Abstract
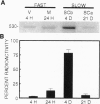
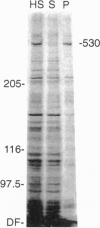
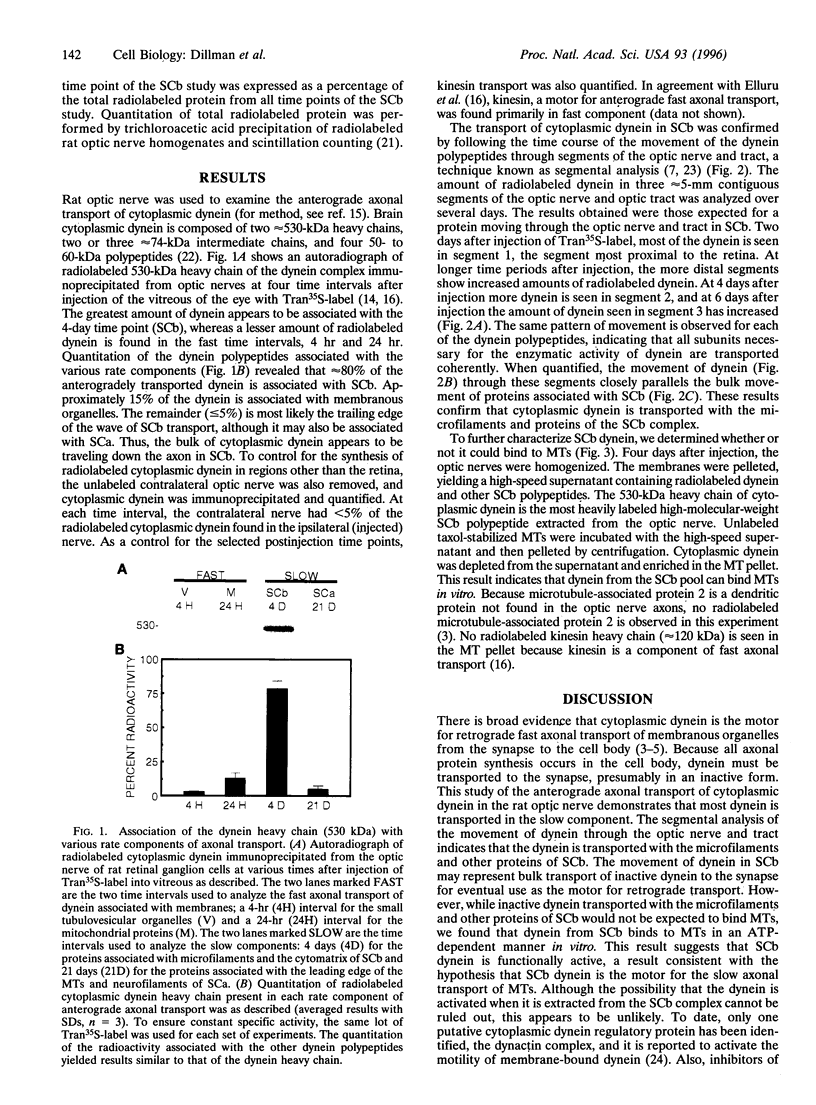
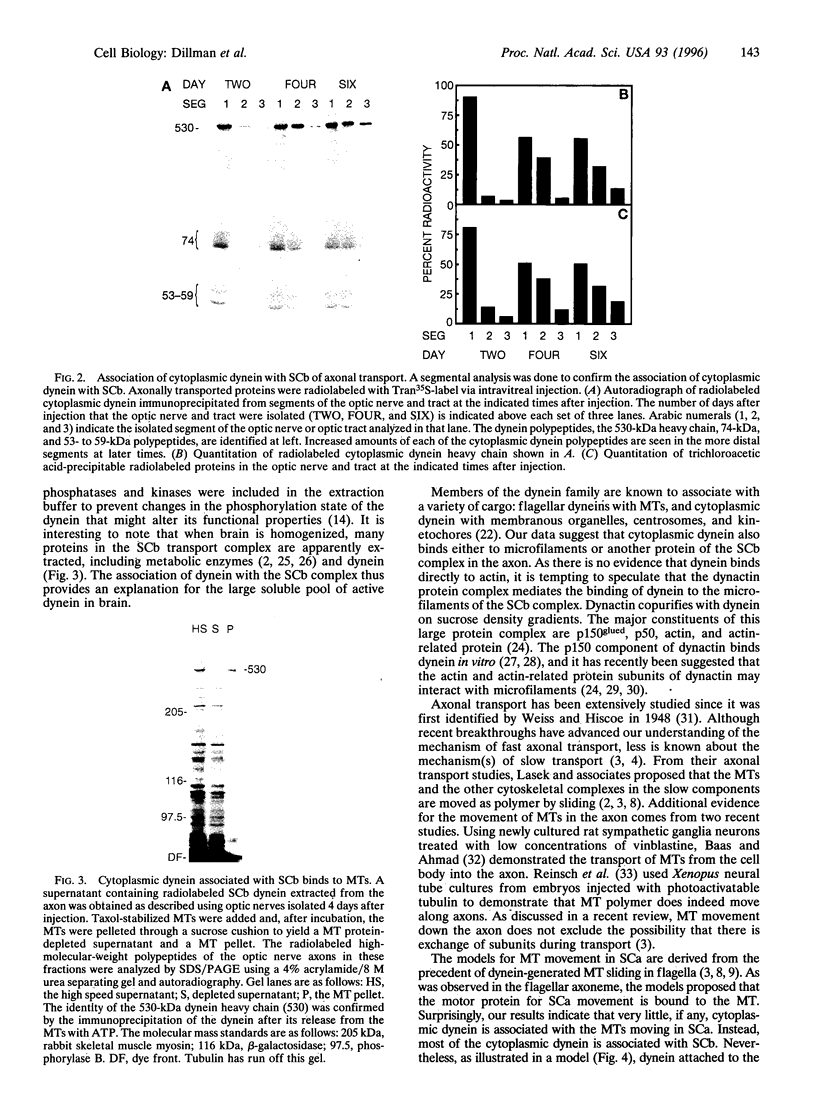
Neuronal function is dependent on the transport of materials from the cell body to the synapse via anterograde axonal transport. Anterograde axonal transport consists of several components that differ in both rate and protein composition. In fast transport, membranous organelles are moved along microtubules by the motor protein kinesin. The cytoskeleton and the cytomatrix proteins move in the two components of slow transport. While the mechanisms underlying slow transport are unknown, it has been hypothesized that the movement of microtubules in slow transport is generated by sliding. To determine whether dynein, a motor protein that causes microtubule sliding in flagella, may play a role in slow axonal transport, we identified the transport rate components with which cytoplasmic dynein is associated in rat optic nerve. Nearly 80% of the anterogradely moving dynein was associated with slow transport, whereas only approximately 15% of the dynein was associated with the membranous organelles of anterograde fast axonal transport. A segmental analysis of the transport of dynein through contiguous regions of the optic nerve and tract showed that dynein is associated with the microfilaments and other proteins of slow component b. Dynein from this transport component has the capacity to bind microtubules in vitro. These results are consistent with the hypothesis that cytoplasmic dynein generates the movement of microtubules in slow axonal transport. A model is presented to illustrate how dynein attached to the slow component b complex of proteins is appropriately positioned to generate force of the correct polarity to slide microtubules down the axon.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baas P. W., Ahmad F. J. The transport properties of axonal microtubules establish their polarity orientation. J Cell Biol. 1993 Mar;120(6):1427–1437. doi: 10.1083/jcb.120.6.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black M. M., Chestnut M. H., Pleasure I. T., Keen J. H. Stable clathrin: uncoating protein (hsc70) complexes in intact neurons and their axonal transport. J Neurosci. 1991 May;11(5):1163–1172. doi: 10.1523/JNEUROSCI.11-05-01163.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brady S. T., Lasek R. J. Axonal transport: a cell-biological method for studying proteins that associate with the cytoskeleton. Methods Cell Biol. 1982;25(Pt B):365–398. doi: 10.1016/s0091-679x(08)61434-x. [DOI] [PubMed] [Google Scholar]
- Brady S. T., Lasek R. J. Nerve-specific enolase and creatine phosphokinase in axonal transport: soluble proteins and the axoplasmic matrix. Cell. 1981 Feb;23(2):515–523. doi: 10.1016/0092-8674(81)90147-1. [DOI] [PubMed] [Google Scholar]
- Brady S. T. Molecular motors in the nervous system. Neuron. 1991 Oct;7(4):521–533. doi: 10.1016/0896-6273(91)90365-7. [DOI] [PubMed] [Google Scholar]
- Cyr J. L., Brady S. T. Molecular motors in axonal transport. Cellular and molecular biology of kinesin. Mol Neurobiol. 1992 Summer-Fall;6(2-3):137–155. doi: 10.1007/BF02780549. [DOI] [PubMed] [Google Scholar]
- Dillman J. F., 3rd, Pfister K. K. Differential phosphorylation in vivo of cytoplasmic dynein associated with anterogradely moving organelles. J Cell Biol. 1994 Dec;127(6 Pt 1):1671–1681. doi: 10.1083/jcb.127.6.1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elluru R. G., Bloom G. S., Brady S. T. Fast axonal transport of kinesin in the rat visual system: functionality of kinesin heavy chain isoforms. Mol Biol Cell. 1995 Jan;6(1):21–40. doi: 10.1091/mbc.6.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbons I. R. Dynein ATPases as microtubule motors. J Biol Chem. 1988 Nov 5;263(31):15837–15840. [PubMed] [Google Scholar]
- Grafstein B., Forman D. S. Intracellular transport in neurons. Physiol Rev. 1980 Oct;60(4):1167–1283. doi: 10.1152/physrev.1980.60.4.1167. [DOI] [PubMed] [Google Scholar]
- Holzbaur E. L., Vallee R. B. DYNEINS: molecular structure and cellular function. Annu Rev Cell Biol. 1994;10:339–372. doi: 10.1146/annurev.cb.10.110194.002011. [DOI] [PubMed] [Google Scholar]
- Karki S., Holzbaur E. L. Affinity chromatography demonstrates a direct binding between cytoplasmic dynein and the dynactin complex. J Biol Chem. 1995 Dec 1;270(48):28806–28811. doi: 10.1074/jbc.270.48.28806. [DOI] [PubMed] [Google Scholar]
- Lasek R. J. Polymer sliding in axons. J Cell Sci Suppl. 1986;5:161–179. doi: 10.1242/jcs.1986.supplement_5.10. [DOI] [PubMed] [Google Scholar]
- Lye R. J., Porter M. E., Scholey J. M., McIntosh J. R. Identification of a microtubule-based cytoplasmic motor in the nematode C. elegans. Cell. 1987 Oct 23;51(2):309–318. doi: 10.1016/0092-8674(87)90157-7. [DOI] [PubMed] [Google Scholar]
- McQuarrie I. G., Brady S. T., Lasek R. J. Diversity in the axonal transport of structural proteins: major differences between optic and spinal axons in the rat. J Neurosci. 1986 Jun;6(6):1593–1605. doi: 10.1523/JNEUROSCI.06-06-01593.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melki R., Vainberg I. E., Chow R. L., Cowan N. J. Chaperonin-mediated folding of vertebrate actin-related protein and gamma-tubulin. J Cell Biol. 1993 Sep;122(6):1301–1310. doi: 10.1083/jcb.122.6.1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paschal B. M., Shpetner H. S., Vallee R. B. MAP 1C is a microtubule-activated ATPase which translocates microtubules in vitro and has dynein-like properties. J Cell Biol. 1987 Sep;105(3):1273–1282. doi: 10.1083/jcb.105.3.1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfister K. K., Wagner M. C., Stenoien D. L., Brady S. T., Bloom G. S. Monoclonal antibodies to kinesin heavy and light chains stain vesicle-like structures, but not microtubules, in cultured cells. J Cell Biol. 1989 Apr;108(4):1453–1463. doi: 10.1083/jcb.108.4.1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinsch S. S., Mitchison T. J., Kirschner M. Microtubule polymer assembly and transport during axonal elongation. J Cell Biol. 1991 Oct;115(2):365–379. doi: 10.1083/jcb.115.2.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sale W. S., Satir P. Direction of active sliding of microtubules in Tetrahymena cilia. Proc Natl Acad Sci U S A. 1977 May;74(5):2045–2049. doi: 10.1073/pnas.74.5.2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer D. A., Gill S. R., Cooper J. A., Heuser J. E., Schroer T. A. Ultrastructural analysis of the dynactin complex: an actin-related protein is a component of a filament that resembles F-actin. J Cell Biol. 1994 Jul;126(2):403–412. doi: 10.1083/jcb.126.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tytell M., Black M. M., Garner J. A., Lasek R. J. Axonal transport: each major rate component reflects the movement of distinct macromolecular complexes. Science. 1981 Oct 9;214(4517):179–181. doi: 10.1126/science.6169148. [DOI] [PubMed] [Google Scholar]
- Vallee R. B., Bloom G. S. Mechanisms of fast and slow axonal transport. Annu Rev Neurosci. 1991;14:59–92. doi: 10.1146/annurev.ne.14.030191.000423. [DOI] [PubMed] [Google Scholar]
- Waterman-Storer C. M., Karki S., Holzbaur E. L. The p150Glued component of the dynactin complex binds to both microtubules and the actin-related protein centractin (Arp-1). Proc Natl Acad Sci U S A. 1995 Feb 28;92(5):1634–1638. doi: 10.1073/pnas.92.5.1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams R. C., Jr, Lee J. C. Preparation of tubulin from brain. Methods Enzymol. 1982;85(Pt B):376–385. doi: 10.1016/0076-6879(82)85038-6. [DOI] [PubMed] [Google Scholar]