Abstract
Eldecalcitol [1α,25-dihydroxy-2β-(3-hydroxypropyloxy)vitamin D3] is an analog of 1α,25-dihydroxyvitamin D3 [1,25(OH)2D3], bearing a hydroxypropyloxy residue at the 2β position. In preclinical studies, eldecalcitol suppressed bone resorption to a greater extent than alfacalcidol but had a similar effect on bone formation and Ca metabolism, resulting in a greater increase in bone mineral density (BMD) in ovariectomized (OVX) rats. Histological analysis in OVX rats immediately after ovariectomy revealed that eldecalcitol reduced osteoclast number and bone resorption parameters with a decrease in bone formation parameters. Eldecalcitol also promoted focal bone formation independent of bone resorption, a process known as bone minimodeling. In clinical studies, eldecalcitol showed stronger effects than alfacalcidol in increasing BMD and reducing bone resorption markers in osteoporotic patients under vitamin D supplementation. A 3-year randomized, double-blind, active-comparator clinical trial demonstrated that once-daily 0.75 μg eldecalcitol reduced vertebral fracture incidence by 26% compared with 1.0 μg alfacalcidol. Eldecalcitol also reduced the incidence of wrist fractures by 71% compared with alfacalcidol. Although this may be due to the previously reported effect of vitamin D in reducing the incidence of falls, it is not known whether eldecalcitol has a stronger effect in preventing falls than alfacalcidol. Because eldecalcitol stimulates intestinal Ca absorption and improves Ca balance in addition to its skeletal effects, combination treatment with antiresorptive agents may be able to show better effects than native vitamin D and Ca supplementation in preventing fractures in osteoporotic patients. Further studies are warranted to clarify these issues.
Introduction
Vitamin D is regarded as an essential nutrient required for increasing intestinal calcium (Ca) and phosphate (Pi) absorption. Vitamin D is converted by 25-hydroxylase in the liver to 25-hydroxyvitamin D [25(OH)D], which is then activated by 1α-hydroxylase in the kidneys to become 1,25-dihydroxyvitamin D [1,25(OH)2D]. The active vitamin D, 1,25(OH)2D, acts in the intestine to stimulate Ca absorption and maintain Ca balance. Under conditions of vitamin D insufficiency or deficiency, intestinal Ca absorption decreases resulting in a subtle reduction in serum Ca, which causes an increase in the secretion of parathyroid hormone. Sustained parathyroid hormone elevation enhances not only activation of vitamin D to 1,25(OH)2D but also bone resorption. As a result, the bone remodeling balance becomes negative under conditions of vitamin D insufficiency or deficiency. Because osteoporosis treatment aims to improve negative remodeling balance in the bone, it is important to make the Ca balance positive. Therefore, vitamin D supplementation is recommended whenever patients are given therapeutic drugs for osteoporosis.
Osteoporosis is characterized by compromised bone strength predisposing to an increased risk of fracture.1 The reduction in bone strength results from an imbalance in bone remodeling, which is caused by various factors, including aging, estrogen deficiency after menopause, glucocorticoid excess and immobilization. Inhibition of bone resorption by treatment with antiresorptive agents can maintain a positive remodeling balance and prevent the reduction in bone strength. In order for these antiresorptive agents to be effective in maintaining bone strength, there needs to be a sufficient supply of Ca and Pi. Although active vitamin D is necessary for the absorption of Ca and Pi from the intestine, the therapeutic efficacy of vitamin D analogs in osteoporosis treatment remains controversial.
A meta-analysis of the effect of native and active forms of vitamin D on bone mineral density (BMD) and fractures found that hydroxylated active vitamin D compounds showed a consistently larger impact on BMD, and that hydroxylated vitamin D doses above 0.5 μg yielded larger effects than native vitamin D on BMD of the lumbar spine and forearms.2 However, those analyses could not address the relative effectiveness of native and hydroxylated vitamin D on fractures. Furthermore, inferences from those analyses were limited by the variability in study designs, methodological weaknesses, small sample sizes, and the inconsistency of results in the primary studies.
The actions of 1,25(OH)2D are exerted via its binding to vitamin D receptor (VDR), a member of the nuclear receptor superfamily. VDR bound to its ligand 1,25(OH)2D forms a heterodimer complex with retinoid X receptor (RXR), and the RXR–VDR heterodimer complex binds to the vitamin D response element with a specific DNA sequence on its target genes. Analogous to selective estrogen receptor modulators, structural modifications of the ligands to nuclear receptors can create tissue-specific agonists or antagonists of the authentic ligands. Thus, there is a possibility that VDR-mediated actions can also be modified in a tissue-selective manner by modulating the structure of its ligands.
In an effort to synthesize new active vitamin D compounds with more potent activity on bone and less calcemic effect than 1α-hydroxy vitamin D3 (alfacalcidol), which is a prodrug of 1α,25-dihydroxyvitamin D3 [1,25(OH)2D3; calcitriol], a number of vitamin D analogs bearing a hydroxyalkoxy group at the 2β position have been synthesized.3 Among them, 1α,25(OH)2-2β-(3-hydroxypropyloxy) vitamin D3 (ED-71; eldecalcitol) showed the strongest activity in preventing bone mineral loss in ovariectomized (OVX) rats.4
Eldecalcitol's affinity for VDR is less than half that of calcitriol, but it has a very high affinity for serum vitamin D-binding protein due to the existence of the hydroxypropyloxy substituent at the 2β position.5 Eldecalcitol's high affinity for serum vitamin D-binding protein appears to be responsible for its unique pharmacokinetics with very long plasma half-life of 53 h in healthy male volunteers.6 Another characteristic of eldecalcitol is its weak suppressive activity for parathyroid hormone secretion.7
In this review, we examine the effects of active vitamin D compounds on bone turnover and fractures in preclinical studies and in clinical studies with osteoporotic patients, with special reference to eldecalcitol.
Preclinical studies
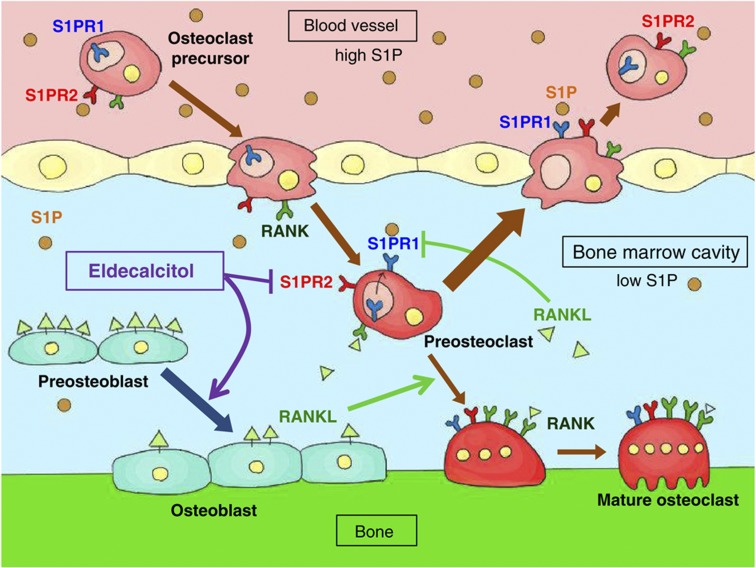
Using OVX rats, Uchiyama et al.8 compared the effects of eldecalcitol with those of alfacalcidol on BMD and bone remodeling processes as a function of their effects on Ca metabolism. Eldecalcitol increased lumbar BMD to a greater extent than did alfacalcidol but increased urinary Ca excretion to an extent similar to that of alfacalcidol. Eldecalcitol reduced the biochemical and histological parameters of bone resorption more potently than did alfacalcidol but maintained bone formation markers. Those results demonstrated that eldecalcitol suppressed bone resorption to a greater extent than alfacalcidol but had a similar effect on bone formation and Ca metabolism, resulting in a greater increase in BMD in OVX rats. Histomorphometric analysis revealed that eldecalcitol increased cancellous bone volume with an increase in bone formation rate/bone surface more potently than alfacalcidol in both normal and OVX rats 3 months after ovariectomy.9 Thus, when the elevation of bone turnover rate was modest, eldecalcitol increased bone formation rate. In contrast, in a detailed histological analysis by de Freitas et al.10 using OVX rats immediately after ovariectomy, eldecalcitol reduced osteoclast number and bone resorption parameters with a decrease in bone formation parameters. In the eldecalcitol-treated group, the preosteoblastic layer was poorly developed with less cell-to-cell contact between preosteoblasts and osteoclast precursors, leading to lower osteoclast number and suppressed bone resorption (Figure 1). In addition, eldecalcitol treatment promoted focal bone formation independent of bone resorption, a process known as bone minimodeling.
Figure 1.
Schematic model of eldecalcitol action on bone cells. Eldecalcitol enhances differentiation of RANKL-rich preosteoblasts to RANKL-poor mature osteoblasts. As a result, the number of RANKL-rich cells is reduced in bone surface. RANKL suppresses the expression of S1PR1 in preosteoclasts. Reduction in RANKL expression by eldecalcitol induces S1PR1. Eldecalcitol also suppresses the expression of S1PR2 in preosteoclasts, and an increase in S1PR1 by eldecalcitol cooperatively stimulates the mobilization of preosteoclasts from bone marrow to blood circulation where S1P concentration is high.
Because eldecalcitol suppresses bone resorption, its effect on callus remodeling was compared with alendronate. Although alendronate treatment induced large calluses, eldecalcitol did not interfere with the fracture healing process and did not affect the structural or material properties of fractured bone.11 Yamane et al.12 examined the effect of eldecalcitol on bone modeling in distraction osteogenesis and found that eldecalcitol increased callus volume during the early period after the completion of lengthening, resulting in thick cortical bone formation. Taken together, these studies demonstrate that, although eldecalcitol suppresses bone resorption, it does not interfere with fracture healing or bone formation processes in distraction osteogenesis.
Although active vitamin D compounds induce receptor activator of NF-κB ligand (RANKL) expression in osteoblastic cells and enhance osteoclast formation and bone resorption in vitro,13 the above OVX rat study demonstrated that both alfacalcidol and eldecalcitol suppress bone resorption in vivo. Harada et al.14 demonstrated that eldecalcitol suppresses bone resorption by reducing the number of RANKL-positive cells on the trabecular bone surface. Osteoclasts are formed from monocytic precursors, and sphingosine-1-phosphate (S1P), a lipid mediator in blood circulation, controls the migration of monocytic osteoclast precursors via binding to its chemoattractive receptor, S1PR1, or chemorepulsive receptor, S1PR2. Ishii et al.15 demonstrated that RANKL stimulation suppresses the expression of S1PR1 and enhances osteoclast formation, whereas calcitriol and eldecalcitol suppress the expression of S1PR2 to enhance migration of osteoclast precursors from bone to blood circulation and thus suppress osteoclast formation (Figure 1).16 Although these results cannot explain the stronger suppressive effect of eldecalcitol on bone resorption, the much higher affinity of eldecalcitol for serum vitamin D-binding protein than alfacalcidol may yield a higher concentration of eldecalcitol in the bone marrow and give it a superior suppressive effect on bone resorption.
Clinical studies
When a once-a-day dose of 0.1, 0.25, 0.5 or 1.0 μg eldecalcitol was given to 40 healthy male volunteers for 15 days, doses of 0.5 to 1.0 μg per day caused a dose-dependent decrease in the bone resorption markers urinary deoxypyridinoline and type I collagen C-telopeptide, with less suppression of the bone formation markers bone-specific alkaline phosphatase and osteocalcin. Although serum and urinary Ca increased slightly with 1.0 μg eldecalcitol, none of the subjects showed sustained increase in serum Ca of over 10.4 mg dl−1 or urinary Ca of over 400 mg per day. Those results demonstrated that eldecalcitol at a dose of 0.5 to 1.0 μg per day can suppress bone resorption without causing sustained hypercalcemia or hypercalciuria.17 The effects of eldecalcitol on bone turnover markers and Ca metabolism were compared with those of alfacalcidol in a randomized open-label study in 59 Japanese postmenopausal women. The subjects were randomly assigned to receive 0.5 or 1.0 μg eldecalcitol or 1.0 μg alfacalcidol once a day for 12 weeks. Both 0.5 and 1.0 μg eldecalcitol suppressed urinary type I collagen N-telopeptide more strongly than did 1.0 μg alfacalcidol. In contrast, changes in serum bone-specific alkaline phosphatase were similar among the three groups. Urinary Ca increased with 0.5 and 1.0 μg eldecalcitol in a dose-dependent manner, and 1.0 μg eldecalcitol increased urinary Ca to an extent similar to 1.0 μg alfacalcidol. There was almost no increase in serum Ca in any of the three study groups throughout the study.18
If the effect of eldecalcitol on bone is stronger than the effect of calcitriol, the skeletal effect of eldecalcitol should be detectable even when there is a sufficient supply of vitamin D. In order to find out the optimal dose of eldecalcitol to increase BMD in osteoporotic patients with sufficient vitamin D supply, a double-blind, placebo-controlled study involving 219 osteoporotic patients was performed. In that study, patients were randomly assigned to receive placebo or 0.5, 0.75 or 1.0 μg eldecalcitol once a day for 12 months under supplementation with 200 IU vitamin D3 for patients with serum 25(OH)D of 20 ng ml−1 or higher and 400 IU vitamin D3 for patients with serum 25(OH)D below 20 ng ml−1. Serum eldecalcitol concentration increased linearly with the increasing dose of eldecalcitol. Serum 25(OH)D was maintained at over 20 ng ml−1 in 92% of the participants. The results demonstrated that 0.75 μg eldecalcitol can effectively and safely increase lumbar and hip BMD.19 The effect of eldecalcitol in increasing BMD was not dependent upon vitamin D status, because the effect of eldecalcitol on lumbar and hip BMD was similar between subjects in the upper tertile of serum 25(OH)D concentration (>29 ng ml−1) and those in the lower tertile (<25 ng ml−1).20
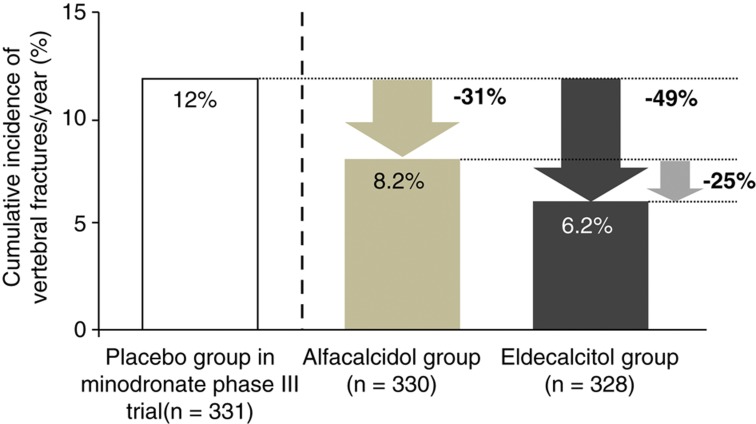
On the basis of those studies, a 3-year double-blind, active comparator clinical trial was conducted to test the superiority of oral 0.75 μg per day eldecalcitol over 1.0 μg per day alfacalcidol for the prevention of osteoporotic fractures. That study involved 1054 patients with osteoporosis, with 400 IU vitamin D3 supplementation given to patients with serum 25(OH)D of less than 20 ng ml−1. Compared with the alfacalcidol group, the incidence of vertebral fractures was significantly lower in the eldecalcitol group after 36 months of treatment (13.4%/17.5%; hazard ratio, 0.74) under the predefined significance level of one-sided alpha of 0.05. The effect of eldecalcitol on vertebral fracture incidence was sustained throughout the 3-year study period, and the annual incidence of vertebral fracture during the 3rd year was significantly lower in the eldecalcitol group than in the alfacalcidol group (3.9% vs 7.0%, respectively). Again, eldecalcitol reduced bone turnover markers and increased BMD more strongly than did alfacalcidol. In addition, while bone resorption markers were suppressed to a similar level throughout the 3-year study period, bone-specific alkaline phosphatase reached a nadir at 1 year after treatment, and gradually increased thereafter. Among patients with two or more prevalent vertebral fractures, the hazard ratio for incident vertebral fractures was 0.61 (95% confidence interval , 0.40–0.93) in favor of eldecalcitol over alfacalcidol. The hazard ratio for incident vertebral fractures among patients with a total hip BMD T-score below −2.5 was 0.56 (95% confidence interval, 0.34–0.90). There were no significant interactions between treatment effect and any baseline clinical findings. These results indicate the superior effect of eldecalcitol on vertebral fractures among patients with multiple prevalent vertebral fractures or low total hip BMD. Eldecalcitol reduced the incidence of three major nonvertebral fractures, which was due to a marked reduction in wrist fractures (1.1 vs 3.6%; hazard ratio, 0.29). The incidence of increase in serum and urinary Ca was higher in the eldecalcitol group, without any difference in the estimated glomerular filtration rate between the two groups. Thus, eldecalcitol is more efficacious than alfacalcidol in preventing vertebral and wrist fractures in osteoporotic patients with vitamin D sufficiency, with a safety profile similar to that of alfacalcidol.21 However, that study was not a placebo-controlled study, and it is difficult to compare the potency of the antifracture effect of eldecalcitol with that of other therapeutic agents of osteoporosis. In this context, Hagino analyzed antifracture effect of alfacalcidol and eldecalcitol in comparison with placebo control of a 2-year fracture prevention trial with minodronate.22 He calculated annual fracture incidence of alfacalcidol and eldecalcitol-treated patients in the eldecalcitol trial who were eligible for the entry criteria of the minodronate trial.23 Although this is not a head-to-head comparison, the annual relative vertebral fracture risk reduction compared with placebo control of minodronate trial was 31% and 49% in alfacalcidol and eldecalcitol groups, respectively (Figure 2).
Figure 2.
Estimation of relative risk reduction of vertebral fractures compared with the placebo group of the minodronate phase III trial. Post hoc analysis was performed in the participants of fracture prevention trial with eldecalcitol who met the entry criteria of minodronate phase III trial (with one to five fragility fractures between the vertebrae Th4 and L4, and L2-4 BMD below T-score -1.7). The annual vertebral fracture incidence in alfacalcidol and eldecalcitol groups of the 3-year eldecalcitol trial was compared with that of placebo-treated patients in 2-year phase III minodronate trial. Relative risk reduction in an eldecalcitol group compared with the placebo group was estimated to be 49%, and from an alfacalcitol group was 25%, which was very similar to the 26% reduction among the whole participants of the eldecalcitol fracture prevention trial. Adapted from reference 23.
It is noteworthy that there was a marked reduction in the incidence of wrist fractures in the eldecalcitol group. Native or active forms of vitamin D have been shown to reduce falls in elderly subjects.24 The health-related quality of life on the patient in this fracture prevention trial was analyzed and showed better improvement by eldecalcitol than alfacalcidol,25 the number of falls was not monitored. Therefore, it is not known whether eldecalcitol has a stronger effect in preventing falls than alfacalcidol, and the reasons for the marked reduction in wrist fractures remain unclear.
The effects of eldecalcitol on the bone geometry and biomechanical properties of the proximal femur were examined by using clinical computed tomography in a 193-patient subgroup of the fracture prevention trial. Multidetector-row CT data were acquired at baseline and the end of the 3-year study. The results revealed that eldecalcitol increased cortical cross-sectional area, volumetric BMD and bone mass, and maintained cortical thickness better than alfacalcidol. As a result, the biomechanical properties of the femoral neck, including cross-sectional moment of inertia and section modulus, were also improved more by eldecalcitol than by alfacalcidol. Although buckling ratio increased in both groups, the increase was less in the eldecalcitol group.26 Taken together, these results suggest that eldecalcitol may have the potential to reduce fracture risk by improving bone geometry and biomechanical properties of the proximal femur.
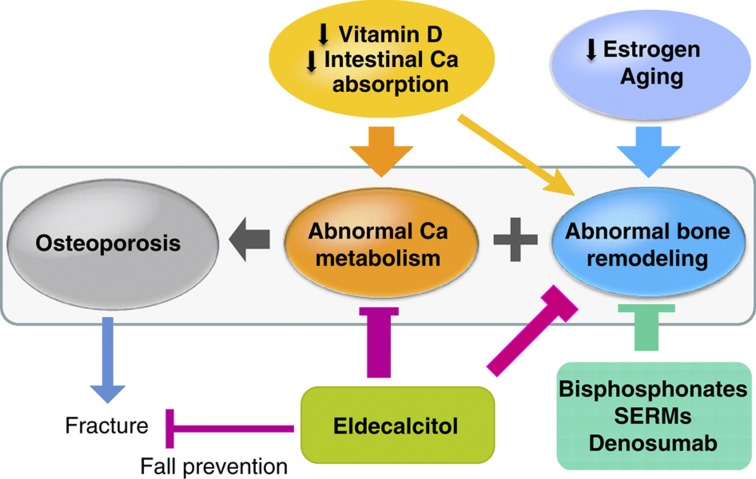
Many questions remain unanswered with respect to the effects and the mechanism of action of eldecalcitol. First of all, the mechanism of action underlying the skeletal effects of eldecalcitol is unclear, and the most important question of how eldecalcitol exhibits a stronger effect on bone relative to its effect on intestinal Ca absorption remains unanswered. Second, although a superior effect of eldecalcitol over alfacalcidol in preventing vertebral fractures has been demonstrated, it is important to know its antifracture effect in comparison with placebo controls and with other drugs used for the treatment of osteoporosis, including bisphosphonates and selective estrogen receptor modulators. Third, the extraskeletal effect of eldecalcitol needs to be examined. It is especially important to examine its effect on falls, muscle strength and physical activity. Fourth, although there was a significant reduction in wrist fractures by eldecalcitol treatment, its effect on other nonvertebral fractures—most importantly, on hip fractures—has not been investigated. Fifth, eldecalcitol stimulates intestinal Ca absorption similarly to 1,25(OH)2D3, and its major side effect is hypercalcemia and hypercalciuria. The safety of eldecalcitol treatment in terms of the risks of hypercalcemia and hypercalciuria needs to be further monitored. Finally, because eldecalcitol stimulates intestinal Ca absorption and improves Ca balance in addition to its skeletal effects, it will be interesting to examine the effect of combined treatment with antiresorptive agents instead of using native vitamin D as a supplement. Because eldecalcitol has so far been approved for the treatment of osteoporosis only in Japan, it is difficult to accumulate the evidence needed to clarify many of the important questions indicated above. However, if one takes into account the fact that eldecalcitol is a drug that has effects in improving both Ca balance and bone remodeling, it is worthwhile to try to answer these questions for further improvement of the treatment of osteoporosis (Figure 3).
Figure 3.
Abnormal bone remodeling and calcium metabolism in patients with osteoporosis. Antiresorptive agents improve abnormal bone remodeling in osteoporotic patients. In order for these agents to enhance bone mineralization and improve bone strength, reduction in intestinal Ca absorption observed in the elderly subjects needs to be corrected. Most of the global fracture prevention studies use native vitamin D and calcium as supplements. However, eldecalcitol is available in Japan, which not only improves negative Ca balance by enhancing intestinal Ca absorption but also improves negative bone remodeling balance, and reduces vertebral fracture incidence. Eldecalcitol may reduce falls, like other active vitamin D compounds that are reported to have a preventive effect on falls. This may also contribute to the reduction in fractures.
Acknowledgments
We thank Mr Satoshi Kondo and Ms Maki Saito for their assistance in manuscript preparation.
Footnotes
TM receives consultant fees from Chugai Pharmaceutical Co., Ltd. TT, HS and FT are employees of Chugai Pharmaceutical Co., Ltd.
References
- NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA 2001;285:785–795. [DOI] [PubMed] [Google Scholar]
- Papadimitropoulos E, Wells G, Shea B, Gillespie W, Weaver B, Zytaruk N et al. Osteoporosis Methodology G, The Osteoporosis Research Advisory Group. Meta-analyses of therapies for postmenopausal osteoporosis. VIII: Meta-analysis of the efficacy of vitamin D treatment in preventing osteoporosis in postmenopausal women. Endocr Rev 2002;23:560–569. [DOI] [PubMed] [Google Scholar]
- Miyamoto K, Murayama E, Ochi K, Watanabe H, Kubodera N. Synthetic studies of vitamin D analogues. XIV. Synthesis and calcium regulating activity of vitamin D3 analogues bearing a hydroxyalkoxy group at the 2 beta-position. Chem Pharm Bull 1993;41:1111–1113. [DOI] [PubMed] [Google Scholar]
- Ono Y, Watanabe H, Shiraishi A, Takeda S, Higuchi Y, Sato K et al. Synthetic studies of vitamin D analogs. XXIV. Synthesis of active vitamin D3 analogs substituted at the 2 beta-position and their preventive effects on bone mineral loss in ovariectomized rats. Chem Pharm Bull 1997;45:1626–1630. [DOI] [PubMed] [Google Scholar]
- Hatakeyama S, Nagashima S, Imai N, Takahashi K, Ishihara J, Sugita A et al. Synthesis and biological evaluation of a 3-positon epimer of 1alpha,25-dihydroxy-2beta-(3-hydroxypropoxy)vitamin D3 (ED-71). J Steroid Biochem Mol Biol 2007;103:222–226. [DOI] [PubMed] [Google Scholar]
- Abe M, Tsuji N, Takahashi F, Tanigawara Y. Overview of the Clinical Pharmacokinetics of Eldecalcitol, a New Active Vitamin D3 Derivative. Jpn Pharmacol Ther 2011;39:261–274. [Google Scholar]
- Harada S, Takeda S, Uno A, Takahashi F, Saito H. Eldecalcitol is less effective in suppressing parathyroid hormone compared to calcitriol in vivo. J Steroid Biochem Mol Biol 2010;121:281–283. [DOI] [PubMed] [Google Scholar]
- Uchiyama Y, Higuchi Y, Takeda S, Masaki T, Shira-Ishi A, Sato K et al. ED-71, a vitamin D analog, is a more potent inhibitor of bone resorption than alfacalcidol in an estrogen-deficient rat model of osteoporosis. Bone 2002;30:582–588. [DOI] [PubMed] [Google Scholar]
- Tsurukami H, Nakamura T, Suzuki K, Sato K, Higuchi Y, Nishii Y. A novel synthetic vitamin D analogue, 2 beta-(3-hydroxypropoxy)1 alpha, 25-dihydroxyvitamin D3 (ED-71), increases bone mass by stimulating the bone formation in normal and ovariectomized rats. Calcif Tissue Int 1994;54:142–149. [DOI] [PubMed] [Google Scholar]
- de Freitas PH, Hasegawa T, Takeda S, Sasaki M, Tabata C, Oda K et al. Eldecalcitol, a second-generation vitamin D analog, drives bone minimodeling and reduces osteoclastic number in trabecular bone of ovariectomized rats. Bone 2011;49:335–342. [DOI] [PubMed] [Google Scholar]
- Cao Y, Mori S, Mashiba T, Kaji Y, Manabe T, Iwata K et al. 1Alpha,25-dihydroxy-2beta(3-hydroxypropoxy)vitamin D3 (ED-71) suppressed callus remodeling but did not interfere with fracture healing in rat femora. Bone 2007;40:132–139. [DOI] [PubMed] [Google Scholar]
- Yamane K, Okano T, Kishimoto H, Hagino H. Effect of ED-71 on modeling of bone in distraction osteogenesis. Bone 1999;24:187–193. [DOI] [PubMed] [Google Scholar]
- Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 1998;95:3597–3602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harada S, Mizoguchi T, Kobayashi Y, Nakamichi Y, Takeda S, Sakai S et al. Daily administration of eldecalcitol (ED-71), an active vitamin D analog, increases bone mineral density by suppressing RANKL expression in mouse trabecular bone. J Bone Miner Res 2012;27:461–473. [DOI] [PubMed] [Google Scholar]
- Ishii M, Egen JG, Klauschen F, Meier-Schellersheim M, Saeki Y, Vacher J et al. Sphingosine-1-phosphate mobilizes osteoclast precursors and regulates bone homeostasis. Nature 2009;458:524–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kikuta J, Kawamura S, Okiji F, Shirazaki M, Sakai S, Saito H et al. Sphingosine-1-phosphate-mediated osteoclast precursor monocyte migration is a critical point of control in antibone-resorptive action of active vitamin D. Proc Natl Acad Sci USA 2013;110:7009–7013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto T, Kubodera N. The ED-71 Study Group. 1a,25-Dihydroxy-2b(3-hydroxypropoxy)vitamin D3 (ED-71): a promising candidate for the treatment of osteoporosis. In: Norman AW, Bouillon R, Thomasset M (eds).Vitamin D Endocrine System: Structural, Biological, Genetic and Clinical Aspects Vitamin D Workshop Inc, University of California: Riverside, 2000; pp985–992. [Google Scholar]
- Matsumoto T, Takano T, Yamakido S, Takahashi F, Tsuji N. Comparison of the effects of eldecalcitol and alfacalcidol on bone and calcium metabolism. J Steroid Biochem Mol Biol 2010;121:261–264. [DOI] [PubMed] [Google Scholar]
- Matsumoto T, Miki T, Hagino H, Sugimoto T, Okamoto S, Hirota T et al. A new active vitamin D, ED-71, increases bone mass in osteoporotic patients under vitamin D supplementation: a randomized, double-blind, placebo-controlled clinical trial. J Clin Endocrinol Metab 2005;90:5031–5036. [DOI] [PubMed] [Google Scholar]
- Matsumoto T, Kubodera N. ED-71 Study Group. ED-71,a new active vitamin D3, increases bone mineral density regardless of serum 25(OH)D levels in osteoporotic subjects. J Steroid Biochem Mol Biol 2007;103:584–586. [DOI] [PubMed] [Google Scholar]
- Matsumoto T, Ito M, Hayashi Y, Hirota T, Tanigawara Y, Sone T et al. A new active vitamin D3 analog, eldecalcitol, prevents the risk of osteoporotic fractures - a randomized, active comparator, double-blind study. Bone 2011;49:605–612. [DOI] [PubMed] [Google Scholar]
- Matsumoto T, Hagino H, Shiraki M, Fukunaga M, Nakano T, Takaoka K et al. Effect of daily oral minodronate on vertebral fractures in Japanese postmenopausal women with established osteoporosis: a randomized placebo-controlled double-blind study. Osteoporos Int 2009;20:1429–1437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagino H. Eldecalcitol: newly developed active vitamin D3 analog for the treatment of osteoporosis. Expert Opin. Pharmacother 2013;14:817–825. [DOI] [PubMed] [Google Scholar]
- Bischoff-Ferrari HA, Dawson-Hughes B, Willett WC, Staehelin HB, Bazemore MG, Zee RY et al. Effect of vitamin D on falls: a meta-analysis. JAMA 2004;291:1999–2006. [DOI] [PubMed] [Google Scholar]
- Hagino H, Takano T, Fukunaga M, Shiraki M, Nakamura T, Matsumoto T. Eldecalcitol reduces the risk of severe vertebral fractures and improves the health-related quality of life in patients with osteoporosis. J Bone Miner Metab 2013;31:183–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito M, Nakamura T, Fukunaga M, Shiraki M, Matsumoto T. Effect of eldecalcitol, an active vitamin D analog, on hip structure and biomechanical properties: 3D assessment by clinical CT. Bone 2011;49:328–334. [DOI] [PubMed] [Google Scholar]