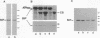Abstract
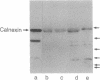
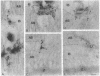
The skeletal muscle sarcoplasmic reticulum (SR) was investigated for the presence of well-known endoplasmic reticulum (ER) markers: the lumenal protein BiP and a group of membrane proteins recognized by an antibody raised against ER membrane vesicles. Western blots of SR fractions revealed the presence of BiP in fast- and slow-twitch muscles of the rabbit as well as in rat and chicken muscles. Analyses of purified SR subfractions, together with cryosection immunofluorescence and immunogold labeling, revealed BiP evenly distributed within the longitudinal SR and the terminal cisternae. Within the terminal cisternae BiP appeared not to be mixed with calsequestrin but to be distributed around the aggregates of the latter Ca2+ binding protein. Of the various membrane markers only calnexin (91 kDa) was found to be distributed within both SR subfractions, whereas the other markers (apparent molecular masses of 64 kDa and 58 kDa and a doublet around 28 kDa) were concentrated in the terminal cisternae. These results suggest that the SR is a specialized ER subcompartment in which general markers, such as the ones we have investigated, coexist with the major SR proteins specifically responsible for Ca2+ uptake, storage, and release. The differential distribution of the ER markers reveals new aspects of the SR molecular structure that might be of importance for the functioning of the endomembrane system.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bole D. G., Dowin R., Doriaux M., Jamieson J. D. Immunocytochemical localization of BiP to the rough endoplasmic reticulum: evidence for protein sorting by selective retention. J Histochem Cytochem. 1989 Dec;37(12):1817–1823. doi: 10.1177/37.12.2685110. [DOI] [PubMed] [Google Scholar]
- Bole D. G., Hendershot L. M., Kearney J. F. Posttranslational association of immunoglobulin heavy chain binding protein with nascent heavy chains in nonsecreting and secreting hybridomas. J Cell Biol. 1986 May;102(5):1558–1566. doi: 10.1083/jcb.102.5.1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cala S. E., Scott B. T., Jones L. R. Intralumenal sarcoplasmic reticulum Ca(2+)-binding proteins. Semin Cell Biol. 1990 Aug;1(4):265–275. [PubMed] [Google Scholar]
- Chyn T. L., Martonosi A. N., Morimoto T., Sabatini D. D. In vitro synthesis of the Ca2+ transport ATPase by ribosomes bound to sarcoplasmic reticulum membranes. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1241–1245. doi: 10.1073/pnas.76.3.1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costello B., Chadwick C., Saito A., Chu A., Maurer A., Fleischer S. Characterization of the junctional face membrane from terminal cisternae of sarcoplasmic reticulum. J Cell Biol. 1986 Sep;103(3):741–753. doi: 10.1083/jcb.103.3.741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duggan P. F., Martonosi A. Sarcoplasmic reticulum. IX. The permeability of sarcoplasmic reticulum membranes. J Gen Physiol. 1970 Aug;56(2):147–167. doi: 10.1085/jgp.56.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezerman E. B., Ishikawa H. Differentiation of the sarcoplasmic reticulum and T system in developing chick skeletal muscle in vitro. J Cell Biol. 1967 Nov 1;35(2):405–420. doi: 10.1083/jcb.35.2.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleischer S., Inui M. Biochemistry and biophysics of excitation-contraction coupling. Annu Rev Biophys Biophys Chem. 1989;18:333–364. doi: 10.1146/annurev.bb.18.060189.002001. [DOI] [PubMed] [Google Scholar]
- Fliegel L., Burns K., MacLennan D. H., Reithmeier R. A., Michalak M. Molecular cloning of the high affinity calcium-binding protein (calreticulin) of skeletal muscle sarcoplasmic reticulum. J Biol Chem. 1989 Dec 25;264(36):21522–21528. [PubMed] [Google Scholar]
- Fliegel L., Burns K., Opas M., Michalak M. The high-affinity calcium binding protein of sarcoplasmic reticulum. Tissue distribution, and homology with calregulin. Biochim Biophys Acta. 1989 Jun 26;982(1):1–8. doi: 10.1016/0005-2736(89)90166-1. [DOI] [PubMed] [Google Scholar]
- Fliegel L., Newton E., Burns K., Michalak M. Molecular cloning of cDNA encoding a 55-kDa multifunctional thyroid hormone binding protein of skeletal muscle sarcoplasmic reticulum. J Biol Chem. 1990 Sep 15;265(26):15496–15502. [PubMed] [Google Scholar]
- Fliegel L., Ohnishi M., Carpenter M. R., Khanna V. K., Reithmeier R. A., MacLennan D. H. Amino acid sequence of rabbit fast-twitch skeletal muscle calsequestrin deduced from cDNA and peptide sequencing. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1167–1171. doi: 10.1073/pnas.84.5.1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flynn G. C., Pohl J., Flocco M. T., Rothman J. E. Peptide-binding specificity of the molecular chaperone BiP. Nature. 1991 Oct 24;353(6346):726–730. doi: 10.1038/353726a0. [DOI] [PubMed] [Google Scholar]
- Franzini-Armstrong C., Kenney L. J., Varriano-Marston E. The structure of calsequestrin in triads of vertebrate skeletal muscle: a deep-etch study. J Cell Biol. 1987 Jul;105(1):49–56. doi: 10.1083/jcb.105.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofmann S. L., Goldstein J. L., Orth K., Moomaw C. R., Slaughter C. A., Brown M. S. Molecular cloning of a histidine-rich Ca2+-binding protein of sarcoplasmic reticulum that contains highly conserved repeated elements. J Biol Chem. 1989 Oct 25;264(30):18083–18090. [PubMed] [Google Scholar]
- Jorgensen A. O., Kalnins V., MacLennan D. H. Localization of sarcoplasmic reticulum proteins in rat skeletal muscle by immunofluorescence. J Cell Biol. 1979 Feb;80(2):372–384. doi: 10.1083/jcb.80.2.372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jorgensen A. O., Shen A. C., Campbell K. P., MacLennan D. H. Ultrastructural localization of calsequestrin in rat skeletal muscle by immunoferritin labeling of ultrathin frozen sections. J Cell Biol. 1983 Nov;97(5 Pt 1):1573–1581. doi: 10.1083/jcb.97.5.1573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller G. A., Tokuyasu K. T., Dutton A. H., Singer S. J. An improved procedure for immunoelectron microscopy: ultrathin plastic embedding of immunolabeled ultrathin frozen sections. Proc Natl Acad Sci U S A. 1984 Sep;81(18):5744–5747. doi: 10.1073/pnas.81.18.5744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leberer E., Charuk J. H., Clarke D. M., Green N. M., Zubrzycka-Gaarn E., MacLennan D. H. Molecular cloning and expression of cDNA encoding the 53,000-dalton glycoprotein of rabbit skeletal muscle sarcoplasmic reticulum. J Biol Chem. 1989 Feb 25;264(6):3484–3493. [PubMed] [Google Scholar]
- Leberer E., Charuk J. H., Green N. M., MacLennan D. H. Molecular cloning and expression of cDNA encoding a lumenal calcium binding glycoprotein from sarcoplasmic reticulum. Proc Natl Acad Sci U S A. 1989 Aug;86(16):6047–6051. doi: 10.1073/pnas.86.16.6047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leberer E., Timms B. G., Campbell K. P., MacLennan D. H. Purification, calcium binding properties, and ultrastructural localization of the 53,000- and 160,000 (sarcalumenin)-dalton glycoproteins of the sarcoplasmic reticulum. J Biol Chem. 1990 Jun 15;265(17):10118–10124. [PubMed] [Google Scholar]
- Louvard D., Reggio H., Warren G. Antibodies to the Golgi complex and the rough endoplasmic reticulum. J Cell Biol. 1982 Jan;92(1):92–107. doi: 10.1083/jcb.92.1.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma J., Fill M., Knudson C. M., Campbell K. P., Coronado R. Ryanodine receptor of skeletal muscle is a gap junction-type channel. Science. 1988 Oct 7;242(4875):99–102. doi: 10.1126/science.2459777. [DOI] [PubMed] [Google Scholar]
- Macer D. R., Koch G. L. Identification of a set of calcium-binding proteins in reticuloplasm, the luminal content of the endoplasmic reticulum. J Cell Sci. 1988 Sep;91(Pt 1):61–70. doi: 10.1242/jcs.91.1.61. [DOI] [PubMed] [Google Scholar]
- Mitchell R. D., Simmerman H. K., Jones L. R. Ca2+ binding effects on protein conformation and protein interactions of canine cardiac calsequestrin. J Biol Chem. 1988 Jan 25;263(3):1376–1381. [PubMed] [Google Scholar]
- PORTER K. R., PALADE G. E. Studies on the endoplasmic reticulum. III. Its form and distribution in striated muscle cells. J Biophys Biochem Cytol. 1957 Mar 25;3(2):269–300. doi: 10.1083/jcb.3.2.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelham H. R. Evidence that luminal ER proteins are sorted from secreted proteins in a post-ER compartment. EMBO J. 1988 Apr;7(4):913–918. doi: 10.1002/j.1460-2075.1988.tb02896.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reithmeier R. A., de Leon S., MacLennan D. H. Assembly of the sarcoplasmic reticulum. Cell-free synthesis of te Ca2+ + Mg2+-adenosine triphosphatase and calsequestrin. J Biol Chem. 1980 Dec 25;255(24):11839–11846. [PubMed] [Google Scholar]
- Saito A., Seiler S., Chu A., Fleischer S. Preparation and morphology of sarcoplasmic reticulum terminal cisternae from rabbit skeletal muscle. J Cell Biol. 1984 Sep;99(3):875–885. doi: 10.1083/jcb.99.3.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiaffino S., Margreth A. Coordinated development of the sarcoplasmic reticulum and T system during postnatal differentiation of rat skeletal muscle. J Cell Biol. 1969 Jun;41(3):855–875. doi: 10.1083/jcb.41.3.855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takei K., Stukenbrok H., Metcalf A., Mignery G. A., Südhof T. C., Volpe P., De Camilli P. Ca2+ stores in Purkinje neurons: endoplasmic reticulum subcompartments demonstrated by the heterogeneous distribution of the InsP3 receptor, Ca(2+)-ATPase, and calsequestrin. J Neurosci. 1992 Feb;12(2):489–505. doi: 10.1523/JNEUROSCI.12-02-00489.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas K., Navarro J., Benson R. J., Campbell K. P., Rotundo R. L., Fine R. E. Newly synthesized calsequestrin, destined for the sarcoplasmic reticulum, is contained in early/intermediate Golgi-derived clathrin-coated vesicles. J Biol Chem. 1989 Feb 25;264(6):3140–3145. [PubMed] [Google Scholar]
- Tooze J., Hollinshead M., Fuller S. D., Tooze S. A., Huttner W. B. Morphological and biochemical evidence showing neuronal properties in AtT-20 cells and their growth cones. Eur J Cell Biol. 1989 Aug;49(2):259–273. [PubMed] [Google Scholar]
- Tooze J., Kern H. F., Fuller S. D., Howell K. E. Condensation-sorting events in the rough endoplasmic reticulum of exocrine pancreatic cells. J Cell Biol. 1989 Jul;109(1):35–50. doi: 10.1083/jcb.109.1.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tougard C., Louvard D., Picart R., Tixier-Vidal A. The rough endoplasmic reticulum and the Golgi apparatus visualized using specific antibodies in normal and tumoral prolactin cells in culture. J Cell Biol. 1983 May;96(5):1197–1207. doi: 10.1083/jcb.96.5.1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villa A., Podini P., Clegg D. O., Pozzan T., Meldolesi J. Intracellular Ca2+ stores in chicken Purkinje neurons: differential distribution of the low affinity-high capacity Ca2+ binding protein, calsequestrin, of Ca2+ ATPase and of the ER lumenal protein, Bip. J Cell Biol. 1991 May;113(4):779–791. doi: 10.1083/jcb.113.4.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volpe P., Alderson-Lang B. H., Madeddu L., Damiani E., Collins J. H., Margreth A. Calsequestrin, a component of the inositol 1,4,5-trisphosphate-sensitive Ca2+ store of chicken cerebellum. Neuron. 1990 Nov;5(5):713–721. doi: 10.1016/0896-6273(90)90225-5. [DOI] [PubMed] [Google Scholar]
- Volpe P., Villa A., Damiani E., Sharp A. H., Podini P., Snyder S. H., Meldolesi J. Heterogeneity of microsomal Ca2+ stores in chicken Purkinje neurons. EMBO J. 1991 Nov;10(11):3183–3189. doi: 10.1002/j.1460-2075.1991.tb04880.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wada I., Rindress D., Cameron P. H., Ou W. J., Doherty J. J., 2nd, Louvard D., Bell A. W., Dignard D., Thomas D. Y., Bergeron J. J. SSR alpha and associated calnexin are major calcium binding proteins of the endoplasmic reticulum membrane. J Biol Chem. 1991 Oct 15;266(29):19599–19610. [PubMed] [Google Scholar]
- Yuan S. H., Arnold W., Jorgensen A. O. Biogenesis of transverse tubules and triads: immunolocalization of the 1,4-dihydropyridine receptor, TS28, and the ryanodine receptor in rabbit skeletal muscle developing in situ. J Cell Biol. 1991 Jan;112(2):289–301. doi: 10.1083/jcb.112.2.289. [DOI] [PMC free article] [PubMed] [Google Scholar]