Abstract
Background:
Brown rice (BR) is unpolished rice with various beneficial compounds such as vitamins, magnesium and other minerals, dietary fiber, essential fatty acids, γ-oryzanol and γ-aminobutyric acid. In the present study, we compared the effects of white rice (WR) and BR on inflammatory marker high-sensitivity C-reactive protein (hs-CRP) and cardiovascular risk factors among non-menopausal overweight or obese female.
Methods:
In a randomized cross-over clinical trial, 40 overweight or obese (body mass index (BMI) >25) women were randomly allocated to group 1 (n = 20): Treatment with BR diet and group 2 (n = 20): Treatment with WR diet for 6 weeks (first intervention period). Two participants in group 2 dropped out during this period. After a 2-week washout period, individuals were switched to the alternate diet for an additional 6 weeks (second intervention period) and three subjects in group 2 did not follow this period and eliminated, finally this study was completed with 35 subjects (group 1 = 20 and group 2 = 15). Each one was instructed to consume 150 g cooked WR or BR daily in each intervention period. Cardiovascular risk factors including BMI, waist and hip circumference, blood pressure, serum lipid profiles, fasting blood glucose (FBG) and hs-CRP as an inflammatory marker, were measured 4 times (in study week 0, 6, 8, 14).
Results:
BR diet in comparison with WR diet could significantly reduce weight, waist and hip circumference, BMI, Diastole blood pressure and hs-CRP. No significant differences between the two diets were found regarding lipid profiles and FBG.
Conclusions:
The present results suggest that BR replacement in the diet may be useful to decrease inflammatory marker level and several cardiovascular risk factors among non-menopausal overweight or obese female.
Keywords: Brown rice, female, inflammation, obese, overweight, white rice
INTRODUCTION
Obesity has been a major world-wide public health challenge.[1] During the past 10 years, there has been a dramatic increase in the prevalence of obesity in both developed and developing countries.[2] It is imperative to identify effective and simple life-style intervention strategies to reduce the risk of obesity.
Adipose tissue as an endocrine organ producing adipokines, are thought to be key regulators of inflammation. Low grade chronic inflammation is a characteristic of obese state and has an important role in pathogenesis and progression of chronic disease such as metabolic syndrome, diabetes and cardiovascular disorders.[3] It is becoming increasingly evident that over nutrition, physical inactivity and aging would result in hypersecretion inflammatory mediators and eventually lead to obesity-induced disorders.[2]
Nutrition has a key role both in prevention and control of obesity.[4] For centuries rice has been traditional staple food in Asian countries and more than 70% of rice consumed is in the form of white rice (WR).[5] Several cohort studies indicate that higher WR intake is associated with elevated risk of diabetes.[6] In a prospective study conducted in Japan, the same result was obtained.[7] Recent studies shown that intake of whole grain as a component of healthy eating pattern has been associated with lower risk of chronic disease.[8]
Brown rice (BR) is the unrefined whole grain contains various and important nutrient in bran layer that is located between white center and outer bran.[9] Removing bran layer results in losing potential health benefits of rice. Due to the milling process and different degree of processing, compared with WR, BR has a greater amount of dietary fiber, magnesium, vitamin B, γ-aminobutyric acid (GABA), γ-oryzanol (γ-ORZ), Phytoestrogens, lignin and essential fatty acid. All of which are potentially protective against metabolic risk factors in obesity adults.[10]
Major previous studies had focused on the effects of BR on diabetic risk factors which show BR is associated whit a lower risk of diabetes.[11,12] In BR, GABA that is known as a neurotransmitter has the ability to regulate the blood pressure and increase insulin secretion.[13] Furthermore, γ-ORZ as a potent anti-oxidant by antihypercholestrolemic effects[14] and vitamin E[15] may prevent the progression of cardiovascular complication and may confer protection against metabolic disorders. Few studies that had considered the influence of substituting WR with BR on metabolic risk factors show no significant result.[9] Previously conflicting results regarding the effect of BR on some inflammation mediators such as plasminogen activator inhibitor type-1 (PAI-1) and tumor necrosis factor-alpha (TNF-α) in rats were reported.[16,17] In the 14 week clinical trial using cross-over design, we aim to determine the effects of BR consumption on high-sensitivity C-reactive protein (hs-CRP) as an inflammatory marker and some cardiovascular risk factors among non-menopausal overweight or obese female adults.
METHODS
Participants
Participants were eligible if they have a body mass index (BMI) more than 25 kg/m2 and non-menopausal, non-pregnant, non-lactating and non-smoking women in the range of 18-50 years old were included in this study. Individual with a history of chronic disease, consuming medicine and flowing special regimen were excluded. The sample size calculation was determined using the serum hs-CRP level as a key dependent variable[18] in the standard formula suggested for cross-over trials:[19] n = [(Z 1 − α/2 + Z 1 − β)2. S2]/2 (ϻ1 − ϻ2 − δ)2; where Z 1 − α/2 was 1.96, Z 1 − β was 0.84, S was 0.01, ϻ1 − ϻ2 was 0 and δ was 0.04. In the present study, 40 participants were needed for adequate power.
A total of 40 eligible volunteers enrolled into the present study and allocated to group 1 (treatment from BR to WR; n = 20) and group 2 (treatment from WR to BR; n = 20). All of them were instructed to use BR in one period of trial and WR in another period. Each patient received two diets. In group 2, 2 subjects in the first intervention period and 3 subjects in the second intervention period were eliminated from this study, due to: Busy schedule: n = 3, intervention unrelated surgery problem: n = 1 and pregnancy: n = 1. Finally this study was conducted with 15 subjects in group 2 and 20 subjects in grup1. Signing the written consent was requested from all of them. The study protocol was approved by Food Security Research Center Isfahan University of Medical Science and Nutrition Department. The participant flow diagram is shown in Figure 1.
Figure 1.

Patients flow diagram in the present study
Study design
In the run in period subjects were allowed to continue performing daily living and normal activities. After 2 week run in, participants were randomly allocated to two experimental periods of WR or BR, each one for 6 weeks in a crossover design. First intervention period was 0-6 week, second intervention period was 8-14 week and 2 week was set for washout phase from 6 to 8 of the study [Figure 2]. In the present work participant were not blinded and all of them were on a weight reducing diet in duration of the study. Two kinds of rice used in this study were from the same Iranian rice variety (Tarom). Raw rice packages were given to each subject and all of them were instructed to eat 150 g of cooked rice daily. Participants were allowed to continue their usual physical activity and prepared their own meal according to prescribed diet.
Figure 2.
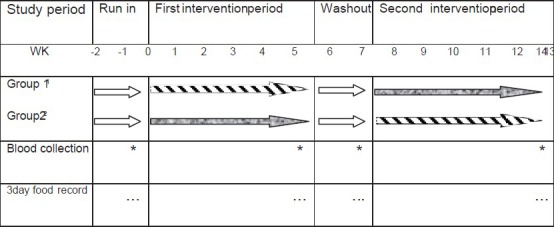
Study cross-over design to observe the effect of white rice and brown rice on inflammatory marker and cardiovascular risk factors among overweight or obese female adults
Diets
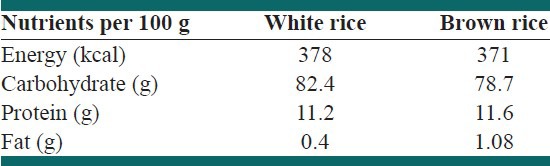
Two diets were prescribed for each patient: (1) Diet with WR (2) diet with BR. The macronutrient composition of both diet were: 50-60% carbohydrate, 15-20% protein and <30% fat respectively. For assessing subject adherence, dietary intake based on 3-day food records, were obtained and analysis at baseline and end of each intervention period. As derived from diet prescription and food records, no significant differences were obtained between consumed amount and prescribed amount from each of the five food group. In the both of intervention periods, we provided weight reducing diet with 200-500 kcal/day deficit for each female based on the BMI range. In BR period 10 spoon (150 g) BR was replaced instead of 10 spoons (150 g) WR. Individual diet was prescribed for each participant and provided an exchange list and educated them how to record their food intake and method of using exchange list. Composition of BR and WR is presented in Table 1.
Table 1.
Composition of white rice and brown rice
Variables assessment
After an overnight fasting, Blood samples were collected from patients between 9 and 11 am. After centrifuging at 4°C and 500 × g for 10 min, tubes were frozen at −80°C until analyzing. Serum glucose, total cholesterol (TC), triacylglycerol (TG), high-density lipoprotein (HDL) and low-density lipoprotein (LDL) were measured by photometric enzyme assay (pars Azmoon, Iran). hs-CRP was determined by latex-immunoturbidemetric assay (Bionic, Iran). Both intra and inter assay coefficients of variation for all of measurements were <5%.
Body weight and height were measured with light clothing and without shoes to the nearest 0.1 kg and 0.1 cm respectively and BMI was calculated. Waist circumference was obtained at the midpoint between the lowest rib and the iliac crest. When participants were in comfortable seat position, after 10 min rest, blood pressure was measured on the right arm using electronic blood monitor. Except height, other physical characteristics and blood pressure were measured 4 times, in study week 0, 6, 8, 14 respectively.
Statistical analysis
N4 was used for analyzing dietary intake. In this study for all variables, different value between first and last intervention in the each period was calculated and then difference between them was determined as mean difference for both group 1 and group 2. Normality of the variables were checked using quantile-quantile-plot. We used paired t-test for comparing means difference to detect any statistical significant level as main effect of BR and WR diets. We also check the period and carry-over effects using independent t-test. Data were analyzed using SPSS (version 15) and a 2-sided P < 0.05 was considered to be significant.
RESULTS
After randomization, the two groups were balanced but during the study, 5 person withdrew duo to busy schedule (n = 3), intervention unrelated surgery problem (n = 1) and pregnancy (n = 1). Thus, a total of 40 participants, 35 non-menopausal over weight and obese female completed the study. The period and carry-over effects were checked and none of them were significant in this study.
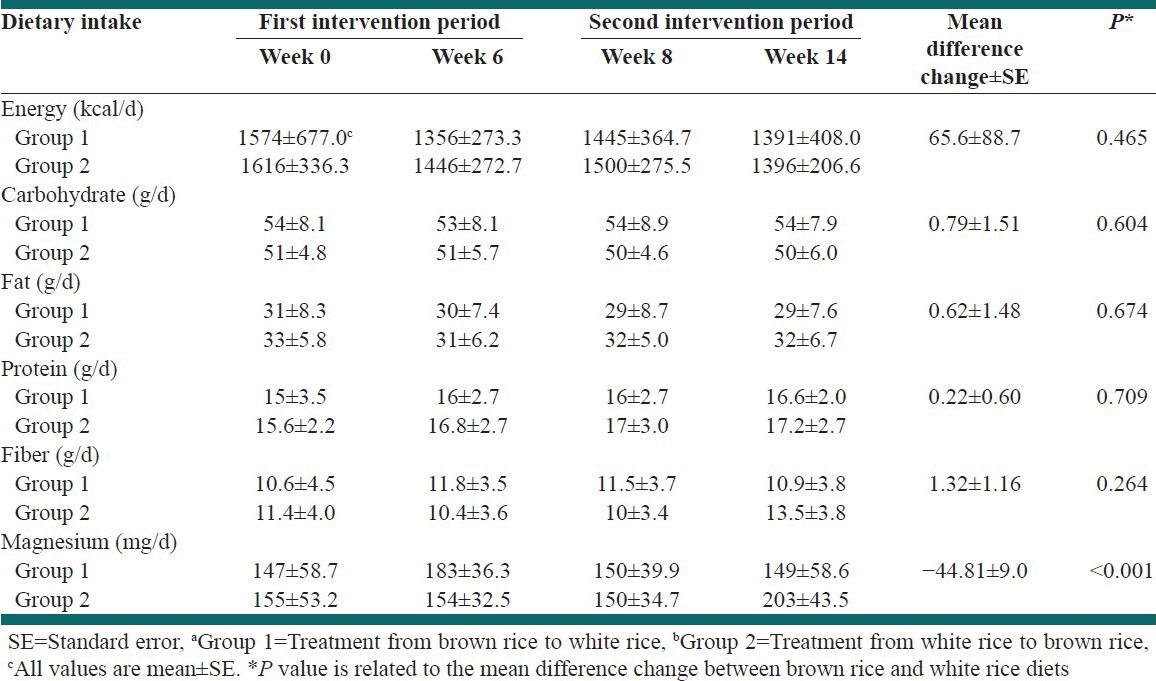
The mean age of the patient was 32.6 ± 6 years and the mean of BMI was 29.38 ± 4. Majority of patients were married. None of them were on specific medication and no one consume any nutritional supplement. Except dietary magnesium content which was significantly (P < 0.001) higher during BR consumption, no significant differences were found between WR and BR diets regarding each of the five food groups, polyunsaturated fatty acid (PUFA), monounsaturated fatty acid (MUFA), saturated fatty acids and majority of micronutrients as derived from the food record. Energy and nutrient intake in the first and second intervention period was shown in Table 2. Participant's physical activity level was moderate and did not have any exercises. They were instructed to avoid change in exercise habits, hence their physical activity was similar and it was checked by researchers.
Table 2.
Energy and nutrient intakes in the first and second intervention periods in Groups 1a and 2b
BR diet in comparison with WR could decrease physical characteristics such as: Weight, waist and hip circumference and BMI significantly. Mean difference changes were 1.2 kg (95% confidence interval [CI] =0.60-1.96, P = 0.001), 2.38 cm (95% CI = 0.51-4.25, P = 0.014), 2.98 cm (95% CI = 1.55-4.42, P ≤ 0.001) and 0.52 kg/m2 (95% CI = 0.26-0.78, P ≤ 0.001) respectively.
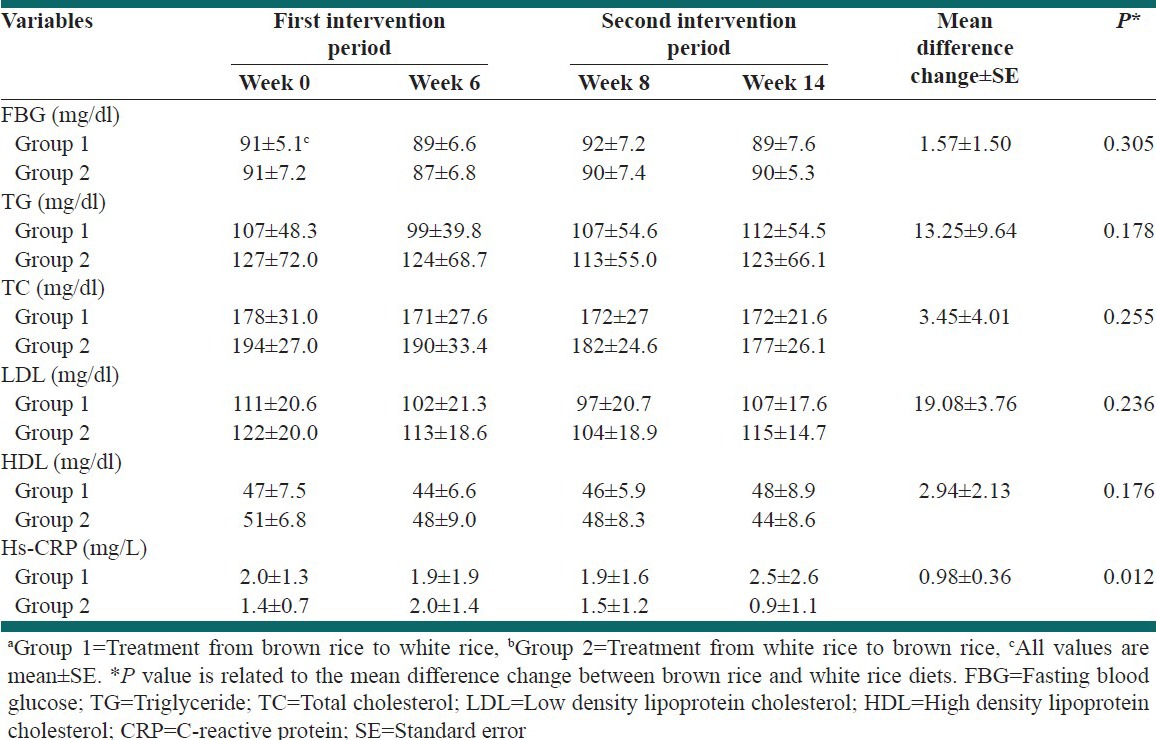
In the BR diet, inflammatory marker (hs-CRP) level decreased significantly in comparison with WR diet. Mean difference changes was 0.98 mg/L (95% CI = 0.23-1.73, P = 0.012). No significant changes were appeared regarding serum lipids concentration such as TG, TC, HDL, LDL and fasting blood glucose (FBG).
Diastole blood pressure level was significantly (P = 0.032) decreased after consuming BR diet compared with WR diet. Physical characteristics and biochemical parameters in first and second intervention phase in group 1 and 2, as shown in Table 3 and Table 4.
Table 3.
Physical characteristics in the first and second intervention period in Groups 1a and 2b
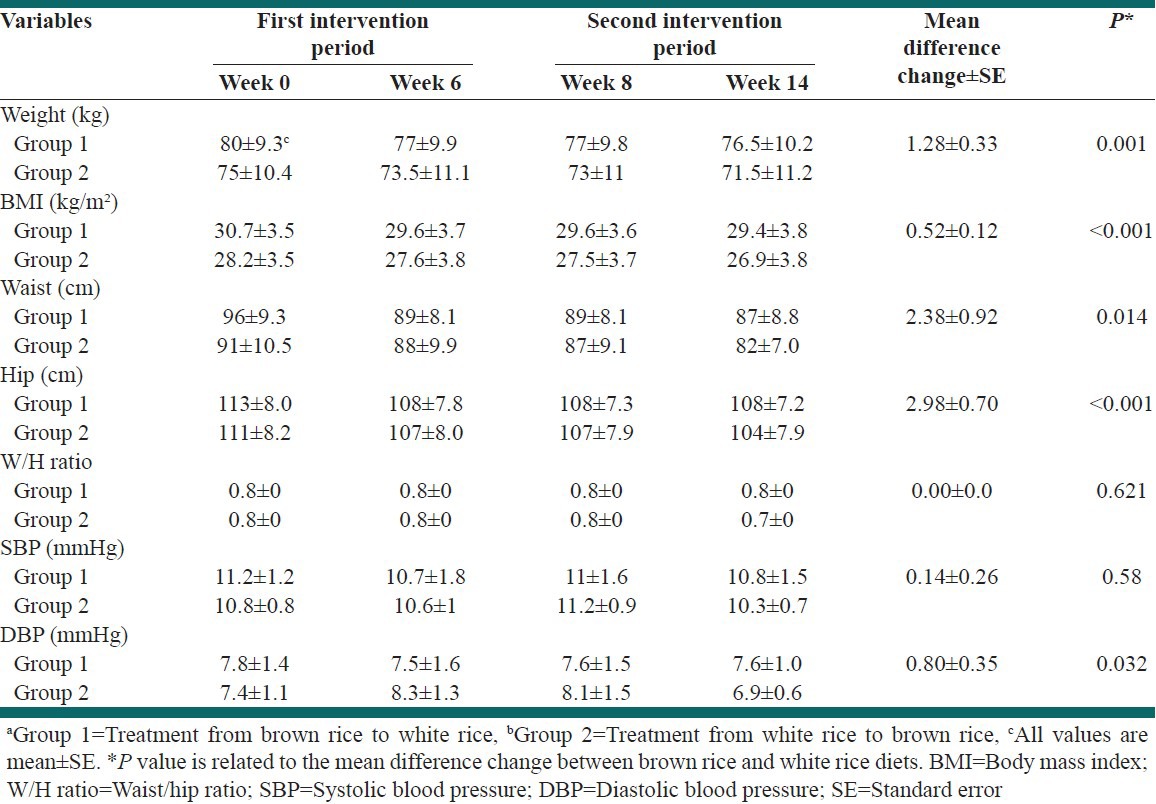
Table 4.
Biochemical parameters in the first and second intervention periods in Groups 1a and 2b
DISCUSSION
After BR consumption magnesium was significantly higher than WR consumption. This is due to the high concentration of this nutrient in bran layer of BR, which has potential benefit for the cardiovascular system.[20]
The present results show that BR diet in comparison with WR diet could significantly reduce weight, waist and hip circumference, BMI and hs-CRP. Diastole blood pressure significantly improved after BR consumption.
BR as unrefined whole grain contain protective factors for diabetics and cardiovascular disease, such as vitamins, magnesium and other minerals, fiber and essential fatty acids.[21] By milling and polishing BR and convert it to WR, 50% fiber 84% magnesium and 69% of total MUFA and PUFA have been eliminated respectively. Despite these benefits, Asian people prefer WR because WR is softer and tastier than BR.[9]
Previous studies indicated no significant changes on body weight following BR consumption[9,22] but the size of adipocytes was reduced by feeding with pre germinated BR in diabetic rats.[16] In this study diet with BR could reduce waist circumference, a strong risk factor for cardiovascular disease[23] and enhanced weight loss. There are many reports which show the ability of fiber to slow down the process of absorption of nutrients by modifying the metabolic response to intake, its ability to lowering fasting insulin level, increase the excretion of biliary acid and the metabolic effects of the short-chain fatty acids (SCFA) produced in the bacterial fermentation of fiber.[24]
BR has 4 times as much dietary fiber as WR.[25] arabinoxylan and β-glucan are growth substrates for beneficial bacteria such as Bifidobacterium and Lactobacillus.[26] Recent human studies reported relationships between the composition of the intestinal microbiota and obesity.[27] In a recent study, BR could increase large bowel SCFA and have a beneficially effect on the gut microbial composition in humans. Hence anti-obesity effects of BR may be related to the profile and activity of the intestinal microbiota.[28]
Leptin is released from adipocytes and controls food intake and energy expenditure by brain mechanisms. GABA, an important component of rice bran, has the ability to prevent obesity by linking to leptin as the co-transmitter.[29] Finding from an animal study indicated that BR has the anti-obesity effect through down regulation of lipogenic gene.[30]
The organelle responsible for protein synthesis, folding and trafficking is endoplasmic reticulum (ER). Creating a toxic condition due to accumulation of newly synthesized unfolded proteins defined as ER stress, which induces apoptotic cell death.[31] hypothalamic ER stress is linked to leptin resistance, which leads to dysregulation of appetite and energy expenditure in obese mice.[32] Furthermore hypothalamic ER stress influences the preference for dietary fat. γ-ORZ (ferulate ester of triterpen alcohols) in bran layer acts as a chemical chaperone; so it can decrease hypothalamic ER stress, ameliorated hypothalamic leptin resistance and attenuate the preference for dietary fat.[33] Overall this evidence highlights that BR may have potential for the enhanced weight loss in humans.
There are conflicting results regarding the effect of BR on serum lipid profiles and FBG. In a recent study substituting WR with BR for 16 weeks was not associated with improvement in fasting serum glucose, insulin, Serum lipids among people with diabetes.[9] Two recent short-term interventions indicated no significant benefits of substituting whole grains for refined grains on fasting glucose, insulin and blood lipids.[34,35] But in the study with cross-over design diabetic patients were instructed to consume 180 g of rice 3 times a day for 6 week and blood glucose management, lipid related markers were improved on BR diet.[11] These results were similar to the results of another study in which blood glucose and insulin reaction were lower for BR when compared with WR.[36]
Past studies have reported the favorable effects of dietary fiber on blood glucose concentration. Higher content of dietary fiber in BR (2 g/100 g) than in WR (0.4 g/100 g) might be responsible for this effect. Fiber in bran layer raps α-amylase, the enzyme to digest starch and substrates (saccharides) in the intestine, ultimately suppresses increasing in postprandial blood glucose levels.[22] GABA that is richer in BR than WR (contents of GABA in raw BR and WR were 7 mg/100 and 3 mg/100 respectively), stimulate the pancreatic beta cell and insulin secretion from pancreas[13] and injection GABA agonist to streptozotocin-induced diabetic rats ameliorated the elevation of blood glucose concentration and increased their insulin levels.[37]
BR contains γ-ORZ that are effective for improving hypercholesterolemia by suppressing cholesterol absorption from intestine, in both human and animal's models.[37,38] BR diet can also suppress hypercholesterolemia via enhancing fecal bile acids secretion without affecting on the cholesterol synthesis of host liver.[39] Comparing with WR, BR contains more GABA with hypocholestrolemic effect, although the exact mechanisms are not yet clear.[40,41] However in our study BR diet in comparison with WR diet, did not have any significant effects on serum lipid profiles and FBG. This might be related to the low amount of BR intake in the current study.
In the present study, we observed that comparing with WR diet; BR diet could reduce diastole blood pressure. This result was similar to the findings of the study which showed the BR intervention had a beneficial effect on diastole pressure among diabetic person.[9] Furthermore in another study consumption of diet with BR/whole wheat for 5 week could reduce blood pressure significantly in hypercholestrolemic patients.[42] Several mechanisms have been proposed to be responsible for the reduction in blood pressure, which occurs with increased fiber intake including increased water and electrolyte excretion with insoluble fiber intake. Past studies have shown that GABA is not only a neurotransmitter but also it can decrease blood pressure.[43] The major bioactive peptide of the renin-angiotensin system, Angiotensin II, is an important growth factor causing hypertrophy and hyperplasia of vascular smooth muscle cells that is believed to be pivotal for progression of hypertension. Extract from the bran layer of BR has a protective effect toward hypertension by interfering with signal transduction induced by Ang II.[44]
Few studies are available about the effects of whole grain or BR on inflammatory risk markers. hs-CRP is a strong inflammatory marker for predicting future cardiovascular events.[45] Compared with WR, BR ameliorated the increase in both plasma TNF-α and PAI-1 and decrease in adiponectin in diabetic rats.[16] This result was similar to another study which showed PAI-1 significantly decreased in diabetic rats by taking PGBR for 7 week.[17] Two previous studies did not find significant benefits of substituting whole grains for refined grains on interleukin-6 and hs-CRP. In a study consumption of 3 portions/d whole-grain foods for 12 week had not any effects on mentioned markers.[32] This result was consistent with results from a 14-week pilot trial using a crossover design in 30 participants.[46]
In our short-term trial BR diet in comparison with WR diet, significantly reduced level of hs-CRP. The probabilistic mechanism can be explained through nuclear factor-kappa (NF-κB). Gene expression of adhesion molecules in vascular endothelium including intercellular adhesion molecule, vascular cell adhesion molecule and E-selection regulate by NF-κB.[47,48]
A recent study showed that γ-ORZ as well as it's component does-dependently reduced adhesion-molecules expression through NF-κB inhibition in vascular endothelium.[49] CRP could be induced from Il-6 through the activation of NF-κB.[50,51]
γ-ORZ probably can have meditative role on CRP expression, at least, partly through the modulation of NF-κB dependent path way, but the exact mechanisms are not known yet.
Previous studies regarding the effect of BR had focused mostly on diabetic markers. However, a few human studies are available about the effect of BR on inflammatory and cardiovascular risk factors. Furthermore, we assessed the effect of BR in cross-over design as the strong method for conducting this trial.
One important difference between our study and the other research was the amount of BR intake. The subjects were instructed to consume only 150 g BR accompany with weigh loss diet and some significantly effects were seen for mentioned variables. In the present study we just recommended food item but BR and WR were provided for participants. This limitation should be considered in interpretation the finding of this research. We analyzed patient food records and their diets controlled exactly.
In non-menopausal female, since lipid profiles variation may be affected by change in blood estrogen level, patient biochemical measurement were assessed based on menstrual date and all of them were in the same hormone phase. This study was conducted on healthy adults without any specific disorder; hence, our trial should have good external validity.
CONCLUSIONS
BR replacement in the diet as a staple food can be useful to improve weight loss, waist and hip circumference, BMI, diastole pressure and hs-CRP level significantly, among over weight and obese female adults. Our findings show that in overweight or obese female adults BR diet in comparison with WR diet resulted in decreased diastole blood pressure and inflammatory marker (hs-CRP). Furthermore, BR replacement in the diet can be useful to improve weight loss and visceral obesity significantly. Replacement of white rice by brown rice did not have any significant effects on serum lipid profiles and FBG. Therefore brown rice may be recommended for more use considering its benefits.
ACKNOWLEDGMENTS
Isfahan University of Medical Sciences and Food Security Research Center supported this research (391213). The writers appreciate the cooperation of MILAD laboratory for providing facilities to do the biochemical experiments. We also would like to thank all the female adults who participated in the present study.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared
REFERENCES
- 1.Martin LG, Schoeni RF, Andreski PM. Trends in health of older adults in the United States: Past, present, future. Demography. 2010;47(Suppl):S17–40. doi: 10.1353/dem.2010.0003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Esposito K, Giugliano G, Scuderi N, Giugliano D. Role of adipokines in the obesity-inflammation relationship: The effect of fat removal. Plast Reconstr Surg. 2006;118:1048–57. doi: 10.1097/01.prs.0000232281.49432.ce. [DOI] [PubMed] [Google Scholar]
- 3.Calder PC, Ahluwalia N, Brouns F, Buetler T, Clement K, Cunningham K, et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br J Nutr. 2011;106(Suppl 3):S5–78. doi: 10.1017/S0007114511005460. [DOI] [PubMed] [Google Scholar]
- 4.Bol’shova OV, Malinovs’ka TM. Diet therapy of obesity in children and adolescents. Lik Sprava. 2008;7-8:70–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19663021 . [PubMed] [Google Scholar]
- 5.Schulze MB, Schulz M, Heidemann C, Schienkiewitz A, Hoffmann K, Boeing H. Fiber and magnesium intake and incidence of type 2 diabetes: A prospective study and meta-analysis. Arch Intern Med. 2007;167:956–65. doi: 10.1001/archinte.167.9.956. [DOI] [PubMed] [Google Scholar]
- 6.Villegas R, Liu S, Gao YT, Yang G, Li H, Zheng W, et al. Prospective study of dietary carbohydrates, glycemic index, glycemic load, and incidence of type 2 diabetes mellitus in middle-aged Chinese women. Arch Intern Med. 2007;167:2310–6. doi: 10.1001/archinte.167.21.2310. [DOI] [PubMed] [Google Scholar]
- 7.Nanri A, Mizoue T, Noda M, Takahashi Y, Kato M, Inoue M, et al. Rice intake and type 2 diabetes in Japanese men and women: The Japan Public Health Center-based Prospective Study. Am J Clin Nutr. 2010;92:1468–77. doi: 10.3945/ajcn.2010.29512. [DOI] [PubMed] [Google Scholar]
- 8.Jang Y, Lee JH, Kim OY, Park HY, Lee SY. Consumption of whole grain and legume powder reduces insulin demand, lipid peroxidation, and plasma homocysteine concentrations in patients with coronary artery disease: Randomized controlled clinical trial. Arterioscler Thromb Vasc Biol. 2001;21:2065–71. doi: 10.1161/hq1201.100258. [DOI] [PubMed] [Google Scholar]
- 9.Zhang G, Pan A, Zong G, Yu Z, Wu H, Chen X, et al. Substituting white rice with brown rice for 16 weeks does not substantially affect metabolic risk factors in middle-aged Chinese men and women with diabetes or a high risk for diabetes. J Nutr. 2011;141:1685–90. doi: 10.3945/jn.111.142224. [DOI] [PubMed] [Google Scholar]
- 10.Roohinejad S, Omidizadeh A, Mirhosseini H, Saari N, Mustafa S, Yusof RM, et al. Effect of pre-germination time of brown rice on serum cholesterol levels of hypercholesterolaemic rats. J Sci Food Agric. 2010;90:245–51. doi: 10.1002/jsfa.3803. [DOI] [PubMed] [Google Scholar]
- 11.Hsu TF, Kise M, Wang MF, Ito Y, Yang MD, Aoto H, et al. Effects of pre-germinated brown rice on blood glucose and lipid levels in free-living patients with impaired fasting glucose or type 2 diabetes. J Nutr Sci Vitaminol (Tokyo) 2008;54:163–8. doi: 10.3177/jnsv.54.163. [DOI] [PubMed] [Google Scholar]
- 12.Sun Q, Spiegelman D, van Dam RM, Holmes MD, Malik VS, Willett WC, et al. White rice, brown rice, and risk of type 2 diabetes in US men and women. Arch Intern Med. 2010;170:961–9. doi: 10.1001/archinternmed.2010.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dong H, Kumar M, Zhang Y, Gyulkhandanyan A, Xiang YY, Ye B, et al. Gamma-aminobutyric acid up-and downregulates insulin secretion from beta cells in concert with changes in glucose concentration. Diabetologia. 2006;49:697–705. doi: 10.1007/s00125-005-0123-1. [DOI] [PubMed] [Google Scholar]
- 14.Cicero AF, Gaddi A. Rice bran oil and gamma-oryzanol in the treatment of hyperlipoproteinaemias and other conditions. Phytother Res. 2001;15:277–89. doi: 10.1002/ptr.907. [DOI] [PubMed] [Google Scholar]
- 15.Blum S, Vardi M, Brown JB, Russell A, Milman U, Shapira C, et al. Vitamin E reduces cardiovascular disease in individuals with diabetes mellitus and the haptoglobin 2-2 genotype. Pharmacogenomics. 2010;11:675–84. doi: 10.2217/pgs.10.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Torimitsu M, Nagase R, Yanagi M, Homma M, Sasai Y, Ito Y, et al. Replacing white rice with pre-germinated brown rice mildly ameliorates hyperglycemia and imbalance of adipocytokine levels in type 2 diabetes model rats. J Nutr Sci Vitaminol (Tokyo) 2010;56:287–92. doi: 10.3177/jnsv.56.287. [DOI] [PubMed] [Google Scholar]
- 17.Hagiwara H, Seki T, Ariga T. The effect of pre-germinated brown rice intake on blood glucose and PAI-1 levels in streptozotocin-induced diabetic rats. Biosci Biotechnol Biochem. 2004;68:444–7. doi: 10.1271/bbb.68.444. [DOI] [PubMed] [Google Scholar]
- 18.Azadbakht L, Kimiagar M, Mehrabi Y, Esmaillzadeh A, Hu FB, Willett WC. Soy consumption, markers of inflammation, and endothelial function: A cross-over study in postmenopausal women with the metabolic syndrome. Diabetes Care. 2007;30:967–73. doi: 10.2337/dc06-2126. [DOI] [PubMed] [Google Scholar]
- 19.Fleiss JL. London: Wiley; 1986. The Design and Analysis of Clinical Experiments; pp. 263–71. [Google Scholar]
- 20.Qu X, Jin F, Hao Y, Li H, Tang T, Wang H, et al. Magnesium and the risk of cardiovascular events: A meta-analysis of prospective cohort studies. PLoS One. 2013;8:e57720. doi: 10.1371/journal.pone.0057720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Slavin JL, Martin MC, Jacobs DR, Jr, Marquart L. Plausible mechanisms for the protectiveness of whole grains. Am J Clin Nutr. 1997;70:S459–63. doi: 10.1093/ajcn/70.3.459s. [DOI] [PubMed] [Google Scholar]
- 22.Madar Z. Effect of brown rice and soybean dietary fiber on the control of glucose and lipid metabolism in diabetic rats. Am J Clin Nutr. 1983;38:388–93. doi: 10.1093/ajcn/38.3.388. [DOI] [PubMed] [Google Scholar]
- 23.Kim TH, Kim EK, Lee MS, Lee HK, Hwang WS, Choe SJ, et al. Intake of brown rice lees reduces waist circumference and improves metabolic parameters in type 2 diabetes. Nutr Res. 2011;31:131–8. doi: 10.1016/j.nutres.2011.01.010. [DOI] [PubMed] [Google Scholar]
- 24.Seki T, Nagase R, Torimitsu M, Yanagi M, Ito Y, Kise M, et al. Insoluble fiber is a major constituent responsible for lowering the post-prandial blood glucose concentration in the pre-germinated brown rice. Biol Pharm Bull. 2005;28:1539–41. doi: 10.1248/bpb.28.1539. [DOI] [PubMed] [Google Scholar]
- 25.Benno Y, Endo K, Miyoshi H, Okuda T, Koishi H, Mitsuoka T. Effect of rice fiber on human fecal microflora. Microbiol Immunol. 1989;33:435–40. doi: 10.1111/j.1348-0421.1989.tb01992.x. [DOI] [PubMed] [Google Scholar]
- 26.Jaskari J, Kontula P, Siitonen A, Jousimies-Somer H, Mattila-Sandholm T, Poutanen K. Oat beta-glucan and xylan hydrolysates as selective substrates for Bifidobacterium and Lactobacillus strains. Appl Microbiol Biotechnol. 1998;49:175–81. doi: 10.1007/s002530051155. [DOI] [PubMed] [Google Scholar]
- 27.Tremaroli V, Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012;489:242–9. doi: 10.1038/nature11552. [DOI] [PubMed] [Google Scholar]
- 28.Diamant M, Blaak EE, de Vos WM. Do nutrient-gut-microbiota interactions play a role in human obesity, insulin resistance and type 2 diabetes? Obes Rev. 2011;12:272–81. doi: 10.1111/j.1467-789X.2010.00797.x. [DOI] [PubMed] [Google Scholar]
- 29.Xu Y, O’Brien WG, 3rd, Lee CC, Myers MG, Jr, Tong Q. Role of GABA release from leptin receptor-expressing neurons in body weight regulation. Endocrinology. 2012;153:2223–33. doi: 10.1210/en.2011-2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ho JN, Son ME, Lim WC, Lim ST, Cho HY. Anti-obesity effects of germinated brown rice extract through down-regulation of lipogenic genes in high fat diet-induced obese mice. Biosci Biotechnol Biochem. 2012;76:1068–74. doi: 10.1271/bbb.110666. [DOI] [PubMed] [Google Scholar]
- 31.Yoshida H. ER stress and diseases. FEBS J. 2007;274:630–58. doi: 10.1111/j.1742-4658.2007.05639.x. [DOI] [PubMed] [Google Scholar]
- 32.Ozcan L, Ergin AS, Lu A, Chung J, Sarkar S, Nie D, et al. Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab. 2009;9:35–51. doi: 10.1016/j.cmet.2008.12.004. [DOI] [PubMed] [Google Scholar]
- 33.Kozuka C, Yabiku K, Takayama C, Matsushita M, Shimabukuro M. Natural food science based novel approach toward prevention and treatment of obesity and type 2 diabetes: Recent studies on brown rice and γ-oryzanol. Obes Res Clin Pract. 2013;7:e165–72. doi: 10.1016/j.orcp.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 34.Tighe P, Duthie G, Vaughan N, Brittenden J, Simpson WG, Duthie S, et al. Effect of increased consumption of whole-grain foods on blood pressure and other cardiovascular risk markers in healthy middle-aged persons: A randomized controlled trial. Am J Clin Nutr. 2010;92:733–40. doi: 10.3945/ajcn.2010.29417. [DOI] [PubMed] [Google Scholar]
- 35.Brownlee IA, Moore C, Chatfield M, Richardson DP, Ashby P, Kuznesof SA, et al. Markers of cardiovascular risk are not changed by increased whole-grain intake: The WHOLE heart study, a randomised, controlled dietary intervention. Br J Nutr. 2010;104:125–34. doi: 10.1017/S0007114510000644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.O’Dea K, Nestel PJ, Antonoff L. Physical factors influencing postprandial glucose and insulin responses to starch. Am J Clin Nutr. 1980;33:760–5. doi: 10.1093/ajcn/33.4.760. [DOI] [PubMed] [Google Scholar]
- 37.Gomez R, Asnis N, Tannhauser SL, Barros HM. GABA agonists differentially modify blood glucose levels of diabetic rats. Jpn J Pharmacol. 1999;80:327–31. doi: 10.1254/jjp.80.327. [DOI] [PubMed] [Google Scholar]
- 38.Wilson TA, Nicolosi RJ, Woolfrey B, Kritchevsky D. Rice bran oil and oryzanol reduce plasma lipid and lipoprotein cholesterol concentrations and aortic cholesterol ester accumulation to a greater extent than ferulic acid in hypercholesterolemic hamsters. J Nutr Biochem. 2007;18:105–12. doi: 10.1016/j.jnutbio.2006.03.006. [DOI] [PubMed] [Google Scholar]
- 39.Miura D, Ito Y, Mizukuchi A, Kise M, Aoto H, Yagasaki K. Hypocholesterolemic action of pre-germinated brown rice in hepatoma-bearing rats. Life Sci. 2006;79:259–64. doi: 10.1016/j.lfs.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 40.Roohinejad S, Omidizadeh A, Mirhosseini H, Rasti B, Saari N, Mustafa S. Effect of pre-germination time of brown rice on serum cholesterol levels of hypercholesterolemic rats. J Sci Food Agric. 2010;90:245–51. doi: 10.1002/jsfa.3803. [DOI] [PubMed] [Google Scholar]
- 41.Ito Y, Mizukuchi A, Kise M, Aoto H, Yamamoto S, Yoshihara R, et al. Postprandial blood glucose and insulin responses to pre-germinated brown rice in healthy subjects. J Med Invest. 2005;52:159–64. doi: 10.2152/jmi.52.159. [DOI] [PubMed] [Google Scholar]
- 42.Hallfrisch J, Scholfield DJ, Behall KM. Blood pressure reduced by whole grain diet containing barley or whole wheat and brown rice in moderately hypercholesterolemic men. Nutr Res. 2003;23:1631–42. [Google Scholar]
- 43.Shizuka F, Kido Y, Nakazawa T, Kitajima H, Aizawa C, Kayamura H, et al. Antihypertensive effect of gamma-amino butyric acid enriched soy products in spontaneously hypertensive rats. Biofactors. 2004;22:165–7. doi: 10.1002/biof.5520220133. [DOI] [PubMed] [Google Scholar]
- 44.Utsunomiya H, Takaguri A, Bourne AM, Elliott KJ, Akazawa S, Okuno Y, et al. An extract from brown rice inhibits signal transduction of angiotensin II in vascular smooth muscle cells. Am J Hypertens. 2011;24:530–3. doi: 10.1038/ajh.2011.10. [DOI] [PubMed] [Google Scholar]
- 45.Ridker PM, Hennekens CH, Buring JE, Rifai N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med. 2000;342:836–43. doi: 10.1056/NEJM200003233421202. [DOI] [PubMed] [Google Scholar]
- 46.Andersson A, Tengblad S, Karlström B, Kamal-Eldin A, Landberg R, Basu S, et al. Whole-grain foods do not affect insulin sensitivity or markers of lipid peroxidation and inflammation in healthy, moderately overweight subjects. J Nutr. 2007;137:1401–7. doi: 10.1093/jn/137.6.1401. [DOI] [PubMed] [Google Scholar]
- 47.De Martin R, Hoeth M, Hofer-Warbinek R, Schmid JA. The transcription factor NF-kappa B and the regulation of vascular cell function. Arterioscler Thromb Vasc Biol. 2000;20:E83–8. doi: 10.1161/01.atv.20.11.e83. [DOI] [PubMed] [Google Scholar]
- 48.Ledebur HC, Parks TP. Transcriptional regulation of the intercellular adhesion molecule-1 gene by inflammatory cytokines in human endothelial cells. Essential roles of a variant NF-kappa B site and p65 homodimers. J Biol Chem. 1995;270:933–43. doi: 10.1074/jbc.270.2.933. [DOI] [PubMed] [Google Scholar]
- 49.Sakai S, Murata T, Tsubosaka Y, Ushio H, Hori M, Ozaki H. γ-Oryzanol reduces adhesion molecule expression in vascular endothelial cells via suppression of nuclear factor-κB activation. J Agric Food Chem. 2012;60:3367–72. doi: 10.1021/jf2043407. [DOI] [PubMed] [Google Scholar]
- 50.Agrawal A, Cha-Molstad H, Samols D, Kushner I. Overexpressed nuclear factor-kappaB can participate in endogenous C-reactive protein induction, and enhances the effects of C/EBPbeta and signal transducer and activator of transcription-3. Immunology. 2003;108:539–47. doi: 10.1046/j.1365-2567.2003.01608.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Barnes PJ, Karin M. Nuclear factor-kappaB: A pivotal transcription factor in chronic inflammatory diseases. N Engl J Med. 1997;336:1066–71. doi: 10.1056/NEJM199704103361506. [DOI] [PubMed] [Google Scholar]


