Abstract
Aims/Introduction: Emerging evidence from observational studies suggests that diabetes mellitus affects the cancer risk. However, whether there are differences in the magnitude of the influence of diabetes among ethnic groups is unknown.
Materials and Methods: We searched MEDLINE and the Cochrane Library for pertinent articles that had been published as of 4 April 2011, and included them in a meta‐analysis of the risk of all‐cancer mortality and incidence in diabetic subjects.
Results: A total of 33 studies were included in the meta‐analysis, and they provided 156,132 diabetic subjects for the mortality analysis and 993,884 for the incidence analysis. Cancer mortality was approximately 3%, and cancer incidence was approximately 8%. The pooled adjusted risk ratio (RR) of all‐cancer mortality was significantly higher than for non‐diabetic people (RR 1.32 [CI 1.20–1.45] for Asians; RR 1.16 [CI 1.01–1.34] for non‐Asians). Diabetes was also associated with an increased RR of incidence across all cancer types (RR 1.23 [CI 1.09–1.39] for Asians; RR 1.15 [CI 0.94–1.43] for non‐Asians). The RR of incident cancer for Asian men was significantly higher than for non‐Asian men (P = 0.021).
Conclusions: Diabetes is associated with a higher risk for incident cancer in Asian men than in non‐Asian men. In light of the exploding global epidemic of diabetes, particularly in Asia, a modest increase in the cancer risk will translate into a substantial socioeconomic burden. Our current findings underscore the need for clinical attention and better‐designed studies of the complex interactions between diabetes and cancer. (J Diabetes Invest, doi: 10.1111/j.2040‐1124.2011.00183.x, 2012)
Keywords: Cancer, Diabetes, Meta‐analysis
Introduction
Emerging evidence from observational data and meta‐analyses of the data suggest that diabetes mellitus is associated with an increased risk of cancer. The mechanisms responsible for the increase in risk have yet to be investigated, but as insulin might have a mitogenic effect through binding the insulin‐like growth factor‐1 receptor1–11, insulin resistance and secondary hyperinsulinemia is the most frequently proposed hypothesis and hyperglycemia itself might promote carcinogenesis12–18. However, the possibility of methodological issues, bias and occult malignant tumors cannot be completely excluded. Meta‐analyses have shown that diabetes increases the risks of total cancer19,20 and of site‐specific cancers of the breast21, endometrium22, bladder23, liver24, colorectum25 and pancreas26,27, and that it decreases the risk of prostate cancer28,29.
The same as in Western countries, the prevalence of diabetes is markedly increasing in Asia. This trend is presumably attributable to the rapid Westernization of people’s lifestyle, a trend that is likely shared by the majority of Asian populations30. Although cardiovascular disease is the main cause of mortality in Western countries and patients with diabetes have a high risk of such disease, cancer is the leading cause of death in Asian countries, including Japan31,32. As the current diabetes epidemic and the higher mortality in cancer patients with diabetes33, particularly in Asia, will translate into crucial clinical and public health consequences on a global scale, attention should be directed to elucidating the association between these diseases in populations with increased risks to make timely, rational and informed decisions, not only in the public health area and socioeconomic area, but also for the prevention and targeted management of diabetes in routine clinical practice both domestically and globally.
The aforementioned circumstances prompted us to more precisely investigate the effect of diabetes on all‐cancer mortality and incidence among Asians and non‐Asians by carefully reviewing pertinent original reports and combining their data in an attempt to obtain meaningful clues to the prevention and management of cancer in diabetes.
Materials and Methods
Data Sources and Searches
Searches of MEDLINE and the Cochrane Library from their inception until 4 April 2011 were carried out, and articles that reported investigations of cancer mortality and incidence in diabetic patients and non‐diabetic subjects were extracted. Relevant reports were identified by using a combination of the following medical subject headings as search terms: ‘diabetes’, ‘cancer’ or ‘neoplasms’, and ‘risk’ or ‘risk factors’. The literature reference lists of the pertinent articles were also examined.
Relevant reports included those of observational studies that evaluated type 2 diabetes, but not reports of studies that focused on impaired glucose tolerance/impaired fasting glucose, or solely type 1 diabetes. Cohort, case–control and cross‐sectional studies carried out to evaluate the risk of cancer based on original data analyses were assessed to determine their eligibility for inclusion in a qualitative analysis, and those of them that reported risk ratios (RR), that is, hazard ratios (HR), relative risks or odds ratios (OR) adjusted for possible confounders with confidence intervals (CI), were eligible for inclusion in the meta‐analysis.
Data Extraction and Quality Assessment
We reviewed each full‐text report to determine its eligibility, and extracted and tabulated all of the relevant data independently. The majority of the studies that were included had been systematically reviewed elsewhere19,20, and the additional studies34–40 used for inclusion in the present analysis were evaluated in the same manner: the data extracted included the subjects’ characteristics (including age, sex and comorbidities), study design, study years, follow‐up period, and the methods used to ascertain the presence or absence of diabetes and cancer. Any disagreement was resolved by consensus among the investigators. To ascertain the validity of the eligible studies, the quality of each report was appraised in reference to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement41.
Data Synthesis and Statistical Analysis
If more than one study was published in regard to the same cohort, the report with information on the most comprehensive population was included to avoid overlapping populations. This process resulted in the exclusion of two articles from the meta‐analysis16,42. Another investigation, carried out on diabetic patients with autopsy‐proven nephropathy43, was also excluded, because cohorts with this condition are rare, and the generalizability of the findings was deemed to be poor.
The reports were summarized quantitatively into a meta‐analysis. The individual RR were combined, and the pooled RR adjusted for possible confounders with 95% CI was calculated by using the random‐effects model with inverse‐variance weighting. If not provided in the original study, the RR for the men and women combined was estimated before pooling. The equality of RR between Asian and non‐Asian studies were assessed by using z‐statistic tests. Heterogeneity among studies was evaluated using I2 statistics. The possibility of a publication bias, which can result from non‐publication of small studies with negative findings, was assessed visually by using a funnel plot for asymmetry. Subgroup analyses for each sex were carried out to further elucidate the impact of the risk of all‐cancer mortality and incidence in diabetic patients. The RevMan software program (version 5.1, The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, Denmark) was used to make all of the calculations. All of the procedures were in accordance with the guidelines for the meta‐analysis of observational studies in epidemiology44 and the Preferred Reporting Items for Systematic Reviews and Meta‐analyses (PRISMA) statement45.
Results
Search Results
A total of 1514 citations were identified during our search, and 45 of them were evaluated as eligible for inclusion in our meta‐analysis aimed at determining the influence of diabetes on all‐cancer mortality and all‐cancer incidence among Asians and non‐Asians (Figure 1). The 33 (31 cohort studies, one cross‐sectional study and one case–control study) of these 45 articles that provided sufficient information were included in the meta‐analysis. The 33 articles15,34–39,46–71 that were selected for inclusion in the meta‐analysis were moderately heterogeneous in terms of the population demographics and assessment of confounding factors, and the methodological quality of the majority of the studies included was fair20 (data not shown for the additional data34–40). The sizes of the diabetic patient samples in the studies ranged from 224 to 594,815. Cancer mortality and cancer incidence were approximately 3 and 8%, respectively.
Figure 1.
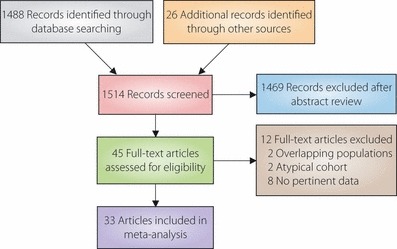
Summary of the procedure used to select studies for inclusion in the meta‐analysis.
Quantitative Summary (Meta‐analysis)
As shown in Figures 2 and 3, the diabetic patients worldwide had a significantly increased risk of all‐cancer mortality in comparison with the non‐diabetic subjects. The adjusted RR for both men and women were also significantly higher, and the RR were consistently higher for Asians than for non‐Asians across the analyses, although they did not reach statistical significance (P = 0.130 for men and women; 0.086 for men; 0.536 for women). As shown in Figures 4 and 5, diabetes was also associated with an increased RR of incidence across all cancer types worldwide, and the RR was significantly higher for Asian men than for non‐Asian men (P = 0.585 for men and women; 0.021 for men; 0.467 for women). Significant heterogeneity was observed across these studies. No clear publication bias was detected by a funnel plot assessment (data not shown).
Figure 2.
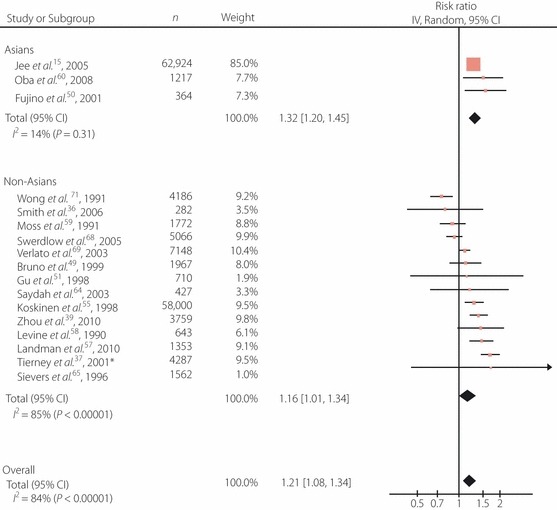
Adjusted risk ratios (RR) for all‐cancer mortality among the subjects with diabetes. *Cross‐sectional study. Boxes, estimated RR; bars, 95% confidence intervals (CI); diamonds, RR; width of diamonds, pooled CI.
Figure 3.

Adjusted risk ratios (RR) for all‐cancer mortality among men and women with diabetes. *Cross‐sectional study. Boxes, estimated RR; bars, 95% confidence intervals (CI); diamonds, RR; width of diamonds, pooled CI. NR, not reported.
Figure 4.
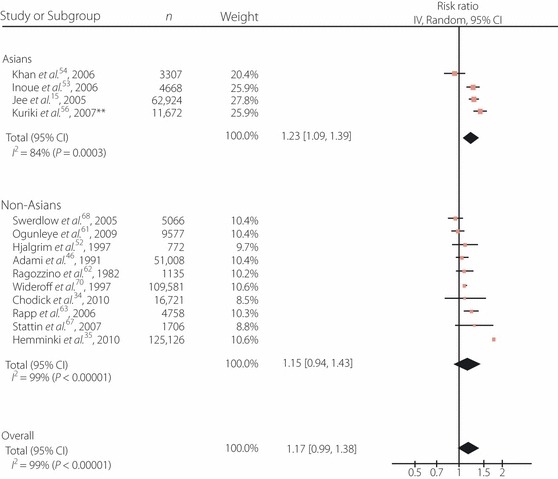
Adjusted risk ratios (RR) for all‐cancer incidence among the subjects with diabetes. **Case–control study. Boxes, estimated RR; bars, 95% confidence intervals (CI); diamonds, RR; width of diamonds, pooled CI.
Figure 5.

Adjusted risk ratios (RR) for all‐cancer incidence among men and women with diabetes. **Case–control study. Boxes, estimated RR; bars, 95% confidence intervals (CI); diamonds, RR; width of diamonds, pooled CI. NR, not reported.
Discussion
We recently showed a worldwide increased risk of all‐cancer mortality and incidence among diabetic patients in a meta‐analysis of population‐based observational reports of epidemiological data20. In the present study we found associations between diabetes and a moderately increased risk of all‐cancer mortality and all‐cancer incidence among both Asians and non‐Asians, and confirmed the worldwide trend20 with the updated data. Few reports have addressed the risk of total cancer in diabetes, and, to the best of our knowledge, ours is the first meta‐analysis to compare the magnitude of risk in different races. Our findings are of considerable clinical and socioeconomic importance, because the cancer risk proved to be significantly increased in the rapidly growing Asian diabetic population as well, and the risk increment in incidence was found to be larger for Asian men than for the diabetic men in the other areas.
The strength of the present study lies in the fact that the analysis regarding overall cancer was mainly based on large population‐based cohorts from several different countries and ethnic groups, and was carried out with high levels of precision and generalizability. Although the pooled RR were robust, the results of the component studies were statistically heterogeneous. The large I2 values showed that the range of plausible risk estimates is wide, but there was very little evidence in our analysis to support a protective effect of diabetes on all‐cancer incidence and mortality. These findings might reflect the different mechanisms of development of cancer at different sites and/or different epidemiological characteristics among the diverse populations.
Type 2 diabetes is characterized by insulin resistance and compensatory hyperinsulinemia, and people with type 2 diabetes are typically obese and lead sedentary lives, both of which also contribute to their hyperinsulinemia. It has been postulated that insulin has a mitogenic effect by multiple and complex mechanisms. First, insulin might bind and activate its related insulin‐like growth factor‐1 receptor, which is the most frequently proposed mechanism to explain the clearly increased risk of cancer in diabetic patients1–11. Second, hyperinsulinemia might increase the risk of certain cancers by increased insulin receptor signaling, leading to proliferative and anti‐apoptotic effects72. Finally, the mitogenic activity of insulin might be enhanced at the cellular level by post‐receptor molecular mechanisms, including insulin residence time on the receptor and the intracellular upregulation of the insulin mitogenic pathway73. It has been reported that this mitogenic pathway, unlike the metabolic pathway, might not be blunted in the condition of insulin resistance. The activated protein kinase (AMPK), mammalian target of rapamycin and insulin‐signaling pathway represent three interrelated components of a complex mechanism controlling cell responses to nutrient availability. It is suggested that metformin might have an anti‐cancer effect by activating AMPK, followed by modulation of downstream tumor gene regulators.
Several findings would seem to support this insulin supply hypothesis. Pancreatic cancer has been reported to be induced more effectively with a carcinogen or by implantation of cancer cells when experimental insulin‐deficient animals are given supplemental insulin74,75. Humans with type 1 diabetes have a lower risk of cancer than humans with type 2 diabetes76,77, although the evidence for a higher risk than in the general population is inconclusive78,79. However, they are derived from retrospective observational studies, and because of the possible existence of confounders and biases in those studies, they do not necessarily indicate causality80,81. In fact, the data from insulin‐treated patients are inconclusive82.
Interestingly, diabetes has been reported to protect against the development of prostate cancer28,29, which is testosterone‐dependent. Testosterone deficiency is common in men with diabetes, because they have low levels of sex‐hormone‐binding globulin, and testosterone levels have been shown to be partly influenced by insulin resistance83–85. The magnitude of the decrease in cancer risk as a result of testosterone deficiency is likely to be higher than the magnitude of the increase in cancer risk as a result of insulin resistance, and thus this favorable effect of diabetes on prostate cancer might have contributed to the attenuation of the increase in cancer risk in men found in the current study and in our preceding report19. However, those meta‐analyses28,29 were mainly based on data for Caucasian men and the reported risks for Asian men have been either significantly elevated in Taiwan86,87 or non‐significant in Japan53,54,56,88,89 and Korea15, which points to the possibility that the effect of diabetes on prostate cancer might not be universal, probably because of genetic/cultural/socioeconomic factors. In fact, the current study showed that the RR for prostate cancer for Asian men were non‐significant (data not shown) and that the RR for total cancer incidence was significantly higher for Asian men than for non‐Asian men.
Hyperglycemia has also been reported to promote tumor cell proliferation and cancer metastasis in type 2 diabetes90,91. Indeed, this forms the basis for 18F‐fluorodeoxyglucose‐positron emission tomography of cancers, which detects tissues with high rates of glucose uptake. In addition, hyperglycemia itself might promote carcinogenesis by generating oxidative stress12–18, which is frequently observed to be increased in diabetes, in a variety of cells. The increase in oxidative stress would cause DNA damage, the initial step in carcinogenesis17. Community‐based prospective surveys have documented associations between plasma glucose levels and the risk of cancer12–15,92. The results of the present study support this hypothesis, because the results showed that the risk of both cancer incidence and mortality is also generally higher among Japanese19,50,53,56,60 and Korean15 patients with diabetes, who have been reported to be insulinopenic30,93–97. However, a meta‐analysis of large randomized‐controlled trials of intensified glycemic control did not support the hypothesis that hyperglycemia is causally linked to increased cancer risk98,99.
Potential risk factors common to both cancer and diabetes need to be addressed, because it remains to be clarified whether the association between diabetes and the risk of cancer is mainly a result of shared risk factors or whether diabetes itself increases the risk of some types of cancer. Those risk factors include age, sex, race/ethnicity, obesity, physical activity, diet, alcohol and smoking100. Clearly, lower levels of adiposity, a healthy diet and regular physical activity are associated with a decreased risk of diabetes and several types of cancer; these factors are generally interrelated and thus it is difficult to assess the contribution of each factor.
It is particularly noteworthy that our meta‐analysis showed that the RR was higher for Asian men than for non‐Asian men. This finding suggests the presence of other factors that promote carcinogenesis, such as susceptibility to insulin/glucose and genetic/environmental factors. The current findings underscore the crucial need to understand the role of glucose metabolism and insulin resistance in carcinogenesis30,101.
Alternative explanations for the increased risk of cancer in diabetic patients should be taken into consideration, because the relationship between the aforementioned factors and increased risk might not be causal. First, several comorbidity confounders exist. For example, coexisting obesity and a sedentary lifestyle, which induce hyperinsulinemia, might be the true causes, and diabetes might merely be an innocent bystander. The other possible confounders include age, sex, diet, consumption of alcoholic beverages, smoking, liver cirrhosis and the indication of insulin usage, which were not fully adjusted for in the present study. The second alternative explanation is that diabetic patients might receive medical care more frequently and thus have more opportunities for cancer detection than non‐diabetic subjects. The third is that diabetes might develop as a consequence of cancer, as cancers generally cause insulin resistance and subsequent hyperglycemia by producing cytokines, such as tumor necrosis‐α102,103. The fourth is that the studies that were included in the meta‐analysis might have left room for confounding by treatment indication: differences between the treatment of cancer according to whether or not they had diabetes might have contributed to the increased mortality of the subjects. Diabetic patients often have other diabetes‐related comorbidities that might influence the treatment decisions and prognosis. For example, diabetes might be accompanied by a higher risk of infection and the diagnosis of cancer might result in inappropriate glucose management, and Caucasians have higher cardiovascular mortality, which might lead to an underestimation of the absolute increment in cancer mortality risk among non‐Asian diabetic people.
Several limitations of our meta‐analysis should be noted. As with any overview, the possibility that relevant research papers were missed and the inability to adjust fully for confounders based on population‐based databases must be recognized. It is also important to realize that the populations in the studies were heterogeneous, most likely because of ethnic diversity or study design variation, and that the risks of site‐specific cancers might have varied. Therefore, an analysis of cancer at any sites might be overly simplistic and dilute the true associations. The small number of articles that reported studies carried out on Asian subjects that were included might have further restricted the generalizability of the results. Despite these limitations, the results of our meta‐analysis should stir health‐care providers, policy makers and patients into devising measures to prevent and manage cancer in diabetic patients. Another limitation is that the methods used to ascertain the presence of diabetes in the studies extracted included self‐reports, which might have led to diagnostic inaccuracies. In addition, the baseline surveillance in most of the studies was carried out when the diagnostic cut‐off value for fasting glucose was higher than the currently accepted value, and the prevalence of diabetes in the control groups most likely increased exponentially during the long follow‐up period. Thus, the true prevalence of diabetes and its impact on cancer risk might have been underestimated. Last, the possibility of modification of cancer risk by diabetes medication cannot be completely excluded in descriptive studies, although relevant data are limited at present, and further investigation is required.
In conclusion, the results of our meta‐analysis strongly suggest that diabetes is associated with an increased risk of all‐cancer incidence and all‐cancer mortality worldwide, and that the RR are higher for Asians. In light of the exploding global epidemic of diabetes, particularly in Asia, a modest increase in the risk of cancer will translate into a substantial socioeconomic burden. Our current findings underscore the need for clinical attention and better‐designed studies of the complex interactions between diabetes and cancer.
Acknowledgements
This study was supported by a Health Sciences Research Grant (Comprehensive Research on Diabetes/Cardiovascular and Life‐Style Related Diseases H22‐019) from the Ministry of Health, Labour and Welfare of Japan, and by a grant from Japan Diabetes Foundation. The authors have no financial conflicts of interest to declare.
References
- 1.White MF. The insulin signalling system and the IRS proteins. Diabetologia 1997; 40(Suppl 2): S2–S17 [DOI] [PubMed] [Google Scholar]
- 2.Kim YI. Diet, lifestyle, and colorectal cancer: is hyperinsulinemia the missing link? Nutr Rev 1998; 56: 275–279 [DOI] [PubMed] [Google Scholar]
- 3.Kaaks R. Nutrition, hormones, and breast cancer: is insulin the missing link? Cancer Causes Control 1996; 7: 605–625 [DOI] [PubMed] [Google Scholar]
- 4.Giovannucci E. Insulin and colon cancer. Cancer Causes Control 1995; 6: 164–179 [DOI] [PubMed] [Google Scholar]
- 5.Yu H, Berkel H. Insulin‐like growth factors and cancer. J La State Med Soc 1999; 151: 218–223 [PubMed] [Google Scholar]
- 6.Zhang W, Thornton WH, MacDonald RS. Insulin‐like growth factor‐I and II receptor expression in rat colon mucosa are affected by dietary lipid intake. J Nutr 1998; 128: 158–165 [DOI] [PubMed] [Google Scholar]
- 7.Bruning PF, Bonfrer JM, van Noord PA, et al. Insulin resistance and breast‐cancer risk. Int J Cancer 1992; 52: 511–516 [DOI] [PubMed] [Google Scholar]
- 8.Hu FB, Manson JE, Liu S, et al. Prospective study of adult onset diabetes mellitus (type 2) and risk of colorectal cancer in women. J Natl Cancer Inst 1999; 91: 542–547 [DOI] [PubMed] [Google Scholar]
- 9.Silverman DT, Schiffman M, Everhart J, et al. Diabetes mellitus, other medical conditions and familial history of cancer as risk factors for pancreatic cancer. Br J Cancer 1999; 80: 1830–1837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wolf I, Sadetzki S, Catane R, et al. Diabetes mellitus and breast cancer. Lancet Oncol 2005; 6: 103–111 [DOI] [PubMed] [Google Scholar]
- 11.Le Roith D. Seminars in medicine of the Beth Israel Deaconess Medical Center. Insulin‐like growth factors. N Engl J Med 1997; 336: 633–640 [DOI] [PubMed] [Google Scholar]
- 12.Barclay AW, Petocz P, McMillan‐Price J, et al. Glycemic index, glycemic load, and chronic disease risk – a meta‐analysis of observational studies. Am J Clin Nutr 2008; 87: 627–637 [DOI] [PubMed] [Google Scholar]
- 13.Gapstur SM, Gann PH, Lowe W, et al. Abnormal glucose metabolism and pancreatic cancer mortality. JAMA 2000; 283: 2552–2558 [DOI] [PubMed] [Google Scholar]
- 14.Seow A, Yuan JM, Koh WP, et al. Diabetes mellitus and risk of colorectal cancer in the Singapore Chinese Health Study. J Natl Cancer Inst 2006; 98: 135–138 [DOI] [PubMed] [Google Scholar]
- 15.Jee SH, Ohrr H, Sull JW, et al. Fasting serum glucose level and cancer risk in Korean men and women. JAMA 2005; 293: 194–202 [DOI] [PubMed] [Google Scholar]
- 16.Stocks T, Rapp K, Bjorge T, et al. Blood glucose and risk of incident and fatal cancer in the metabolic syndrome and cancer project (me‐can): analysis of six prospective cohorts. PLoS Med 2009; 6: e1000201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Abe R, Yamagishi S. AGE‐RAGE system and carcinogenesis. Curr Pharm Des 2008; 14: 940–945 [DOI] [PubMed] [Google Scholar]
- 18.Inoue M, Kurahashi N, Iwasaki M, et al. Metabolic factors and subsequent risk of hepatocellular carcinoma by hepatitis virus infection status: a large‐scale population‐based cohort study of Japanese men and women (JPHC Study Cohort II). Cancer Causes Control 2009; 20: 741–750 [DOI] [PubMed] [Google Scholar]
- 19.Noto H, Osame K, Sasazuki T, et al. Substantially increased risk of cancer in patients with diabetes mellitus: a systematic review and meta‐analysis of epidemiologic evidence in Japan. J Diabetes Complications 2010; 24: 345–353 [DOI] [PubMed] [Google Scholar]
- 20.Noto H, Tsujimoto T, Sasazuki T, et al. Significantly increased risk of cancer in patients with diabetes mellitus. Endocr Pract 2011; 17: 616–628 [DOI] [PubMed] [Google Scholar]
- 21.Larsson SC, Mantzoros CS, Wolk A. Diabetes mellitus and risk of breast cancer: a meta‐analysis. Int J Cancer 2007; 121: 856–862 [DOI] [PubMed] [Google Scholar]
- 22.Friberg E, Orsini N, Mantzoros CS, et al. Diabetes mellitus and risk of endometrial cancer: a meta‐analysis. Diabetologia 2007; 50: 1365–1374 [DOI] [PubMed] [Google Scholar]
- 23.Larsson SC, Orsini N, Brismar K, et al. Diabetes mellitus and risk of bladder cancer: a meta‐analysis. Diabetologia 2006; 49: 2819–2823 [DOI] [PubMed] [Google Scholar]
- 24.El‐Serag HB, Hampel H, Javadi F. The association between diabetes and hepatocellular carcinoma: a systematic review of epidemiologic evidence. Clin Gastroenterol Hepatol 2006; 4: 369–380 [DOI] [PubMed] [Google Scholar]
- 25.Larsson SC, Orsini N, Wolk A. Diabetes mellitus and risk of colorectal cancer: a meta‐analysis. J Natl Cancer Inst 2005; 97: 1679–1687 [DOI] [PubMed] [Google Scholar]
- 26.Huxley R, Ansary‐Moghaddam A, Berrington de Gonzalez A, et al. Type‐II diabetes and pancreatic cancer: a meta‐analysis of 36 studies. Br J Cancer 2005; 92: 2076–2083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Everhart J, Wright D. Diabetes mellitus as a risk factor for pancreatic cancer. A meta‐analysis. JAMA 1995; 273: 1605–1609 [PubMed] [Google Scholar]
- 28.Bonovas S, Filioussi K, Tsantes A. Diabetes mellitus and risk of prostate cancer: a meta‐analysis. Diabetologia 2004; 47: 1071–1078 [DOI] [PubMed] [Google Scholar]
- 29.Kasper JS, Giovannucci E. A meta‐analysis of diabetes mellitus and the risk of prostate cancer. Cancer Epidemiol Biomarkers Prev 2006; 15: 2056–2062 [DOI] [PubMed] [Google Scholar]
- 30.Chan JC, Malik V, Jia W, et al. Diabetes in Asia: epidemiology, risk factors, and pathophysiology. JAMA 2009; 301: 2129–2140 [DOI] [PubMed] [Google Scholar]
- 31.Hotta N, Nakamura J, Iwamoto Y, et al. Causes of death in Japanese diabetics based on the results of a survey of 18,385 diabetics during 1991–2000. Report of Committee on Cause of Death in Diabetes Mellitus. J Jpn Diabetes Soc 2007; 50: 47–61 [Google Scholar]
- 32.Ministry of Health LaWoJ. Patient Survey, 2005.
- 33.Barone BB, Yeh HC, Snyder CF, et al. Long‐term all‐cause mortality in cancer patients with preexisting diabetes mellitus: a systematic review and meta‐analysis. JAMA 2008; 300: 2754–2764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chodick G, Heymann AD, Rosenmann L, et al. Diabetes and risk of incident cancer: a large population‐based cohort study in Israel. Cancer Causes Control 2010; 21: 879–887 [DOI] [PubMed] [Google Scholar]
- 35.Hemminki K, Li X, Sundquist J, et al. Risk of cancer following hospitalization for type 2 diabetes. Oncologist 2010; 15: 548–555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Smith NL, Barzilay JI, Kronmal R, et al. New‐onset diabetes and risk of all‐cause and cardiovascular mortality: the Cardiovascular Health Study. Diabetes Care 2006; 29: 2012–2017 [DOI] [PubMed] [Google Scholar]
- 37.Tierney EF, Geiss LS, Engelgau MM, et al. Population‐based estimates of mortality associated with diabetes: use of a death certificate check box in North Dakota. Am J Public Health 2001; 91: 84–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Atchison EA, Gridley G, Carreon JD, et al. Risk of cancer in a large cohort of U.S. veterans with diabetes. Int J Cancer 2011; 128: 635–643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhou XH, Qiao Q, Zethelius B, et al. Diabetes, prediabetes and cancer mortality. Diabetologia 2010; 53: 1867–1876 [DOI] [PubMed] [Google Scholar]
- 40.Driver JA, Yung R, Gaziano JM, et al. Chronic disease in men with newly diagnosed cancer: a nested case–control study. Am J Epidemiol 2010; 172: 299–308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 2008; 61: 344–349 [DOI] [PubMed] [Google Scholar]
- 42.de Marco R, Locatelli F, Zoppini G, et al. Cause‐specific mortality in type 2 diabetes. The Verona Diabetes Study. Diabetes Care 1999; 22: 756–761 [DOI] [PubMed] [Google Scholar]
- 43.Nerlich AG, Hagedorn HG, Boheim M, et al. Patients with diabetes‐induced microangiopathy show a reduced frequency of carcinomas. In Vivo 1998; 12: 667–670 [PubMed] [Google Scholar]
- 44.Stroup DF, Berlin JA, Morton SC, et al. Meta‐analysis of observational studies in epidemiology: a proposal for reporting. Meta‐analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000; 283: 2008–2012 [DOI] [PubMed] [Google Scholar]
- 45.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta‐analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med 2009; 151: W65–W94 [DOI] [PubMed] [Google Scholar]
- 46.Adami HO, McLaughlin J, Ekbom A, et al. Cancer risk in patients with diabetes mellitus. Cancer Causes Control 1991; 2: 307–314 [DOI] [PubMed] [Google Scholar]
- 47.Adlerberth AM, Rosengren A, Wilhelmsen L. Diabetes and long‐term risk of mortality from coronary and other causes in middle‐aged Swedish men. A general population study. Diabetes Care 1998; 21: 539–545 [DOI] [PubMed] [Google Scholar]
- 48.Balkau B, Eschwege E, Ducimetiere P, et al. The high risk of death by alcohol related diseases in subjects diagnosed as diabetic and impaired glucose tolerant: the Paris Prospective Study after 15 years of follow‐up. J Clin Epidemiol 1991; 44: 465–474 [DOI] [PubMed] [Google Scholar]
- 49.Bruno G, Merletti F, Boffetta P, et al. Impact of glycaemic control, hypertension and insulin treatment on general and cause‐specific mortality: an Italian population‐based cohort of type II (non‐insulin‐dependent) diabetes mellitus. Diabetologia 1999; 42: 297–301 [DOI] [PubMed] [Google Scholar]
- 50.Fujino Y, Mizoue T, Tokui N, et al. Prospective study of diabetes mellitus and liver cancer in Japan. Diabetes Metab Res Rev 2001; 17: 374–379 [DOI] [PubMed] [Google Scholar]
- 51.Gu K, Cowie CC, Harris MI. Mortality in adults with and without diabetes in a national cohort of the U.S. population, 1971–1993. Diabetes Care 1998; 21: 1138–1145 [DOI] [PubMed] [Google Scholar]
- 52.Hjalgrim H, Frisch M, Ekbom A, et al. Cancer and diabetes – a follow‐up study of two population‐based cohorts of diabetic patients. J Intern Med 1997; 241: 471–475 [DOI] [PubMed] [Google Scholar]
- 53.Inoue M, Iwasaki M, Otani T, et al. Diabetes mellitus and the risk of cancer: results from a large‐scale population‐based cohort study in Japan. Arch Intern Med 2006; 166: 1871–1877 [DOI] [PubMed] [Google Scholar]
- 54.Khan M, Mori M, Fujino Y, et al. Site‐specific cancer risk due to diabetes mellitus history: evidence from the Japan Collaborative Cohort (JACC) Study. Asian Pac J Cancer Prev 2006; 7: 253–259 [PubMed] [Google Scholar]
- 55.Koskinen SV, Reunanen AR, Martelin TP, et al. Mortality in a large population‐based cohort of patients with drug‐treated diabetes mellitus. Am J Public Health 1998; 88: 765–770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kuriki K, Hirose K, Tajima K. Diabetes and cancer risk for all and specific sites among Japanese men and women. Eur J Cancer Prev 2007; 16: 83–89 [DOI] [PubMed] [Google Scholar]
- 57.Landman GW, Kleefstra N, van Hateren KJ, et al. Metformin associated with lower cancer mortality in type 2 diabetes: ZODIAC‐16. Diabetes Care 2010; 33: 322–326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Levine W, Dyer AR, Shekelle RB, et al. Post‐load plasma glucose and cancer mortality in middle‐aged men and women. 12‐year follow‐up findings of the Chicago Heart Association Detection Project in Industry. Am J Epidemiol 1990; 131: 254–262 [DOI] [PubMed] [Google Scholar]
- 59.Moss SE, Klein R, Klein BE. Cause‐specific mortality in a population‐based study of diabetes. Am J Public Health 1991; 81: 1158–1162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Oba S, Nagata C, Nakamura K, et al. Self‐reported diabetes mellitus and risk of mortality from all causes, cardiovascular disease, and cancer in Takayama: a population‐based prospective cohort study in Japan. J Epidemiol 2008; 18: 197–203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ogunleye AA, Ogston SA, Morris AD, et al. A cohort study of the risk of cancer associated with type 2 diabetes. Br J Cancer 2009; 101: 1199–1201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ragozzino M, Melton LJ 3rd, Chu CP, et al. Subsequent cancer risk in the incidence cohort of Rochester, Minnesota, residents with diabetes mellitus. J Chronic Dis 1982; 35: 13–19 [DOI] [PubMed] [Google Scholar]
- 63.Rapp K, Schroeder J, Klenk J, et al. Fasting blood glucose and cancer risk in a cohort of more than 140,000 adults in Austria. Diabetologia 2006; 49: 945–952 [DOI] [PubMed] [Google Scholar]
- 64.Saydah SH, Loria CM, Eberhardt MS, et al. Abnormal glucose tolerance and the risk of cancer death in the United States. Am J Epidemiol 2003; 157: 1092–1100 [DOI] [PubMed] [Google Scholar]
- 65.Sievers ML, Nelson RG, Bennett PH. Sequential trends in overall and cause‐specific mortality in diabetic and nondiabetic Pima Indians. Diabetes Care 1996; 19: 107–111 [DOI] [PubMed] [Google Scholar]
- 66.Smith GD, Egger M, Shipley MJ, et al. Post‐challenge glucose concentration, impaired glucose tolerance, diabetes, and cancer mortality in men. Am J Epidemiol 1992; 136: 1110–1114 [DOI] [PubMed] [Google Scholar]
- 67.Stattin P, Bjor O, Ferrari P, et al. Prospective study of hyperglycemia and cancer risk. Diabetes Care 2007; 30: 561–567 [DOI] [PubMed] [Google Scholar]
- 68.Swerdlow AJ, Laing SP, Qiao Z, et al. Cancer incidence and mortality in patients with insulin‐treated diabetes: a UK cohort study. Br J Cancer 2005; 92: 2070–2075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Verlato G, Zoppini G, Bonora E, et al. Mortality from site‐specific malignancies in type 2 diabetic patients from Verona. Diabetes Care 2003; 26: 1047–1051 [DOI] [PubMed] [Google Scholar]
- 70.Wideroff L, Gridley G, Mellemkjaer L, et al. Cancer incidence in a population‐based cohort of patients hospitalized with diabetes mellitus in Denmark. J Natl Cancer Inst 1997; 89: 1360–1365 [DOI] [PubMed] [Google Scholar]
- 71.Wong JS, Pearson DW, Murchison LE, et al. Mortality in diabetes mellitus: experience of a geographically defined population. Diabet Med 1991; 8: 135–139 [DOI] [PubMed] [Google Scholar]
- 72.Gallagher EJ, Leroith D. Minireview: IGF, insulin, and cancer. Endocrinology 2011; 152: 2546–2551 [DOI] [PubMed] [Google Scholar]
- 73.Vigneri P, Frasca F, Sciacca L, et al. Diabetes and cancer. Endocr Relat Cancer 2009; 16: 1103–1123 [DOI] [PubMed] [Google Scholar]
- 74.Fisher WE, Boros LG, O’Dorisio TM, et al. GI hormonal changes in diabetes influence pancreatic cancer growth. J Surg Res 1995; 58: 754–758 [DOI] [PubMed] [Google Scholar]
- 75.Bell RH Jr, McCullough PJ, Pour PM. Influence of diabetes on susceptibility to experimental pancreatic cancer. Am J Surg 1988; 155: 159–164 [DOI] [PubMed] [Google Scholar]
- 76.Lindblad P, Chow WH, Chan J, et al. The role of diabetes mellitus in the aetiology of renal cell cancer. Diabetologia 1999; 42: 107–112 [DOI] [PubMed] [Google Scholar]
- 77.Brinton LA, Berman ML, Mortel R, et al. Reproductive, menstrual, and medical risk factors for endometrial cancer: results from a case–control study. Am J Obstet Gynecol 1992; 167: 1317–1325 [DOI] [PubMed] [Google Scholar]
- 78.Zendehdel K, Nyren O, Ostenson CG, et al. Cancer incidence in patients with type 1 diabetes mellitus: a population‐based cohort study in Sweden. J Natl Cancer Inst 2003; 95: 1797–1800 [DOI] [PubMed] [Google Scholar]
- 79.Shu X, Ji J, Li X, et al. Cancer risk among patients hospitalized for Type 1 diabetes mellitus: a population‐based cohort study in Sweden. Diabet Med 2010; 27: 791–797 [DOI] [PubMed] [Google Scholar]
- 80.Johnson JA, Gale EA. Diabetes, insulin use, and cancer risk: are observational studies part of the solution‐or part of the problem? Diabetes 2010; 59: 1129–1131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pocock SJ, Elbourne DR. Randomized trials or observational tribulations? N Engl J Med 2000; 342: 1907–1909 [DOI] [PubMed] [Google Scholar]
- 82.McFarland MS, Cripps R. Diabetes mellitus and increased risk of cancer: focus on metformin and the insulin analogs. Pharmacotherapy 2010; 30: 1159–1178 [DOI] [PubMed] [Google Scholar]
- 83.Grossmann M, Thomas MC, Panagiotopoulos S, et al. Low testosterone levels are common and associated with insulin resistance in men with diabetes. J Clin Endocrinol Metab 2008; 93: 1834–1840 [DOI] [PubMed] [Google Scholar]
- 84.Dhindsa S, Prabhakar S, Sethi M, et al. Frequent occurrence of hypogonadotropic hypogonadism in type 2 diabetes. J Clin Endocrinol Metab 2004; 89: 5462–5468 [DOI] [PubMed] [Google Scholar]
- 85.Ding EL, Song Y, Malik VS, et al. Sex differences of endogenous sex hormones and risk of type 2 diabetes: a systematic review and meta‐analysis. JAMA 2006; 295: 1288–1299 [DOI] [PubMed] [Google Scholar]
- 86.Tseng CH. Prostate cancer mortality in Taiwanese men: increasing age‐standardized trend in general population and increased risk in diabetic men. Ann Med 2011; 43: 142–150 [DOI] [PubMed] [Google Scholar]
- 87.Tseng CH. Diabetes and risk of prostate cancer: a study using the National Health Insurance. Diabetes Care 2011; 34: 616–621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mishina T, Watanabe H, Araki H, et al. Epidemiological study of prostatic cancer by matched‐pair analysis. Prostate 1985; 6: 423–436 [DOI] [PubMed] [Google Scholar]
- 89.Li Q, Kuriyama S, Kakizaki M, et al. History of diabetes mellitus and the risk of prostate cancer: the Ohsaki Cohort Study. Cancer Causes Control 2010; 21: 1025–1032 [DOI] [PubMed] [Google Scholar]
- 90.Richardson LC, Pollack LA. Therapy insight: influence of type 2 diabetes on the development, treatment and outcomes of cancer. Nat Clin Pract Oncol 2005; 2: 48–53 [DOI] [PubMed] [Google Scholar]
- 91.Morss AS, Edelman ER. Glucose modulates basement membrane fibroblast growth factor‐2 via alterations in endothelial cell permeability. J Biol Chem 2007; 282: 14635–14644 [DOI] [PubMed] [Google Scholar]
- 92.Seshasai SR, Kaptoge S, Thompson A, et al. Diabetes mellitus, fasting glucose, and risk of cause‐specific death. N Engl J Med 2011; 364: 829–841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kadowaki T, Miyake Y, Hagura R, et al. Risk factors for worsening to diabetes in subjects with impaired glucose tolerance. Diabetologia 1984; 26: 44–49 [DOI] [PubMed] [Google Scholar]
- 94.Fukushima M, Usami M, Ikeda M, et al. Insulin secretion and insulin sensitivity at different stages of glucose tolerance: a cross‐sectional study of Japanese type 2 diabetes. Metabolism 2004; 53: 831–835 [DOI] [PubMed] [Google Scholar]
- 95.Kuroe A, Fukushima M, Usami M, et al. Impaired beta‐cell function and insulin sensitivity in Japanese subjects with normal glucose tolerance. Diabetes Res Clin Pract 2003; 59: 71–77 [DOI] [PubMed] [Google Scholar]
- 96.Boyko EJ, Fujimoto WY, Leonetti DL, et al. Visceral adiposity and risk of type 2 diabetes: a prospective study among Japanese Americans. Diabetes Care 2000; 23: 465–471 [DOI] [PubMed] [Google Scholar]
- 97.Chan WB, Tong PC, Chow CC, et al. The associations of body mass index, C‐peptide and metabolic status in Chinese Type 2 diabetic patients. Diabet Med 2004; 21: 349–353 [DOI] [PubMed] [Google Scholar]
- 98.Stefansdottir G, Zoungas S, Chalmers J, et al. Intensive glucose control and risk of cancer in patients with type 2 diabetes. Diabetologia 2011; 54: 1608–1614 [DOI] [PubMed] [Google Scholar]
- 99.Johnson JA, Bowker SL. Intensive glycaemic control and cancer risk in type 2 diabetes: a meta‐analysis of major trials. Diabetologia 2011; 54: 25–31 [DOI] [PubMed] [Google Scholar]
- 100.Giovannucci E, Harlan DM, Archer MC, et al. Diabetes and cancer: a consensus report. Diabetes Care 2010; 33: 1674–1685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Karin M, Lawrence T, Nizet V. Innate immunity gone awry: linking microbial infections to chronic inflammation and cancer. Cell 2006; 124: 823–835 [DOI] [PubMed] [Google Scholar]
- 102.McCall JL, Tuckey JA, Parry BR. Serum tumour necrosis factor alpha and insulin resistance in gastrointestinal cancer. Br J Surg 1992; 79: 1361–1363 [DOI] [PubMed] [Google Scholar]
- 103.Noguchi Y, Yoshikawa T, Marat D, et al. Insulin resistance in cancer patients is associated with enhanced tumor necrosis factor‐alpha expression in skeletal muscle. Biochem Biophys Res Commun 1998; 253: 887–892 [DOI] [PubMed] [Google Scholar]


