Glucose is essential for energy production in the living body; the glucose transporter plays a critical role in various organs. Glucose transporters are classified into two families: facilitative glucose transporters (GLUTs) and sodium‐dependent glucose transporters (SGLTs), through which glucose is transported by facilitated diffusion, and Na+/glucose are co‐transported by an electrochemical gradient across the membrane, respectively. Six isoforms of the SGLT gene belong to the SLC5 gene family and they consist of 15 exons (Table 1). All SGLT proteins have 14 transmembrane helices in topology. SGLT1 and SGLT2 function as a glucose/galactose transporter and a glucose transporter across the membrane, respectively. SGLT3 is not a transporter in some species; for example, in humans, in which SGLT3 is thought to be a glucose sensor expressed in the enteric nervous system and muscle, whereas the functions of SGLT4, SGLT5, and SGLT6 are not known.
Table 1. Subtypes of human sodium‐dependent glucose transporter and tissue distribution.
| SGLT family (gene) | Function | Tissue distribution |
|---|---|---|
| SGLT1 (SLC5A1) | Glucose/galactose transporter | Intestine, trachea, kidney, heart, brain, testis, prostate |
| SGLT2 (SLC5A2) | Glucose transporter | Kidney, liver, thyroid, muscle, heart |
| SGLT3 (SLC5A4) | Glucose sensor? | Intestine, testis, uterus, lung, brain, thyroid |
| SGLT4 (SLC5A9) | ? | Intestine, kidney, liver, brain, trachea, lung, uterus, pancreas |
| SGLT5 (SLC5A10) | ? | Kidney, cortex |
| SGLT6 (SLC5A11) | ? | Spinal cord, kidney, brain |
SGLT, sodium‐dependent glucose transporter.
SGLT1 is expressed mainly in the intestine and kidney; SGLT2 is expressed highly in the kidney (Figure 1). Recently, Gorboulev et al.1 reported that SGLT1 is expressed in the brush border membrane (BBM) of the intestine, and that glucose absorption across the BBM disappeared in SGLT1‐deficient mice, which indicates that intestinal glucose absorption in the intestine is mediated predominantly by SGLT1.
Figure 1.
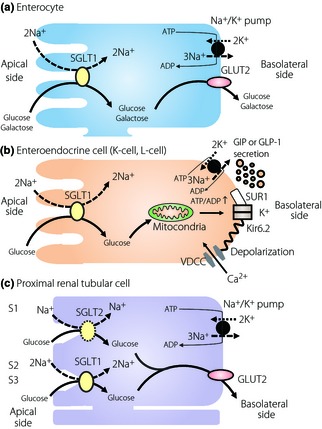
Glucose absorption through sodium‐dependent glucose transporter 1 (SGLT1) in (a) an enterocyte and (b) an enteroendocrine cell, and (c) glucose reabsorption through SGLT1 and sodium‐dependent glucose transporter 2 (SGLT2) in a proximal renal tubular cell. ADP, adenosine diphosphate; ATP, adenosine triphosphate; GIP, gastric inhibitory polypeptide; GLP‐1, glucagon‐like peptide‐1; GLUT, glucose transporter; SUR1, sulfonylurea receptor 1; VDCC, voltage‐dependent calcium channel.
Some studies found that SGLT1 is expressed in incretin‐secreting cells and is involved in incretin secretion2. In addition, it is reported that the SGLT inhibitor, phrorizin, reduces incretin secretion3. Incretin is the intestinal hormone that is secreted on meal ingestion and potentiates insulin secretion from pancreatic β‐cells in a glucose‐dependent manner. Gastric inhibitory polypeptide (GIP) and glucagon‐like peptide‐1 (GLP‐1), the major incretins, are secreted from K‐cells in the proximal small intestine, and from L‐cells in the distal small intestine and colon, respectively. Glucose, protein and fat all induce incretin secretions, but fat especially induces GIP secretion in human studies4. Gorboulev et al.1 reported that fat ingestion stimulates both GIP and GLP‐1 secretion in both wild‐type and SGLT1‐deficient mice, but that glucose does not in SGLT1‐deficient mice, clearly showing that SGLT1 plays a critical role in incretin secretion in response to glucose in vivo. Glucokinase, sulfonylurea receptor 1 (SUR1), and Kir6.2, which are associated with glucose‐induced insulin secretion in β‐cells, are reported to be expressed in both K‐cells and L‐cells, suggesting that incretin secretion is exerted by glucose metabolism in the cells. In contrast, it has been reported that a‐methyl‐D‐glucopyranoside, which is not metabolized in the glycolytic pathway or tricarboxylic acid cycle, induces incretin secretion5. In such a case, membrane depolarization as a result of sodium ion uptake with unmetabolizable glucose into the cell through SGLT1 would be critical for incretin secretion. However, the relative importance in incretin secretion of glucose absorption from the lumen side and from glucose metabolism in the cells remains unclear.
In the kidney, glucose filtered in glomeruli is reabsorbed in proximal renal tubules, and is not usually secreted in the urine. However, when the blood glucose level is over 160~180 mg/dL, glucose reabsorption exceeds reabsorption capacity and glucose does appear in the urine. SGLT1 is expressed mainly in the S2 and S3 segments of the proximal renal tubules, and reabsorbs one glucose molecule coupled with two sodium ions. SGLT2 has 60% homology with SGLT1 and is highly expressed in the BBM of the S1 segment of the proximal renal tubules. SGLT2 has a low affinity to glucose and reabsorbs one glucose molecule coupled with one sodium ion. It is thought that SGLT1 and SGLT2 reabsorb 10 and 90% of filtered glucose, respectively, in the kidney6. Considering that SGLT2 is largely involved in glucose reabsorption and that SGLT2 expression is upregulated in the diabetic rat7, inhibition of SGLT2 might well be a new therapeutic approach to excrete glucose into the urine and manage blood glucose levels in type 2 diabetes mellitus patients. To date, various SGLT2 inhibitors have been developed for treatment of type 2 diabetes mellitus. Interestingly, Gorboulev et al. showed that SGLT1‐deficient mice lose just ~3% of the filtered glucose into the urine, whereas SGLT2‐deficient mice lose ~60% of the filtered glucose into the urine, suggesting that wild‐type mice do not use the maximal transport capacity of SGLT1 under normoglycemic conditions1. In diabetic patients, the glucose concentration is overwhelming in early proximal tubules and is even more in the patients with a SGLT2‐specific inhibitor. In this condition, SGLT1 transporter might be performing at full capacity and minimizes the effects of the drug. In this context, SGLT1 inhibition might have therapeutic potential. However, it might reduce incretin secretion and induce side‐effects, such as diarrhea and polyurea, the main symptoms of glucose–galactose malabsorption in patients with SGLT1 gene mutations.
Acknowledgements
Nobuya Inagaki has received research grants and consultancy fees from Astellas Pharma Inc., Taisho Pharmaceutical Co., Ltd., and Mitsubishi Tanabe Pharma Corporation. Norio Harada declares no conflict of interest.
References
- 1.Gorboulev V, Schürmann A, Vallon V, et al Na(+)‐D‐glucose cotransporter SGLT1 is pivotal for intestinal glucose absorption and glucose‐dependent incretin secretion. Diabetes 2012; 61: 187–196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Reimann F, Habib AM, Tolhurst G, et al Glucose sensing in L cells: a primary cell study. Cell Metab 2008; 8: 532–539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gribble FM, Williams L, Simpson AK, et al A novel glucose‐sensing mechanism contributing to glucagon‐like peptide‐1 secretion from the GLUTag cell line. Diabetes 2003; 52: 1147–1154 [DOI] [PubMed] [Google Scholar]
- 4.Yamane S, Harada N, Hamasaki A, et al Effects of glucose and meal ingestion on incretin secretion in Japanese subjects with normal glucose tolerance. J Diabetes Invest 2012; 3: 80–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moriya R, Shirakura T, Ito J, et al Activation of sodium–glucose cotransporter 1 ameliorates hyperglycemia by mediating incretin secretion in mice. Am J Physiol Endocrinol Metab 2009; 297: E1358–E1365 [DOI] [PubMed] [Google Scholar]
- 6.Ferrannini E, Solini A. SGLT2 inhibition in diabetes mellitus: rationale and clinical prospects. Nat Rev Endocrinol 2012; (Epub ahead of print). doi: 10.1038/nrendo.2011.243 [DOI] [PubMed] [Google Scholar]
- 7.Freitas HS, Anhê GF, Melo KF, et al Na(+)‐glucose transporter‐2 messenger ribonucleic acid expression in kidney of diabetic rats correlates with glycemic levels: involvement of hepatocyte nuclear factor‐1alpha expression and activity. Endocrinology 2008; 149: 717–724 [DOI] [PubMed] [Google Scholar]


