 125I-containing acylating agent. Biochem J. 1973 Jul;133(3):529–539. doi: 10.1042/bj1330529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitfeld P. P., Simmons C. F., Jr, Strous G. J., Geuze H. J., Schwartz A. L. Cell biology of the asialoglycoprotein receptor system: a model of receptor-mediated endocytosis. Int Rev Cytol. 1985;97:47–95. doi: 10.1016/s0074-7696(08)62348-7. [DOI] [PubMed] [Google Scholar]
- Bretscher M. S., Lutter R. A new method for detecting endocytosed proteins. EMBO J. 1988 Dec 20;7(13):4087–4092. doi: 10.1002/j.1460-2075.1988.tb03302.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis C. G., Lehrman M. A., Russell D. W., Anderson R. G., Brown M. S., Goldstein J. L. The J.D. mutation in familial hypercholesterolemia: amino acid substitution in cytoplasmic domain impedes internalization of LDL receptors. Cell. 1986 Apr 11;45(1):15–24. doi: 10.1016/0092-8674(86)90533-7. [DOI] [PubMed] [Google Scholar]
- Davis C. G., van Driel I. R., Russell D. W., Brown M. S., Goldstein J. L. The low density lipoprotein receptor. Identification of amino acids in cytoplasmic domain required for rapid endocytosis. J Biol Chem. 1987 Mar 25;262(9):4075–4082. [PubMed] [Google Scholar]
- Drickamer K., Mamon J. F., Binns G., Leung J. O. Primary structure of the rat liver asialoglycoprotein receptor. Structural evidence for multiple polypeptide species. J Biol Chem. 1984 Jan 25;259(2):770–778. [PubMed] [Google Scholar]
- Goldstein J. L., Brown M. S., Anderson R. G., Russell D. W., Schneider W. J. Receptor-mediated endocytosis: concepts emerging from the LDL receptor system. Annu Rev Cell Biol. 1985;1:1–39. doi: 10.1146/annurev.cb.01.110185.000245. [DOI] [PubMed] [Google Scholar]
- Halberg D. F., Wager R. E., Farrell D. C., Hildreth J., 4th, Quesenberry M. S., Loeb J. A., Holland E. C., Drickamer K. Major and minor forms of the rat liver asialoglycoprotein receptor are independent galactose-binding proteins. Primary structure and glycosylation heterogeneity of minor receptor forms. J Biol Chem. 1987 Jul 15;262(20):9828–9838. [PubMed] [Google Scholar]
- Kornfeld S. Trafficking of lysosomal enzymes. FASEB J. 1987 Dec;1(6):462–468. doi: 10.1096/fasebj.1.6.3315809. [DOI] [PubMed] [Google Scholar]
- Lazarovits J., Roth M. A single amino acid change in the cytoplasmic domain allows the influenza virus hemagglutinin to be endocytosed through coated pits. Cell. 1988 Jun 3;53(5):743–752. doi: 10.1016/0092-8674(88)90092-x. [DOI] [PubMed] [Google Scholar]
- Loeb J. A., Drickamer K. The chicken receptor for endocytosis of glycoproteins contains a cluster of N-acetylglucosamine-binding sites. J Biol Chem. 1987 Mar 5;262(7):3022–3029. [PubMed] [Google Scholar]
- McPhaul M., Berg P. Formation of functional asialoglycoprotein receptor after transfection with cDNAs encoding the receptor proteins. Proc Natl Acad Sci U S A. 1986 Dec;83(23):8863–8867. doi: 10.1073/pnas.83.23.8863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearse B. M. Clathrin and coated vesicles. EMBO J. 1987 Sep;6(9):2507–2512. doi: 10.1002/j.1460-2075.1987.tb02536.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearse B. M. Receptors compete for adaptors found in plasma membrane coated pits. EMBO J. 1988 Nov;7(11):3331–3336. doi: 10.1002/j.1460-2075.1988.tb03204.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth J., Bendayan M., Orci L. Ultrastructural localization of intracellular antigens by the use of protein A-gold complex. J Histochem Cytochem. 1978 Dec;26(12):1074–1081. doi: 10.1177/26.12.366014. [DOI] [PubMed] [Google Scholar]
- Sawyer J. T., Sanford J. P., Doyle D. Identification of a complex of the three forms of the rat liver asialoglycoprotein receptor. J Biol Chem. 1988 Jul 25;263(21):10534–10538. [PubMed] [Google Scholar]
- Schwartz A. L., Bolognesi A., Fridovich S. E. Recycling of the asialoglycoprotein receptor and the effect of lysosomotropic amines in hepatoma cells. J Cell Biol. 1984 Feb;98(2):732–738. doi: 10.1083/jcb.98.2.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
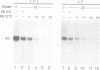
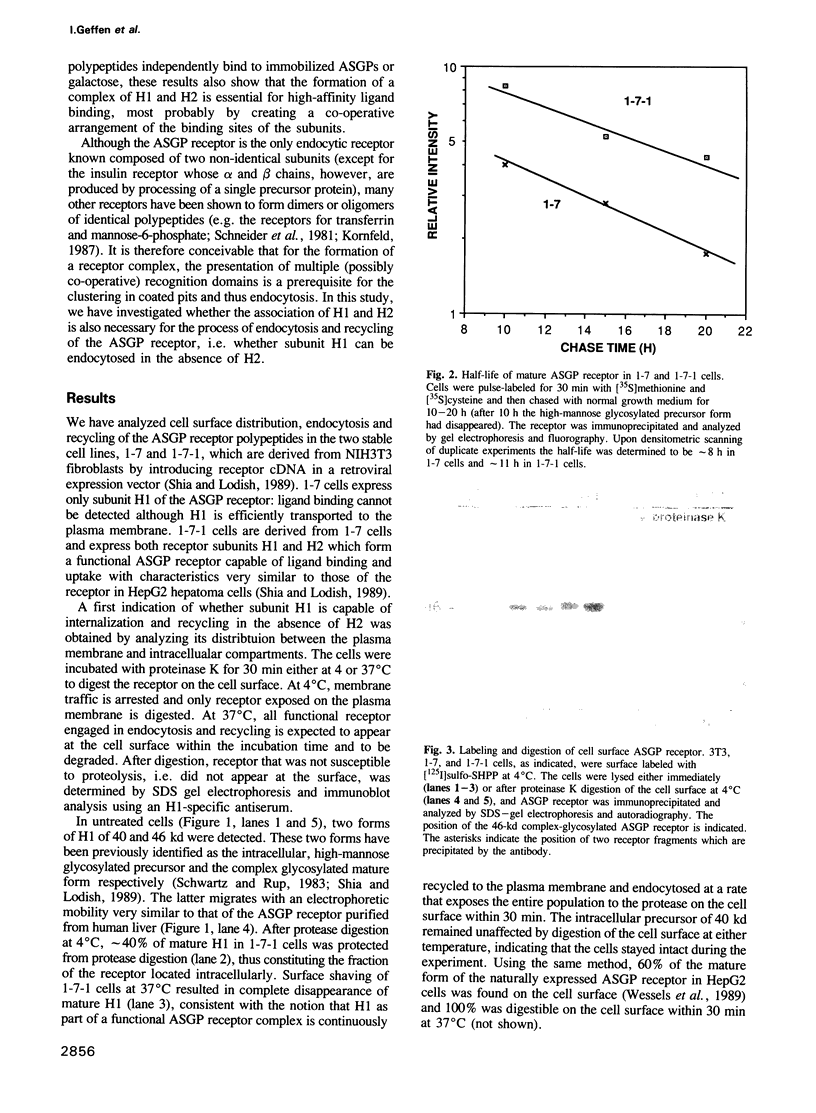
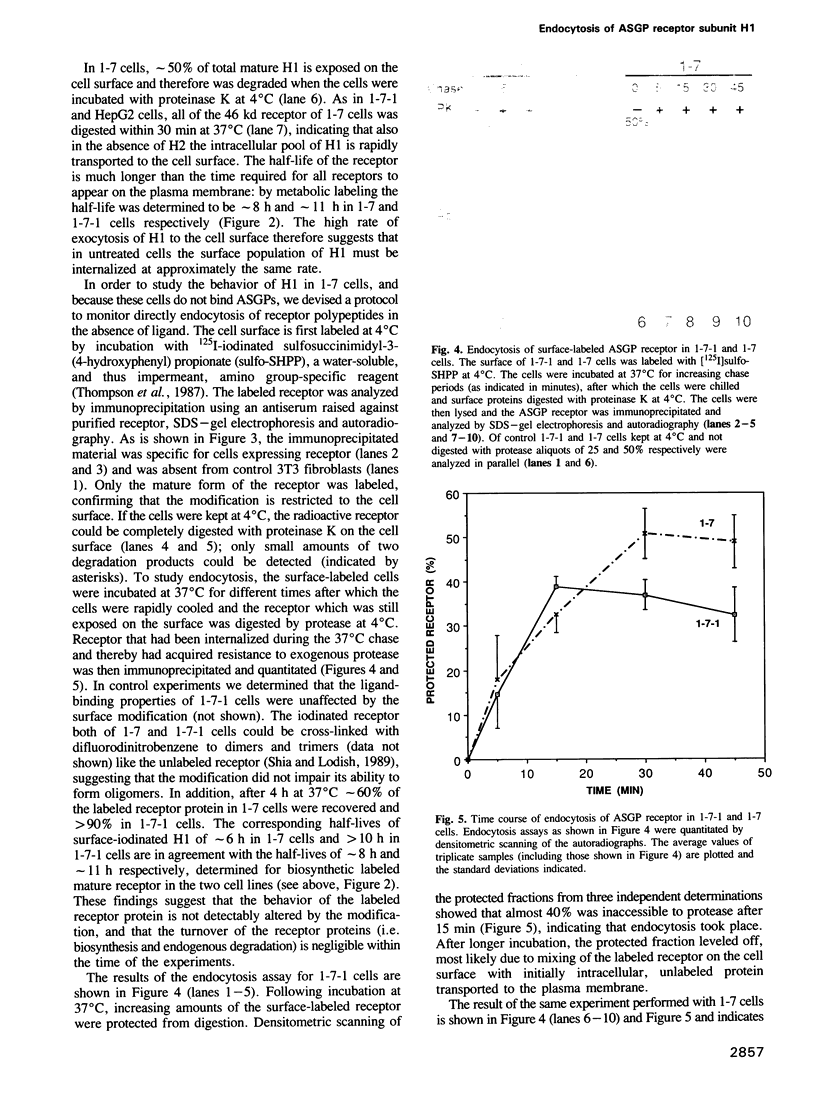
- Schwartz A. L., Rup D. Biosynthesis of the human asialoglycoprotein receptor. J Biol Chem. 1983 Sep 25;258(18):11249–11255. [PubMed] [Google Scholar]
- Shia M. A., Lodish H. F. The two subunits of the human asialoglycoprotein receptor have different fates when expressed alone in fibroblasts. Proc Natl Acad Sci U S A. 1989 Feb;86(4):1158–1162. doi: 10.1073/pnas.86.4.1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiess M., Lodish H. F. Sequence of a second human asialoglycoprotein receptor: conservation of two receptor genes during evolution. Proc Natl Acad Sci U S A. 1985 Oct;82(19):6465–6469. doi: 10.1073/pnas.82.19.6465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson J. A., Lau A. L., Cunningham D. D. Selective radiolabeling of cell surface proteins to a high specific activity. Biochemistry. 1987 Feb 10;26(3):743–750. doi: 10.1021/bi00377a014. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wall D. A., Hubbard A. L. Galactose-specific recognition system of mammalian liver: receptor distribution on the hepatocyte cell surface. J Cell Biol. 1981 Sep;90(3):687–696. doi: 10.1083/jcb.90.3.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessels H. P., Geffen I., Spiess M. A hepatocyte-specific basolateral membrane protein is targeted to the same domain when expressed in Madin-Darby canine kidney cells. J Biol Chem. 1989 Jan 5;264(1):17–20. [PubMed] [Google Scholar]