Abstract

Gilenya (fingolimod, FTY720) was recently approved by the U.S. FDA for the treatment of patients with remitting relapsing multiple sclerosis (RRMS). It is a potent agonist of four of the five sphingosine 1-phosphate (S1P) G-protein-coupled receptors (S1P1 and S1P3−5). It has been postulated that fingolimod's efficacy is due to S1P1 agonism, while its cardiovascular side effects (transient bradycardia and hypertension) are due to S1P3 agonism. We have discovered a series of selective S1P1 agonists, which includes 3-[6-(5-{3-cyano-4-[(1-methylethyl)oxy]phenyl}-1,2,4-oxadiazol-3-yl)-5-methyl-3,4-dihydro-2(1H)-isoquinolinyl]propanoate, 20, a potent, S1P3-sparing, orally active S1P1 agonist. Compound 20 is as efficacious as fingolimod in a collagen-induced arthritis model and shows excellent pharmacokinetic properties preclinically. Importantly, the selectivity of 20 against S1P3 is responsible for an absence of cardiovascular signal in telemetered rats, even at high dose levels.
Keywords: Fingolimod, S1P, agonism, bradycardia, lymphopenia, multiple sclerosis
Fingolimod (1)1 was developed as an analogue of the natural product myriocin 2, a potent inhibitor of palmitoyltransferase that demonstrates immunosuppressant activity (Figure 1). Compound 1 has displayed clinical efficacy in transplantation2 and remitting relapsing multiple sclerosis3 trials and is now marketed in the United States for the latter indication. Administration of 1 leads to the sequestration of lymphocytes in secondary lymphoid organs and consequently to a reduction of lymphocyte counts in the peripheral blood. The unique pharmacological profile of 1 is not due to any related myriocin-like activity. Indeed, 1 is enantioselectively phosphorylated in vivo4,5 to give phosphate 3, a potent agonist of four of the five G-protein-coupled receptors (S1P1 and S1P3−5) for sphingosine 1-phosphate (S1P) 4. S1P has several physiological roles, but only agonism of S1P1 is required to induce egress of T cells from lymphoid organs.6,7 On the other hand, it has been demonstrated using selective tool compounds8,9 or transgenic mice10 that the unwanted cardiovascular effects seen with 1 are related to its agonism of the S1P3 receptor, although selectivity against S1P3 may not preclude bradycardia.11
Figure 1.
Structures of fingolimod 1 and its phosphate 3, myriocin 2, and S1P 4.
Understanding of the unique mode of action of 1 has triggered intensive effort toward the discovery of S1P1 agonists with an increased degree of selectivity versus S1P3,12,13 either as pro-drugs such as KRP-203145 or as direct agonists such as ACT-128800156 (Figure 2). Zwitterions such as 7(16) or propionic acids such as 8(17) are likely to interact with S1P1 in a similar fashion to S1P itself.18,18b Agonist 8 was of particular interest to us because biaryl oxadiazoles 9(19) and 10(20) (Figure 3) are in clinical trials (not as S1P1 agonists), suggesting good developability properties. Our strategy to develop novel orally active S1P1 selective agonists focused on drugability:21 Several reports22,22b have highlighted that the attrition rate of oral compounds in clinical trials can be correlated with compound properties such as (high) molecular weight (MW),23 (high) lipophilicity (cLogP, LogD), (low) polar surface area (PSA), or the number of rotable bounds.24 Moreover, the odds of toxicity are minimal when compounds have cLogP < 3 and PSA > 75 Å2.25 We focused on these two parameters, in particular on lipophilicity, as the pharmacophore of S1P precludes the discovery of compounds with a MW < 350. Inspired by the structures of S1P agonists 7 and 8, we pursued the design and synthesis of constrained triaryl scaffolds incorporating a zwitterionic moiety. The two charged residues should increase the PSA and hydrophilicity (Scheme 1).26 The substitution pattern on the distal ring was influenced by the reported structure−activity relationship (SAR),17 as it was likely to be optimal for in vitro potency.
Figure 2.
Structures of known S1P1 agonists.
Figure 3.
Structure of biaryl oxadiazoles.
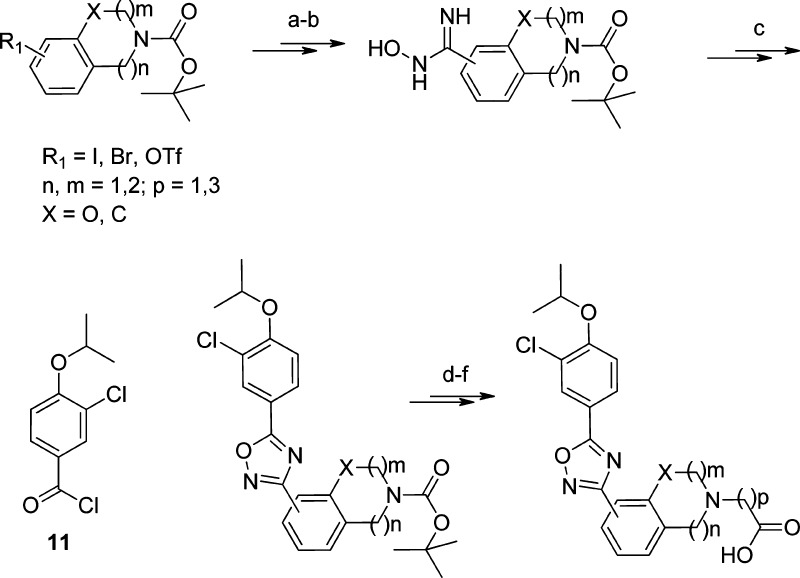
Scheme 1. Lead Generation Strategy Toward Druglike S1P1 Agonist Leads.
Reagents and conditions: (a) Palladium-mediated nitrile formation. (b) Addition of hydroxylamine. (c) Compound 11, base, heat. (d) HCl. (e) Alkylation. (f) Saponification.
Among several bicyclic rings/bqn containing a basic nitrogen that we explored, the C-5 substituted tetrahydroisoquinoline (THIQ) appeared most promising and was used for further optimization. A representative synthesis of these agonists is depicted in Scheme 2: The α,β-unsaturated ketone 13 was accessed via reaction of an enamine derived from ketone 12. Its palladium-mediated oxidation led to the corresponding phenol, which was transformed into 14 via triflation followed by cyanation. The addition of hydroxylamine to 14 and reaction of the corresponding hydroxy-amidine with 15 followed by dehydration of the noncyclized intermediate gave oxadiazole 16. Deprotection in acidic media of the secondary amine and Michael addition to ethyl acrylate followed by saponification led to agonist 20.
Scheme 2. Synthesis of 20.
Reagents and conditions: (a) Pyrrolidine, toluene, Dean−Stark, reflux. (b) Pent-1-en-3-one, hydroquinone, 59% (2 steps). (c) Lithium bis(trimethylsilyl)amide (LiHMDS), THF, −63 °C and then trimethylsilyl chloride (TMSCl). (d) Pd(OAc)2, CH3CN, T < 35 °C, and then tetra-n-butylammonium fluoride (TBAF), 55% (2 steps). (e) Tf2O, pyridine, CH2Cl2, −30 °C. (f) Zn(CN)2, Pd(PPh3)4, DMF, 100 °C, 92% (2 steps). (g) Aqueous NH2OH, EtOH, 80 °C, 86%. (h) Compound 15, pyridine, toluene, 0−110 °C, 51% (2 steps). (i) HCl, dioxane, room temperature, 98%. (j) Ethyl acrylate, diaza(1,3)bicycle[5.4.0]undecane (DBU), CH3CN, room temperature, 94%. (k) NaOH, EtOH/water, room temperature, 91%.
The key SAR findings are summarized in Table 1 and show (1) the need for a meta-substituent on the distal aromatic ring (cf. 18 vs 19 and 20); lipophilic functionalities (19) and substituents with some polarity (20) are tolerated in this position, but the nitrile group proved optimal in terms of potency, selectivity, and lipophilicity (cf. 20 vs 19). (2) The isopropoxy para-substituent on this ring was optimal for potency (cf. 21 vs 20)27 and S1P3 selectivity (cf. 22, 23 vs 20). (3) Replacement of the phenyl ring with electron-poor aromatic leads to a significant loss of potency (24 and 19 as representative examples).28 (4) Introduction of a C-4 substituent on the THIQ aromatic ring is beneficial for S1P3 selectivity without compromising S1P1 activity (cf. 25 vs 20). (5) Introduction of an acid group as a phosphate mimetic29 on the N-substituent is beneficial for activity and selectivity (cf. 26 vs 20, 27, and 28), but the length of the chain (1−3 carbons) has no impact on these parameters (cf. 20 vs 27 and 28).
Table 1. SAR Data in the Triaryl THIQ S1P1 Agonist Series.

| pEC50 (n) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|
| compd | R1 | R2 | R3 | X | n | S1P1 GTPγS | S1P1 β-arrestin | S1P3 GTPγS | CHROM LogDa at pH 7.4 |
| 3 | 8.4 ± 0.31 (130) | 7.7 ± 0.17 (44) | 8.3 ± 0.31 (38) | ||||||
| 18 | H | −CH(CH3)2 | CH3 | CH | 2 | 6.5 ± 0.25 (7) | 6.5 ± 0.30 (10) | 4.9 (1) | 3.81 |
| 19 | Cl | −CH(CH3)2 | CH3 | CH | 2 | 7.9 ± 0.24 (13) | 8.5 ± 0.12 (11) | 5.2 ± 0.27 (5) | 4.35 |
| 20 | CN | − CH(CH3)2 | CH3 | CH | 2 | 7.5 ± 0.24 (8) | 8.3 ± 0.11 (11) | <4.4 (8) | 3.41 |
| 21 | CN | −n-C2H5 | CH3 | CH | 2 | 7.3 ± 0.31 (7) | 7.5 ± 0.14 (10) | <4 (9) | 3.01 |
| 22 | CN | −n-C3H7 | CH3 | CH | 2 | 7.5 ± 0.22 (7) | 7.8 ± 0.15 (10) | 5.1 ± 0.21 (4) | 3.54 |
| 23 | CN | −n-C4H9 | CH3 | CH | 2 | 7.2 ± 0.30 (7) | 7.7 ± 0.15 (9) | 5 ± 0.34 (8) | 4.06 |
| 24 | Cl | −CH(CH3)2 | CH3 | N | 2 | 7.1 ± 0.17 (5) | 7.6 ± 0.49 (5) | 4.7 ± 0.04 (2) | 4.46 |
| 25 | CN | −CH(CH3)2 | H | CH | 2 | 8.2 ± 0.23 (11) | 8.5 ± 0.11 (8) | 5.4 ± 0.14 (11) | 3.30 |
| 26 | CN | −CH(CH3)2 | CH3 | CH | 0 (NH) | 7.6 ± 0.3 (10) | 7.2 ± 0.89 (27) | 4.9 ± 0.43 (4) | 4.21 |
| 27 | CN | −CH(CH3)2 | CH3 | CH | 1 | 7.8 ± 0.3 (9) | 8.3 ± 0.1 (8) | <4 (7) | 3.32 |
| 28 | CN | −CH(CH3)2 | CH3 | CH | 3 | 7.7 ± 0.27 (9) | 8.2 ± 0.09 (8) | <4 (11) | 3.51 |
CHROM LogD = chromatographic hydrophobicity index [CHI] × 0.0857 − 2; for comparison, CHI LogD = CHI × 0.0525 − 1.467. See Valko K.; Bevan C.; Reynolds D.. Anal. Chem. 1997, 69, 2022−2029.
With these data in hand, the most potent and selective compounds were screened in our pharmacodynamic (PD) lymphocyte reduction model in rats following oral administration. Compound 20 proved to deliver full lymphopenia at the lowest oral dose (0.1 mg/kg po, Figure 4). As opposed to 1, this reduction of lymphocyte count is reversible within 24 h. Our pharmacokinetic (PK)/PD modeling shows that this differentiation is due to the much shorter half-life of agonist 20 in rats (vide infra). A head-to-head comparison with 1 in a collagen-induced arthritis model was performed (Figure 5). At a dose of 3 mg/kg po, agonist 20 shows a clear dose-dependent reduction of paw volume and similar efficacy to 1.30 We next turned our attention to the effect on heart rate of our compounds following oral administration. To our delight, 20 did not show any statistically significant effect on heart rate at doses as high as 100 mg/kg po31 and therefore clearly differentiates it from 1 (Figure 6).
Figure 4.

Lymphocyte reduction over time following oral administration of 20 (as sodium salt) in rats.
Figure 5.

Effect of once daily dosing of 20 (as free base) on paw volume in rat collagen-induced arthritis model. *p < 0.05, **<0.01, and ***<0.001 vs arthritic control ANOVA, posthoc LS means.
Figure 6.

Comparison of the effect of 20 (sodium salt) and 1 on heart rate over time in rats following oral administration.
Because of these promising data, further profiling of 20 was implemented. This compound has excellent intrinsic properties (Table 2), and its poor solubility is compensated by excellent permeability, allowing linear PK in rats up to 300 mg/kg po. No significant CYP inhibition was observed with this molecule, and no covalent adducts were detected in glutathione trapping experiments (nor time-dependent inhibition of CYP 3A4 and 2D6). Low in vitro hepatocyte clearance also translated into low in vivo clearance in three preclinical species (Table 3). Agonist 20 showed good distribution in tissues translating into moderate half-life; oral bioavailability was excellent in all species tested (mouse, rat, and dog).
Table 2. In Vitro Profile of 20.
| MW, PSA, cLogP | 446, 112, 1.94 |
| CHI LogD at pH 2.0, 7.4, and 10.5 | 1.25, 1.72, 1.41 |
| solubility (FeSSIF, mg/mL) | 0.16 |
| permeability (MDCK type 2, nm/s) | 200 |
| hepatocyte CLi (mg/min/g liver; rat, dog, mouse, human) | <0.85, <1.70, <0.85, <0.85 |
| CYP IC50 (μM, 1A2, 2C9, 2C19, 2D6, 3A4VG, 3A4 VR, n = 4) | >50, >50, >50, >50, >50, 29 ± 15 |
Table 3. In Vivo PK of 20 (Sodium Salt).
| species | mouse | rat | dog |
|---|---|---|---|
| strain | CD-1 | CD | beagle |
| dose iv,a po (mg/kg) | 1, 3 | 1, 3 | 1, 2 |
| CLbb (mL/min/kg), % liver blood flow | 18 ± 4,c 15% | 5 ± 1, 6% | 10 ± 3, 25% |
| Vss (L/kg) | 1.9 ± 0.6c | 1.1 ± 0.1 | 2.2 ± 0.6 |
| t1/2 (iv, h) | 1.6 ± 0.7c | 3.0 ± 0.1 | 4.8 ± 3.0 |
| F, po %d | 64 ± 17 | 98 ± 13 | 53 ± 11 |
iv dose was 1 h of infusion in DMSO:10% (w/v) Kleptose HPB (2:98).
Values are means (n = 3) ± SD unless otherwise noted.
iv value for n = 4.
Dose vehicle: 1% (w/v) methylcellulose (400 cps) (aq).
In conclusion, we have identified a druglike S1P3-sparing S1P1 agonist32 showing similar efficacy to 1 at low doses in a model of arthritis. This compound, unlike 1, does not cause bradycardia in rats even at high oral doses. Its excellent PK suggest low human therapeutic doses (<10 mg/kg po once daily).
Acknowledgments
We thank Dr. Richard Upton and Nick Waite for NMR support, Dr. Bill Leavens for recording HRMS spectra, and Dr. Eric Rossman, Jason Payseur, and Xuejun Wu for supporting the cardiovascular studies. Dr. Tom Heightman and Dr. Karen Philpott and their co-workers are gratefully acknowledged for their contribution to the early phase of this effort.
Supporting Information Available
Experimental procedures for the synthesis of compounds 12−28, in vitro assay protocols for the determination of EC50, and protocols for in vivo studies (lymphocyte reduction and CIA model). This material is available free of charge via the Internet at http://pubs.acs.org.
Supplementary Material
References
- Martini S.; Peters H.; Böhler T.; Budde K. Current Perspectives on FTY720. Expert Opin. Invest. Drugs 2007, 16, 505–518. [DOI] [PubMed] [Google Scholar]
- Budde K.; Schütz M.; Glander P.; Peters H.; Waiser J.; Liefeldt L.; Neumayer H-H; Böhler T. FTY720 (fingolimod) in renal transplantation. Clin. Transplant. 2006, 20Suppl. 1717–24. [DOI] [PubMed] [Google Scholar]
- Kappos L.; Antel J.; Comi G.; Montalban X.; O'Connor P.; Polman C. H.; Haas T.; Korn A. A.; Karlsson G.; Radue E. W. Oral Fingolimod (FTY720) for Relapsing Multiple Sclerosis. N. Engl. J. Med. 2006, 355, 1124–1139. [DOI] [PubMed] [Google Scholar]
- Billich A.; Bornancin F.; Dévay P.; Mechtcheriakova D.; Urtz N.; Baumruker T. Phosphorylation of the Immunomodulatory Drug FTY720 by Sphingosine Kinases. J. Biol. Chem. 2003, 278, 47408–47415. [DOI] [PubMed] [Google Scholar]
- Albert R.; Hinterding K.; Brinkmann V.; Guerini D.; Müller-Hartwieg C.; Knecht H.; Simeon C.; Streiff M.; Wagner T.; Welzenbach K.; Zécri F.; Zollinger M.; Cooke N.; Francotte E. Novel Immunomodulator FTY720 Is Phosphorylated in Rats and Humans to Form a Single Stereoisomer. Identification, Chemical Proof, and Biological Characterisation of the Biologically Active Species and Its Enantiomer. J. Med. Chem. 2005, 48, 5373–5377. [DOI] [PubMed] [Google Scholar]
- Matloubian M.; Lo C. G.; Cinamon G.; Lesneski M. J.; Xu Y.; Brinkmann V.; Allende M. L.; Proia R. L.; Cyster J. G. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 2004, 427, 355–360. [DOI] [PubMed] [Google Scholar]
- Wei S. H.; Rosen H.; Matheu M. P.; Sanna M. G.; Wang S.-K.; Jo E.; Wong C.-H.; Parker I.; Cahalan M. D. Sphingosine 1-Phosphate Type 1 receptor Agonism Inhibits Transendothelial Migration of Medullary T Cells to Lymphatic Sinuses. Nature Immunol. 2005, 6, 1228–1235. [DOI] [PubMed] [Google Scholar]
- Hale J. J.; Doherty G.; Toth L.; Mills S. G.; Hajdu R.; Keohane C. A.; Rosenbach M.; Milligan J.; Shei G.-J.; Chrebet G.; Bergstrom J.; Card D.; Forrest M.; Sun S.-Y.; West S.; Xie H.; Nomura N.; Rosen H.; Mandala S. Selecting against S1P3 enhances the acute cardiovascular tolerability of 3-(N-benzyl)aminopropylphosphonic acid S1P receptor agonists. Bioorg. Med. Chem. Lett. 2004, 14, 3501–3505. [DOI] [PubMed] [Google Scholar]
- Forrest M.; Sun S.-Y.; Hajdu R.; Bergstrom J.; Card D.; Doherty G.; Hale J.; Keohane C.; Meyers C.; Milligan J.; Mills S.; Nomura N.; Rosen H.; Rosenbach M.; Shei G.-J.; Singer I. I.; Tian M.; West S.; White V.; Xie J.; Proia R. L.; Mandala S. Immune Cell Regulation and Cardiovascular Effects of Sphingosine 1-Phosphate Receptor Agonists in Rodents Are Mediated via Distinct Receptor Subtypes. J. Pharmacol. Exp. Ther. 2004, 309, 758–768. [DOI] [PubMed] [Google Scholar]
- Sanna M. G.; Liao J.; Jo E.; Alfonso C.; Ahn M.-Y.; Peterson M. S.; Webb B.; Lefebvre S.; Chun J.; Gray N.; Rosen H. Sphingosine 1-Phosphate (S1P) Receptor Subtypes S1P1 and S1P3, Respectively, Regulate Lymphocyte Recirculation and Heart Rate. J. Biol. Chem. 2004, 279, 13839–13848. [DOI] [PubMed] [Google Scholar]
- Hamada M.; Nakamura M.; Kiuchi M.; Marukawa K.; Tomatsu A.; Shimano K.; Sato N.; Sugahara K.; Asayama M.; Takagi K.; Adachi K. Removal of Sphingosine 1-Phosphate receptor-3 (S1P3) Agonism is Essential, But Inadequate to Obtain Immunomodulating 2-Aminopropane-1,3-diol S1P1 Agonists with Reduced Effect on Heart Rate. J. Med. Chem. 2010, 53, 3154–3168. [DOI] [PubMed] [Google Scholar]
- Buzard D. J.; Thatte J.; Lerner M.; Edwards J.; Jones R. M. Recent Progress in the Development of Selective S1P1 Receptor Agonists for the Treatment of Inflammatory and Autoimmune disorders. Expert Opin. Ther. Patents 2008, 18, 1141–1159. [Google Scholar]
- Crosignani S.; Bombrun A.; Covini D.; Maio M.; Marin D.; Quattropani A.; Swinnen D.; Simpson D.; Sauer W.; Françon B.; Martin T.; Cambet Y.; Nichols A.; Martinou I.; Burgat-Charvillon F.; Rivron D.; Donini C.; Schott O.; Eligert V.; Novo-Perez L.; Vitte P.-A.; Arrighi J.-F. Discovery of a Novel Series of Potent S1P1 Agonists. Bioorg. Med. Chem. Lett. 2010, 20, 1516–1519. [DOI] [PubMed] [Google Scholar]
- Shimizu H.; Takahashi M.; Kaneko T.; Murakami T.; Hakamata Y.; Kudou S.; Kishi T.; Fukuchi K.; Iwanami S.; Kuriyama K.; Yasue T.; Enosawa S.; Matsumoto K.; Takeyoshi I.; Morishita Y.; Kobayashi E. KRP-203, a Novel Synthetic Immunosuppressant, Prolongs Graft Survival and Attenuates Chronic Rejection in Rat Skin and Heart Allografts. Circulation 2005, 111, 222–229. [DOI] [PubMed] [Google Scholar]
- Bolli M. H.; Abele S.; Binkert C.; Bravo R.; Buchmann S.; Bur D.; Gatfield J.; Hess P.; Kohl C.; Mangold C.; Mathys B.; Menyhart K.; Müller C.; Nayler O.; Scherz M.; Schmidt G.; Sippel V.; Steiner B.; Strasser D.; Treiber A.; Weller T. 2-Imino-thiazolidin-4-one Derivatives as Potent, Orally Active S1P1 Receptor Agonists. J. Med. Chem. 2010, 53, 4198–4211. [DOI] [PubMed] [Google Scholar]
- Hale J. J.; Lynch C. L.; Neway W.; Mills S. G.; Hajdu R.; Keohane C. A.; Rosenbach M. J.; Milligan J. A.; Shei G.-J.; Parent S. A.; Chrebet G.; Bergstrom J.; Card D.; Ferrer M.; Hodder P.; Strulovici B.; Rosen H.; Mandala S. A Rational Utilisation of High-Throughput Screening Affords Selective, Orally Bioavailable 1-Benzyl-3-carboxyazetidine Sphingosine-1-phosphate-1 Receptor Agonists. J. Med. Chem. 2004, 47, 6662–6665. [DOI] [PubMed] [Google Scholar]
- Yan L.; Huo P.; Hale J. J.; Mills S. G.; Hajdu R.; Keohane C. A.; Rosenbach M. J.; Milligan J. A.; Shei G.-J.; Chrebet G.; Bergstrom J.; Card D.; Mandala S. M. SAR studies of 3-Arylpropionic Acids as Potent and Selective Agonists of Sphingosine-1-phosphate Receptor-1 (S1P1) with Enhanced Pharmacokinetics Properties. Bioorg. Med. Chem. Lett. 2007, 17, 828–831. [DOI] [PubMed] [Google Scholar]
- During the preparation of this manuscript, S1P1 agonists analogous to 7 have been reported as follows:Lanman B. A.; Cee V. J.; Cheruku S. R.; Frohn M.; Golden J.; Lin J.; Lobera M.; Marantz Y.; Muller K. M.; Neira S. C.; Pickrell A. J.; Rivenzon-Segal D.; Schutz N.; Sharadendu A.; Yu X.; Zhang Z.; Buys J.; Fiorino M.; Gore A.; Horner M.; Itano A.; McElvain M.; Middleton S.; Schrag M.; Vargas H. M.; Xu H.; Xu Y.; Zhang X.; Siu J.; Bürli R. W. Discovery of a Potent S1P3-Sparing Benzothiazole Agonist of Sphingosine-1 Phosphate Receptor 1 (S1P1). ACS Med. Chem. Lett. 2011, 2, 102–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saha A. K.; Yu X.; Lin J.; Lobera M.; Sharadendu A.; Chereku S.; Schutz N.; Segal D.; Marantz Y.; McCauley D.; Middleton S.; Siu J.; Bürli R. W.; Buys J.; Horner M.; Salyers K.; Schrag M.; Vargas H. M.; Xu Y.; McElvain M.; Xu H. Benzofuran Derivatives as Potent, Orally Active S1P1 Receptor Agonists: A Preclinical Lead Molecule for MS. ACS Med. Chem. Lett. 2011, 2, 97–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feigin V. Irampanel. Curr. Opin. Invest. Drugs. 2002, 3, 908–910. [PubMed] [Google Scholar]
- Hirawat S.; Welch E. M.; Elfring G. L.; Northcutt V. J.; Paushkin S.; Hwang S.; Leonard E. M.; Almstead N. G.; Ju W.; Peltz S. W.; Miller L. L. Safety, Tolerability, and Pharmacokinetics of PCT124, a Nonaminoglycoside Nonsense Mutation Suppressor, Following Single- and Multiple-Dose Administration to Healthy Male and Female Adult Volunteers. J. Clin. Pharmacol. 2007, 47, 430–444. [DOI] [PubMed] [Google Scholar]
- Leeson P. D.; Springthorpe B. The Influence of Drug-like Concepts on Decision-making in Medicinal Chemistry. Nat. Rev. Drug Discovery 2007, 6, 881–890. [DOI] [PubMed] [Google Scholar]
- See, for example:Lipinski C. A. Lead- and Drug-like Compounds: The Rule-of-five Revolution. Drug Discovery Today Technol. 2004, 1, 337–341. [DOI] [PubMed] [Google Scholar]
- See also:Lipinski C. A.; Lombardo F.; Dominy B. W.; Feeney P. J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Delivery Rev. 1997, 23, 3–25. [DOI] [PubMed] [Google Scholar]
- Wenlock M. C.; Austin R. P.; Barton P.; Davis A. M.; Leeson P. D. A Comparison of Physiochemical Property Profiles of Development and Marketed Oral Drugs. J. Med. Chem. 2003, 46, 1250–1256. [DOI] [PubMed] [Google Scholar]
- Veber D. F.; Johnson S. R.; Cheng H.-Y.; Smith B. R.; Ward K. W.; Kopple K. D. Molecular Properties That Influence The Oral Bioavailability of Drug Candidates. J. Med. Chem. 2002, 45, 2615–2623. [DOI] [PubMed] [Google Scholar]
- Hughes J. D.; Blagg J.; Price D. A.; Bailey S.; DeCrescenzo G. A.; Devraj R. V.; Ellsworth E.; Fobian Y. M.; Gibbs M. E.; Gilles R. W.; Greene N.; Huang E.; Krieger-Burke T.; Loesel J.; Wager T.; Whiteley L.; Zhang Y. Physiochemical Drug Properties Associated With In Vivo Toxicological Outcomes. Bioorg. Med. Chem. Lett. 2008, 18, 4872–4875. [DOI] [PubMed] [Google Scholar]
- Reported SAR suggests that the three aryl rings present in agonist 8 are the frame critical to place the substituent in the right position to see agonism; therefore, no attempt to modify these vectors was initiated. See ref (17) and references cited therein.
- S1P1 is coupled to Gi so the GTPγS assay was used to confirm agonist function and reflects proximal signaling events induced by agonist engagement. Loss of S1P1 function following agonist-induced receptor internalization is the proposed mechanism of action of these synthetic S1P1 ligands, so we also confirmed S1P1 receptor internalisation using a β-arrestin assay (a surrogate of S1P1 internalization). For S1P3, an absence of activity was required for our compounds. In this case, a GTPγS assay was used.
- A few examples with substituted 5-membered ring heterocycles in this position were made and were all very significantly less active than the phenyl derivative.
- It has not been unambiguously proven that the acid functionality in this template interacts with the same residues as do the phosphates of 3 and S1P. See the following:Gonzalez-Cabrera P. J.; Jo E.; Sanna M. G.; Brown S.; Leaf N.; Marsolais D.; Schaeffer M.-T.; Chapman J.; Cameron M.; Guerrero M.; Roberts E.; Rosen H. Full Pharmacological Efficacy of a Novel S1P1 Agonist That Does Not Require S1P-Like Headgroup Interactions. Mol. Pharmacol. 2008, 74, 1308–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- In this experiment, as the free base of 20 was used rather than the sodium salt, maximal lymphopenia was observed only at the 3 mg/kg po dose (in sharp contrast with what was observed in the lymphocyte count experiment using the sodium salt; Figure 4). See the experimental part for details.
- The exposure of 20 depends linearly of the dose given between 0.1 and 300 mg/kg po in rats.
- Compound 20 was inactive at S1P2 (pEC50 < 4.48, n = 6) and S1P4 (pEC50 < 4.38, n = 4) and a partial agonist at S1P5 (pEC50 = 6.8 ± 0.16, 77% of maximum response, n = 6). See the experimental part for assay protocols.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.