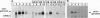Abstract
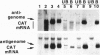
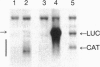
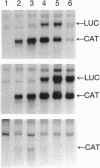
RNA synthesis by the paramyxovirus respiratory syncytial virus, a ubiquitous human pathogen, was found to be more complex than previously appreciated for the nonsegmented negative-strand RNA viruses. Intracellular RNA replication of a plasmid-encoded "minigenome" analog of viral genomic RNA was directed by coexpression of the N, P, and L proteins. But, under these conditions, the greater part of mRNA synthesis terminated prematurely. This difference in processivity between the replicase and the transcriptase was unanticipated because the two enzymes ostensively shared the same protein subunits and template. Coexpression of the M2 gene at a low level of input plasmid resulted in the efficient production of full-length mRNA and, in the case of a dicistronic minigenome, sequential transcription. At a higher level, coexpression of the M2 gene inhibited transcription and RNA replication. The M2 mRNA contains two overlapping translational open reading frames (ORFs), which were segregated for further analysis. Expression of the upstream ORF1, which encoded the previously described 22-kDa M2 protein, was associated with transcription elongation. A model involving this protein in the balance between transcription and replication is proposed. ORF2, which lacks an assigned protein, was associated with inhibition of RNA synthesis. We propose that this activity renders nucleocapsids synthetically quiescent prior to incorporation into virions.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beckes J. D., Haller A. A., Perrault J. Differential effect of ATP concentration on synthesis of vesicular stomatitis virus leader RNAs and mRNAs. J Virol. 1987 Nov;61(11):3470–3478. doi: 10.1128/jvi.61.11.3470-3478.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins P. L., Hill M. G., Camargo E., Grosfeld H., Chanock R. M., Murphy B. R. Production of infectious human respiratory syncytial virus from cloned cDNA confirms an essential role for the transcription elongation factor from the 5' proximal open reading frame of the M2 mRNA in gene expression and provides a capability for vaccine development. Proc Natl Acad Sci U S A. 1995 Dec 5;92(25):11563–11567. doi: 10.1073/pnas.92.25.11563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins P. L., Hill M. G., Johnson P. R. The two open reading frames of the 22K mRNA of human respiratory syncytial virus: sequence comparison of antigenic subgroups A and B and expression in vitro. J Gen Virol. 1990 Dec;71(Pt 12):3015–3020. doi: 10.1099/0022-1317-71-12-3015. [DOI] [PubMed] [Google Scholar]
- Collins P. L., Mink M. A., Stec D. S. Rescue of synthetic analogs of respiratory syncytial virus genomic RNA and effect of truncations and mutations on the expression of a foreign reporter gene. Proc Natl Acad Sci U S A. 1991 Nov 1;88(21):9663–9667. doi: 10.1073/pnas.88.21.9663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emerson S. U. Reconstitution studies detect a single polymerase entry site on the vesicular stomatitis virus genome. Cell. 1982 Dec;31(3 Pt 2):635–642. doi: 10.1016/0092-8674(82)90319-1. [DOI] [PubMed] [Google Scholar]
- Fuerst T. R., Niles E. G., Studier F. W., Moss B. Eukaryotic transient-expression system based on recombinant vaccinia virus that synthesizes bacteriophage T7 RNA polymerase. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8122–8126. doi: 10.1073/pnas.83.21.8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García J., García-Barreno B., Vivo A., Melero J. A. Cytoplasmic inclusions of respiratory syncytial virus-infected cells: formation of inclusion bodies in transfected cells that coexpress the nucleoprotein, the phosphoprotein, and the 22K protein. Virology. 1993 Jul;195(1):243–247. doi: 10.1006/viro.1993.1366. [DOI] [PubMed] [Google Scholar]
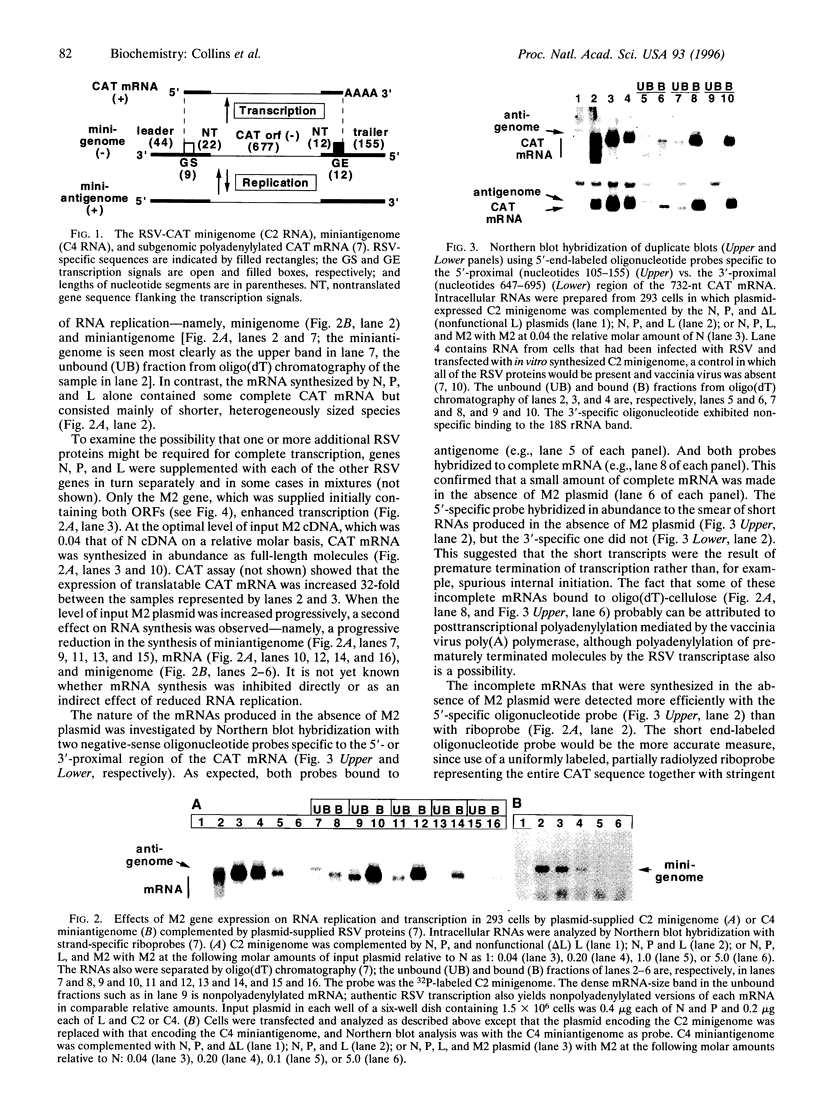
- Grosfeld H., Hill M. G., Collins P. L. RNA replication by respiratory syncytial virus (RSV) is directed by the N, P, and L proteins; transcription also occurs under these conditions but requires RSV superinfection for efficient synthesis of full-length mRNA. J Virol. 1995 Sep;69(9):5677–5686. doi: 10.1128/jvi.69.9.5677-5686.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iverson L. E., Rose J. K. Localized attenuation and discontinuous synthesis during vesicular stomatitis virus transcription. Cell. 1981 Feb;23(2):477–484. doi: 10.1016/0092-8674(81)90143-4. [DOI] [PubMed] [Google Scholar]
- Ling R., Easton A. J., Pringle C. R. Sequence analysis of the 22K, SH and G genes of turkey rhinotracheitis virus and their intergenic regions reveals a gene order different from that of other pneumoviruses. J Gen Virol. 1992 Jul;73(Pt 7):1709–1715. doi: 10.1099/0022-1317-73-7-1709. [DOI] [PubMed] [Google Scholar]
- Marciniak R. A., Sharp P. A. HIV-1 Tat protein promotes formation of more-processive elongation complexes. EMBO J. 1991 Dec;10(13):4189–4196. doi: 10.1002/j.1460-2075.1991.tb04997.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrault J., Clinton G. M., McClure M. A. RNP template of vesicular stomatitis virus regulates transcription and replication functions. Cell. 1983 Nov;35(1):175–185. doi: 10.1016/0092-8674(83)90220-9. [DOI] [PubMed] [Google Scholar]
- Routledge E. G., Willcocks M. M., Morgan L., Samson A. C., Scott R., Toms G. L. Heterogeneity of the respiratory syncytial virus 22K protein revealed by Western blotting with monoclonal antibodies. J Gen Virol. 1987 Apr;68(Pt 4):1209–1215. doi: 10.1099/0022-1317-68-4-1209. [DOI] [PubMed] [Google Scholar]
- Vidal S., Kolakofsky D. Modified model for the switch from Sendai virus transcription to replication. J Virol. 1989 May;63(5):1951–1958. doi: 10.1128/jvi.63.5.1951-1958.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright S. Regulation of eukaryotic gene expression by transcriptional attenuation. Mol Biol Cell. 1993 Jul;4(7):661–668. doi: 10.1091/mbc.4.7.661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Q., Davis P. J., Brown T. D., Cavanagh D. Sequence and in vitro expression of the M2 gene of turkey rhinotracheitis pneumovirus. J Gen Virol. 1992 Jun;73(Pt 6):1355–1363. doi: 10.1099/0022-1317-73-6-1355. [DOI] [PubMed] [Google Scholar]
- Yu Q., Hardy R. W., Wertz G. W. Functional cDNA clones of the human respiratory syncytial (RS) virus N, P, and L proteins support replication of RS virus genomic RNA analogs and define minimal trans-acting requirements for RNA replication. J Virol. 1995 Apr;69(4):2412–2419. doi: 10.1128/jvi.69.4.2412-2419.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]