ABSTRACT
Due to the essential role macrophages play in antiviral immunity, it is important to understand the intracellular and molecular processes that occur in macrophages following infection with various strains of vaccinia virus, particularly those used as vaccine vectors. Similarities as well as differences were found in macrophages infected with different poxvirus strains, particularly at the level of virus-induced apoptosis and the expression of immunomodulatory genes, as determined by microarray analyses. Interestingly, the attenuated modified vaccinia Ankara virus (MVA) was particularly efficient in triggering apoptosis and beta interferon (IFN-β) secretion and in inducing changes in the expression of genes associated with increased activation of innate immunity, setting it apart from the other five vaccinia virus strains tested. Taken together, these results increase our understanding of how these viruses interact with human macrophages, at the cellular and molecular levels, and suggest mechanisms that may underlie their utility as recombinant vaccine vectors.
IMPORTANCE Our studies clearly demonstrate that there are substantial biological differences in the patterns of cellular gene expression between macrophages infected with different poxvirus strains and that these changes are due specifically to infection with the distinct viruses. For example, a clear induction in IFN-β mRNA was observed after infection with MVA but not with other poxviruses. Importantly, antiviral bioassays confirmed that MVA-infected macrophages secreted a high level of biologically active type I IFN. Similarly, the phagocytic capacity of macrophages was also specifically increased after infection with MVA. Although the main scope of this study was not to test the vaccine potential of MVA as there are several groups in the field working extensively on this aspect, the characteristics/phenotypes we observed at the in vitro level clearly highlight the inherent advantages that MVA possesses in comparison to other poxvirus strains.
INTRODUCTION
Efficient antiviral immunity involves both innate and adaptive immune responses. Macrophages and dendritic cells (DCs) are specialized antigen-presenting cells (APCs) that play an important role in the innate defense against infection, clearance of infected host cells and molecules, and presentation of viral antigens. Being widely dispersed throughout the body, DCs and macrophages participate in the initial capture and processing of foreign antigens (innate immunity), leading to the subsequent activation of specific T and B cell effector mechanisms (adaptive immunity). In addition to their efficient phagocytic and antigen presentation activities, these phagocytes are also strong regulators of local and systemic immune responses through the secretion of chemokines and cytokines (1). For example, macrophages are equipped with a broad range of pathogen recognition receptors that make them efficient at recognizing foreign viral antigens and producing inflammatory cytokines (2).
The Poxviridae are a family of linear double-stranded DNA viruses that replicate entirely in the cytoplasm of infected cells. While smallpox has been eradicated, interest in poxvirus-derived vectors remains high due to their inherent immunogenic properties, including the ability to induce long-term humoral and cell-mediated immunity (2). Modified vaccinia Ankara virus (MVA), a highly attenuated strain of the poxvirus vaccinia virus (VACV) generated after more than 500 passages in chicken embryo fibroblasts, is considered to be a potent vaccine vector candidate due to its high immunogenic properties (3–5). MVA replicates well in vitro in avian cells but is largely replication deficient in human and other mammalian cells, including HeLa and BSC40 cells (3). Thus, MVA-based recombinants have been effectively used in mammals to induce protective immunity against a wide spectrum of pathogens (6, 7), and their immunogenic potential is currently being tested in phase I/II clinical trials against HIV-1, malaria, and numerous solid tumors (8–11). MVA is also a potentially safe vaccine candidate for smallpox should this virus reemerge as a bioterrorist weapon (12). NYVAC is an attenuated derivative of the VACV Copenhagen strain (CopV), from which 18 open reading frames (ORFs) were specifically deleted from the parental viral genome, including those involved in host range, virulence, and pathogenesis (13). NYVAC-derived vectors are able to express antigens from a broad range of species (13) and have been used as recombinant vaccines against numerous pathogens and tumors (1, 14–16), and phase I/II clinical trials using NYVAC against HIV-1 have shown strong and specific immunogenicity and a good safety profile (17). The most frequently studied nonattenuated VACV strain is Western Reserve (WR), a mouse brain-passaged derivative of the New York Board of Health (NYBH) vaccinia virus (18), one of the most widely used vaccinia viruses in the smallpox eradication program and the basis of the only commercially approved smallpox vaccine (Dryvax) available for limited use in the United States (19).
Since macrophages are one of the key cell types for initiating inflammatory and immune responses to numerous viruses (20, 21), we aimed to study, in these professional APCs, the virus cycle of poxvirus-derived vectors due to their multiple applications in immunology and vaccinology. Specifically, we characterized the impact of infection of primary human macrophages with a panel of six poxvirus vectors. Our studies show that there are substantial biological differences in the patterns of cellular proteins and genes expressed between macrophages infected with different poxvirus strains, and the changes observed differed between nonattenuated and attenuated vaccinia vectors. For example, a strong and clear induction in beta interferon (IFN-β) mRNA expression was observed in macrophages after infection with the attenuated strain MVA and to a lesser degree with NYVAC but not with other nonattenuated poxviruses. Importantly, antiviral bioassays confirmed that MVA-infected macrophages secreted high levels of biologically active type I IFN, and the phagocytic capacity of macrophages was also specifically increased after infection with MVA. Thus, our results clearly highlight the inherent immuno-stimulatory advantages that attenuated vaccinia virus vectors, specifically MVA, possess in comparison to other poxvirus strains.
MATERIALS AND METHODS
Cells, viruses, and infection conditions.
BSC40 and BHK-21 cells have been previously described (22). Human blood was obtained from healthy donors, and peripheral blood mononuclear cells (PBMCs) were isolated from buffy coats following standard protocols (2). (This study received the approval of the Committee of Ethics and Clinical Investigation of the Universidad Autónoma de Madrid [Spain].) Briefly, whole blood was layered on top of a Ficoll cushion (Ficoll-Plaque Plus; GE Healthcare) and centrifuged at 1,800 rpm for 30 min at room temperature. The lymphocyte/monocytic fraction was isolated, washed with 1× phosphate-buffered saline (PBS) and subjected to red blood cell lysis (ammonium chloride solution; Stem cell Technologies) for 5 min at room temperature, and following an additional wash with 1× PBS, monocytes were cultured under adherent conditions in 1× RPMI medium (Gibco) supplemented with 10% fetal bovine serum (FBS) in a humidified atmosphere with 5% CO2 at 37°C. Adhered monocytes were cultured for 7 to 10 days to allow for differentiation into macrophages.
The poxvirus strains used in this work were kindly provided by Mariano Esteban and Antonio Alcamí. NYVAC and MVA strains were propagated in BHK-21 cells. WR, vaccinia virus Ankara (ANK), CopV, and NYCBH were grown in BSC40 cells. All of the strains were purified by centrifugation through a sucrose cushion, followed by sucrose gradient fractionation (23), and then titers were determined by plaque assay on BSC40 cells for all strains except MVA, for which titers were determined by immunostaining on BHK-21 cells with a polyclonal anti-VACV antibody as previously described (22).
Analysis of virus growth.
To determine virus growth profiles, macrophages grown in 24-well tissue culture plates were infected in triplicate with MVA, ANK, WR, NYBH, NYVAC, or CopV at a multiplicity of infection (MOI) of 0.1 PFU/cell. Following virus adsorption for 60 min at 37°C, the inoculum was removed, and infected cultures were incubated with fresh Dulbecco's modified Eagle's medium (DMEM) containing 2% fetal calf serum (FCS) at 37°C in a 5% CO2 atmosphere. At different times postinfection (p.i.) (0, 24, and 48 h), cells were removed by scraping, subjected to three freeze-thaw cycles, and briefly sonicated, and virus yields were determined by plaque assay on BSC40 cells for each virus except MVA, for which the titers were determined by immunostaining on BHK-21 cells.
Metabolic labeling of proteins.
Macrophages were mock infected or infected with MVA, ANK, WR, NYBH, NYVAC, or CopV (5 PFU/cell), and at the indicated times (see Fig. 3A), 1 × 106 cells were washed with methionine-free medium and incubated in methionine-free medium containing [35S]methionine (50 μCi/well) for 30 min at 37°C. Proteins from cell extracts, prepared in lysis buffer, were fractionated by 12% SDS-PAGE and developed by autoradiography.
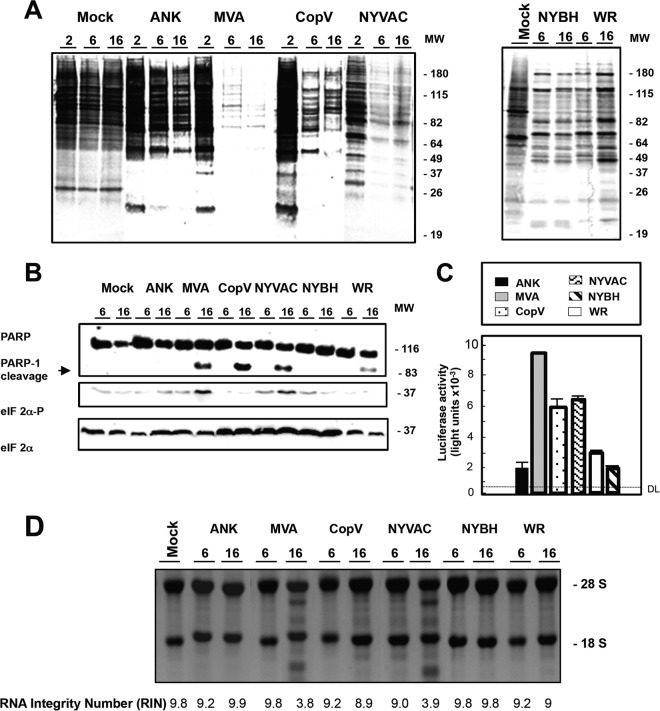
FIG 3.
Protein synthesis inhibition, apoptosis induction, and rRNA breakdown during poxvirus infection of primary human macrophages. (A) Metabolic labeling of proteins during MVA, ANK, WR, NYBH, NYVAC, or CopV infection. Human macrophages were mock infected or infected with the indicated poxviruses (5 PFU/cell); at the times indicated, cells were labeled (30 min) with [35S]Met-Cys Promix (50 μCi/ml), and equal amounts of proteins were subsequently analyzed by SDS-PAGE (10%) and autoradiography. Molecular weight (MW; in thousands) is indicated based on protein standards. (B) Time course of PARP-1 cleavage during MVA, ANK, WR, NYBH, NYVAC, or CopV infection. Human macrophages were mock infected or infected (5 PFU/cell) with the indicated poxviruses, and at the times indicated, total protein (100 μg) was fractionated by SDS-PAGE, transferred to nitrocellulose membranes, and immunoblotted with anti-PARP-1. An 89-kDa PARP-1 cleavage product was observed at 16 h p.i. Molecular weight standards (MW; in thousands) are indicated. eIF-2α or phosphorylated eIF-2α (eIF-2α-P) at Ser51 was measured using specific antibodies. (C) Quantification of cells undergoing apoptosis after infection with MVA, ANK, WR, NYBH, NYVAC, or CopV. At 24 h hours after mock infection or infection with the indicated viruses (5 PFU/cell), cells were fixed, and apoptosis was measured using a Caspase-Glo 3/7 assay kit. Results represent the means ± the standard deviations of three independent experiments. P values from a two-tailed t test assuming nonequal variance were determined. In all the cases, P is <0.05. DL, detection limit. (D) MVA or NYVAC infection of primary human macrophages causes rRNA breakdown. Total rRNA was isolated from uninfected (mock) or poxvirus-infected macrophages (5 PFU/cell). At indicated times p.i., 2 μg of total RNA was separated by electrophoresis and subsequently stained with ethidium bromide. RNAs were also analyzed using an Agilent 2100 Bioanalyzer. RNA integrity numbers are listed under each corresponding sample.
Western blotting.
A total of 1 × 106 macrophages were infected with MVA, ANK, WR, NYBH, NYVAC, or CopV at an MOI of 5 PFU/cell, and at 2, 6, and 16 h p.i., cells were collected and lysed in lysis buffer (50 mM Tris-HCl, pH 8.0, 0.5 M NaCl, 10% NP-40, 1% SDS) for 5 min on ice. Protein lysates (100 μg) were fractionated by 14% or 8% SDS-PAGE, transferred to nitrocellulose membranes, and incubated with anti-poly(ADP-ribose) polymerase (PARP; Cell Signaling, Boston, MA), anti-actin (Santa Cruz, Santa Cruz, CA), anti-E3L (kindly provide by the Beltram Jacobs lab), anti-B5R (Bei Resources), anti-A27L (Bei Resources), anti-phospho-eIF-2α (eIF-2α-P; Biosource, Camarillo, CA), or anti-eIF-2α (Santa Cruz) antibody, followed by secondary antibodies (mouse and rabbit peroxidase conjugates). Protein expression was detected using enhanced chemiluminescence (ECL) reagents (Amersham, Uppsala, Sweden).
Microarray labeling.
Ultraspect-II RNA (Biotecx, Houston, TX) was used to isolate total RNA from primary human macrophages infected with MVA, ANK, WR, NYBH, NYVAC, or CopV (3 × 106 cells/time p.i.; 5 PFU/cell) or mock infected (mock). RNA was then purified with Megaclear (Ambion, Foster City, CA), and integrity was confirmed using an Agilent 2100 Bioanalyzer (Agilent, Santa Clara, CA). Total RNA (1.5 μg) was amplified with an Amino Allyl MessageAmp aRNA kit (Ambion); 54 to 88 μg of amplified RNA (aRNA) was obtained. The mean RNA size was 1,500 nucleotides, as observed using an Agilent 2100 Bioanalyzer. For each sample, 6 μg of aRNA was labeled with Cy3 or Cy5 mono-NHS (N-hydroxysuccinimide) ester (CyDye Postlabeling Reactive Dye Pack; GE Healthcare) and purified using Megaclear. Cy5 and Cy3 incorporation was measured using 1 μl of probe in a Nanodrop spectrophotometer (Nanodrop Technologies). For each hybridization, Cy5 and Cy3 probes (150 mol each) were mixed and dried by speed vacuum and resuspended in 9 μl of RNase-free water. Labeled aRNA was fragmented by adding 1 μl of 10× fragmentation buffer (Ambion), followed by incubation at 70°C for 15 min. The reaction was terminated with 1 μl of stop solution (Ambion). Two dye-swapped hybridizations were performed for each comparison: in one, the mock-infected sample was Cy3 labeled, and the MVA-infected sample was Cy5 labeled; in the second, labeling was reversed. Double labeling was used to abolish dye-specific labeling and hybridization differences.
Slide treatment and hybridization.
Slides containing 22,264 spots (19,256 different oligonucleotides) corresponding to the Human Genome Oligo set, version 2.2 (Qiagen, Hilden, Germany), were obtained from the Genomic and Microarrays Laboratory (Cincinnati University, OH). Information about printing and the oligonucleotide set can be found at http://microarray.uc.edu. Slides were prehybridized and hybridized as described previously (24–27). Images from Cy3 and Cy5 channels were equilibrated and captured with an Axon 4000B scanner, and spots were quantified using GenePix, version 5.1, software. Data for replicates were analyzed using Almazen software (Bioalma, Spain). In general, lowest normalization was applied to each replicate, and the log ratios were merged with the corresponding standard deviation and z-score.
Gene expression analysis.
The original data set contained 19,256 oligonucleotides per slide. In each analysis, genes with an interreplicate mean signal of <100 or an interreplicate standard deviation of >1 were filtered out. Genes were considered to be differently expressed if the fold change in expression was <−2 (downregulated) or >2 (upregulated). Functional analyses of regulated genes were generated by Ingenuity Pathways Analysis (Ingenuity Systems, Redwood City, CA). Hierarchical clustering was carried out using the software SpotFire Decision Site for Functional Genomics. Ward's method with the average value ordering function and half squared Euclidean distance function was used.
Latex bead phagocytosis.
Human macrophages were seeded in eight-well μ-Slide microscopy chambers (Ibidi, Germany) and filmed after incubation with 1-μm-diameter latex beads conjugated to green fluorescent protein (GFP) (Sigma) at a ratio of 10 beads per cell. Alternatively, cells were cultured on coverslips and incubated for 1 h with the beads (20 or 10 beads per cell, depending on the experiment), washed three times with 1× PBS, and incubated with DMEM for an additional hour as described previously (28). Cells were fixed with 4% paraformaldehyde (PFA) and processed for immunofluorescence analysis. Briefly, cells were washed with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde, and permeabilized with 0.1% Triton X-100 in PBS at room temperature for 10 min, and DNA was stained with ToPro 3 (Life Technologies). Images were obtained using a Bio-Rad Radiance 2100 confocal laser microscope.
Determination of caspase 3/7 activation.
Apoptosis quantification was carried out using the Caspase-Glo 3/7 assay kit (Promega) according to the manufacturer's protocol. Briefly, human macrophage monolayers, grown in 96-well plates, were infected at an MOI of 5 PFU/cell, and at the indicated times p.i., 100 μl of Caspase-Glo 3/7 reagent was added to the wells. Plates were gently shaken and then incubated in the dark at 20°C for 60 min before luciferase activity was recorded using an Orion microplate luminometer (Berthold Technologies).
Quantitative real-time RT-PCR.
Total RNA was isolated from macrophages uninfected or infected with MVA, ANK, WR, NYBH, NYVAC, or CopV (5 PFU/cell) with Ultraspect-II RNA reagent (Biotecx), according to the manufacturer's instructions. One microgram of RNA was reverse transcribed using SuperScript (Invitrogen) with oligo(dT) as a primer. A 1:40 dilution of the reverse transcription (RT) reaction mixture was used for quantitative PCR (qRT-PCR). Primers and probe sets used to amplify tumor necrosis factor alpha (TNF-α; probe Hs00174128_m1), IFN-stimulated gene 15 (ISG15; Hs00192713_m1), interleukin-7 (IL-7; Hs00978525_m1), IFN-α (Hs01652729_s1), and NF-κ2 (Hs0128892_g1) were purchased from Applied Biosystems. RT-PCRs were performed according to Assay-on-Demand, optimized for TaqMan Universal PCR MasterMix, no AmpErase UNG, as described previously (26). All samples were assayed in triplicate. Threshold cycle (CT) values were used to plot a standard curve in which the CT value decreased in linear proportion to the log of the template copy number. The correlation values of standard curves were always >99%.
VSV antiviral assay.
Vero cells left either untreated or pretreated with either IFN-α (1,000 U ml−1 for 16 h) or with the supernatant obtained from MVA-infected macrophages from three independent donors were infected with vesicular stomatitis virus (VSV; 0.1 PFU/cell). VSV titers were quantified by plaque assay on Vero cells as previously described (29).
RESULTS
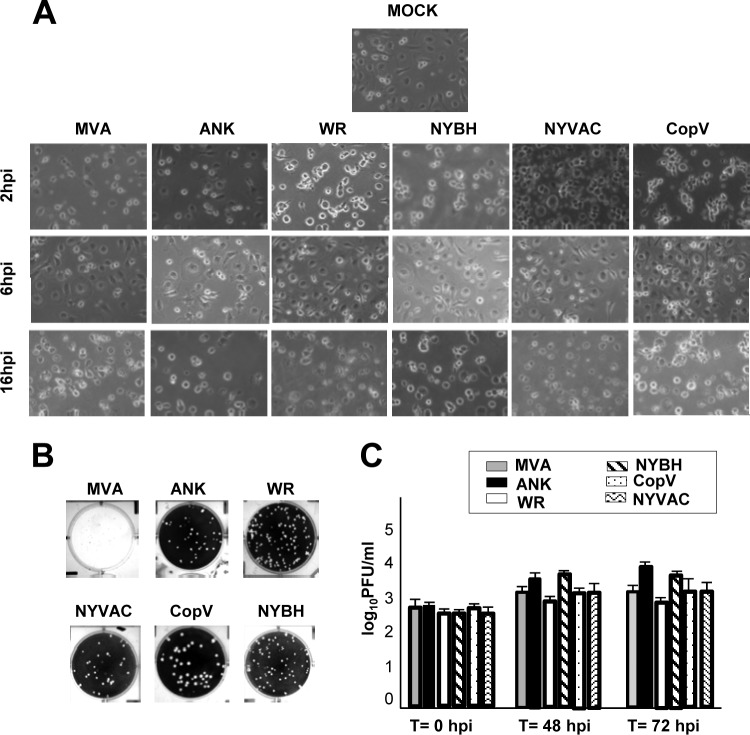
Attenuated and nonattenuated poxvirus vectors induce CPE in primary human macrophages independent of viral replication.
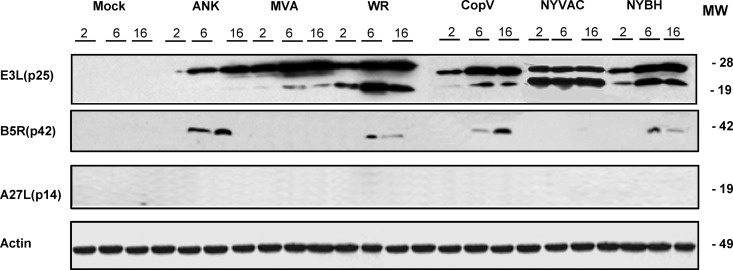
Due to the facts that (i) macrophages are one of the first immune cells to encounter viral pathogens and (ii) these professional APCs are critically important in activating the immune system, it is important to study the biology of viral infections in these cells in order to understand how macrophages might participate in mediating an effective immune response against viruses such as poxviruses. To this end, we characterized the phenotypic, intracellular, and molecular changes in macrophages following infection with the attenuated vaccine vectors MVA and NYVAC and the nonattenuated wild-type strains ANK and CopV as well as WR and its derivative strain NYBH. We first defined the induced cytopathic effect (CPE) in macrophages at 2, 6, and 16 h postinfection (p.i.) with all six virus strains. Peripheral blood mononuclear cells (PBMCs) were isolated from buffy coats, and macrophages were enriched by growth under adherent conditions. The purity of monocyte cultures was confirmed by CD14 labeling and flow cytometry analysis (average, 89% of CD14-positive cells) (data not shown). Primary macrophages were infected with all six VACVs at an MOI of 5 PFU/cell. As early as 2 h p.i., we observed in all cases various degrees of CPE, characterized by changes in cell morphology, including cell rounding and cytoplasmic contraction (Fig. 1A), with more severe effects noted at 16 h p.i. To evaluate whether the differences in CPE were due to variations in viral production, we determined the intracellular titers of infectious virus at 48 and 72 h following infection of human macrophages with all six VACV strains at an MOI of 0.1 PFU/cell. Titers were determined using a standard plaque-forming assay for all VACVs tested except MVA, for which the titers were determined by immunohistochemical staining as previously described (24) (Fig. 1B). Quantification of our titer results clearly demonstrated that poxvirus infection in human macrophages is abortive as none of the viral strains was able to produce de novo infectious virions (Fig. 1C). To analyze which step in the viral cycle was blocked, we examined viral protein synthesis in infected macrophages by Western blotting using antibodies specific for early p25 (E3L) and late p42 (B5R) and p14 (A27L) viral proteins (Fig. 2). The early p25 (E3L) protein was detected in lysates from macrophages infected with all of the viruses, indicating that all VACV strains could synthesize early viral proteins regardless of their attenuation state. In contrast, the late protein encoded by the B5R gene was detectable only in lysates from macrophage infected with the nonattenuated strains ANK, WR, CopV, and NYBH, and the late protein p14 (A27L) was not detected in any of the lysates examined (Fig. 2). Importantly, these results were validated by immunofluorescence analysis (data not shown) and differ from previous results obtained in DCs infected with MVA, in which the late proteins encoded by the A17L and A27L genes were detected in infected DC lysates (30).
FIG 1.
Cellular changes in primary human macrophages following infection with different poxvirus vectors. (A) Morphological changes in human macrophages mock infected or infected with MVA, ANK, WR, NYBH, NYVAC, or CopV (5 PFU/cell) in six-well plates. Changes were examined by phase-contrast microscopy at 2, 6, and 16 h p.i. (hpi). For each group of infections, representative fields are shown at a magnification of ×40. (B) Titration of each viral strain. MVA titers were determined by immunostaining using BHK-21 cells; ANK, WR, NYBH, NYVAC, and CopV titers were determined by plaque assay on BSC40 cells. The images show differential plaque sizes produced by the different poxvirus-derived vectors. (C) VACV growth in infected human macrophages. Cells were infected with MVA, ANK, WR, NYBH, NYVAC, or CopV (0.1 PFU/cell), and at the times indicated, cells were harvested, and virus yields were determined by plaque assay for each virus except MVA, for which the titer was determined by immunostaining. Results represent the means ± the standard deviations of three independent experiments. P values from a two-tailed t test assuming nonequal variance were determined. In all the cases, P is <0.01. T, time.
FIG 2.
Viral protein expression during MVA, ANK, WR, NYBH, NYVAC, or CopV infection. Primary human macrophages were mock infected or infected with the indicated poxviruses (5 PFU/cell), and at the indicated times p.i., equal amounts of proteins from cell extracts were fractionated by SDS-PAGE, transferred to nitrocellulose membranes, and treated with antibodies to specific virus early (p25) and late proteins (p64 and p14). Molecular weight (MW; in thousands) is indicated based on protein standards. In all blots, detection of actin was used as a loading control.
Attenuated and nonattenuated poxvirus vectors differentially induce protein translation shutoff and apoptosis in primary human macrophages.
We next examined the effect of poxvirus infection on protein translation. In line with previous observations (24), we observed total protein translational shutoff following infection of macrophages with VACV. Specifically, macrophages were metabolically labeled with [35S]methionine-cysteine at 2, 6, and 16 h p.i. with all six VACV strains, and protein lysates were analyzed by SDS-PAGE and autoradiography. We observed a severe translational block in protein synthesis by 16 h p.i. in cells infected with all viruses (Fig. 3A); however, the effect was earlier (∼6 h p.i.) and more pronounced following infection with MVA and NYVAC. While this blockade in protein translation coincided with phosphorylation of eIF-2α (Fig. 3B) under all conditions tested, the levels of phospho-eIF-2α were increased in macrophages following infection with MVA or NYVAC (Fig. 3B).
Apoptosis is a very important innate immune response against viral infections and is generally considered a self-defense mechanism (17) as loss of infected cells should impair virus propagation and further spread. Moreover, phagocytosis of apoptotic bodies by APCs is an important mechanism for cross-presentation as foreign antigens gain access to major histocompatibility complex class I (MHC-I) molecules for priming of cytolytic T cells (31). Viruses, however, resist this host defense mechanism by inhibiting apoptosis via antiapoptotic viral proteins (32). Previously, we reported that the attenuated poxviruses MVA and NYVAC induce apoptosis in HeLa (27) and DCs (30); thus, DCs infected with these viruses could putatively initiate an immune response through cross-priming (i.e., with T cells) (33). To analyze whether apoptosis also occurs in poxvirus-infected macrophages, we used an antibody that recognizes both full-length and cleaved poly(ADP-ribose) polymerase-1 (PARP-1) to assess apoptosis in human macrophages following infection with all six poxvirus strains. In MVA-, NYVAC-, CopV-, and WR-infected macrophages, the 89-kDa cleaved PARP-1 protein was evident at 16 h p.i. (Fig. 3B), indicating that these four poxviruses are capable of inducing apoptosis in macrophages. In order to better appreciate and measure the level of apoptosis activation in these cells, a Caspase-Glo 3/7 assay kit was used as per the manufacturer's instructions. As shown in Fig. 3C, MVA, CopV, and NYVAC markedly induced apoptosis in comparison to ANK, WR, and NYBH, where little to no caspase 3/7 activation was detected. Along these lines, we also examined rRNA integrity in poxvirus-infected macrophages and observed pronounced RNA degradation in macrophages infected with MVA and NYVAC at 16 h p.i. (Fig. 3D). Together, these results reveal that of the poxvirus vectors tested, MVA is the most potent inducer of apoptosis, while NYVAC and CopV can also induce apoptosis to different degrees and likely via different mechanisms.
Differential gene regulation in macrophages infected with attenuated and nonattenuated poxvirus vectors.
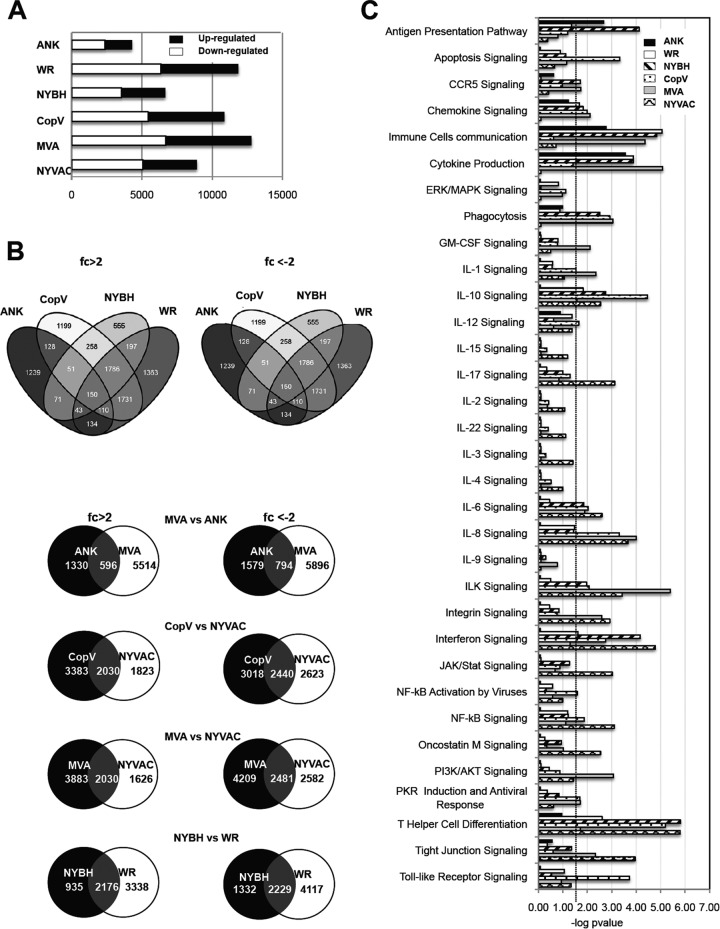
To study the cellular and molecular changes mediated by each poxvirus vector in more detail, microarray analyses were performed. Since the majority of the cellular changes we observed (Fig. 1 and 2) occurred by 6 h p.i. and before the onset of rRNA degradation (Fig. 3D), cDNA from 6-h mock-infected or 6-h poxvirus-infected macrophages was hybridized to SurePrint G3 Human Gene Expression Microarrays (Agilent), and results were analyzed as described previously (34). Using this approach, we identified genes up- and downregulated in macrophages infected with each poxvirus vector (see Table S1 in the supplemental material) and subsequently determined pathways that were similarly modulated by the majority of the poxvirus strains tested and some that were distinctly altered using the Ingenuity Pathways Analysis software (Ingenuity Systems, Redwood City, CA).
Of the six poxvirus vectors tested, MVA modulated the largest number of genes, while ANK modulated the least number of genes (Fig. 4A). When the four nonattenuated vectors were compared, we observed that while WR, NYBH, and CopV modulated similar genes, the genes modulated by ANK infection were distinct and different. Furthermore, when we compared the nonattenuated poxvirus vectors ANK and CopV to their respective attenuated derivatives MVA and NYVAC, respectively, we observed that of the 6,110 genes upregulated and 6,690 genes downregulated by MVA, only 596 (∼9%) and 794 (∼12%) genes were also modulated by ANK, respectively, while NYVAC and CopV and even MVA and NYVAC modulated many of the same genes (>35%) (Fig. 4B). Thus, excluding ANK, the infection-induced gene expression changes in WR-, NYBH-, CopV-, MVA-, and NYVAC-infected cells were largely similar. In the case of WR infection, the gene-specific changes observed in this study are quite different from those previously observed in HeLa cells (26).
FIG 4.
Number of host genes and pathway analysis of genes differentially regulated by poxvirus infection. (A) Shown are the numbers of cellular genes that exhibited expression changes in our microarray analyses larger than 2-fold or less than 2-fold at 6 h after infection with each virus. (B) Intersections of gene upregulation (fold change [fc] of >2) or downregulation (fold change of <2) in macrophages infected with the indicated viruses are shown in the Venn diagrams. (C) Ingenuity Pathway analysis showing selected canonical pathways significantly modulated in macrophages infected with the indicated poxviruses compared to naive macrophage controls (P < 0.05). The dashed line indicates the threshold set at 1.5 log (P value). ERK, extracellular signal-regulated kinase; MAPK, mitogen-activated protein kinase; GM-CSF, granulocyte-macrophage colony-stimulating factor.
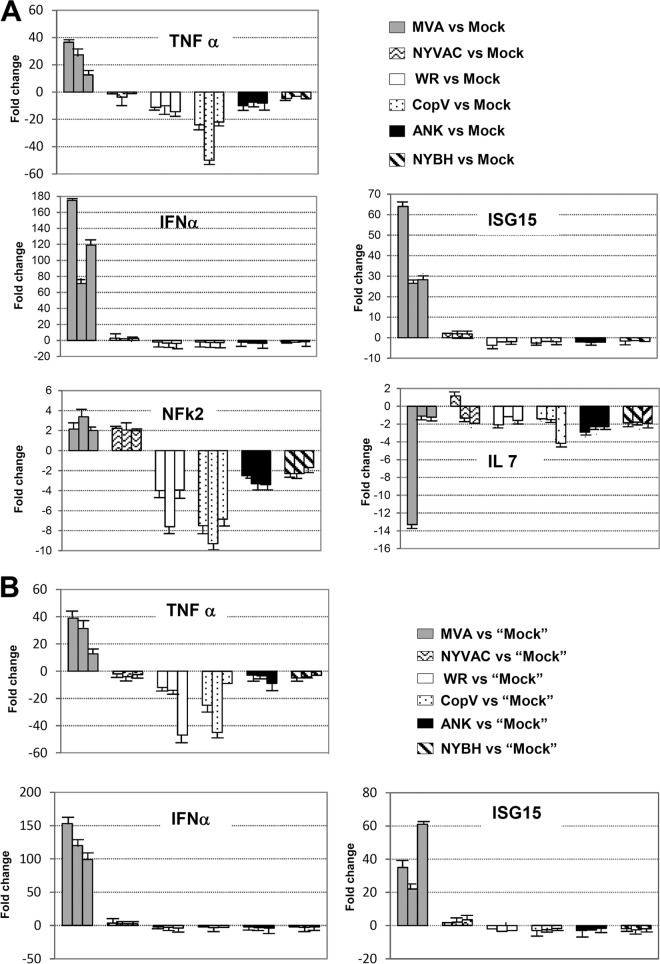
As the goal of our study was to identify poxvirus vectors that modulate immune signaling pathways in a way that would support activation of an antiviral immune response, we next assessed the pathways modulated by each poxvirus, focusing on genes and pathways involved in IFN, Toll-like receptor (TLR), and interleukin signaling, as well as apoptosis and NF-κB and phosphatidylinositol 3-kinase (PI3K)/AKT signaling. Compared to the nonattenuated poxvirus strains, probably due to the absence of certain viral genes, infection with MVA and NYVAC was associated with the modulation of a larger number of pathways and genes involved in immune signaling and activation (Fig. 4C), such as IFN and interleukin signaling as well as NF-κB- and PI3K/AKT-mediated pathways (Table 1; see also Fig. S1 in the supplemental material). At the gene level, beta 1 interferon (IFNβ1), tumor and proinflammatory cytokines such as TNF-α and IL-29, chemokine receptors CXCR5 and CXCR6, and antiviral response genes such as CD70 and interferon-induced transmembrane protein 1 (IFITM1) were uniquely upregulated or significantly higher in MVA- and NYVAC-infected macrophages. Interestingly, while IL-29 was upregulated, other interleukins such as IL-1, IL-7, and IL-6 were strongly downregulated in all samples (Fig. 5A; see also Fig. S1), which interestingly differs from our previous studies in DCs where IL-6 was upregulated (30).
TABLE 1.
Representative genes regulated by poxviruses in infected macrophages involved in immune responsea
| Pathway and gene symbol | Descriptionb | Fold change in expressionc |
|||||
|---|---|---|---|---|---|---|---|
| MVA | NYVAC | WR | NYBH | CopV | ANK | ||
| Apoptosis inhibitors | |||||||
| BCL2 | B-cell CLL/lymphoma 2 | −9.000 | −8.786 | −4.828 | −4.931 | −7.981 | NS |
| BCL2A1 | BCL2-related protein A1 | −4.175 | −2.544 | −4.471 | −3.388 | −4.715 | NS |
| BCL2L1 | BCL2-like 1 | −3.799 | −3.966 | −2.121 | −3.740 | −2.608 | NS |
| MCL1 | Myeloid cell leukemia sequence 1 (BCL2-related) | 2.782 | −2.041 | NS | NS | NS | NS |
| NAIP | NLR family, apoptosis-inhibitory protein | −2.113 | 2.092 | NS | 2.308 | 4.057 | NS |
| Apoptosis activators | |||||||
| FASLG | Fas ligand (TNF superfamily, member 6) | 39.393 | −10.768 | NS | 2.364 | −6.786 | 3.856 |
| BAK1 | BCL2-antagonist/killer 1 | −2.866 | −2.866 | NS | NS | NS | NS |
| CASP3 | Caspase 3, apoptosis-related cysteine peptidase | 2.285 | NS | NS | 2.341 | NS | NS |
| CASP8 | Caspase 8, apoptosis-related cysteine peptidase | 2.331 | 2.141 | NS | NS | NS | NS |
| CASP10 | Caspase 10, apoptosis-related cysteine peptidase | 2.887 | NS | NS | NS | NS | NS |
| DIABLO | Diablo, IAP-binding mitochondrial protein | 2.691 | NS | 3.967 | 2.251 | 3.654 | NS |
| IFN signaling | |||||||
| IFNB1 | Interferon beta 1, fibroblast | 403.148 | 2.369 | NS | NS | NS | NS |
| IFNG | Interferon gamma | 23.457 | −5.430 | −3.656 | −7.697 | −10.565 | NS |
| MX1 | Myxovirus (influenza virus) resistance 1 | −3.426 | −13.306 | −7.458 | −8.109 | −6.898 | NS |
| OAS1 | 2′-5′-Oligoadenylate synthetase 1, 40/46 kDa | −3.932 | −6.136 | −11.467 | −3.997 | −7.923 | NS |
| PIAS1 | Protein inhibitor of activated STAT, 1 | 3.446 | 2.159 | 2.748 | 2.828 | 4.030 | NS |
| TLR signaling | |||||||
| MYD88 | Myeloid differentiation primary response 88 | −4.343 | −10.603 | −3.042 | −4.086 | −2.476 | NS |
| TLR4 | Toll-like receptor 4 | −6.389 | −4.153 | −5.010 | −2.736 | −3.323 | NS |
| TLR7 | Toll-like receptor 7 | −11.177 | −5.374 | −12.849 | −4.313 | −25.369 | NS |
| NF-κB signaling | |||||||
| MAPK8 | Mitogen-activated protein kinase 8 | 2.073 | 2.314 | NS | NS | NS | NS |
| NFKBIA | Nuclear factor of kappa light polypeptide gene enhancer in B cells inhibitor, alpha | 4.035 | 2.579 | NS | NS | NS | NS |
| TNF | Tumor necrosis factor | 2.560 | −4.075 | −7.124 | −7.025 | −12.289 | NS |
| TAB3 | MAP3K7 binding protein 3 | 5.469 | 6.867 | 7.504 | 2.340 | 6.524 | NS |
| PI3K-AKT signaling | |||||||
| EIF4 | Eukaryotic translation initiation factor 4E | −2.350 | 2.114 | NS | NS | NS | NS |
| IKBKB | Inhibitor of kappa light polypeptide gene enhancer in B cells, kinase beta | 2.061 | NS | 4.732 | NS | 3.521 | NS |
| ITG3A | Integrin, alpha 3 | −5.736 | −4.779 | −5.489 | −2.574 | −2.741 | 2.224 |
| PDPK1 | 3-Phosphoinositide dependent protein kinase 1 | −3.446 | −2.185 | NS | NS | NS | NS |
Comparison of gene expression profiling for representative human genes specifically regulated by each vector according to predicted biological function.
CLL, chronic lymphocytic leukemia; NLR, NOD-like receptor; IAP, inhibitor of apoptosis.
NS, no significant modulation in gene expression.
FIG 5.
Validation of microarray data at the mRNA level. (A) RT-PCR analysis of the relative expression levels of immune response genes. Macrophages from three donors were mock, MVA, NYVAC, WR, CopV, ANK, or NYBH infected and at 6 h p.i. were processed for RT-PCR analysis. The name of each gene product obtained by RT-PCR analysis is indicated. Three independent experiments were carried out, and a representative experiment is shown. Error bars indicate the standard deviations of the means. (B). Macrophages from three donors were MVA, NYVAC, WR, CopV, ANK, or NYBH infected. Mock inoculum is the uninfected cellular inoculum purified using the same protocol for the purification of vaccinia virus. The purified mock inoculum from cultures of BHK-21 cells was used for comparisons with MVA and NYVAC infections, and the purified mock inoculum from BSC40 cells was used for comparisons with the other infections (WR, CopV, ANK, and NYBH). The name of each gene product, microarray data values at 6 h p.i., and values obtained by RT-PCR analysis are indicated. Three independent experiments were carried out, and a representative experiment is shown. Error bars indicate the standard deviations of the means.
Importantly we validated many of the genes shown in Table 1 by real-time RT-PCR (Fig. 5A). In addition, to discard the possibility that cellular debris present in the virus stock could account for the observed changes in gene expression in macrophages following infection, mock inoculum was purified from uninfected cells using the same protocol as for the purification of vaccinia viruses. Mock inocula prepared from cultures of BHK-21 cells were used as controls for MVA and NYVAC infections, and mock inocula from BSC40 cells were used as controls for the other infections. Infections of human macrophages with all the virus strains and their respective mock controls were then carried out, and the expression levels of a select group of genes (IFN-β, TNF-α, and ISG15) were analyzed by real-time RT-PCR. Compared to mock-infected cells, gene expression patterns similar to our previous results (Fig. 5A) were observed (Fig. 5B), indicating that the changes in the expression levels of genes postinfection are due to intrinsic differences among the virus strains and not to the presence of contaminants in the viral preparations.
Biological effects of IFN pathway activation in MVA-infected macrophages.
Our microarray experiments indicated that infection of macrophages with the attenuated VACV strain MVA resulted in a significant increase in the expression of a number of cytokines (Table 1). For example, MVA elicited high induction of IFN-β, an important protein involved in the innate antiviral immune response. To validate this result in vitro, we functionally measured the presence of IFN-β in the supernatant of MVA-infected macrophages using a vesicular stomatitis virus (VSV)-based antiviral assay. Specifically, VSV-infected Vero cells were incubated with the supernatants of MVA-infected macrophages from three donors, and at 48 h posttreatment, VSV titers were determined. A clear inhibition of VSV infection in vitro was observed after incubation with medium from MVA-infected macrophages (Fig. 6). These results clearly demonstrate that the increased mRNA expression of type I IFN observed in MVA-infected macrophages corresponds to increased translation and release of this antiviral protein.
FIG 6.
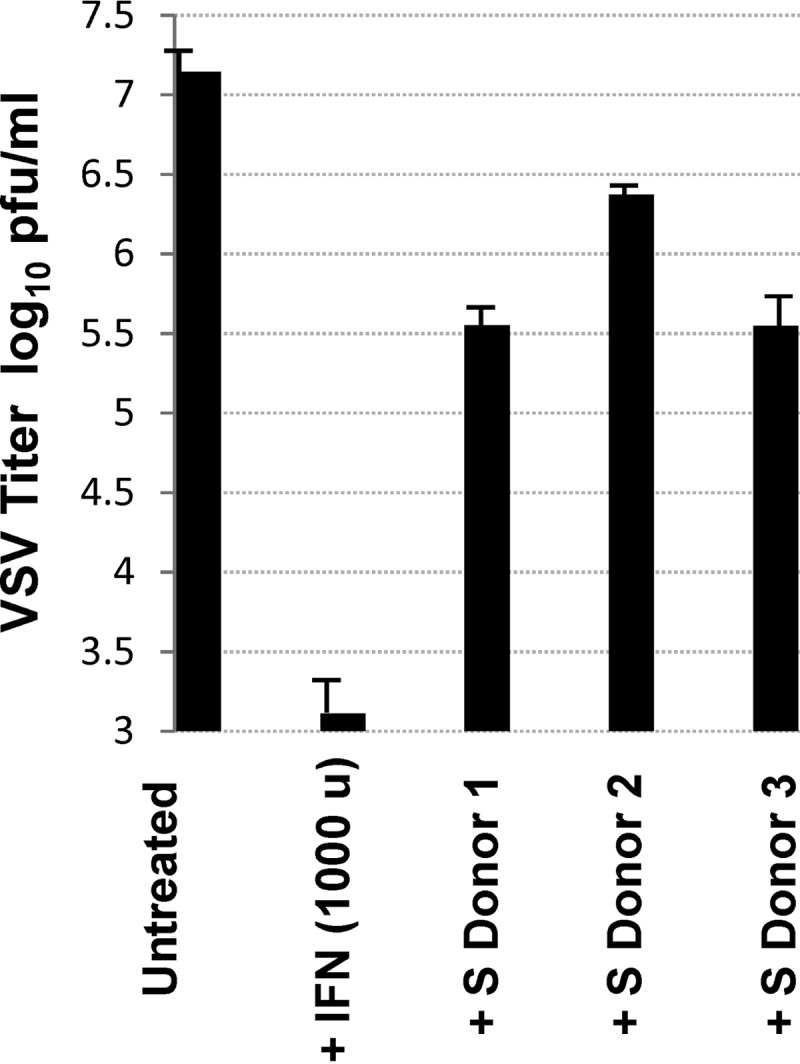
Antiviral bioassays confirm that MVA-infected macrophages secreted high levels of biologically active type I IFN. Plaque assay analysis of VSV-infected Vero cells left either untreated or pretreated with either IFN-α (1,000 U ml−1 for 16 h) or with the supernatant (S) obtained from MVA-infected macrophages from three independent donors. Results represent the means ± the standard deviations of three independent experiments.
A major function of macrophages is the phagocytosis of pathogens, antigens, and infected or apoptotic cells, which is critical for innate as well as for adaptive immunity. Recently, we reported that the phagocytic capacity of macrophages is highly regulated by IFN (35). Latex beads are a common substrate used in biochemical studies to study phagocytosis in macrophages (28). Thus, to determine whether the IFN produced in MVA-infected macrophages is critical for the phagocytic capacity of macrophages, we analyzed the intake of GFP-labeled latex beads by confocal and time-lapse microscopy in naive macrophages mock treated or treated with supernatants from MVA-infected macrophages from three donors. Time-lapse microscopy analyses showed a marked increase in the phagocytosis of latex beads in macrophages treated with supernatants from MVA-infected macrophages compared to levels in untreated controls (Fig. 7 A). Quantification of these results revealed that the phagocytic capacity of treated macrophages was ∼100 times higher than that observed in untreated cultures (Fig. 7B). Taken together, these results strongly suggest that the IFN produced from MVA-infected macrophages may have an important immunogenic role, such as decreasing viral replication and increasing the phagocytic capacity of APCs, two vital processes necessary for the development of an effective antiviral immune response.
FIG 7.
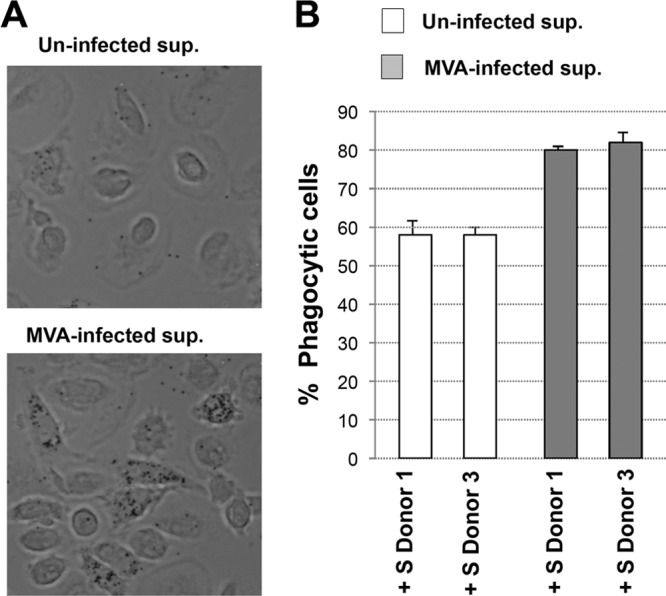
MVA infection increases the phagocytosis of GFP-labeled latex beads in human macrophages. (A) Phagocytosis of GFP-latex beads by human macrophages. Cells were seeded in eight-well tissue culture plates and treated with supernatant (sup) from mock-infected (Uninfected) or MVA-infected macrophages for 16 h. After that, the cells were incubated with 1-μm-diameter latex beads conjugated to GFP in a ratio of 10 latex beads per cell. Phagocytized beads and cells were visualized 2 h after latex bead incubation by phase-contrast microscopy. Representative fields are shown at a magnification of ×40. (B) Cells having phagocytized GFP-labeled latex beads were quantified by immunofluorescence microscopy. Representative phase-contrast images are shown. The graph shows the quantification of phagocytic cells in macrophage cultures treated with supernatant (sup) from mock-infected (Uninfected) or MVA-infected macrophages. Results represent the means ± the standard deviations of three independent experiments. P values from a two-tailed t test assuming nonequal variance were determined. In all cases, P is <0.05.
DISCUSSION
Prior to the development of an effective adaptive immune response, the innate immune system provides the first line of immunological defense against a wide range of microorganisms, including bacteria and viruses (36). Macrophages and dendritic cells (DCs) are specialized phagocytes that play an important role in innate immunity and in the clearance of virus-infected cells. These professional APCs are widely dispersed throughout the body and are located in main portals of entry of microbes, such as the skin and mucosal epithelia. They participate in the initial capture and processing of potential antigens (innate immunity) and then in the activation of specific T and B lymphocyte effector mechanisms (adaptive immunity), resulting in immunological memory. A comprehensive understanding of how immunity is generated, the role of APCs in this process, and how the efficiency of this process could be enhanced is essential for vaccine development.
Poxvirus vectors are efficient activators of host immune responses to virally expressed antigens of distinct origin and therefore are being used as potential vaccines for several pathogens and tumors (6, 17, 37). In the present study, we have characterized the infection of macrophages with different poxvirus vectors to gain a better understanding of how these vectors activate specific immune responses in these APCs. We determined that poxvirus infection in human macrophages is abortive as none of the viral strains was able to produce de novo infectious virus; however, we observed that macrophages infected with all the poxvirus strains underwent extensive morphological damage at late times postinfection, which, at the intracellular level, was characterized by severe inhibition of protein synthesis by 6 h p.i. and the induction of apoptosis and rRNA degradation at 16 h p.i. In spite of a general translational block induced by these viruses, some of the early (p25) and late (p42, p14) viral proteins examined were produced in infected cultures although differences were observed between the attenuated and nonattenuated vectors (Fig. 2). For example, in contrast to the nonattenuated poxvirus strains analyzed, MVA- and NYVAC-infected macrophages did not produce the late viral protein p42 encoded by the viral gene B5R. This clear difference might be related to the degree and extent of eIF-2α phosphorylation and RNA degradation induced during infection with these two attenuated strains as well as the presence or absence of certain viral genes in the poxvirus virions. Perhaps for MVA and NYVAC, the absence of B5R translation (Fig. 2) may be due to early eIF-2α phosphorylation and subsequent inactivation of the cellular translational machinery, while in macrophages infected with the nonattenuated poxviruses, the delayed phosphorylation of eIF-2α may have allowed for the translation of the protein encoded by the B5R gene. Deciphering the putative viral genetic elements that are responsible for these differences may prove potentially useful for developing vaccinia vectors that require early protein translational shutoff.
We next performed genomic studies to dissect the impact of infection of human macrophages with each poxvirus strain. Having observed profound morphological and intracellular effects (e.g., apoptosis and rRNA degradation) in human macrophages at a late time postinfection (i.e., 16 h p.i.), we performed gene expression profiling analyses in primary human macrophages at 6 h postinoculation with each vaccinia virus vector. Our gene profiling analyses clearly demonstrated substantial biological differences in the expression patterns of cellular genes between macrophages infected with different poxvirus strains, and the observed changes were specifically due to infection with each distinct viral vector (Fig. 4 and Table 1; see also Table S1 and Fig. S1 in the supplemental material).
While genes involved in hundreds of pathways were similarly and differentially modulated by each poxvirus, we were specifically interested in determining if genes involved in innate immune responses were differentially regulated based on the vaccinia virus vector utilized. For example, proinflammatory cytokines such TNF-α and type I IFN and TLR signaling represent first-line defenses against viral infections by generating an intracellular environment that restricts viral replication (38–40). Type I IFN signaling is considered the most effective antiviral immune response mechanism of the innate immune system. IFN gene expression is regulated by interferon regulatory factor 3 (IRF-3) phosphorylation, homodimerization, and nuclear translocation (41), leading to the transcriptional activation of IRF-responsive genes, such as IL-29 and IFNβ1. Our microarray experiments showed clear upregulation of IFNβ1 levels after MVA and NYVAC infection (403- and 2.4-fold, respectively) compared with WR, NYBH, CopV, and ANK infection, where no change in IFNβ1 gene expression was detected (Table 1). Likewise, we also observed a significant upregulation of IL-29 (∼43-fold) but only in MVA-infected macrophages. This apparent and specific activation of type I IFN by MVA was validated at the gene level by quantitative RT-PCR (qRT-PCR) analyses and at the protein level using an antiviral assay to assess IFN-mediated inhibition of VSV, as previously described (42) (Fig. 5A and B and 6A). We also assessed the expression of other IFN-related genes, such ISG15, which we have previously shown to be an important antiviral player in VAVC infections and macrophage phagocytosis (35). We have previously reported that type I IFN treatment increases latex bead uptake in macrophages (35); therefore, to determine if the IFN signaling stimulation produced after MVA infection could also potentiate the phagocytic potential of naive macrophages, we treated primary cultures of macrophages with supernatant from uninfected or MVA-infected macrophages and performed a phagocytosis assay using GFP-labeled latex beads. Time-lapse microscopy images indicated that naive macrophages incubated with supernatants from MVA-infected macrophages increased their phagocytic capacity compared to macrophages incubated with control medium alone (data not shown). Quantification of these results revealed that after incubation with MVA-infected macrophage-conditioned supernatant, 80% of the macrophages showed clear phagocytosis, while the amount of phagocytosis observed in cells treated with supernatant from mock-infected cells was ∼58% (Fig. 7B). These results indicate that soluble factors released after MVA infection enhance the phagocytic activity of macrophages, and based on our previous studies, we postulate that this factor is likely type I IFN (35). Reinforcing the biological relevance of phagocytosis for innate as well as for adaptive immunity, the upregulation of this mechanism after MVA infection could be important in modulating the immune response, which in turn can influence the extent and quality of the host response and protective efficacy after vaccination.
Compared to IFN signaling, however, TLR signaling was not activated in VACV-infected macrophages. TLRs are membrane-bound receptors that recognize highly conserved molecular patterns among pathogens, such as viral double-stranded RNA (dsRNA), which is recognized by TLR3. Activation of these receptors leads to a cascade of intracellular events that result in the activation of the innate immune system (reviewed in reference 43). We observed that all of the poxvirus vectors tested downregulated genes involved in TLR signaling, such as Myd88 and TLRs (Table 1). This finding, however, was not surprising as VACV subverts many innate immune response mechanisms by producing viral proteins that counteract these pathways (44–47), and all of the vectors used encode the VACV protein A46R that blunts TLR signaling (48).
Finally, in full agreement with our in vitro apoptosis results, all of the poxvirus vectors downregulated antiapoptotic genes such as BCL2 and upregulated inducers of apoptosis, such as Fas ligand or caspase genes (Table 1). Apoptosis is very important for the efficacy of the innate immune response against pathogen infection and is generally considered a self-defense mechanism (17) as loss of host cell activity should impair virus propagation. For instance, apoptotic cells are engulfed and digested in lysosomes of phagocytes. This mode of phagocytic elimination of invading microbes represents an important innate immune mechanism. In contrast, viruses appear to resist this host action by inhibiting apoptosis using antiapoptotic proteins encoded by viral genes (32). Moreover, the phagocytosis of apoptotic bodies seems to be an important mechanism to cross-prime macrophages as extracellular antigens gain access to MHC-I molecules for priming of cytolytic T cells (31). Importantly, MVA and NYVAC modulated significantly more apoptosis inhibitors and inducers, again highlighting their enhanced potential to induce apoptosis via genetic and cellular processes. These results, together with our observations that infection with MVA and NYVAC led to the modulation of considerably more genes involved in immunomodulation, would suggest that these attenuated vectors are more potent activators of different components of the innate immune system. Specifically, MVA induced the greatest activation of ISG and type I IFN gene expression, which was also confirmed at the protein level using media from MVA-infected macrophages of different donors to inhibit VSV infection in vitro (Fig. 6) and induce phagocytosis in naive macrophages (Fig. 7). Likewise, MVA also induced the greatest amount of apoptosis in infected macrophages, which we hypothesize could putatively activate the immune system in vivo via cross-priming with T cells. It is important to note that while MVA can be genetically modified to reverse its inhibitory effects on apoptosis signaling and/or to modulate its ability to activate the innate immune system, as has been shown by other investigators (49), our study suggests that there is a correlation between stimulation of innate responses, such as IFN production or apoptosis, and immune stimulation by the currently available attenuated poxvirus vectors.
In summary, the data presented here, studying a large panel of different poxvirus vectors, should serve to advance our understating of these vectors at the cellular, immune, and molecular levels. Apart from having conducted an extensive study of the viral life cycle of nonattenuated and attenuated vaccinia virus vectors in primary human macrophages, we performed in-depth analyses of the transcriptional changes that occur within infected cells and discovered many immune-related genes and pathways that are differentially regulated by each poxvirus. Thus, we hope that the knowledge gained from these genetic studies will aid vaccine development groups in improving or developing new vaccinia virus-based vaccine vectors which can specifically activate key innate immune responses, such as type I IFN signaling, apoptosis, and phagocytosis.
Supplementary Material
ACKNOWLEDGMENTS
We thank Mariano Esteban and Antonio Alcamí for providing us with the different poxvirus strains and Beltram L. Jacobs for the generous gift of the rabbit E3 antibody. We also thank Sylvia Gutierrez for her help with the phagocytosis assays and Beatriz Martín and Victor Toledano for excellent technical assistance.
This work was supported by grants from the Spanish Ministry of Health, FIS2011-00127 (S.G.) and FISPI11/00350 (E.L.-C), and by the Universidad Autónoma de Madrid (UAM)-Banco de Santander (S.G.).
Footnotes
Published ahead of print 5 March 2014
S.G. dedicates this article to the memory of her father-in-law.
Supplemental material for this article may be found at http://dx.doi.org/10.1128/JVI.00468-14.
REFERENCES
- 1.Franchini G, Gurunathan S, Baglyos L, Plotkin S, Tartaglia J. 2004. Poxvirus-based vaccine candidates for HIV: two decades of experience with special emphasis on canarypox vectors. Expert Rev. Vaccines 3:S75–S88. 10.1586/14760584.3.4.S75 [DOI] [PubMed] [Google Scholar]
- 2.Crotty S, Felgner P, Davies H, Glidewell J, Villarreal L, Ahmed R. 2003. Cutting edge: long-term B cell memory in humans after smallpox vaccination. J. Immunol. 171:4969–4973 [DOI] [PubMed] [Google Scholar]
- 3.Antoine G, Scheiflinger F, Dorner F, Falkner FG. 1998. The complete genomic sequence of the modified vaccinia Ankara strain: comparison with other orthopoxviruses. Virology 244:365–396. 10.1006/viro.1998.9123 [DOI] [PubMed] [Google Scholar]
- 4.Gomez CE, Perdiguero B, Garcia-Arriaza J, Esteban M. 2013. Clinical applications of attenuated MVA poxvirus strain. Expert Rev. Vaccines 12:1395–1416. 10.1586/14760584.2013.845531 [DOI] [PubMed] [Google Scholar]
- 5.Meyer H, Sutter G, Mayr A. 1991. Mapping of deletions in the genome of the highly attenuated vaccinia virus MVA and their influence on virulence. J. Gen. Virol. 72:1031–1038. 10.1099/0022-1317-72-5-1031 [DOI] [PubMed] [Google Scholar]
- 6.Moss B. 1996. Genetically engineered poxviruses for recombinant gene expression, vaccination, and safety. Proc. Natl. Acad. Sci. U. S. A. 93:11341–11348. 10.1073/pnas.93.21.11341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schneider J, Gilbert SC, Blanchard TJ, Hanke T, Robson KJ, Hannan CM, Becker M, Sinden R, Smith GL, Hill AV. 1998. Enhanced immunogenicity for CD8+ T cell induction and complete protective efficacy of malaria DNA vaccination by boosting with modified vaccinia virus Ankara. Nat. Med. 4:397–402. 10.1038/nm0498-397 [DOI] [PubMed] [Google Scholar]
- 8.Cebere I, Dorrell L, McShane H, Simmons A, McCormack S, Schmidt C, Smith C, Brooks M, Roberts JE, Darwin SC, Fast PE, Conlon C, Rowland-Jones S, McMichael AJ, Hanke T. 2006. Phase I clinical trial safety of DNA- and modified virus Ankara-vectored human immunodeficiency virus type 1 (HIV-1) vaccines administered alone and in a prime-boost regime to healthy HIV-1-uninfected volunteers. Vaccine 24:417–425. 10.1016/j.vaccine.2005.08.041 [DOI] [PubMed] [Google Scholar]
- 9.Corona Gutierrez CM, Tinoco A, Navarro T, Contreras ML, Cortes RR, Calzado P, Reyes L, Posternak R, Morosoli G, Verde ML, Rosales R. 2004. Therapeutic vaccination with MVA E2 can eliminate precancerous lesions (CIN 1, CIN 2, and CIN 3) associated with infection by oncogenic human papillomavirus. Hum. Gene Ther. 15:421–431. 10.1089/10430340460745757 [DOI] [PubMed] [Google Scholar]
- 10.Gilbert SC, Moorthy VS, Andrews L, Pathan AA, McConkey SJ, Vuola JM, Keating SM, Berthoud T, Webster D, McShane H, Hill AV. 2006. Synergistic DNA-MVA prime-boost vaccination regimes for malaria and tuberculosis. Vaccine 24:4554–4561. 10.1016/j.vaccine.2005.08.048 [DOI] [PubMed] [Google Scholar]
- 11.Harrop R, Connolly N, Redchenko I, Valle J, Saunders M, Ryan MG, Myers KA, Drury N, Kingsman SM, Hawkins RE, Carroll MW. 2006. Vaccination of colorectal cancer patients with modified vaccinia Ankara delivering the tumor antigen 5T4 (TroVax) induces immune responses which correlate with disease control: a phase I/II trial. Clin. Cancer Res. 12:3416–3424. 10.1158/1078-0432.CCR-05-2732 [DOI] [PubMed] [Google Scholar]
- 12.Belyakov IM, Earl P, Dzutsev A, Kuznetsov VA, Lemon M, Wyatt LS, Snyder JT, Ahlers JD, Franchini G, Moss B, Berzofsky JA. 2003. Shared modes of protection against poxvirus infection by attenuated and conventional smallpox vaccine viruses. Proc. Natl. Acad. Sci. U. S. A. 100:9458–9463. 10.1073/pnas.1233578100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tartaglia J, Perkus ME, Taylor J, Norton EK, Audonnet JC, Cox WI, Davis SW, van der Hoeven J, Meignier B, Riviere M, Languet B, Paoletti E. 1992. NYVAC: a highly attenuated strain of vaccinia virus. Virology 188:217–232. 10.1016/0042-6822(92)90752-B [DOI] [PubMed] [Google Scholar]
- 14.Kanesa-thasan N, Smucny JJ, Hoke CH, Marks DH, Konishi E, Kurane I, Tang DB, Vaughn DW, Mason PW, Shope RE. 2000. Safety and immunogenicity of NYVAC-JEV and ALVAC-JEV attenuated recombinant Japanese encephalitis virus-poxvirus vaccines in vaccinia-nonimmune and vaccinia-immune humans. Vaccine 19:483–491. 10.1016/S0264-410X(00)00191-2 [DOI] [PubMed] [Google Scholar]
- 15.Myagkikh M, Alipanah S, Markham PD, Tartaglia J, Paoletti E, Gallo RC, Franchini G, Robert-Guroff M. 1996. Multiple immunizations with attenuated poxvirus HIV type 2 recombinants and subunit boosts required for protection of rhesus macaques. AIDS Res. Hum. Retroviruses 12:985–992. 10.1089/aid.1996.12.985 [DOI] [PubMed] [Google Scholar]
- 16.Sivanandham M, Shaw P, Bernik SF, Paoletti E, Wallack MK. 1998. Colon cancer cell vaccine prepared with replication-deficient vaccinia viruses encoding B7.1 and interleukin-2 induce antitumor response in syngeneic mice. Cancer Immunol. Immunother. 46:261–267. 10.1007/s002620050486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ockenhouse CF, Sun PF, Lanar DE, Wellde BT, Hall BT, Kester K, Stoute JA, Magill A, Krzych U, Farley L, Wirtz RA, Sadoff JC, Kaslow DC, Kumar S, Church LW, Crutcher JM, Wizel B, Hoffman S, Lalvani A, Hill AV, Tine JA, Guito KP, de Taisne C, Anders R, Horii T, Paoletti E, Ballou WR. 1998. Phase I/IIa safety, immunogenicity, and efficacy trial of NYVAC-Pf7, a pox-vectored, multiantigen, multistage vaccine candidate for Plasmodium falciparum malaria. J. Infect. Dis. 177:1664–1673. 10.1086/515331 [DOI] [PubMed] [Google Scholar]
- 18.Parker RF, Bronson LH, Green RH. 1941. Further studies of the infectious unit of vaccinia. J. Exp. Med. 74:263–281. 10.1084/jem.74.3.263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Buller RM, Palumbo GJ. 1991. Poxvirus pathogenesis. Microbiol. Rev. 55:80–122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Martinez O, Leung LW, Basler CF. 2012. The role of antigen-presenting cells in filoviral hemorrhagic fever: gaps in current knowledge. Antiviral Res. 93:416–428. 10.1016/j.antiviral.2012.01.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rodgers B, Mims CA. 1981. Interaction of influenza virus with mouse macrophages. Infect. Immun. 31:751–757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rodriguez D, Rodriguez JR, Rodriguez JF, Trauber D, Esteban M. 1989. Highly attenuated vaccinia virus mutants for the generation of safe recombinant viruses. Proc. Natl. Acad. Sci. U. S. A. 86:1287–1291. 10.1073/pnas.86.4.1287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rodriguez D, Esteban M, Rodriguez JR. 1995. Vaccinia virus A17L gene product is essential for an early step in virion morphogenesis. J. Virol. 69:4640–4648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guerra S, Lopez-Fernandez LA, Conde R, Pascual-Montano A, Harshman K, Esteban M. 2004. Microarray analysis reveals characteristic changes of host cell gene expression in response to attenuated modified vaccinia virus Ankara infection of human HeLa cells. J. Virol. 78:5820–5834. 10.1128/JVI.78.11.5820-5834.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guerra S, Lopez-Fernandez LA, Garcia MA, Zaballos A, Esteban M. 2006. Human gene profiling in response to the active protein kinase, interferon-induced serine/threonine protein kinase (PKR), in infected cells. Involvement of the transcription factor ATF-3 IN PKR-induced apoptosis. J. Biol. Chem. 281:18734–18745. 10.1074/jbc.M511983200 [DOI] [PubMed] [Google Scholar]
- 26.Guerra S, Lopez-Fernandez LA, Pascual-Montano A, Munoz M, Harshman K, Esteban M. 2003. Cellular gene expression survey of vaccinia virus infection of human HeLa cells. J. Virol. 77:6493–6506. 10.1128/JVI.77.11.6493-6506.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Guerra S, Lopez-Fernandez LA, Pascual-Montano A, Najera JL, Zaballos A, Esteban M. 2006. Host response to the attenuated poxvirus vector NYVAC: upregulation of apoptotic genes and NF-κB-responsive genes in infected HeLa cells. J. Virol. 80:985–998. 10.1128/JVI.80.2.985-998.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Marti-Lliteras P, Regueiro V, Morey P, Hood DW, Saus C, Sauleda J, Agusti AG, Bengoechea JA, Garmendia J. 2009. Nontypeable Haemophilus influenzae clearance by alveolar macrophages is impaired by exposure to cigarette smoke. Infect. Immun. 77:4232–4242. 10.1128/IAI.00305-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sharma S, tenOever BR, Grandvaux N, Zhou GP, Lin R, Hiscott J. 2003. Triggering the interferon antiviral response through an IKK-related pathway. Science 300:1148–1151. 10.1126/science.1081315 [DOI] [PubMed] [Google Scholar]
- 30.Guerra S, Najera JL, Gonzalez JM, Lopez-Fernandez LA, Climent N, Gatell JM, Gallart T, Esteban M. 2007. Distinct gene expression profiling after infection of immature human monocyte-derived dendritic cells by the attenuated poxvirus vectors MVA and NYVAC. J. Virol. 81:8707–8721. 10.1128/JVI.00444-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Joffre OP, Segura E, Savina A, Amigorena S. 2012. Cross-presentation by dendritic cells. Nat. Rev. Immunol. 12:557–569. 10.1038/nri3254 [DOI] [PubMed] [Google Scholar]
- 32.Ichikawa T, Nakao K, Nakata K, Hamasaki K, Takeda Y, Kajiya Y, Higashi S, Ohkubo K, Kato Y, Ishii N, Eguchi K. 2001. Geranylgeranylacetone induces antiviral gene expression in human hepatoma cells. Biochem. Biophys. Res. Commun. 280:933–939. 10.1006/bbrc.2000.4228 [DOI] [PubMed] [Google Scholar]
- 33.Aderem A, Underhill DM. 1999. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 17:593–623. 10.1146/annurev.immunol.17.1.593 [DOI] [PubMed] [Google Scholar]
- 34.Smyth GK, Altman NS. 2013. Separate-channel analysis of two-channel microarrays: recovering inter-spot information. BMC Bioinformatics 14:165. 10.1186/1471-2105-14-165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yanguez E, Garcia-Culebras A, Frau A, Llompart C, Knobeloch KP, Gutierrez-Erlandsson S, Garcia-Sastre A, Esteban M, Nieto A, Guerra S. 2013. ISG15 regulates peritoneal macrophages functionality against viral infection. PLoS Pathog. 9:e1003632. 10.1371/journal.ppat.1003632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Klotman ME, Chang TL. 2006. Defensins in innate antiviral immunity. Nat. Rev. Immunol. 6:447–456. 10.1038/nri1860 [DOI] [PubMed] [Google Scholar]
- 37.Deng L, Dai P, Ding W, Granstein RD, Shuman S. 2006. Vaccinia virus infection attenuates innate immune responses and antigen presentation by epidermal dendritic cells. J. Virol. 80:9977–9987. 10.1128/JVI.00354-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Grandvaux N, tenOever BR, Servant MJ, Hiscott J. 2002. The interferon antiviral response: from viral invasion to evasion. Curr. Opin. Infect. Dis. 15:259–267. 10.1097/00001432-200206000-00008 [DOI] [PubMed] [Google Scholar]
- 39.Hotchin J, Seegal R. 1977. Virus-induced behavioral alteration of mice. Science 196:671–674. 10.1126/science.854742 [DOI] [PubMed] [Google Scholar]
- 40.Yoneyama M, Kikuchi M, Natsukawa T, Shinobu N, Imaizumi T, Miyagishi M, Taira K, Akira S, Fujita T. 2004. The RNA helicase RIG-I has an essential function in double-stranded RNA-induced innate antiviral responses. Nat. Immunol. 5:730–737. 10.1038/ni1087 [DOI] [PubMed] [Google Scholar]
- 41.Servant MJ, tenOever B, LePage C, Conti L, Gessani S, Julkunen I, Lin R, Hiscott J. 2001. Identification of distinct signaling pathways leading to the phosphorylation of interferon regulatory factor 3. J. Biol. Chem. 276:355–363. 10.1074/jbc.M007790200 [DOI] [PubMed] [Google Scholar]
- 42.Vogel SN, Friedman RM, Hogan MM. 2001. Measurement of antiviral activity induced by interferons alpha, beta, and gamma. Curr. Protoc. Immunol. Chapter 6:Unit 6.9. 10.1002/0471142735.im0609s37 [DOI] [PubMed] [Google Scholar]
- 43.Perales-Linares R, Navas-Martin S. 2013. Toll-like receptor 3 in viral pathogenesis: friend or foe? Immunology 140:153–167. 10.1111/imm.12143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Alcami A. 2003. Viral mimicry of cytokines, chemokines and their receptors. Nat. Rev. Immunol. 3:36–50. 10.1038/nri980 [DOI] [PubMed] [Google Scholar]
- 45.Beattie E, Paoletti E, Tartaglia J. 1995. Distinct patterns of IFN sensitivity observed in cells infected with vaccinia K3L- and E3L-mutant viruses. Virology 210:254–263. 10.1006/viro.1995.1342 [DOI] [PubMed] [Google Scholar]
- 46.Kotwal GJ, Isaacs SN, McKenzie R, Frank MM, Moss B. 1990. Inhibition of the complement cascade by the major secretory protein of vaccinia virus. Science 250:827–830. 10.1126/science.2237434 [DOI] [PubMed] [Google Scholar]
- 47.Symons JA, Alcami A, Smith GL. 1995. Vaccinia virus encodes a soluble type I interferon receptor of novel structure and broad species specificity. Cell 81:551–560. 10.1016/0092-8674(95)90076-4 [DOI] [PubMed] [Google Scholar]
- 48.Stack J, Haga IR, Schroder M, Bartlett NW, Maloney G, Reading PC, Fitzgerald KA, Smith GL, Bowie AG. 2005. Vaccinia virus protein A46R targets multiple Toll-like-interleukin-1 receptor adaptors and contributes to virulence. J. Exp. Med. 201:1007–1018. 10.1084/jem.20041442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Perdiguero B, Gomez CE, Najera JL, Sorzano CO, Delaloye J, Gonzalez-Sanz R, Jimenez V, Roger T, Calandra T, Pantaleo G, Esteban M. 2012. Deletion of the viral anti-apoptotic gene F1L in the HIV/AIDS vaccine candidate MVA-C enhances immune responses against HIV-1 antigens. PLoS One 7:e48524. 10.1371/journal.pone.0048524 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.