Abstract
Background
Mechanisms predisposing HIV-infected patients to increased CVD risk remain unclear.
Objective
To determine the interrelationship between arterial inflammation and high-risk coronary plaque morphology in HIV-infected patients with subclinical coronary atherosclerosis.
Methods
41 HIV-infected patients on stable ART without known CVD but with atherosclerotic plaque on coronary CT angiography were evaluated with 18F-FDG-PET. Patients were stratified into two groups based on relative degree of arterial inflammation (aortic target-to-background-ratio, or TBR). High-risk coronary atherosclerotic plaque morphology features were compared between groups.
Results
HIV-infected patients with higher and lower TBR’s were similar with respect to traditional CVD risk parameters. Among HIV-infected patients with higher TBR, an increased percentage of patients demonstrated at least one low attenuation coronary atherosclerotic plaque (40% vs. 10%, p = 0.02) and at least one coronary atherosclerotic plaque with both low attenuation and positive remodeling (35% vs. 10%, p = 0.04). Moreover, in the higher TBR group, both the number of low attenuation plaques (LAP’s) per patient (p = 0.02) and the number of vulnerability features in the most vulnerable plaque (p = 0.02) were increased. TBR grouping remained significantly related to the number of LAP’s/subject (β=0.35, p = 0.004), controlling for age, gender, LDL, duration HIV, and CD4.
Conclusion
These data demonstrate a relationship between arterial inflammation on 18F-FDG-PET and high-risk coronary atherosclerotic plaque features among HIV-infected patients with sublclinical coronary atherosclerosis. Further studies are needed to determine whether arterial inflammation and related high-risk coronary morphology increase the risk of clinical CVD events in the HIV population.
Keywords: HIV, Arterial Inflammation, Coronary Atherosclerotic Plaque, Cardiovascular Disease
Introduction
HIV-infected patients demonstrate increased cardiovascular disease (CVD) risk compared to non HIV-infected patients, and this increased risk is not fully accounted for by traditional risk factors1,2. Heightened CVD risk in HIV has been attributed, in part, to increased inflammation and immune activation3–5.
18F-fluorodeoxyglucose-positon emission tomography (18F-FDG-PET) is a functional imaging technique used to assess arterial inflammation. Arterial FDG uptake provides a measure of macrophage infiltration within atheromatous plaques6. Increased arterial inflammation on 18F-FDG-PET has been shown to significantly predict increased CVD events in non HIV-infected patients7,8. We have previously utilized 18F-FDG-PET to assess arterial inflammation among HIV-infected patients. We demonstrated increased arterial inflammation in association with indices of increased immune activation using this technique among HIV-infected patients with good virological control and low Framingham Risk Scores (FRS)9.
Among HIV-infected patients with good virological control and low FRS, we have also shown increased high-risk coronary plaque morphology marked by low attenuation plaque and positive remodeling using coronary computed tomography angiography (CTA)10. The presence of these high-risk morphological features is known to predict increased CVD events in non HIV-infected patients11, as such features are thought to demonstrate plaque more vulnerable to rupture. However, the degree to which indices of arterial inflammation are associated with these features in HIV-infected patients remains unknown.
In the current study, we test the hypothesis that increased systemic arterial inflammation relates to and potentially provides a substrate for the development of high-risk morphology coronary atherosclerotic plaque. We evaluated HIV-infected patients without known CVD using coronary CTA and 18F-FDG-PET. We then compared, amongst those patients with demonstrated subclinical atherosclerosis, the degree of arterial inflammation on 18F-FDG-PET to specific, standardized high-risk plaque morphology features on coronary CTA.
Methods
Study Participants
82 HIV-infected subjects were screened for the study. These subjects were being evaluated for inclusion in an ongoing trial of statin therapy to reduce arterial inflammation in HIV. Subjects with known CVD (myocardial infarction, arrhythmias, valvular heart disease, pericarditis, congestive heart failure, stroke and peripheral vascular disease), or symptoms suggestive of CVD (angina), use of statins, or recent acute infection were excluded. HIV-infected subjects were on stable doses of antiretroviral therapy (ART) (i.e. no change in prescribed ART agent or dosing) for at least 3 months prior to the time of the study. 53 HIV-infected subjects meeting these inclusion criteria underwent coronary CTA to assess coronary atherosclerotic plaque burden and morphology and 18F-FDG-PET to assess arterial inflammation in the aorta. Data on demographics, traditional CVD risk factors, and immunologic parameters were collected. Of these subjects, 12 had no atherosclerotic plaque. In the current study, we investigate the relationship of arterial inflammation to plaque morphology among the 41 subjects with subclinical coronary atherosclerosis as per our hypothesis that increased systemic arterial inflammation may provide a substrate for the development of high-risk plaque morphology among those with subclinical plaque. These data extend the data previously published on a subset of 27 of these subjects9 and now add plaque morphology to the analysis, which was not included in our prior study. The data in this study represent screening data, prior to randomization into the statin trial.
Measurement of Arterial Inflammation with 18F-FDG-PET
Participating HIV-infected subjects underwent 18F-FDG-PET imaging after an overnight fast to reduce myocardial FDG uptake as previously described9. The PET imaging was performed 3 h after administration of 13 mCi of 18F-FDG (Siemens ECAT Exact HR+ PET or biograph 64 system, Knoxville, Tennessee)8. The system provides 63 planes, a 15.5-cm field of view, and a maximum 4.2-mm intrinsic resolution at the center of the field of view. Images were acquired in 3 dimensional mode over 20 minutes with an effective resolution of 5mm. All image analysis was performed while blinded to clinical data. The ascending aorta was chosen for measurement of arterial inflammation. Arterial inflammation is expressed in terms of aortic target-to-background ratio (TBR), which in turn is calculated by dividing the mean arterial standardized uptake value (SUV) by the mean venous SUV. The mean arterial SUV is derived from the average of the maximum SUV values in serial axial measurements9.
Assessment of High-risk Atherosclerotic Plaque Morphology by Coronary CTA
All coronary arterial segments were assessed for presence of plaque. Among those with plaque, trained experts evaluated detailed morphological characteristics (S.A., B.W.). Cross-sectional and multiplanar reconstructed images were acquired and assessed for 18 coronary artery segments: left main coronary artery, proximal, mid, and distal left anterior descending artery segments, first to third diagonal branches, ramus intermedius, proximal, mid, and distal left circumflex coronary artery segments, first and second obtuse marginal branches, posterior left ventricular branch, proximal, mid, and distal right coronary artery segments, and posterior descending artery, as previously described 10. Thresholds for definitions of high-risk atherosclerotic plaque morphology features were as per previously published clinical studies12: Specifically, low attenuation plaque was defined as plaque having a mean minimal attenuation < 40 Hounsfield Units. For determination of low attenuation plaque, 5 regions of interest (area = 1 mm2) were placed on each plaque and the smallest average value within the regions of interest was recorded to represent the mean minimal attenuation. Positive remodeling was defined as [plaque segment diameter/ reference segment diameter] > 1.05. Image analysis was performed while blinded to clinical data, and blinding was maintained between investigators analyzing the 18F-FDG PET data, on the one hand, and the coronary CTA data on the other.
Assessment of Demographic and Cardiometabolic Risk Factors
Demographic parameters including age, gender, race, family history of premature heart disease, smoking status, current diabetes, and use of antihypertensive medication were assessed by history. Body mass index (BMI) and systolic blood pressure were assessed using standard techniques. A screening lipid panel was obtained and analyzed using standard techniques. LDL cholesterol level was calculated via the Friedwald equation.
Assessment of HIV Disease-related Parameters
HIV viral load was determined by ultrasensitive RT PCR (Roche Amplicor Monitor). HIV testing was performed using ELISA (Abbott) and confirmed by Western blot. CD4 cell count was assessed by flow cytometry.
Statistical Methods
HIV-infected subjects with plaque were stratified into two groups based on degree of arterial inflammation, above and below the median aortic target-to-background ratio (TBR) value of 2.17. For each group, data on demographic and cardiovascular risk factors, HIV-disease related parameters, and atherosclerotic plaque morphologic features were analyzed. Comparison of atherosclerotic plaque morphology features between two groups of HIV-infected subjects with relatively higher and lower degrees of arterial inflammation was chosen to clearly delineate differences in plaque morphology between two subgroups with different physiology. Data were tested for normality by the Shapiro-Wilk test. Normally distributed data are presented as mean ± SD while non-normally distributed data are presented as median (95% CI). 95% CI are also reported for dichotomous parameters. Between-group comparisons of variables in the aforementioned categories were performed using the Student’s t-test for normally distributed variables and using the Wilcoxon rank sum test for non-normally distributed variables. Dichotomous parameters were compared between groups using Chi squared test. High-risk atherosclerotic plaque morphology features were non-normally distributed and compared by the Wilcoxon test. Descriptive data are provided for the mean and median for these parameters to provide additional information. Number of overall plaque segments per patient, number of calcified plaque segments per patient, and per patient coronary artery calcium score were also compared by the Wilcoxon test. Additionally, descriptive data on demographic and CVD risk parameters including arterial inflammation are provided for the 12 HIV-infected subjects who underwent coronary CTA, in addition to 18F-FDG-PET, but who were found to have no coronary atherosclerotic plaque. In a sensitivity analysis, we re-categorized the high and low inflammation groupings above and below the median including in the analysis those 12 subjects found not to have coronary atherosclerotic plaque on coronary CTA. We classified these subjects as having no high-risk morphology plaque and performed a two-group comparison of high-risk morphology plaque among those in the larger group with relatively higher and lower levels of arterial inflammation. In an exploratory analysis among those subjects with plaque, we split the subjects into those with and without at least 1 high-risk low attenuation coronary atherosclerotic plaque and compared arterial inflammation between groups using the Wilcoxon test. Finally, among those patients with plaque, a multivariate linear regression model was constructed for one key measure of high-risk atherosclerotic plaque morphology - number of low attenuation plaques per subject. Independent variables included in the model included arterial inflammation category (higher versus lower), as well as those parameters which were significant on univariate comparison between high and low TBR groups (e.g. gender and CD4 count). Also included as independent variables were age, duration of HIV, and LDL cholesterol levels, which could logically be expected to contribute to high-risk atherosclerotic plaque morphology based on current understandings of atherogenesis. These parameters were important to control for in assessing the independent relationship of TBR to high-risk plaque morphology. A second model was constructed for number of low attenuation plaques per patient in the group with plaque substituting nadir CD4 count for CD4 count as an independent variable.
Results
12 HIV-infected subjects had no coronary atherosclerotic plaque on coronary CTA. Among these subjects, TBR was 2.26 (2.05, 2.60) median (95% CI), similar to the TBR in the group of 41 HIV-infected subjects with coronary atherosclerotic plaque, which was 2.17 (1.45, 3.40) (p = 0.42). Demographic data on the 12 subjects without plaque are reported in Table 1. HIV-infected subjects with plaque were stratified into two groups according to degree of arterial inflammation (quantified by aortic target-to-background ratio, or TBR). TBR in the group below the median was 1.95 (1.45, 2.17) and TBR in the group above the median was 2.41 (2.18, 3.42).
Table 1.
Baseline Demographics
| All HIV- infected subjects with plaque (n= 41) |
HIV-infected subjects with plaque and aortic TBR below median (n = 21) |
HIV-infected subjects with plaque and aortic TBR above median (n=20) |
p-value for comparison by TBR stratification among those with plaque |
HIV-infected subjects without plaque (n=12) |
|
|---|---|---|---|---|---|
| Demographics and Cardiometabolic Risk Parameters | |||||
| Age, y | 51± 5 | 52 ± 5 | 50 ± 5 | 0.25 | 47 ± 5 |
| Gender | |||||
| % Male | 80% (33/41) (65%, 91%) | 67% (14/21) (43%, 85%) | 95% (19/20) (75%, 100%) | 83% (10/12) (52%, 98%) | |
| % Female | 20% (8/41) | 33% (7/21) | 5% (1/20) | 0.02* | 17% (2/12) |
| Race | |||||
| % White | 66% (27/41) | 67% (14/21) | 65% (13/20) | 58% | |
| % Black | 20% (8/41) | 19% (4/21) | 20% (4/20) | 33% | |
| % Hispanic | 7% (3/41) | 9% (2/21) | 5% (1/20) | 8% | |
| % Other | 7% (3/41) | 5% (1/21) | 10% (2/20) | 0.78 | 0% |
| Family history of premature heart disease, % * ± | 39% (7/18) (17%, 64%) | 40% (2/5) (5%, 85%) | 38% (5/13) (14%–68%) | 0.95 | 17% (1/6) (0.4%, 64%) |
| Current smoker, % | 30% (12/40) (17%, 47%) | 40% (8/20) (19%, 64%) | 20% (4/20) (6%, 44%) | 0.16 | 0% (0/9) (0%, 34%) |
| Diabetes mellitus, % | 10% (4/40) (3%, 24%) | 10% (2/20) (1%, 32%) | 10% (2/20) (1%, 32%) | 1.0 | 0% (0/9) (0%, 34%) |
| Use of antihypertensive medications, % | 20% (8/40) (9%, 36%) | 10% (2/20) (1%, 32%) | 30% (6/20) (12%, 54%) | 0.11 | 11% (1/9) (0.3%, 48%) |
| 10 year Framingham risk for coronary heart disease, % ±± | 6 (0.5, 16) | 7 (0.5, 16) | 5 (1, 16) | 0.90 | 3 (0.5, 6) |
| Body mass index (BMI), kg/m2 | 25.9 ± 3.9 | 26.2 ± 4.8 | 25.6 ± 2.9 | 0.64 | 25.7 ± 4.4 |
| Systolic blood pressure, mm Hg | 122 ± 17 | 122 ± 17 | 122 ± 17 | 0.98 | 116 ± 12.3 |
| LDL cholesterol level, mg/dL | 114 ± 27 | 116 ± 27 | 111 ± 28 | 0.58 | 91 ± 29 |
| HDL cholesterol level, mg/dL | 52 ± 17 | 50 ± 15 | 54 ± 18 | 0.53 | 49 ± 15 |
| Triglyceride level, mg/dL | 113 (55, 494) | 121 (55, 248) | 106 (67, 498) | 0.68 | 137 ± 62 |
| HIV disease-related parameters | |||||
| Duration of HIV infection, y | 15 ± 6 | 17 ± 6 | 14 ± 6 | 0.10 | 13 ± 5 |
| Duration of antiretroviral therapy, y | 11 ± 4 | 12 ± 4 | 11 ± 4 | 0.39 | 7 ± 3 |
| current PI treatment, % | 47% (19/40) (32%, 64%) | 45% (9/20) (23%, 69%) | 50% (10/20) (27%, 73%) | 0.75 | 56% (5/9) (21%, 86%) |
| Current NRTI treatment, % | 95% (38/40) (83%, 99%) | 95% (19/20) (75%, 100%) | 95% (19/20) (75%, 100%) | 1.0 | 100% (9/9) (66%, 100%) |
| Current NNRTI treatment, % | 47% (19/40) (32%, 64%) | 50% (10/20) (27%, 73%) | 45% (9/20) (23%, 69%) | 0.75 | 22% (2/9) (3%, 60%) |
| nadir CD4 count (self-reported), cells/mm3 | 199 (10, 700) | 200 (50, 620) | 120 (10, 700) | 0.23 | 222 (9, 300) |
| CD4 count, cells/mm3 | 582 ± 270 | 669 ± 278 | 485 ± 232 | 0.04* | 483 ± 227 |
| HIV RNA level (viral load), copies/ml | 47** (47, 355) | 47 (47, 355) | 47 (47, 204) | 0.32 | 47 (47, 528) |
| undetectable viral load, % | 78% (25/32) (60%, 91%) | 71% (12/17) (44%, 90%) | 87% (13/15) (60%, 98%) | 0.26 | 71% (5/7) (29%, 96%) |
Data on family history of premature heart disease not available on all studied subjects.
Family history of premature heart disease is based on the NCEP definition - i.e. CHD in male first degree relative <55 years; CHD in female first degree relative <65 years.
Framingham 10-year risk scores are calculated as per the NHLBI website (http://www.nhlbi.nih.gov/guidelines/cholesterol/risk_tbl.htm).
47 = lower limit of detection of HIV RNA level (viral load), according to assay
Demographic and Cardiometabolic Risk Parameters among HIV-Infected Subjects with Plaque Stratified by Degree of Arterial Inflammation
Age, race, family history of premature coronary heart disease, smoking status, diabetes, use of antihypertensive medications, body mass index, systolic blood pressure, and lipid levels were not significantly different between the groups stratified by TBR status (Table 1). No patients were receiving statin therapy. Slightly fewer subjects in the higher TBR group were women (20% versus 33%, p = 0.02). The Framingham 10-year risk scores were low and not statistically different between the groups (5% (1, 16) median (95% CI) versus 7% (0.5, 16), p = 0.9; higher TBR group versus lower TBR group) (Table 1).
HIV-Disease Related Parameters Among HIV-Infected Subjects with Plaque Stratified by Degree of Arterial Inflammation
Duration of HIV and duration of ART were similar between groups, as were current use therapy with a protease inhibitor (PI), nucleoside reverse transcriptase inhibitor (NRTI), and non-nucleoside reverse transcriptase inhibitor (NNRTI) (Table 1). There was no statistically significant difference between nadir CD4 count (120 cells/mm3 (10, 700) median (95% CI) versus 200 cells/mm3 (50, 620), p = 0.23; higher TBR group versus lower TBR group) and viral load (47 copies/ml (47, 204) versus 47 copies/ml (47, 355), p = 0.32; higher TBR group versus lower TBR group) between groups. Current CD4 count was lower in the higher TBR group relative to the lower TBR group (485 ± 232 cells/mm3 versus 669 ± 278 cells/mm3 mean ± SD, p = 0.04) (Table 1).
Measures of High-risk Coronary Plaque Morphology among HIV-Infected Subjects with Plaque Stratified by Degree of Arterial Inflammation
There was no significant difference between groups in the overall number of plaques per patient (3 (1, 11) median (95%CI) [3.6 ± 2.9] mean ± SD versus 2 (1, 11) [3.6 ± 2.9] p=0.93, lower TBR group versus higher TBR group). Among subjects in the higher TBR group, there was an increased number of low attenuation plaques per subject (0 (0, 2) median (95% CI) [0.5 ± 0.7] mean ± SD versus 0 (0, 1) [0.1 ± 0.3], p = 0.02) and an increased number of high-risk morphology features in the most high-risk coronary atherosclerotic plaque per subject (1 (0, 2) [1.3 ± 0.6] versus 1 (0, 2) [0.8 ± 0.6], p = 0.02). A representative image of a plaque with low attenuation and positive remodeling in a subject with increased TBR above the median is shown (Figure 1). The higher TBR group also featured an increased percentage of subjects with at least one low attenuation plaque (40% versus 10%, p = 0.02) and an increased percentage of subjects with at least one plaque characterized by both low attenuation and positive remodeling (35% versus 10%, p = 0.04) (Figure 2). There was not a statistically significant difference in the number of positively remodeled plaques per subject (1 (0, 5) [1.8 ± 1.4] versus 1 (0, 7) [1.6 ± 1.8], p = 0.50; higher TBR group versus lower TBR group) or in the percentage of subjects with at least one positively remodeled plaque in the TBR groups (85% versus 67%, p = 0.17; higher TBR group versus lower TBR group)(Table 2). There was no significant difference between groups in the number of calcified plaque segments per patient (p=0.85) or in the overall coronary artery calcium score (p=0.88). In a sensitivity analysis among all HIV-infected subjects (including those without plaque), the number of low attenuation plaques per patient remained significantly higher in the high TBR group (p=0.04). For this analysis, subjects without plaque were categorized as having no high-risk morphology plaque. In a further exploratory analysis among those subjects with plaque, we split the subjects into those with and without at least 1 high-risk low attenuation coronary atherosclerotic plaque. There was no significant difference in arterial inflammation between the group with and without at least one high-risk plaque of this nature (p=0.24).
Figure 1.
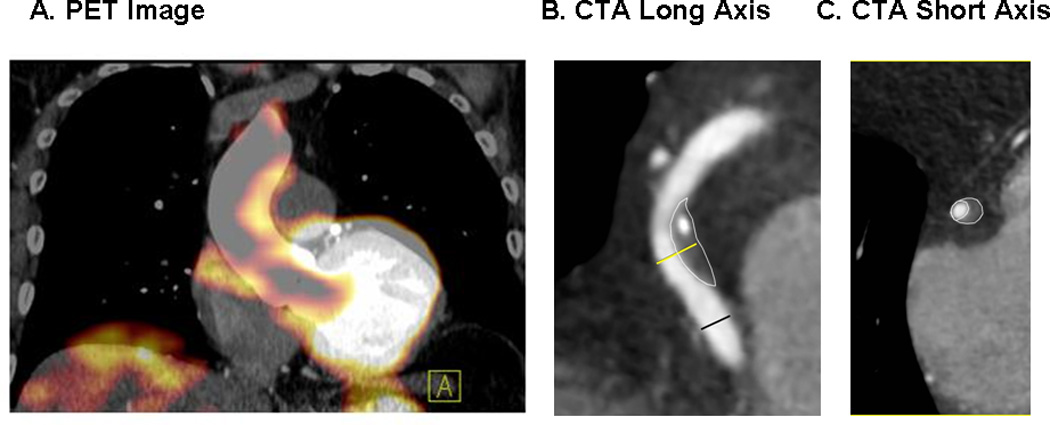
Panel A. Representative 18F-FDG-PET image from a subject with increased (2.36) aortic target to background ratio (TBR) demonstrated on coronal imaging. Panels B and C. Representative coronary computed tomography (CTA) long-axis (B) and short-axis (C) images in the same subject as Panel A, demonstrating a low attenuation, positively remodelled plaque with spotty calcification in the mid right coronary. The diameter measurements at the plaque (yellow line) and at distal normal reference segment (black line) are shown for the determination of remodeling index. Short axis image is at level of plaque.
Figure 2.
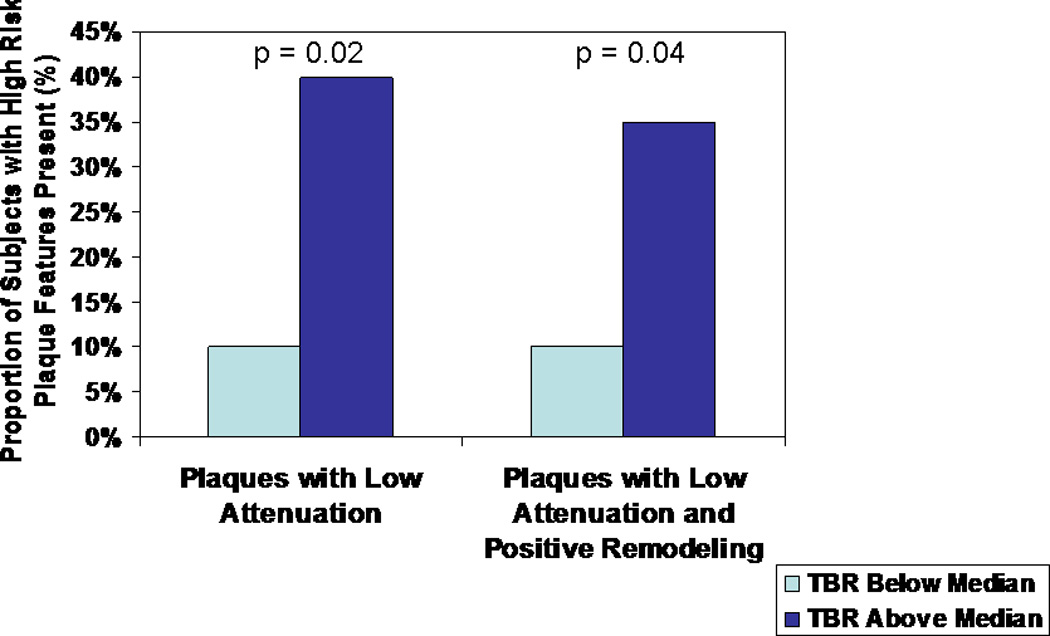
Comparison of percent of subjects with at least one low attenuation coronary atherosclerotic plaque and comparison of percent of subjects with at least one coronary atherosclerotic plaque characterized by both low attenuation and positive remodeling among HIV-infected subjects with aortic TBR (a measure of arterial inflammation) above and below the group median.
Table 2.
Measures of high risk coronary plaque morphology among HIV-infected subjects grouped by degree of atherosclerotic plaque inflammation
| All HIV-infected subjects with plaque (n= 41) |
HIV-infected subjects with plaque and aortic TBR below median (n = 21) |
HIV-infected subjects with plaque and aortic TBR above median (n=20) |
p-value for comparison by TBR stratification among those with plaque |
|
|---|---|---|---|---|
| number of low attenuation plaques per subject | 0 (0, 2) 0.3 ±0.6 |
0 (0, 1) 0.1 ± 0.3 |
0 (0, 2) 0.5 ± 0.7 |
0.02 |
| number of positively remodeled plaques per subject | 1 (0, 6.9) 1.7 ± 1.6 |
1 (0, 7) 1.6 ± 1.8 |
1 (0, 5) 1.8 ± 1.4 |
0.50 |
| % of subjects with at least one low attenuation plaque | 24% (10/41) (12%, 40%) | 10% (2/21) (1%, 30%) | 40% (8/20) (19%, 64%) | 0.02 |
| % of subjects with at least one positively remodeled plaque | 76% (31/41) (60%, 88%) | 67% (14/21) (43%, 85%) | 85% (17/20) (62%, 97%) | 0.17 |
| % of subjects with at least one plaque that is either low attenuation or positively remodeled | 78% (32/41) (62%, 89%) | 67% (14/21) (43%, 85%) | 90 % (18/20) (68%, 99%) | 0.06 |
| % of subjects with at least one plaque that is both low attenuation and positively remodeled | 22% (9/41) (11%, 38%) | 10% (2/21) (1%, 30%) | 35% (7/20) (15%, 59%) | 0.04 |
| number of high risk morphology features (0, 1, or 2) in the most high risk coronary atherosclerotic plaque per subject | 1 (0,2) 1.0 ± 0.7 |
1 (0, 2) 0.8 ± 0.6 |
1 (0, 2) 1.3 ± 0.6 |
0.02 |
Multivariate Linear Regression Modeling for High-risk Coronary Plaque Morphology among HIV-infected Subjects with Plaque
A multivariate linear regression model for number of low attenuation plaques per subject - a key measure of high-risk coronary atherosclerotic plaque morphology - was performed. In this model, TBR categorization (higher versus lower) was entered as per the a priori hypothesis of this study. Independent variables in the model included those parameters which were significant on univariate comparison between high and low TBR groups (e.g. gender and CD4 count), as well as parameters which could be expected to contribute to high-risk morphology coronary plaque - namely, age, duration of HIV, and LDL cholesterol levels. In this model controlling for traditional and HIV-specific risk factors, TBR categorization remained significantly associated with high-risk morphological features, with 0.35 more low attenuation plaques/patient in the higher TBR group (ß estimate 0.35, SE 0.11, p = 0.004; R2 for overall model 0.40, p = 0.03). Age, gender, LDL cholesterol level, and CD4 count were not significant in the model. In contrast, duration of HIV was associated with increased high-risk morphology in the model (ß estimate 0.04, SE 0.02, p = 0.05) (Table 3a). TBR grouping was also significantly related to high-risk plaque morphology when nadir CD4 count was substituted for CD4 count in the model (Table 3b).
Table 3.
| Multivariate model for number of low attenuation plaques per subject, a measure of high risk coronary plaque morphology, among subjects with plaque R2 for overall model 0.40, p = 0.03 | |||
|---|---|---|---|
| β estimate | standard error | p value | |
|
median TBR (high TBR group) |
0.35 | 0.11 | 0.004 |
|
age (years) |
0.02 | 0.02 | 0.42 |
|
Duration of HIV infection (years) |
0.04 | 0.02 | 0.05 |
|
gender (male gender) |
0.08 | 0.12 | 0.49 |
|
LDL cholesterol level (mg/dl) |
−0.002 | 0.004 | 0.61 |
|
CD4 count (cells/mm3) |
0.0006 | 0.0004 | 0.17 |
| a. Multivariate model for number of low attenuation plaques per subject, a measure of high risk coronary plaque morphology among subjects with plaque R2 for overall model 0.49, p = 0.007 | |||
|---|---|---|---|
| β estimate | standard error | p value | |
|
median TBR (high TBR group) |
0.32 | 0.09 | 0.002 |
|
age (years) |
0.02 | 0.02 | 0.31 |
|
duration of HIV infection (years) |
0.03 | 0.02 | 0.06 |
|
gender (male gender) |
0.01 | 0.11 | 0.91 |
|
LDL cholesterol level (mg/dl) |
−0.003 | 0.003 | 0.37 |
|
nadir CD4 count, self-reported (cells/mm3) |
−0.0003 | 0.0005 | 0.56 |
Discussion
Recent data suggest that both arterial inflammation and high-risk coronary plaque morphology are increased in HIV-infected patients without known CVD and minimal traditional CVD risk factors 9,10. Increased arterial inflammation and high-risk coronary plaque morphology have been independently linked to increased CVD risk in non-HIV populations7,8,11,13. However, degree of arterial inflammation has not before been compared to presence of high-risk coronary plaque morphology features among HIV-infected patients with subclinical coronary atherosclerotic plaque. The significant association we show in this study between high-risk morphology and arterial inflammation among subjects with plaque provides further evidence for the important role of inflammation as a potential factor contributing to increased CVD among HIV-infected patients.
In this study, we identified a group of HIV-infected patients without known clinical CVD or significant traditional CVD risk factors, who demonstrated coronary plaque on CTA. We stratified this group into two groups - one group with a relatively lower degree of arterial inflammation (aortic TBR on 18F-FDG-PET below the median) and another group with a relatively higher degree of vascular inflammation (aortic TBR on 18F-FDG-PET above the median) for comparison of plaque features. Our analysis shows these two groups were relatively balanced on key demographic/cardiometabolic risk parameters (e.g. age, race, family history of premature coronary heart disease, smoking status, diabetes, use of antihypertensive medications, body mass index, systolic blood pressure, and lipid levels) and key HIV-disease related parameters (duration of HIV, duration of ART, use of main categories of ART, viral load, and nadir CD4 count). There were subtle but statistically significant differences in gender composition and current CD4 count between groups. Of note, the median TBR in the group as a whole was increased at 2.17, consistent with our prior published data in a subset of these patients9, and therefore the differences between the two groups are relative in nature, among a group with overall increases in TBR. In our prior study, we showed that increased TBR was common among HIV-infected patients9. We did not select for patients with known CVD, but rather studied HIV-infected subjects without clinical symptoms or increased traditional risk, representing the large majority of those at increased risk in contemporary HIV cohorts1,2, increasing the generalizability of our findings. Results might differ among those with clinical CVD or increased traditional risk factors.
Though the groups of HIV-infected patients with higher and lower levels of arterial inflammation in our study were largely balanced on traditional CVD risk factors HIV disease-specific factors and overall number of plaques per patient, there were key differences between groups in measures of high-risk coronary atherosclerotic plaque morphology. We demonstrate for the first time that HIV-infected patients with increased arterial inflammation on 18F-FDG-PET (aortic TBR above the median) have a higher number of low attenuation plaques per subject. Moreover, we show that among the high TBR group, a higher percentage of subjects have at least one low attenuation plaque. Pathologic studies have shown that low attenuation plaques contain lipid-rich necrotic cores infiltrated by macrophages14. Such studies lend biologic plausibility to the relationship we observed in our HIV-infected subjects between 18F-FDG-PET uptake - reflecting atherosclerotic macrophage infiltration - and increased number/heightened prevalence of low attenuation coronary atherosclerotic plaques.
Modeling for number of low attenuation plaques per subject as a measure of high-risk plaque morphology, we further show that the relationship with aortic TBR categorization (lower versus higher) remains significant even when controlling for age, gender, LDL cholesterol level, duration of HIV infection, and CD4 count or nadir CD4 count. Such modeling reinforces the significance of the observed relationship between arterial inflammation and high-risk plaque morphology. In this modelling, gender, age, LDL and CD4 count were not significantly related to high-risk plaque morphology. Although there were differences between CD4 and gender in the TBR categorization, these differences do not appear to drive the relationship with high-risk morphology based on our modelling. In contrast, duration of HIV infection remained related to high-risk morphology suggesting more high-risk plaque with longer duration of disease. Further studies, with longitudinal follow up of HIV patients, are required to fully elucidate the nature of the relationship between duration of HIV infection, degree of arterial inflammation, and development of high-risk morphology, rupture-prone coronary atherosclerotic plaque.
Another important observation from our study is that among subjects in the higher TBR group, an increased number of high-risk morphology features was noted in the most high-risk coronary atherosclerotic plaque per subject. Moreover, we noted an increased percentage of subjects with at least one plaque characterized by both low attenuation and positive remodelling in the higher TBR group. In a seminal study by Motoyama et. al. among non-HIV patients, acute coronary syndrome developed in 22% of patients with coronary atherosclerotic plaques characterized by both low attenuation and positive remodeling versus 3.7% of patients with plaques characterized by either low attenuation or positive remodeling versus 0.5% of patients with plaques characterized by neither low attenuation nor positive remodeling11. In our study, the relationship between prevalence of coronary atherosclerotic plaque characterized by two high-risk morphologic features, on the one hand, and elevated arterial inflammation, on the other hand suggests a potential for arterial inflammation to predict CVD risk among HIV-infected subjects.
Of note, we also separately report arterial inflammation and demographic parameters among the subgroup of HIV-infected subjects found to have no coronary atherosclerotic plaque on coronary CTA. In this group, TBR was increased, similar to that in the larger group with plaque. These data suggest that a high degree of arterial inflammation may provide the substrate for the development of high-risk morphology coronary atherosclerotic plaque. Although there was insufficient power to formally compare demographic parameters in the group without plaque to those in the group with plaque, we note that the group without plaque had relatively lower levels of traditional CVD risk factors. This may suggest that not only degree of arterial inflammation but potential synergy with traditional CVD risk factors may be important for the development of high-risk coronary atherosclerotic plaque. Moreover, we found in an exploratory analysis among those HIV-infected subjects with plaque, that there was no significant difference in arterial inflammation between those with and without at least one high-risk low attenuation coronary plaque. These data are consistent with the hypothesis increased systemic arterial inflammation may provide a substrate for the development of high-risk morphology coronary plaque, but not vice-versa.
One limitation of this study is that arterial inflammation quantified in the aorta is compared to atherosclerotic plaque vulnerability in the coronary arteries. Of note, direct measurement of arterial inflammation in the coronaries by 18F-FDG-PET, particularly beyond the proximal segments, has not yet been standardized due to analytic challenges in interpretation. However, aortic inflammation has been correlated in non-HIV populations to CVD events 7,8 and is thought to represent the degree of inflammation throughout the arterial tree. Another limitation is that the cross-sectional nature of the current study prevents definitive conclusions as to whether arterial inflammation predisposes to the development of high-risk morphology features of coronary atherosclerotic plaque. Moreover, it limits interpretation as to the significance of duration of arterial inflammation to the development of high-risk morphology plaque. The sample size of the study, 41 subjects, is relatively small but robust relative to other 18F-FDG-PET studies and sufficient to detect significant differences in high-risk coronary atherosclerotic plaque morphology between HIV-infected subjects with higher and lower levels of arterial inflammation. The small size of the study may have limited power to detect differences in other parameters between the groups and the relationship between such parameters to the number of low attenuation plaques per patient in multivariate linear regression modeling.
In conclusion, these data suggest that increased arterial inflammation is associated with high-risk coronary plaque morphology among HIV-infected patients on ART with good virological control and low Framingham Risk Score. Further studies are needed to determine whether immune activation and attendant arterial inflammation in HIV renders coronary atherosclerotic plaque more likely to rupture, resulting in myocardial infarction. If so, clinical strategies aimed at reducing the magnitude and chronicity of arterial inflammation may be tested for their potential to stabilize coronary atherosclerotic plaques in HIV-infected patients.
Acknowledgments
Grant Support: This work was conducted with the support of the NIH R01 HL095123 to Dr. Grinspoon and NIH K23 HL092792 to Dr. Lo.
Footnotes
Clinical Trial Registration Number: NCT00965185
Author contributions: Study design (A.T., J.L., M.Z., S.G.), data collection (A.T., J.L., M.Z., E.M., J.I., M.M., B.W., U.H., S.A., S.G.), data interpretation (A.T., J.L., M.Z., E.M., B.W., S.A., S.G.), drafting of manuscript (A.T., U.H., S.G.), critical revision of manuscript (A.T., J.L., M.Z., E.M., J.I., M.M., B.W., U.H., S.A., S.G.). All authors have read and approved the text submitted.
Disclosures: None related to this work.
References
- 1.Freiberg MS, Chang CC, Kuller LH, et al. HIV Infection and the Risk of Acute Myocardial Infarction. JAMA Intern Med. 2013 Mar 4;:1–9. doi: 10.1001/jamainternmed.2013.3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Triant VA, Lee H, Hadigan C, Grinspoon SK. Increased acute myocardial infarction rates and cardiovascular risk factors among patients with human immunodeficiency virus disease. J Clin Endocrinol Metab. 2007 Jul;92(7):2506–2512. doi: 10.1210/jc.2006-2190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Duprez DA, Neuhaus J, Kuller LH, et al. Inflammation, coagulation and cardiovascular disease in HIV-infected individuals. PLoS One. 2012;7(9):e44454. doi: 10.1371/journal.pone.0044454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kelesidis T, Kendall MA, Yang OO, Hodis HN, Currier JS. Biomarkers of microbial translocation and macrophage activation: association with progression of subclinical atherosclerosis in HIV-1 infection. J Infect Dis. 2012 Nov 15;206(10):1558–1567. doi: 10.1093/infdis/jis545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baker J, Hullsiek KH, Singh A, et al. CROI. Atlanta, GA: 2013. Monocyte Activation, but not T cell Activation, Predicts Progression of Coronary Artery Calcium in a Contemporary HIV Cohort; p. 66. LB. [Google Scholar]
- 6.Tawakol A, Migrino RQ, Bashian GG, et al. In vivo 18F-fluorodeoxyglucose positron emission tomography imaging provides a noninvasive measure of carotid plaque inflammation in patients. J Am Coll Cardiol. 2006 Nov 7;48(9):1818–1824. doi: 10.1016/j.jacc.2006.05.076. [DOI] [PubMed] [Google Scholar]
- 7.Figueroa ALAA, Truong QA, Corsini E, MacNabb MH, Lavendar ZR, et al. Measurement of Arterial Activity on Routine FDG PET/CT Images Improves Prediction of Risk of Future Cardiovascular Events. Journal of the American College of Cardiology: Cardiovascular Imaging. 2013 doi: 10.1016/j.jcmg.2013.08.006. In Press. [DOI] [PubMed] [Google Scholar]
- 8.Rominger A, Saam T, Wolpers S, et al. 18F-FDG PET/CT identifies patients at risk for future vascular events in an otherwise asymptomatic cohort with neoplastic disease. J Nucl Med. 2009 Oct;50(10):1611–1620. doi: 10.2967/jnumed.109.065151. [DOI] [PubMed] [Google Scholar]
- 9.Subramanian S, Tawakol A, Burdo TH, et al. Arterial inflammation in patients with HIV. JAMA. 2012 Jul 25;308(4):379–386. doi: 10.1001/jama.2012.6698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zanni MV, Abbara S, Lo J, et al. Increased Coronary Atherosclerotic Plaque Vulnerability by Coronary Computed Tomography Angiography in HIV-Infected Men. AIDS. 2013 Jan 16; doi: 10.1097/QAD.0b013e32835eca9b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Motoyama S, Sarai M, Harigaya H, et al. Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J Am Coll Cardiol. 2009 Jun 30;54(1):49–57. doi: 10.1016/j.jacc.2009.02.068. [DOI] [PubMed] [Google Scholar]
- 12.Kitagawa T, Yamamoto H, Horiguchi J, et al. Characterization of noncalcified coronary plaques and identification of culprit lesions in patients with acute coronary syndrome by 64-slice computed tomography. JACC Cardiovasc Imaging. 2009 Feb;2(2):153–160. doi: 10.1016/j.jcmg.2008.09.015. [DOI] [PubMed] [Google Scholar]
- 13.Motoyama S, Kondo T, Sarai M, et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J Am Coll Cardiol. 2007 Jul 24;50(4):319–326. doi: 10.1016/j.jacc.2007.03.044. [DOI] [PubMed] [Google Scholar]
- 14.Virmani R, Burke AP, Farb A, Kolodgie FD. Pathology of the Vulnerable Plaque. J Am College of Cardiology. 2006;47:C13–C18. doi: 10.1016/j.jacc.2005.10.065. [DOI] [PubMed] [Google Scholar]


