Abstract
Angiogenin (ANG) is reportedly multifunctional, with roles in angiogenesis and autoimmune diseases. This protein is involved in the innate immune system and has been implicated in several inflammatory diseases. Although ANG may be involved in the anti-inflammatory response, there is no evidence that it has direct anti-inflammatory effects. In this study we sought to determine whether ANG has an anti-inflammatory effect in human corneal fibroblasts (HCFs) exposed to media containing tumor necrosis factor-alpha (TNF-α). We found that ANG reduced the mRNA expression of interleukin-1 beta (IL-1β), -6, -8 and TNF-α receptors (TNFR) 1 and 2. In contrast, ANG increased the mRNA expression of IL-4 and -10. Protein levels of TANK-binding kinase 1 (TBK1) were reduced by ANG in HCFs treated with TNF-α. Moreover, ANG diminished the expression of IL-6 and -8 and monocyte chemotactic protein- (MCP-) 1. The protein expression of nuclear factor-κB (NF-κB) was downregulated by ANG treatment. These findings suggest that ANG suppressed the TNF-α-induced inflammatory response in HCFs through inhibition of TBK1-mediated NF-κB nuclear translocation. These novel results are likely to play a significant role in the selection of immune-mediated inflammatory therapeutic targets and may shed light on the pathogenesis of immune-mediated inflammatory diseases.
1. Introduction
Ocular inflammation is one of the main causes of blindness and visual disturbance. A number of ocular inflammatory diseases cause visual impairment and chronic immune-mediated inflammation in the eye can lead to blindness [1–4]. Inflammation is a main component in the pathophysiology of several ocular diseases including corneal and autoimmune diseases. It has generally been accepted that inflammatory cytokines and chemokines are significantly increased in many ocular diseases and immune rejection of corneal transplantation [5–8].
The corneal stroma is a thick, transparent layer that is frequently subjected to the inflammatory response [9]. Chronic inflammation of corneal fibroblasts causes corneal scarring, neovascularization, edema, opacity, injury, ulceration, and ultimately impaired vision and blindness [4, 9–11]. Human corneal fibroblasts (HCFs) in the corneal stroma perform a significant function in the control of local immune and inflammation [12]. It has been widely reported that corneal fibroblast cells act as sentinel cells of the immune system and participate in the regulation of stromal inflammation through the production of cytokines and chemokines [13–15]. Corneal fibroblast cells secrete proinflammatory cytokines and chemokines including interleukin (IL)-6 and IL-8 in response to external inflammatory stimuli [12, 15, 16].
Tumor necrosis factor-α (TNF-α) is a cytokine involved in the inflammatory response that binds two receptors and tumor necrosis factor receptors (TNFR) 1 and 2. It has been demonstrated that TNF-α plays a critical role in corneal inflammation [17]. TNF-α promotes nuclear factor-κB (NF-κB) translocation, and the NF-κB signaling pathway, which is found in all mammalian cell types, comprises several genes that influence immune and inflammatory responses [18, 19]. NF-κB is activated by phosphorylation of the regulatory protein I-kappa-B (IκB) and TANK-binding kinase 1 (TBK1). TBK1 promotes NF-κB activation and may act downstream of the NF-κB transcriptional pathway [20, 21]. It is well documented that TBK1 inhibitors decrease inflammation [22].
Angiogenin (ANG) is a 14.4 kDa single chain protein containing 123 amino acids. Several studies have demonstrated its function with respect to immunity. This protein is a component of tears and protects the ocular surface as it is an antimicrobial peptide [23, 24]. ANG mRNA expression and ANG protein concentrations in serum are increased during the inflammatory response [25, 26]. Serum ANG was found to be increased in patients with inflammatory bowel disease, and ANG mRNA expression was shown to be elevated by TNF-α and IL-1β [27, 28]. ANG was also shown to have bactericidal activity [29] and has been reported to be a microbial recognition protein related to innate immunity [30]. This evidence suggests that ANG may play a role in modulating the inflammatory response.
We hypothesized that ANG has an inhibitory effect on the inflammatory response in the ocular surface. The aim of this study was to determine whether ANG has an anti-inflammatory effect in HCFs treated with TNF-α. Thus, we attempted to clarify the molecular activity of ANG underlying its inhibition TNF-α mediated transduction, which involves the production of TBK1 and nuclear translocation of NF-κB.
2. Methods
2.1. Isolation and Primary Culture of Human Corneal Fibroblast Cells
Human corneal donor tissues were obtained during penetrating keratoplasty. The corneal epithelium was eliminated and then the corneal fibroblast cells were detached from explant tissue. The corneal tissues were rinsed with phosphate-buffered saline (PBS) mixed with 5% penicillin-streptomycin. After the corneal epithelium was eliminated, the corneal stroma was cut into explants of approximately 1 mm3. Each piece of explant was placed on a culture dish and immersed in culture medium for one week. The HCFs were then subcultured by trypsin digestion. HCFs were cultured in alpha-minimum essential medium (α-MEM) (Invitrogen-Gibco, USA) containing 10% FBS and 1% penicillin-streptomycin. The cells were maintained at 37°C under 5% CO2 and used for experiments after three to five passages. The study protocol and informed consent were approved by the institutional review board of the Chung-Ang University Hospital. This study conformed to the tenets of the Declaration of Helsinki.
2.2. Cell Treatment
HCFs were cultured in six-well plates for three days. They were washed twice with PBS. The medium of confluent corneal fibroblast cells was changed to serum-free MEM for one day before treatment. The cells were treated with TNF-α purchased from PROSPEC (20 ng/mL) for eight hours, and with or without ANG (2 μg/mL) at the last 30 minutes of incubation with TNF-α. ANG was obtained from the Department of Biochemistry at Chungbuk National University and the identity of the purified ANG has been confirmed by western blotting with ANG specific antibodies by methods described in a previous report [31]. The biological activity of the purified ANG has also been confirmed by its nuclear translocation in HUVE cells by procedure described in detail [31]. The purification and endotoxin levels of recombinant ANG expressed in E. coli are shown in Supplementary Figures 1 and 2 (see the Supplememtery Material available online at http://dx.doi.org/10.1155/2014/861435). The cells were then collected for total RNA isolation and protein extraction.
2.3. RNA Isolation and Real-Time RT-PCR
Total RNA was isolated from cultured HCFs using FavorPrep Tri-RNA reagent, according to the manufacturer's protocols. The quantity and quality of the RNA were determined using a NanoDrop ND-1000 spectrophotometer (ND-1000, Nano-Drop Technologies, Inc. Wilmington, DE, USA). Single-stranded complementary DNA (cDNA) was synthesized from 500 ng of total RNA using a cDNA synthesis kit (Takara Bio Inc., Otsu, Japan). Real-time RT-PCR was conducted using the CFX96 Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA) in a total volume of 20 μL containing 10 μL of SYBR Premix Ex Taq (Takara Bio Inc., Otsu, Japan), diluted cDNA template, and forward and reverse primers. The primer sequences and product size are listed in Table 1. The PCR amplification for selected genes was run for 40 cycles. Gene expression was analyzed by real-time reverse transcriptase polymerase chain reaction (RT-PCR). Real-time PCR quantification was done in triplicate for each sample and the mean was calculated. Expression levels were analyzed by RT-PCR using values of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as a reference.
Table 1.
PCR primers used in this study.
| Gene | Sense primer (5′→3′) | Antisense primer (3′→5′) | PCR product size (bp) |
|---|---|---|---|
| GAPDH | CGAGATCCCTCCAAAATCAA | TGTGGTCATGAGTCCTTCCA | 294 |
| IL-1β | CCTGTCCTGCGTGTTGAAAGA | GGGAACTGGGCAGACTCAAA | 150 |
| IL-4 | TGTCTGTTACGGTCAACTCG | ACATTGTCACTGCAAATCGA | 195 |
| IL-6 | TTCGGTCCAGTTGCCTTCTC | GAGGTGAGTGGCTCTCTGTG | 122 |
| IL-8 | ACATGACTTCCAAGCTGGCCG | TTTATGAATTCTCAGCCCTC | 303 |
| IL-10 | GCCTAACATGCTTCGAGATC | TGATGTCTGGGTCTTGGTTC | 206 |
| TNFR1 | GTGCTGTTGCCCCTGGTCAT | GCTTAGTAGTAGTTCCTTCA | 163 |
| TNFR2 | AAACTCAAGCCTGCACTC | GGATGAAGTCGTGTTGGAGA | 209 |
| TBK1 | TTCTGGAAGTCCATACGCAT | ACTGGTGATCTCTATGCTGT | 237 |
2.4. Immunodot Blot Assay
The expression of 42 human cytokines and chemokines was assessed using a commercially available cytokine assay (RayBio Human Cytokine Antibody Array 3, RayBiotech, Norcross, GA, USA) that utilizes membrane-bound cytokine-specific antibodies to assess the presence of several cytokines in biological fluids. The analysis was conducted according to the manufacturer's instructions. Briefly, membranes were blocked for 30 minutes and then incubated with HCFs culture supernatant for two hours at room temperature. The membranes were washed with Wash Buffer I three times for five minutes each and then with Wash Buffer II twice for five minutes each. After washing, the membranes were incubated with a biotin-conjugated antibody mix for two hours, and then streptavidin-conjugated peroxidase was added for two hours at room temperature. The membranes were subsequently washed thoroughly and exposed to chemiluminescence. The membranes were visualized using the ECL Plus detection system and ChemiDoc XRS (Bio-Rad Laboratories, Inc., Berkeley, CA, USA). The densities for individual spots were calculated using ImageJ software (Wayne Rasband, National Institutes of Health, USA). The relative expression ratio was determined by subtraction of the background signal and comparison with positive controls on the membrane. Positive controls visible within each array were used for comparison.
2.5. Nuclear and Cytosolic Protein Extractions
HCFs were washed and scraped with cold PBS. The cells were lysed in buffer A (10 mM HEPES (pH 7.9), 10 mM KCl, 0.1 mM EDTA, 0.1 mM EGTA, 1 mM DTT, 0.5 mM PMSF, and 5 μg/mL Leupeptin) and left on ice for 15 min. After 10% NP-40 was added to the sample, the cytosolic fraction was collected by centrifugation at 14,000 rpm for 5 min at 4°C. The nuclear fraction was resuspended in buffer C (20 mM HEPES (pH7.9), 0.4 n NaCl, 1 mM EDTA, 1 mM EGTA, 1 mM DTT, 1 mM PMSF, and 10 μg/mL Leupeptin) and left on ice for 30 min, then the nuclear fraction was collected by centrifugation at 14,000 rpm for 5 min at 4°C.
2.6. Western Blot Analysis
Nuclear proteins and total cell lysates were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and electrophoretically transferred to a polyvinylidene fluoride membrane (PVDF; Merck Millipore, Billerica, MA, USA) at 100 V (1 h) in buffer containing 0.3% Tris, 1.4% glycine, and 20% methanol using a wet-blotting apparatus (Mini-PROTEAN Tetra cell; Bio-Rad, Hercules, CA, USA). The PVDF membrane containing the transferred proteins was blocked with 5% BSA in PBS for one hour at room temperature. Primary monoclonal antibodies against human TBK1 (Abcam, Inc.) and NF-κB (Bioworld Technology, Inc.) diluted in PBS (1 : 1000) were applied to the PVDF membrane and incubated overnight at 4°C. Secondary antibodies diluted in PBS (1 : 2000) were subsequently applied to the PVDF membrane and incubated for 1 h at room temperature. The PVDF membrane was washed four times (10 min each) with Tris-buffered saline (TBS; 50 mM Tris HCl pH 7.5, 150 mM NaCl) containing 0.1% Tween 20. The binding of specific antibodies was visualized using an enhanced chemiluminescence western blotting detection kit (Pierce Biotechnology, Inc., Rockford, IL, USA). Densitometric quantification of the immunoblot was carried out using ImageJ software. The value of each band was normalized to β-actin or lamin.
2.7. Immunocytochemistry
The HCFs cultured on glass slides were treated with TNF-α (20 ng/mL) for eight hours. Cells were also treated with or without ANG (2 μg/mL) for 0.5 hours. Cells were then fixed in 4% paraformaldehyde for 15 min at room temperature. After being permeabilized by incubation with 0.5% Triton X-100 for 15 min at room temperature, the slides were incubated with anti-NF-κB (diluted to 1 : 50 in PBS, Bioworld Technology, Inc.) for 1 h at room temperature. Glass slides were incubated with secondary antibody for 1 h at room temperature. At each step slides were washed three times (5 min each) with PBS. Cover slips were mounted on the slides using Vectashield (Vector Laboratories, Burlingame, CA, USA) containing 40,6-diamidino-2-phenylindole (DAPI).
2.8. Statistical Analysis
Data are expressed as the mean ± standard error (SE). Statistical analysis of three separate experiments was conducted using one-way ANOVA followed by a post hoc pairwise comparison adjusted with a Bonferroni correction. Statistical analyses were performed using SPSS software version 19.0 (SPSS Inc., Chicago, IL, USA). Differences were considered statistically significant at P < 0.05.
3. Results
3.1. ANG Inhibits mRNA Expression of Proinflammatory Cytokines and Promotes mRNA Expression of Anti-Inflammatory Cytokines in HCFs
In order to determine whether ANG can reduce the inflammatory response in HCFs, TNF-α (20 ng/mL, 8 h) was added to the culture media and then cells were cultured in the presence or absence of ANG (2 μg/mL, 30 min). Real-time PCR was conducted to investigate the effects of ANG treatment on the mRNA expression of proinflammatory (IL-1β, -6, and -8) and anti-inflammatory cytokines (IL-4 and -10). The expression of proinflammatory cytokines (IL-1β, -6, and -8) induced by TNF-α treatment was reduced significantly in cells treated with ANG (Figure 1(a)). ANG treatment alone did not exert influence on the mRNA expression of proinflammatory cytokines. Moreover, the mRNA expression of anti-inflammatory cytokines (IL-4 and -10) was increased significantly after ANG treatment (Figure 1(b)). ANG treatment alone increased the mRNA expression of IL-4 and -10.
Figure 1.
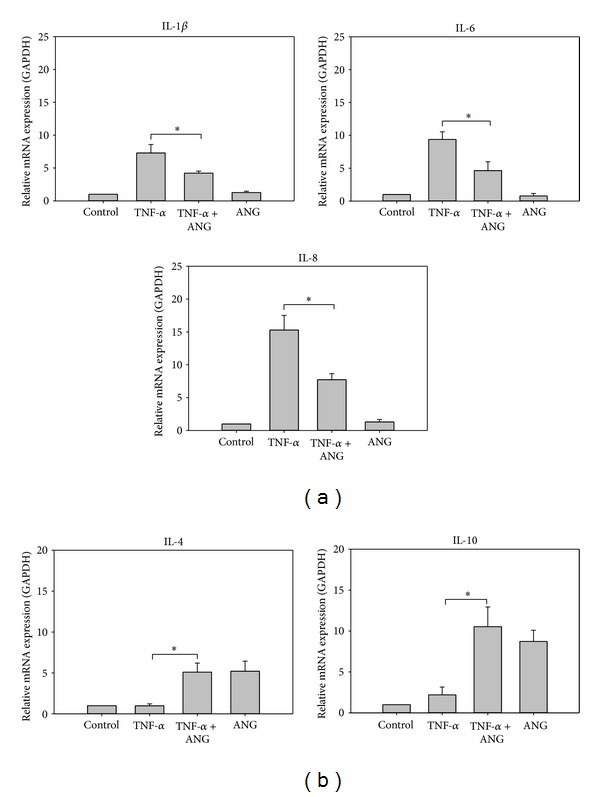
Real-time PCR analyses of proinflammatory cytokines and anti-inflammatory cytokines in HCFs. (a) The relative level of IL-1β, -6, and -8 mRNA was diminished by ANG treatment. ANG treatment alone did not affect the mRNA expression of proinflammatory cytokines. (b) The relative expression of IL-4 and -10 mRNA was increased by ANG treatment. ANG treatment alone increased the mRNA expression of IL-4 and -10. The experiments were performed in triplicate (*P < 0.05).
3.2. ANG Suppresses the Expression of Inflammatory Cytokines and Chemokines in HCFs
Immunodot blot assays were conducted to determine whether ANG decreases inflammatory cytokines and chemokines in media. Treatment with TNF-α promoted the expression of inflammatory cytokines and chemokines such as IL-6 and -8, growth-related proteins (GRO), growth-related proteins-alpha (GRO-α), and monocyte chemotactic protein- (MCP-) 1. Production of these cytokines and chemokines was downregulated in the presence of ANG, but expression of ANG was upregulated by ANG treatment (Figures 2(a) and 2(b)). We detected significant differences when comparing media before and after ANG treatment with respect to the presence of IL-6 and -8, MCP-1, and ANG (Figure 2(c)). ANG treatment alone increased only ANG expression. The expression of another cytokines was not affected by ANG treatment alone.
Figure 2.
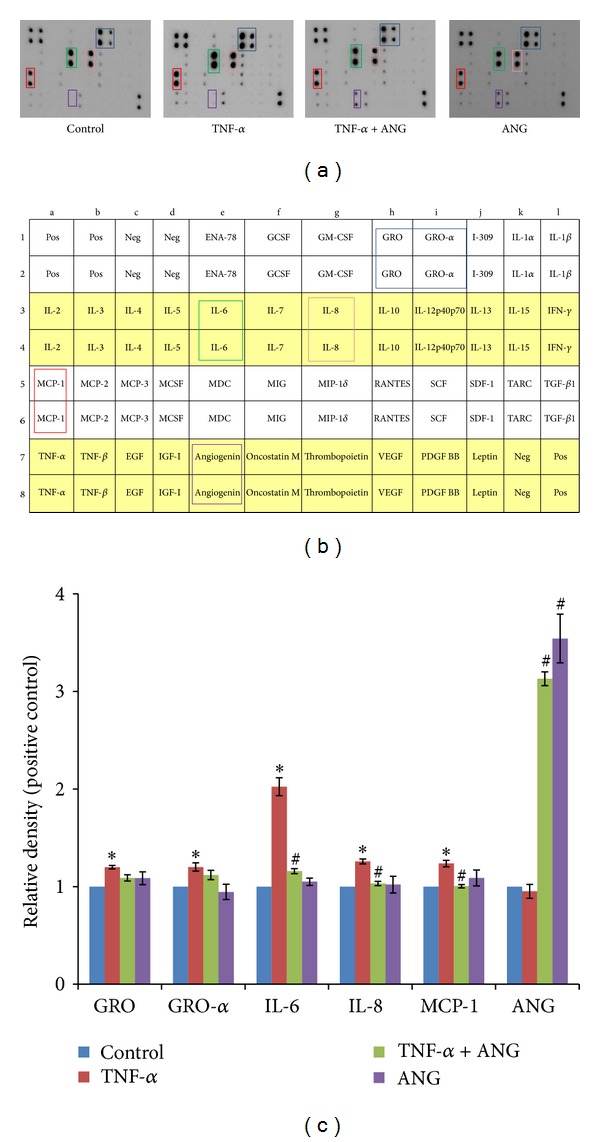
Inflammatory cytokine profiles in HCF culture medium. (a) Treatment with TNF-α (20 ng/mL) resulted in amplification of five inflammatory cytokines and chemokines (GRO and -α, IL-6 and -8, and MCP-1). Treatment with ANG (2 μg/mL) resulted in reduction of these inflammatory cytokines and chemokines. Treatment with ANG alone only induced upregulation of ANF area. (b) Custom human growth factor antibody array map. Pos: positive control; Neg: negative control. (c) Relative density of inflammatory cytokines and chemokines. The signal intensities were observed and quantified using a chemiluminescence imaging device. The values presented in the bar graph are the mean ± SE from triplicate experiments (*P < 0.05 versus control cells; # P < 0.05 versus TNF-α treated cells). GRO: growth regulated oncogene; MCP: monocyte chemotactic protein.
3.3. ANG Suppresses TNFR mRNA Expression and TBK1 Production
Real-time PCR was performed to determine whether ANG decreases TBK1 and TNFR1 and 2. TNF-α treatment increased the mRNA expression of TNFR1 and 2, but a significant downregulation was noted after ANG treatment (Figure 3). ANG treatment alone did not affect the mRNA expression of TBK1 and TNFR1 and 2. Western blot analysis showed that ANG reduces the expression of TBK1. TNF-α treatment produced a dose-dependent increase in TBK1 phosphorylation and increased TBK1 expression. After ANG treatment, TBK1 expression was decreased (Figure 4). ANG treatment alone has little effect on the expression of TBK1.
Figure 3.
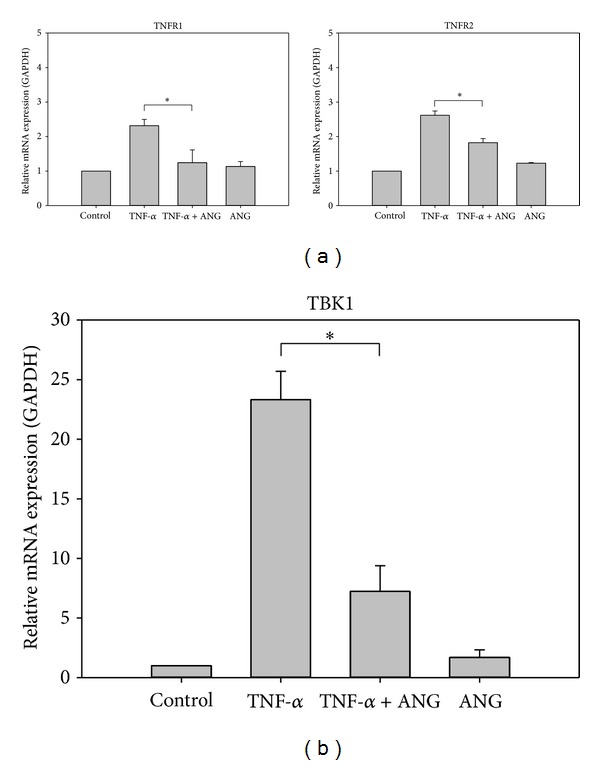
Real-time PCR analysis of TNFR1 and 2 and TBK1 in HCFs. (a) The relative level of TNFR1 and 2 mRNA was diminished by ANG treatment. (b) The relative level of TBK1 mRNA was diminished by ANG treatment. The experiments were performed in triplicate (*P < 0.05).
Figure 4.
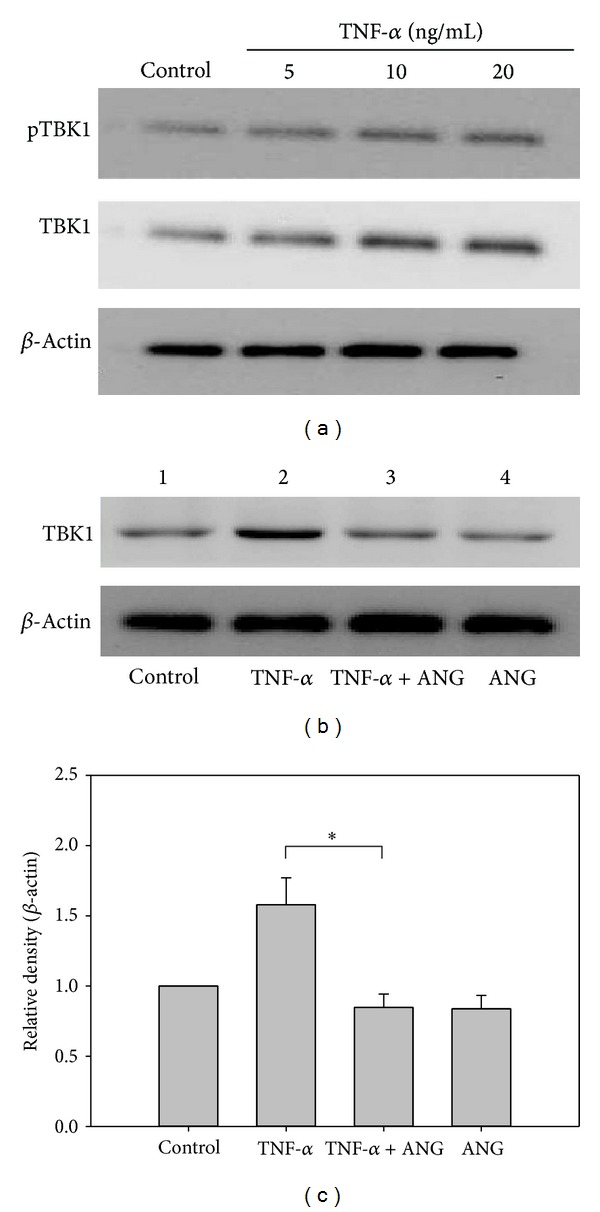
Western blot analyses of TBK1 protein expression and TBK1 phosphorylation (pTBK1). (a) TNF-α treatment increased the expression of TBK1 and TBK1 phosphorylation in HCFs. (b) After ANG treatment, TBK1 protein expression was decreased in HCFs. ANG treatment did not affect TBK1 protein expression. (c) Densitometric analysis of the relative ratio of TBK1 in cells treated with TNF-α only to those treated with TNF-α+ANG. The experiments were performed in triplicate (*P < 0.05).
3.4. ANG Inhibits Nuclear Translocation of NF-κB
HCFs were either cultured with TNF-α (20 ng/mL, 8 h) or treated with ANG (2 μg/mL, 0.5 h) to examine whether ANG inhibits nuclear translocation of NF-κB. The cells were subjected to immunofluorescent localization of NF-κB. Treatment with TNF-α induced the translocation of NF-κB from the cytosol to nucleus. However, the presence of ANG inhibited the translocation of NF-κB (Figure 5). ANG treatment alone did not influence the expression of NF-κB in nucleus.
Figure 5.
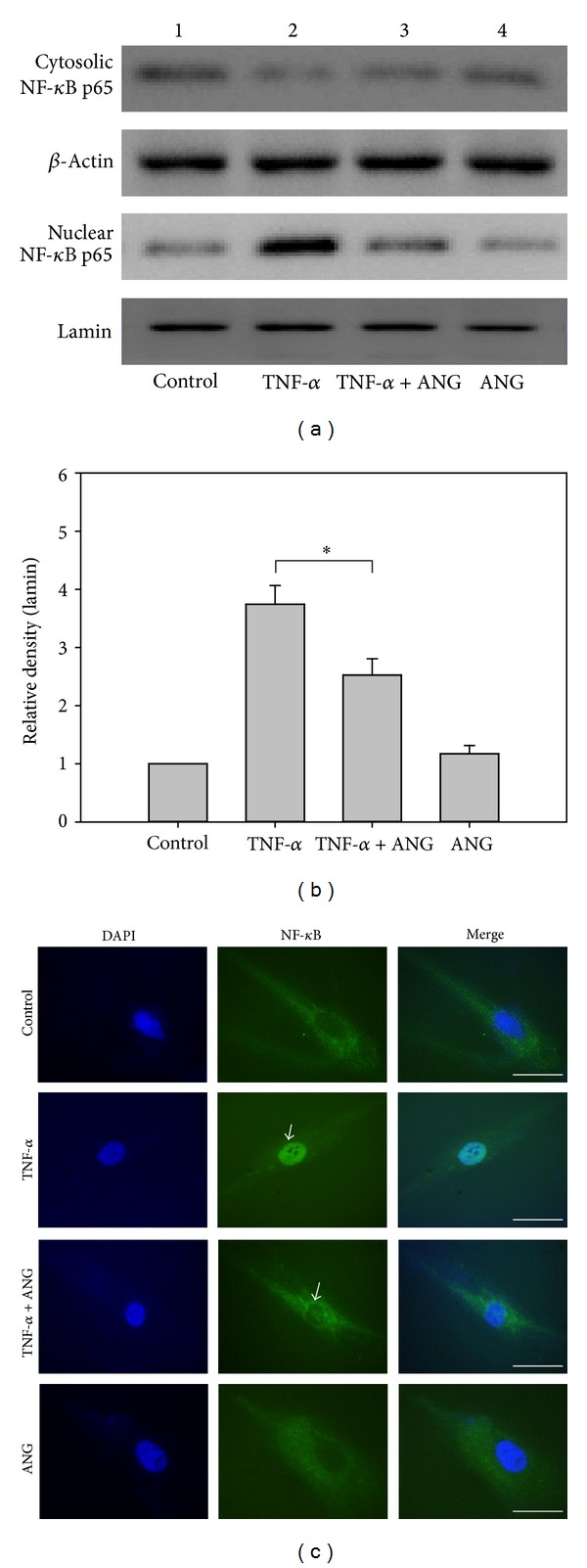
Western blot analyses of NF-κB in the nucleus and cytoplasm. (a) NF-κB nuclear translocation induced by TNF-α was diminished after ANG treatment. ANG treatment alone did not affect NF-κB nuclear translocation. (b) Densitometric analysis of the relative ratio of NF-κB in cells treated with TNF-α alone to those treated with TNF-α+ANG. The experiments were performed in triplicate (*P < 0.05). (c) Immunocytochemistry of NF-κB in the nuclei and cytoplasm of HCFs. After treatment with ANG, the cells were fixed and then labeled with an anti-NF-κB antibody. Immunofluorescent images at higher magnification demonstrate attenuation of the expression of NF-κB in nucleus after treatment with ANG ((c) arrow) in cells treated with TNF-α (scale bar, 100 μm).
4. Discussion
It is generally believed that ANG is an angiogenic molecule, inducing angiogenesis, cell migration, proliferation, and tumor growth [32–36]. Thus, previous studies have focused on its role in a possible antiangiogenin-based cure for cancer [37–39]. However, ANG is known to function as an antimicrobial peptide and is related to diverse inflammatory diseases and innate immunity [23, 28, 40–42]. Although ANG may have anti-inflammatory activity, there is currently no evidence in the literature to support this notion. In this study, we investigated the effect of ANG on inflamed HCFs and discovered a previously unidentified function of ANG in the anti-inflammatory mechanism.
The most substantial finding in our study is that ANG reduced the TBK1-mediated inflammatory response induced by TNF-α treatment. TBK1 is a member of the IκB kinase family and is involved in the inflammatory response as it is related to NF-κB activation [21]. TBK1 also has the ability to modulate the expression of IL-6 and TNF-α [43, 44]. It has been widely demonstrated that TBK1 plays a critical role in inflammatory diseases [45–47]. We have shown that ANG treatment decreased the expression of TBK1 in inflamed HCFs, suggesting that ANG may have possibility of anti-inflammatory activity.
It has been suggested that TNF-α induces the production of proinflammatory cytokines and chemokines including IL-6 and -8 and MCP-1 [48, 49]. These cytokines and chemokines are known to cause corneal inflammation [12, 50–54]. IL-6 produced by T cells and macrophages act as a proinflammatory cytokine. There is evidence that IL-6 plays a significant role in the acute inflammatory response, escalation of autoimmune reactions, and chronic inflammatory diseases [55–57]. IL-8 is a chemokine and secreted by macrophages. It has been reported that IL-8 is involved in acute inflammation and plays a central role in the initiation and maintenance of the inflammatory response in various inflammatory diseases [58–60]. MCP-1 recruits monocytes and memory T cells to the inflammation area. It has been well documented that MCP-1 mediates both acute and chronic inflammation [61]. MCP-1 has also been shown to stimulate IL-6 secretion and NF-κB activation [62]. One significant finding of this investigation is that IL-6 and -8 and MCP-1 expression was decreased after ANG treatment, indicating that ANG likely participates in attenuation of the inflammatory response through inhibition of TBK1 expression.
IL-4 and -10 are known to suppress the inflammatory response. IL-4 causes vitalization of triggered B-cell and T-cell proliferation, and IL-10 improves B-cell survival and proliferation. It has been suggested that IL-4 and -10 have anti-inflammatory activity and repress the expression of proinflammatory cytokines [63–65]. In this study, we found that ANG treatment increased mRNA expression of both IL-4 and IL-10. It can therefore be presumed that the expression of IL-6 and -8 was downregulated by ANG.
TNF-α binds TNFR 1 and 2 and triggers the activation of NF-κB [66]. Phosphorylation of TBK1 and TNFR allows NF-κB nuclear translocation [21], which results in the inflammatory response cascade including amplification of TBK1, IL-6 and -8, and MCP-1 [22, 67, 68]. NF-κB plays an important role in regulating the immune response and inflammation. It has been reported that NF-κB is involved in several inflammatory diseases [69–72]. The activation of NF-κB is inhibited by IκB proteins such as TBK1, which deactivate NF-κB by arresting it in the cytoplasm. Our results suggest that ANG may reduce NF-κB nuclear translocation through inhibition of TBK1 expression and reduction of TNFR1 and 2 mRNA expression.
A schematic illustration of the anti-inflammatory signaling pathway induced by ANG treatment of HCFs inflamed by TNF-α is shown in Figure 6. TNF-α induces an inflammatory signal by binding TNFR1 and 2. The inflammatory response is mediated by activation of TBK1, which is required for NF-κB nuclear translocation. ANG downregulates mRNA expression of IL-1β, -6, and -8. Moreover, ANG upregulates mRNA expression of IL-4 and -10. ANG also inhibits NF-κB nuclear translocation through inhibition of TBK1 production. The anti-inflammatory effect induced by ANG results in a reduction of proinflammatory cytokines and chemokines such as MCP-1 and IL-6 and -8.
Figure 6.
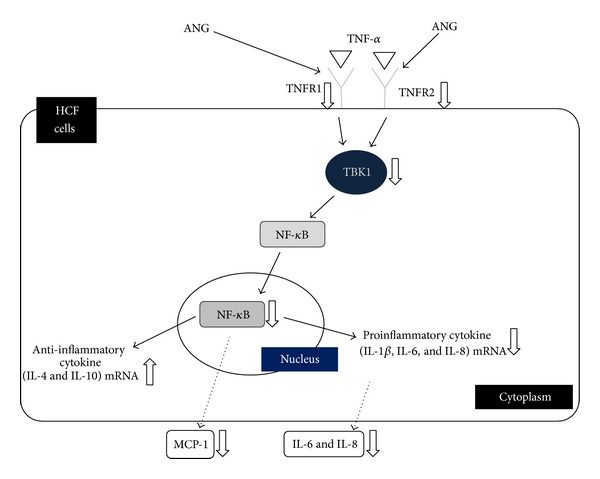
Schematic model illustrating the signaling pathway by which ANG suppresses the inflammatory response involving TBK1-mediated NF-κB nuclear translocation in HCFs inflamed by TNF-α. ANG reduces the mRNA expression of proinflammatory cytokines (IL-1β, -6, and -8) and enhances the mRNA expression of anti-inflammatory cytokines (IL-4 and -10). ANG also inhibits NF-κB nuclear translocation through a reduction in TNFR1 and 2 mRNA expression and TBK1 production. The cascade underlying the effect of ANG results in a decrease in inflammatory cytokines and chemokines such as MCP-1 and IL-6 and -8.
Since ANG is an inducer of new blood vessel growth, ANG treatment may possibly generate injection on the ocular surface. Because this study is confined to in vitro tests, we cannot exclude the possible adverse effects of ANG in vivo. However, this investigation is the first to report the function of ANG in the inflammatory response. In vivo animal studies are needed to further validate our hypothesis prior to the clinical application of ANG in ocular inflammation.
There is little information in the literature regarding the anti-inflammatory effects of ANG. The ANG functions newly identified in this study indicate that it likely plays a critical role in attenuating inflammation. The findings pointing to the anti-inflammatory effects of ANG in inflamed HCFs shed new light on the treatment of ocular inflammation. We have demonstrated that ANG suppresses the TNF-α-induced inflammatory response and NF-κB nuclear translocation through inhibition of TBK1 expression in HCFs. This preliminary evidence suggests that ANG could be a strong candidate for the treatment of corneal inflammation.
Supplementary Material
The data about identity of the purified ANG was obtained from the Department of Biochemistry at Chungbuk National University. They had already confirmed the purity and effect of angiogenin by western blotting with ANG specific antibodies in previous article (Srisa-Art M et al. Analysis of protein-protein interactions by using droplet-based microfluidics. Chembiochem : a European journal of chemical biology. 2009). The biological activity of the purified ANG was confirmed by its nuclear translocation in human umbilical vein endothelial (HUVE) cells. The enzymatic activities of the purified ANG toward poly (C) were measured and compared it with RNase A as shown in Supplementary Figure 1. ANG catalyzes the cleavage of poly (C), and the ribonucleolytic activities of ANG were determined by measuring the rate of formation of perchloric acid-soluble products in poly (C) precipitation assay. The endotoxin concentrations in ANG preparation were determined using the Limulus amebocyte lysate (LAL) assay. A standard curve was used to determine the endotoxin quantities presented in the purified angiogenin as shown in Supplementary Figure 2. Endotoxin level was determined to be 0.011 ng per μg of the purified angiogenin (0.11 EU/ μg). The results suggest that the purified ANG has both the biological and enzymatic activities and contains verified low level of endotoxin for its use both in vitro and in vivo experiments.
Acknowledgments
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2011-0016922), and by a Grant from the Korea Healthcare Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (A121487).
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
References
- 1.Mintz R, Feller E, Bahr RL, Shah SA. Ocular manifestations of inflammatory bowel disease. Inflammatory Bowel Diseases. 2004;10(2):135–139. doi: 10.1097/00054725-200403000-00012. [DOI] [PubMed] [Google Scholar]
- 2.Rothova A, Suttorp-van Schulten MSA, Frits Treffers W, Kijlstra A. Causes and frequency of blindness in patients with intraocular inflammatory disease. British Journal of Ophthalmology. 1996;80(4):332–336. doi: 10.1136/bjo.80.4.332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zoukhri D. Effect of inflammation on lacrimal gland function. Experimental Eye Research. 2006;82(5):885–898. doi: 10.1016/j.exer.2005.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wilson SE, Mohan RR, Mohan RR, Ambrósio R, Jr., Hong J, Lee J. The corneal wound healing response: cytokine-mediated interaction of the epithelium, stroma, and inflammatory cells. Progress in Retinal and Eye Research. 2001;20(5):625–637. doi: 10.1016/s1350-9462(01)00008-8. [DOI] [PubMed] [Google Scholar]
- 5.Na KS, Mok JW, Kim JY, Rho CR, Joo CK. Correlations between tear cytokines, chemokines, and soluble receptors and clinical severity of dry eye disease. Investigative Ophthalmology & Visual Science. 2012;53(9):5443–5450. doi: 10.1167/iovs.11-9417. [DOI] [PubMed] [Google Scholar]
- 6.Valentincic NV, de Groot-Mijnes JDF, Kraut A, Korosec P, Hawlina M, Rothova A. Intraocular and serum cytokine profiles in patients with intermediate uveitis. Molecular Vision. 2011;17:2003–2010. [PMC free article] [PubMed] [Google Scholar]
- 7.VanDerMeid KR, Su SP, Ward KW, Zhang J-Z. Correlation of tear inflammatory cytokines and matrix metalloproteinases with four dry eye diagnostic tests. Investigative Ophthalmology & Visual Science. 2012;53(3):1512–1518. doi: 10.1167/iovs.11-7627. [DOI] [PubMed] [Google Scholar]
- 8.King WJ, Comer RM, Hudde T, Larkin DFP, George AJT. Cytokine and chemokine expression kinetics after corneal transplantation. Transplantation. 2000;70(8):1225–1233. doi: 10.1097/00007890-200010270-00017. [DOI] [PubMed] [Google Scholar]
- 9.Mahajan VB, Wei C, McDonnell PJ., III Microarray analysis of corneal fibroblast gene expression after interleukin-1 treatment. Investigative Ophthalmology and Visual Science. 2002;43(7):2143–2151. [PubMed] [Google Scholar]
- 10.Xi X, McMillan DH, Lehmann GM, et al. Ocular fibroblast diversity: implications for inflammation and ocular wound healing. Investigative Ophthalmology & Visual Science. 2011;52(7):4859–4865. doi: 10.1167/iovs.10-7066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Streilein JW, Dana MR, Ksander BR. Immunity causing blindness: five different paths to herpes stromal keratitis. Immunology Today. 1997;18(9):443–449. doi: 10.1016/s0167-5699(97)01114-6. [DOI] [PubMed] [Google Scholar]
- 12.Liu Y, Kimura K, Yanai R, Chikama T-I, Nishida T. Cytokine, chemokine, and adhesion molecule expression mediated by MAPKs in human corneal fibroblasts exposed to poly(I:C) Investigative Ophthalmology and Visual Science. 2008;49(8):3336–3344. doi: 10.1167/iovs.07-0972. [DOI] [PubMed] [Google Scholar]
- 13.Fini ME. Keratocyte and fibroblast phenotypes in the repairing cornea. Progress in Retinal and Eye Research. 1999;18(4):529–551. doi: 10.1016/s1350-9462(98)00033-0. [DOI] [PubMed] [Google Scholar]
- 14.Daniels JT, Geerling G, Alexander RA, Murphy G, Khaw PT, Saarialho-Kere U. Temporal and spatial expression of matrix metalloproteinases during wound healing of human corneal tissue. Experimental Eye Research. 2003;77(6):653–664. doi: 10.1016/j.exer.2003.08.010. [DOI] [PubMed] [Google Scholar]
- 15.Smith RS, Smith TJ, Blieden TM, Phipps RP. Fibroblasts as sentinel cells. Synthesis of chemokines and regulation of inflammation. The American Journal of Pathology. 1997;151(2):317–322. [PMC free article] [PubMed] [Google Scholar]
- 16.Maertzdorf J, Osterhaus ADME, Verjans GMGM. IL-17 expression in human herpetic stromal keratitis: modulatory effects on chemokine production by corneal fibroblasts. Journal of Immunology. 2002;169(10):5897–5903. doi: 10.4049/jimmunol.169.10.5897. [DOI] [PubMed] [Google Scholar]
- 17.Pfister RR, Hadclox JL, Sommers CI. Injection of chemoattractants into normal cornea: a model of inflammation after alkali injury. Investigative Ophthalmology and Visual Science. 1998;39(9):1744–1750. [PubMed] [Google Scholar]
- 18.Hayden MS, Ghosh S. Shared principles in NF-κB signaling. Cell. 2008;132(3):344–362. doi: 10.1016/j.cell.2008.01.020. [DOI] [PubMed] [Google Scholar]
- 19.Miyamoto S, Maki M, Schmitt MJ, Hatanaka M, Verma IM. Tumor necrosis factor α-induced phosphorylation of IκBα is a signal for its degradation but not dissociation from NF-κB. Proceedings of the National Academy of Sciences of the United States of America. 1994;91(26):12740–12744. doi: 10.1073/pnas.91.26.12740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kravchenko VV, Mathison JC, Schwamborn K, Mercurio F, Ulevitch RJ. IKKi/IKKε plays a key role in integrating signals induced by pro-inflammatory stimuli. Journal of Biological Chemistry. 2003;278(29):26612–26619. doi: 10.1074/jbc.M303001200. [DOI] [PubMed] [Google Scholar]
- 21.Pomerantz JL, Baltimore D. NF-κB activation by a signaling complex containing TRAF2, TANK and TBK1, a novel IKK-related kinase. EMBO Journal. 1999;18(23):6694–6704. doi: 10.1093/emboj/18.23.6694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reilly SM, Chiang SH, Decker SJ, et al. An inhibitor of the protein kinases TBK1 and IKK-varepsilon improves obesity-related metabolic dysfunctions in mice. Nature Medicine. 2013;19(3):313–321. doi: 10.1038/nm.3082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bräuer L, Paulsen FP. Tear film and ocular surface surfactants. Journal of Epithelial Biology and Pharmacology. 2008;1:62–67. [Google Scholar]
- 24.Sack RA, Conradi L, Krumholz D, Beaton A, Sathe S, Morris C. Membrane array characterization of 80 chemokines, cytokines, and growth factors in open- and closed-eye tears: angiogenin and other defense system constituents. Investigative Ophthalmology and Visual Science. 2005;46(4):1228–1238. doi: 10.1167/iovs.04-0760. [DOI] [PubMed] [Google Scholar]
- 25.Xu Z-P, Monti DM, Hu G-F. Angiogenin activates human umbilical artery smooth muscle cells. Biochemical and Biophysical Research Communications. 2001;285(4):909–914. doi: 10.1006/bbrc.2001.5255. [DOI] [PubMed] [Google Scholar]
- 26.Hartmann A, Kunz M, Köstlin S, et al. Hypoxia-induced up-regulation of angiogenin in human malignant melanoma. Cancer Research. 1999;59(7):1578–1583. [PubMed] [Google Scholar]
- 27.Etoh T, Shibuta K, Barnard GF, Kitano S, Mori M. Angiogenin expression in human colorectal cancer: the role of focal macrophage infiltration. Clinical Cancer Research. 2000;6(9):3545–3551. [PubMed] [Google Scholar]
- 28.Oikonomou KA, Kapsoritakis AN, Kapsoritaki AI, et al. Angiogenin, angiopoietin-1, angiopoietin-2, and endostatin serum levels in inflammatory bowel disease. Inflammatory Bowel Diseases. 2011;17(4):963–970. doi: 10.1002/ibd.21410. [DOI] [PubMed] [Google Scholar]
- 29.Osorio DS, Antunes A, Ramos MJ. Structural and functional implications of positive selection at the primate angiogenin gene. BMC Evolutionary Biology. 2007;7, article 167 doi: 10.1186/1471-2148-7-167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hooper LV, Stappenbeck TS, Hong CV, Gordon JI. Angiogenins: a new class of microbicidal proteins involved in innate immunity. Nature Immunology. 2003;4(3):269–273. doi: 10.1038/ni888. [DOI] [PubMed] [Google Scholar]
- 31.Srisa-Art M, Kang D-K, Hong J, et al. Analysis of protein-protein interactions by using droplet-based microfluidics. ChemBioChem. 2009;10(10):1605–1611. doi: 10.1002/cbic.200800841. [DOI] [PubMed] [Google Scholar]
- 32.Wiȩdłocha A. Following angiogenin during angiogenesis: a journey from the cell surface to the nucleolus. Archivum Immunologiae et Therapiae Experimentalis. 1999;47(5):299–305. [PubMed] [Google Scholar]
- 33.Kishimoto K, Liu S, Tsuji T, Olson KA, Hu G-F. Endogenous angiogenin in endothelial cells is a general requirement for cell proliferation and angiogenesis. Oncogene. 2005;24(3):445–456. doi: 10.1038/sj.onc.1208223. [DOI] [PubMed] [Google Scholar]
- 34.Wei S, Gao X, Du J, Su J, Xu Z. Angiogenin enhances cell migration by regulating stress fiber assembly and focal adhesion dynamics. PLoS ONE. 2011;6(12) doi: 10.1371/journal.pone.0028797.e28797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fett JW, Strydom DJ, Lobb RR, et al. Isolation and characterization of angiogenin, an angiogenic protein from human carcinoma cells. Biochemistry. 1985;24(20):5480–5486. doi: 10.1021/bi00341a030. [DOI] [PubMed] [Google Scholar]
- 36.Riordan JF, Vallee BL. Human angiogenin, an organogenic protein. British Journal of Cancer. 1988;57(6):587–590. doi: 10.1038/bjc.1988.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yoshioka N, Wang L, Kishimoto K, Tsuji T, Hu G-F. A therapeutic target for prostate cancer based on angiogenin-stimulated angiogenesis and cancer cell proliferation. Proceedings of the National Academy of Sciences of the United States of America. 2006;103(39):14519–14524. doi: 10.1073/pnas.0606708103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nilsson UW, Abrahamsson A, Dabrosin C. Angiogenin regulation by estradiol in breast tissue: tamoxifen inhibits angiogenin nuclear translocation and antiangiogenin therapy reduces breast cancer growth in vivo. Clinical Cancer Research. 2010;16(14):3659–3669. doi: 10.1158/1078-0432.CCR-10-0501. [DOI] [PubMed] [Google Scholar]
- 39.Li S, Ibaragi S, Hu G-F. Angiogenin as a molecular target for the treatment of prostate cancer. Current Cancer Therapy Reviews. 2011;7(2):83–90. doi: 10.2174/1573394711107020083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lioté F, Champy R, Moenner M, Boval-Boizard B, Badet J. Elevated angiogenin levels in synovial fluid from patients with inflammatory arthritis and secretion of angiogenin by cultured synovial fibroblasts. Clinical and Experimental Immunology. 2003;132(1):163–168. doi: 10.1046/j.1365-2249.2003.02117.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Koutroubakis IE, Xidakis C, Karmiris K, Sfiridaki A, Kandidaki E, Kouroumalis EA. Serum angiogenin in inflammatory bowel disease. Digestive Diseases and Sciences. 2004;49(11-12):1758–1762. doi: 10.1007/s10620-004-9565-4. [DOI] [PubMed] [Google Scholar]
- 42.Hooper LV, Stappenbeck TS, Hong CV, Gordon JI. Angiogenins: a new class of microbicidal proteins involved in innate immunity. Nature Immunology. 2003;4(3):269–273. doi: 10.1038/ni888. [DOI] [PubMed] [Google Scholar]
- 43.Xie X-H, Zang N, Li S-M, et al. Resveratrol inhibits respiratory syncytial virus-induced IL-6 production, decreases viral replication, and downregulates TRIF expression in airway epithelial cells. Inflammation. 2012;35(4):1392–1401. doi: 10.1007/s10753-012-9452-7. [DOI] [PubMed] [Google Scholar]
- 44.Marchlik E, Thakker P, Carlson T, et al. Mice lacking Tbk1 activity exhibit immune cell infiltrates in multiple tissues and increased susceptibility to LPS-induced lethality. Journal of Leukocyte Biology. 2010;88(6):1171–1180. doi: 10.1189/jlb.0210071. [DOI] [PubMed] [Google Scholar]
- 45.Yu T, Yi YS, Yang Y, Oh J, Jeong D, Cho JY. The pivotal role of TBK1 in inflammatory responses mediated by macrophages. Mediators of Inflammation. 2012;2012:8 pages. doi: 10.1155/2012/979105.979105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hammaker D, Boyle DL, Firestein GS. Synoviocyte innate immune responses: tank-binding kinase-1 as a potential therapeutic target in rheumatoid arthritis. Rheumatology. 2012;51(4):610–618. doi: 10.1093/rheumatology/ker154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Migita K, Nakamura T. TBK1: a potential therapeutic target in RA. Rheumatology. 2012;51(4):588–589. doi: 10.1093/rheumatology/ker207. [DOI] [PubMed] [Google Scholar]
- 48.Zhu J, Yong W, Wu X, et al. Anti-inflammatory effect of resveratrol on TNF-α-induced MCP-1 expression in adipocytes. Biochemical and Biophysical Research Communications. 2008;369(2):471–477. doi: 10.1016/j.bbrc.2008.02.034. [DOI] [PubMed] [Google Scholar]
- 49.Deshmane SL, Kremlev S, Amini S, Sawaya BE. Monocyte chemoattractant protein-1 (MCP-1): an overview. Journal of Interferon and Cytokine Research. 2009;29(6):313–325. doi: 10.1089/jir.2008.0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ooi KG-J, Galatowicz G, Calder VL, Lightman SL. Cytokines and chemokines in uveitis—is there a correlation with clinical phenotype? Clinical Medicine and Research. 2006;4(4):294–309. doi: 10.3121/cmr.4.4.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ghasemi H, Ghazanfari T, Yaraee R, Faghihzadeh S, Hassan ZM. Roles of IL-8 in ocular inflammations: a review. Ocular Immunology and Inflammation. 2011;19(6):401–412. doi: 10.3109/09273948.2011.618902. [DOI] [PubMed] [Google Scholar]
- 52.Lema I, Sobrino T, Durán JA, Brea D, Díez-Feijoo E. Subclinical keratoconus and inflammatory molecules from tears. British Journal of Ophthalmology. 2009;93(6):820–824. doi: 10.1136/bjo.2008.144253. [DOI] [PubMed] [Google Scholar]
- 53.Tuaillon N, Shen DF, Berger RB, Lu B, Rollins BJ, Chan C-C. MCP-1 expression in endotoxin-induced uveitis. Investigative Ophthalmology and Visual Science. 2002;43(5):1493–1498. [PubMed] [Google Scholar]
- 54.Rojas M, Zhang W, Lee DL, et al. Role of IL-6 in angiotensin II-induced retinal vascular inflammation. Investigative Ophthalmology and Visual Science. 2010;51(3):1709–1718. doi: 10.1167/iovs.09-3375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gabay C. Interleukin-6 and chronic inflammation. Arthritis Research and Therapy. 2006;8(supplement 2, article S3):1–6. doi: 10.1186/ar1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Akira S, Taga T, Kishimoto T. Interleukin-6 in biology and medicine. Advances in Immunology. 1993;54:1–78. doi: 10.1016/s0065-2776(08)60532-5. [DOI] [PubMed] [Google Scholar]
- 57.Kishimoto T. Interleukin-6: from basic science to medicine—40 years in immunology. Annual Review of Immunology. 2005;23:1–21. doi: 10.1146/annurev.immunol.23.021704.115806. [DOI] [PubMed] [Google Scholar]
- 58.Harada A, Sekido N, Akahoshi T, Wada T, Mukaida N, Matsushima K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. Journal of Leukocyte Biology. 1994;56(5):559–564. [PubMed] [Google Scholar]
- 59.Izzo RS, Witkon K, Chen AI, Hadjiyane C, Weinstein MI, Pellecchia C. Neutrophil-activating peptide (interleukin-8) in colonic mucosa from patients with Crohn’s disease. Scandinavian Journal of Gastroenterology. 1993;28(4):296–300. doi: 10.3109/00365529309090244. [DOI] [PubMed] [Google Scholar]
- 60.Gibson P, Rosella O. Interleukin 8 secretion by colonic crypt cells in vitro: response to injury suppressed by butyrate and enhanced in inflammatory bowel disease. Gut. 1995;37(4):536–543. doi: 10.1136/gut.37.4.536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Conti P, DiGioacchino M. MCP-1 and RANTES are mediators of acute and chronic inflammation. Allergy and Asthma Proceedings. 2001;22(3):133–137. doi: 10.2500/108854101778148737. [DOI] [PubMed] [Google Scholar]
- 62.Viedt C, Dechend R, Fei J, Hänsch GM, Kreuzer J, Orth SR. MCP-1 induces inflammatory activation of human tubular epithelial cells: involvement of the transcription factors, nuclear factor-κB and activating protein-1. Journal of the American Society of Nephrology. 2002;13(6):1534–1547. doi: 10.1097/01.asn.0000015609.31253.7f. [DOI] [PubMed] [Google Scholar]
- 63.Marie C, Pitton C, Fitting C, Cavaillon J-M. Regulation by anti-inflammatory cytokines (IL-4, IL-10, IL-13, TGFβ) of interleukin-8 production by LPS- and/or TNFα-activated human polymorphonuclear cells. Mediators of Inflammation. 1996;5(5):334–340. doi: 10.1155/S0962935196000488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lee SW, Hong YS, Chun CM, et al. Anti-inflammatory effects of IL-4 and IL-10 on human polymorphonuclear leukocytes. Journal of Korean medical science. 2002;17(1):7–14. doi: 10.3346/jkms.2002.17.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.de Waal Malefyt R, Haanen J, Spits H, et al. Interleukin 10 (IL-10) and viral IL-10 strongly reduce antigen-specific human T cell proliferation by diminishing the antigen-presenting capacity of monocytes via downregulation of class II major histocompatibility complex expression. Journal of Experimental Medicine. 1991;174(4):915–924. doi: 10.1084/jem.174.4.915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mak TW, Yeh WC. Signaling for survival and apoptosis in the immune system. Arthritis Research. 2002;4(supplement 3):S243–S252. doi: 10.1186/ar569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Blackwell TS, Christman JW. The role of nuclear factor-κB in cytokine gene regulation. American Journal of Respiratory Cell and Molecular Biology. 1997;17(1):3–9. doi: 10.1165/ajrcmb.17.1.f132. [DOI] [PubMed] [Google Scholar]
- 68.Muñoz MC, Giani JF, Mayer MA, Toblli JE, Turyn D, Dominici FP. TANK-binding kinase 1 mediates phosphorylation of insulin receptor at serine residue 994: a potential link between inflammation and insulin resistance. Journal of Endocrinology. 2009;201(2):185–197. doi: 10.1677/JOE-08-0276. [DOI] [PubMed] [Google Scholar]
- 69.Hart LA, Krishnan VL, Adcock IM, Barnes PJ, Chung KF. Activation and localization of transcription factor, nuclear factor-κB, in asthma. American Journal of Respiratory and Critical Care Medicine. 1998;158(5):1585–1592. doi: 10.1164/ajrccm.158.5.9706116. [DOI] [PubMed] [Google Scholar]
- 70.Tak PP, Firestein GS. NF-κB: a key role in inflammatory diseases. Journal of Clinical Investigation. 2001;107(1):7–11. doi: 10.1172/JCI11830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Collins T, Cybulsky MI. NF-κB: pivotal mediator or innocent bystander in atherogenesis? Journal of Clinical Investigation. 2001;107(3):255–264. doi: 10.1172/JCI10373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Asahara H, Asanuma M, Ogawa N, Nishibayashi S, Inoue H. High DNA-binding activity of transcription factor NF-κB in synovial membranes of patients with rheumatoid arthritis. Biochemistry and Molecular Biology International. 1995;37(5):827–832. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The data about identity of the purified ANG was obtained from the Department of Biochemistry at Chungbuk National University. They had already confirmed the purity and effect of angiogenin by western blotting with ANG specific antibodies in previous article (Srisa-Art M et al. Analysis of protein-protein interactions by using droplet-based microfluidics. Chembiochem : a European journal of chemical biology. 2009). The biological activity of the purified ANG was confirmed by its nuclear translocation in human umbilical vein endothelial (HUVE) cells. The enzymatic activities of the purified ANG toward poly (C) were measured and compared it with RNase A as shown in Supplementary Figure 1. ANG catalyzes the cleavage of poly (C), and the ribonucleolytic activities of ANG were determined by measuring the rate of formation of perchloric acid-soluble products in poly (C) precipitation assay. The endotoxin concentrations in ANG preparation were determined using the Limulus amebocyte lysate (LAL) assay. A standard curve was used to determine the endotoxin quantities presented in the purified angiogenin as shown in Supplementary Figure 2. Endotoxin level was determined to be 0.011 ng per μg of the purified angiogenin (0.11 EU/ μg). The results suggest that the purified ANG has both the biological and enzymatic activities and contains verified low level of endotoxin for its use both in vitro and in vivo experiments.


