Abstract
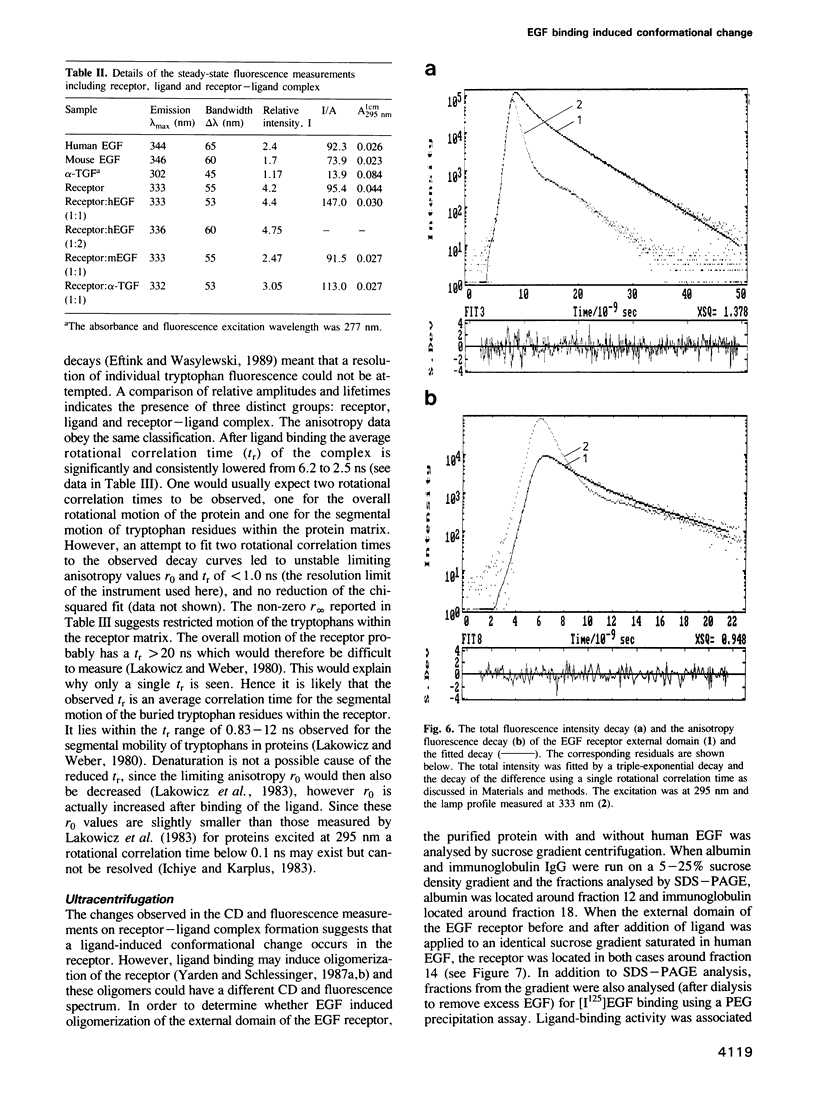
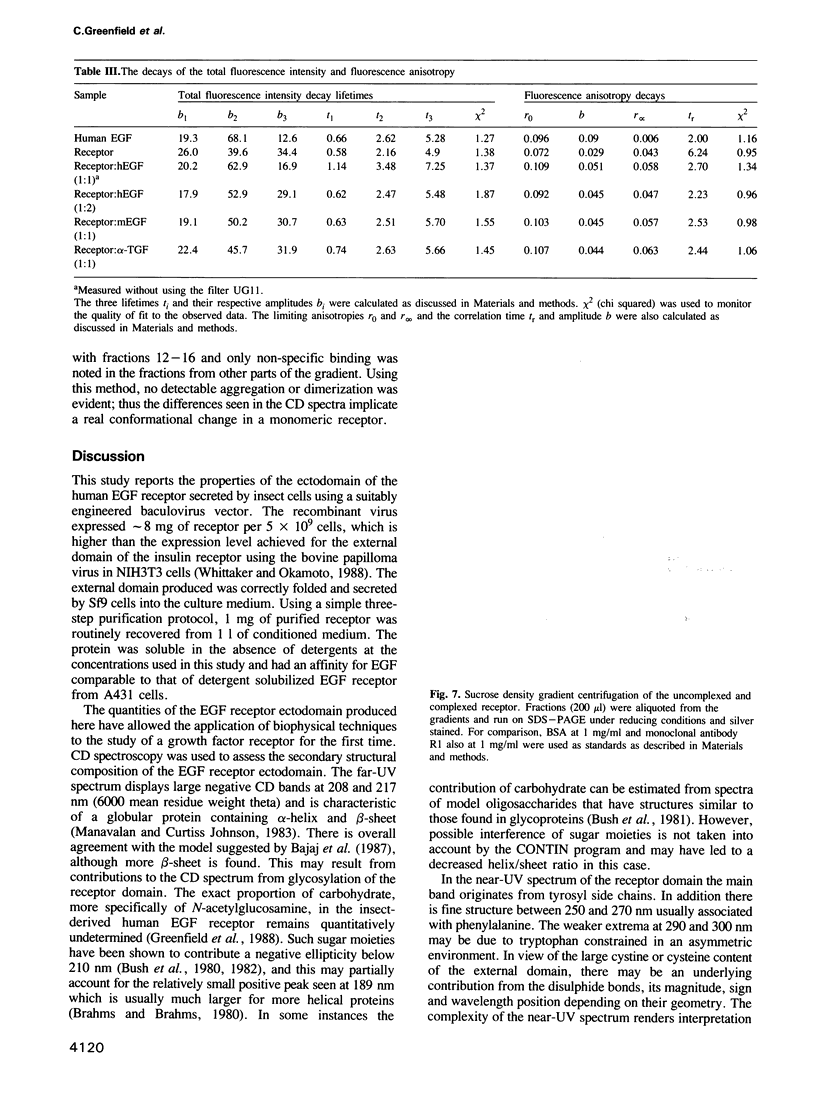
To study the properties of the extracellular epidermal growth factor (EGF) binding domain of the human EGF receptor, we have infected insect cells with a suitably engineered baculovirus vector containing the cDNA encoding the entire ectodomain of the parent molecule. This resulted in a correctly folded, stable, 110 kd protein which possessed an EGF binding affinity of 200 nM. The protein was routinely purified in milligram amounts from 1 litre insect cell cultures using a series of three standard chromatographic steps. The properties of the ectodomain were studied before and after the addition of different EGF ligands, using both circular dichroism and fluorescence spectroscopic techniques. A secondary structural analysis of the far UV CD spectrum of the ectodomain indicated significant proportions of alpha-helix and beta-sheet in agreement with a published model of the EGF receptor. The ligand additions to the receptor showed differences in both the near- and far-UV CD spectra, and were similar for each ligand used, suggesting similar conformational differences between uncomplexed and complexed receptor. Steady-state fluorescence measurements indicated that the tryptophan residues present in the ectodomain are buried and that the solvent-accessible tryptophans in the ligands become buried on binding the receptor. The rotational correlation times measured by fluorescence anisotropy decay for the receptor-ligand complexes were decreased from 6 to 2.5 ns in each case. This may indicate a perturbation of the tryptophan environment of the receptor on ligand binding. Ultracentrifugation studies showed that no aggregation occurred on ligand addition, so this could not explain the observed differences from CD or fluorescence.(ABSTRACT TRUNCATED AT 250 WORDS)
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bajaj M., Waterfield M. D., Schlessinger J., Taylor W. R., Blundell T. On the tertiary structure of the extracellular domains of the epidermal growth factor and insulin receptors. Biochim Biophys Acta. 1987 Nov 26;916(2):220–226. doi: 10.1016/0167-4838(87)90112-9. [DOI] [PubMed] [Google Scholar]
- Brahms S., Brahms J. Determination of protein secondary structure in solution by vacuum ultraviolet circular dichroism. J Mol Biol. 1980 Apr;138(2):149–178. doi: 10.1016/0022-2836(80)90282-x. [DOI] [PubMed] [Google Scholar]
- Burstein E. A., Vedenkina N. S., Ivkova M. N. Fluorescence and the location of tryptophan residues in protein molecules. Photochem Photobiol. 1973 Oct;18(4):263–279. doi: 10.1111/j.1751-1097.1973.tb06422.x. [DOI] [PubMed] [Google Scholar]
- Bush C. A., Dua V. K., Ralapati S., Warren C. D., Spik G., Strecker G., Montreuil J. Conformation of the complex oligosaccharides of glycoproteins. A vacuum ultraviolet circular dichroism study. J Biol Chem. 1982 Jul 25;257(14):8199–8204. [PubMed] [Google Scholar]
- Bush C. A., Duben A., Ralapati S. Conformation of the glycopeptide linkage in asparagine-linked glycoproteins. Biochemistry. 1980 Feb 5;19(3):501–504. doi: 10.1021/bi00544a017. [DOI] [PubMed] [Google Scholar]
- Bush C. A., Feeney R. E., Osuga D. T., Ralapati S., Yeh Y. Antifreeze glycoprotein. Conformational model based on vacuum ultraviolet circular dichroism data. Int J Pept Protein Res. 1981 Jan;17(1):125–129. doi: 10.1111/j.1399-3011.1981.tb01975.x. [DOI] [PubMed] [Google Scholar]
- Böni-Schnetzler M., Pilch P. F. Mechanism of epidermal growth factor receptor autophosphorylation and high-affinity binding. Proc Natl Acad Sci U S A. 1987 Nov;84(22):7832–7836. doi: 10.1073/pnas.84.22.7832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carpenter G. Receptors for epidermal growth factor and other polypeptide mitogens. Annu Rev Biochem. 1987;56:881–914. doi: 10.1146/annurev.bi.56.070187.004313. [DOI] [PubMed] [Google Scholar]
- Chen Y. H., Yang J. T., Martinez H. M. Determination of the secondary structures of proteins by circular dichroism and optical rotatory dispersion. Biochemistry. 1972 Oct 24;11(22):4120–4131. doi: 10.1021/bi00772a015. [DOI] [PubMed] [Google Scholar]
- Cooke R. M., Wilkinson A. J., Baron M., Pastore A., Tappin M. J., Campbell I. D., Gregory H., Sheard B. The solution structure of human epidermal growth factor. 1987 May 28-Jun 3Nature. 327(6120):339–341. doi: 10.1038/327339a0. [DOI] [PubMed] [Google Scholar]
- Cooke R. M., Wilkinson A. J., Baron M., Pastore A., Tappin M. J., Campbell I. D., Gregory H., Sheard B. The solution structure of human epidermal growth factor. 1987 May 28-Jun 3Nature. 327(6120):339–341. doi: 10.1038/327339a0. [DOI] [PubMed] [Google Scholar]
- DiPaola M., Maxfield F. R. Conformational changes in the receptors for epidermal growth factor and asialoglycoproteins induced by the mildly acidic pH found in endocytic vesicles. J Biol Chem. 1984 Jul 25;259(14):9163–9171. [PubMed] [Google Scholar]
- Downward J., Parker P., Waterfield M. D. Autophosphorylation sites on the epidermal growth factor receptor. Nature. 1984 Oct 4;311(5985):483–485. doi: 10.1038/311483a0. [DOI] [PubMed] [Google Scholar]
- Eftink M. R., Wasylewski Z. Fluorescence lifetime and solute quenching studies with the single tryptophan containing protein parvalbumin from codfish. Biochemistry. 1989 Jan 10;28(1):382–391. doi: 10.1021/bi00427a053. [DOI] [PubMed] [Google Scholar]
- Ellis L., Levitan A., Cobb M. H., Ramos P. Efficient expression in insect cells of a soluble, active human insulin receptor protein-tyrosine kinase domain by use of a baculovirus vector. J Virol. 1988 May;62(5):1634–1639. doi: 10.1128/jvi.62.5.1634-1639.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabricant R. N., De Larco J. E., Todaro G. J. Nerve growth factor receptors on human melanoma cells in culture. Proc Natl Acad Sci U S A. 1977 Feb;74(2):565–569. doi: 10.1073/pnas.74.2.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenfield C., Patel G., Clark S., Jones N., Waterfield M. D. Expression of the human EGF receptor with ligand-stimulatable kinase activity in insect cells using a baculovirus vector. EMBO J. 1988 Jan;7(1):139–146. doi: 10.1002/j.1460-2075.1988.tb02793.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hillman G. M., Schlessinger J. Lateral diffusion of epidermal growth factor complexed to its surface receptors does not account for the thermal sensitivity of patch formation and endocytosis. Biochemistry. 1982 Mar 30;21(7):1667–1672. doi: 10.1021/bi00536a030. [DOI] [PubMed] [Google Scholar]
- Ichiye T., Karplus M. Fluorescence depolarization of tryptophan residues in proteins: a molecular dynamics study. Biochemistry. 1983 Jun 7;22(12):2884–2893. doi: 10.1021/bi00281a017. [DOI] [PubMed] [Google Scholar]
- Johnson J. D., Wong M. L., Rutter W. J. Properties of the insulin receptor ectodomain. Proc Natl Acad Sci U S A. 1988 Oct;85(20):7516–7520. doi: 10.1073/pnas.85.20.7516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson W. C., Jr Secondary structure of proteins through circular dichroism spectroscopy. Annu Rev Biophys Biophys Chem. 1988;17:145–166. doi: 10.1146/annurev.bb.17.060188.001045. [DOI] [PubMed] [Google Scholar]
- Kahn P. C. The interpretation of near-ultraviolet circular dichroism. Methods Enzymol. 1979;61:339–378. doi: 10.1016/0076-6879(79)61018-2. [DOI] [PubMed] [Google Scholar]
- Koland J. G., Cerione R. A. Growth factor control of epidermal growth factor receptor kinase activity via an intramolecular mechanism. J Biol Chem. 1988 Feb 15;263(5):2230–2237. [PubMed] [Google Scholar]
- Lakowicz J. R. Fluorescence studies of structural fluctuations in macromolecules as observed by fluorescence spectroscopy in the time, lifetime, and frequency domains. Methods Enzymol. 1986;131:518–567. doi: 10.1016/0076-6879(86)31054-1. [DOI] [PubMed] [Google Scholar]
- Lakowicz J. R., Freshwater G., Weber G. Nanosecond segmental mobilities of tryptophan residues in proteins observed by lifetime-resolved fluorescence anisotropies. Biophys J. 1980 Oct;32(1):591–601. doi: 10.1016/S0006-3495(80)84992-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakowicz J. R., Maliwal B. P., Cherek H., Balter A. Rotational freedom of tryptophan residues in proteins and peptides. Biochemistry. 1983 Apr 12;22(8):1741–1752. doi: 10.1021/bi00277a001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lax I., Bellot F., Howk R., Ullrich A., Givol D., Schlessinger J. Functional analysis of the ligand binding site of EGF-receptor utilizing chimeric chicken/human receptor molecules. EMBO J. 1989 Feb;8(2):421–427. doi: 10.1002/j.1460-2075.1989.tb03393.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lax I., Burgess W. H., Bellot F., Ullrich A., Schlessinger J., Givol D. Localization of a major receptor-binding domain for epidermal growth factor by affinity labeling. Mol Cell Biol. 1988 Apr;8(4):1831–1834. doi: 10.1128/mcb.8.4.1831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montelione G. T., Wüthrich K., Nice E. C., Burgess A. W., Scheraga H. A. Solution structure of murine epidermal growth factor: determination of the polypeptide backbone chain-fold by nuclear magnetic resonance and distance geometry. Proc Natl Acad Sci U S A. 1987 Aug;84(15):5226–5230. doi: 10.1073/pnas.84.15.5226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philips A. V., Coleman M. S., Maskos K., Barkley M. D. Time-resolved fluorescence spectroscopy of human adenosine deaminase: effects of enzyme inhibitors on protein conformation. Biochemistry. 1989 Mar 7;28(5):2040–2050. doi: 10.1021/bi00431a012. [DOI] [PubMed] [Google Scholar]
- Provencher S. W., Glöckner J. Estimation of globular protein secondary structure from circular dichroism. Biochemistry. 1981 Jan 6;20(1):33–37. doi: 10.1021/bi00504a006. [DOI] [PubMed] [Google Scholar]
- Riedel H., Schlessinger J., Ullrich A. A chimeric, ligand-binding v-erbB/EGF receptor retains transforming potential. Science. 1987 Apr 10;236(4798):197–200. doi: 10.1126/science.3494307. [DOI] [PubMed] [Google Scholar]
- Savage C. R., Jr, Inagami T., Cohen S. The primary structure of epidermal growth factor. J Biol Chem. 1972 Dec 10;247(23):7612–7621. [PubMed] [Google Scholar]
- Schlessinger J. Signal transduction by allosteric receptor oligomerization. Trends Biochem Sci. 1988 Nov;13(11):443–447. doi: 10.1016/0968-0004(88)90219-8. [DOI] [PubMed] [Google Scholar]
- Skehel J. J., Bayley P. M., Brown E. B., Martin S. R., Waterfield M. D., White J. M., Wilson I. A., Wiley D. C. Changes in the conformation of influenza virus hemagglutinin at the pH optimum of virus-mediated membrane fusion. Proc Natl Acad Sci U S A. 1982 Feb;79(4):968–972. doi: 10.1073/pnas.79.4.968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith G. E., Summers M. D., Fraser M. J. Production of human beta interferon in insect cells infected with a baculovirus expression vector. Mol Cell Biol. 1983 Dec;3(12):2156–2165. doi: 10.1128/mcb.3.12.2156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turkewitz A. P., Schwartz A. L., Harrison S. C. A pH-dependent reversible conformational transition of the human transferrin receptor leads to self-association. J Biol Chem. 1988 Nov 5;263(31):16309–16315. [PubMed] [Google Scholar]
- Ullrich A., Coussens L., Hayflick J. S., Dull T. J., Gray A., Tam A. W., Lee J., Yarden Y., Libermann T. A., Schlessinger J. Human epidermal growth factor receptor cDNA sequence and aberrant expression of the amplified gene in A431 epidermoid carcinoma cells. 1984 May 31-Jun 6Nature. 309(5967):418–425. doi: 10.1038/309418a0. [DOI] [PubMed] [Google Scholar]
- Whittaker J., Okamoto A. Secretion of soluble functional insulin receptors by transfected NIH3T3 cells. J Biol Chem. 1988 Mar 5;263(7):3063–3066. [PubMed] [Google Scholar]
- Yarden Y., Schlessinger J. Epidermal growth factor induces rapid, reversible aggregation of the purified epidermal growth factor receptor. Biochemistry. 1987 Mar 10;26(5):1443–1451. doi: 10.1021/bi00379a035. [DOI] [PubMed] [Google Scholar]
- Yarden Y., Schlessinger J. Self-phosphorylation of epidermal growth factor receptor: evidence for a model of intermolecular allosteric activation. Biochemistry. 1987 Mar 10;26(5):1434–1442. doi: 10.1021/bi00379a034. [DOI] [PubMed] [Google Scholar]
- Yarden Y., Ullrich A. Growth factor receptor tyrosine kinases. Annu Rev Biochem. 1988;57:443–478. doi: 10.1146/annurev.bi.57.070188.002303. [DOI] [PubMed] [Google Scholar]
- Zidovetzki R., Yarden Y., Schlessinger J., Jovin T. M. Rotational diffusion of epidermal growth factor complexed to cell surface receptors reflects rapid microaggregation and endocytosis of occupied receptors. Proc Natl Acad Sci U S A. 1981 Nov;78(11):6981–6985. doi: 10.1073/pnas.78.11.6981. [DOI] [PMC free article] [PubMed] [Google Scholar]