Abstract
Gastric hamartomatous inverted polyps (GHIP) are difficult to diagnose accurately because of inversion into the submucosal layer. GHIP are diagnosed using the pathological characteristics of the tumor, including the fibroblast cells, smooth muscle, nerve components, glandular hyperplasia, and cystic gland dilatation. Although Peutz-Jeghers syndrome, juvenile polyposis, and Cowden disease are hereditary, it is rare to encounter 2 cases of monostotic and asymptomatic gastric hamartomas. The pathogeneses of hamartomatous inverted polyps and inverted hyperplastic polyps remain controversial because of the paucity of reported cases. There are 3 hypotheses regarding the pathogenesis of complete gastric inverted polyps. Based on our experience with 2 successive, rare GHIP cases, we affirm the hypothesis that after a hamartomatous change occurs in the submucosal layer, some of these components are exposed to the gastric mucosa and, consequently, form a hypertrophic lesion. In Case 1, our hypothesis explains why a tiny hypertrophic change was first detected on the top of the submucosal tumor using a detailed narrow band imaging-magnified endoscopy. There was no confirmation that the milky white mucous and calcification structures were exuding directly from the biopsy site like Case 1, and in Case 2 the presence of this mucous was indirectly confirmed during an endoscopic submucosal dissection (ESD). Regarding the pathogenesis of GHIP, a submucosal hamartomatous change may occur prior to the growth of hypertrophic portions. An en bloc resection using ESD is recommended for treatment.
Keywords: Hamartomatous inverted polyps, Hypotheses of pathogenesis, Milky white mucous, Endoscopic submucosal dissection, Pathological findings
Core tip: The pathogeneses of hamartomatous inverted polyps (HIP) and inverted hyperplastic polyps remain controversial. Our experience with 2 rare successive cases of HIP suggests that after a hamartomatous change occurs in the submucosal layer, some of these components are exposed to the gastric mucosa and, consequently, form a hypertrophic lesion. An en bloc resection by endoscopic submucosal dissection is recommended for treatment.
INTRODUCTION
Gastric hamartomatous polyps (GHP) have been reported as GHIP because of inversion into the submucosal layer of the main body[1]. GHIP located in the submucosal layer and gastric submucosal tumors (SMT) are rarely reported, making an accurate diagnosis difficult[2]. GHIP are diagnosed using the pathological characteristics of the tumor, including the fibroblast cells, smooth muscle proliferation, nerve components, vasoformative tissue, glandular hyperplasia, and cystic gland dilatation[2,3]. GHIP > 20 mm in diameter are recommended to be treated with an en bloc resection with laparoscopic wedge resection of the stomach or endoscopic submucosal resection (ESD) rather than a conventional polypectomy, endoscopic mucosal resection (EMR) with piecemeal resection, or subtotal or total gastrectomy because there is a paucity of studies regarding the long term outcomes of GHIP and these polyps occasionally contain paracancerous portions (approximately 20% of GHIP)[2-4]. Furthermore, there are some controversial reports regarding the pathogenesis or natural growth history of GHIP. One hypothesis suggests that the submucosal hamartomatous or heterogenic gastric mucosa components grow up to the mucosal surface; however, another hypothesis suggests that hyperplastic components or the mucosa is inverted into the submucosa[5,6].
Here, we report 2 cases (31-year-old and 65-year-old patients) that will aid in the understanding of the pathogenesis and growth process of GHIP.
CASE REPORT
A 31-year-old woman was found to have a gastric SMT in the great curvature of the upper body of the upper GI tract during her medical exam. She underwent an esophagogastroduodenoscopy (EGD) and endoscopic ultrasound (EUS) (radial scan, 20 MHz). The EGD showed a gastric SMT measuring 30 mm in diameter in the greater curvature of upper body with bridging folds and a tiny reddish spot on the top (Figure 1A). With the exception of the tiny reddish spot, the SMT surface was normal gastric mucosa. Using narrow band imaging-magnified endoscopy (NBI-ME), the tiny reddish area was shown to be dilated with banded marginal crypt epithelium that differed from the surrounding mucosa and consisted of small round pits of normal fundic glands (Figure 1B). A biopsy specimen taken from the tiny reddish spot showed hyperplastic gastric mucosa. Milky white mucous and calcification structures were found to be exuding from the biopsy site (Figure 1C). After draining the mucous, a solid portion with small cystic changes was found inside the SMT wall using forceps (Figure 1D). An EUS (radial scan, 20 MHz) before the biopsy revealed a heterogeneous tumor with a cystic area and calcification spots with acoustic shadow (Figure 2A). After the biopsy and draining of the mucous, we conducted an EUS from inside the SMT by filling the tumor cavity with distilled water and inserting the EUS probe (Figure 2B). The EUS from inside the SMT cavity showed 5 specific structural layers (high-low-high-low-high). The EUS findings suggested that the 5 layers (high-low-high-low-high) were mucosa, muscularis mucosa, submucosa, muscularis mucosa, and mucosa, similar to a reverse layer pattern. The patient had no clinical symptoms, no family history, and no laboratory data abnormalities. Based on the EGD and EUS findings, including the heterogeneous echo pattern, small cystic structure, and spotty calcification, we suspected the presence of GHIP and lymphangioma with inflammatory changes. As it was difficult to make a definitive diagnosis and disprove the likelihood of a paracancerous lesion, we believed that an en bloc resection of the SMT was necessary to obtain an accurate diagnosis. After obtaining written informed consent from the patient, we resected the SMT by endoscopic submucosal dissection (ESD). A hematoxylin and eosin (HE) stain of the resected SMT is shown in Figure 2C (× 20) and Figure 2D (× 100). Histologically, the 5 structural layers (high-low-high-low-high) shown in the EUS and with the HE stain (× 20, Figure 2B and C red circle) consisted of a normal gastric mucosal layer of columnar epithelium of pyloric glands and fundic glands (high) (Figure 2D, yellow arrow), an immature fibroblast cell rich layer (low) (Figure 2D, black arrow), a pyloric gland rich layer (high) (Figure 2D, yellow arrow), muscularis mucosa (low) (Figure 2D, black arrow), and a normal gastric mucosal layer of columnar epithelium (high) (Figure 2D, yellow arrow) from inside the stomach. As shown in Figure 1C, the milky white mucous and calcification structures were not confirmed. The high echo solid portion of the SMT with tiny low echoic cystic spots (Figure 2B, blue circle) were shown to be the proliferation of pseudo-pylorus glands, cystic glands without cytological atypia, fibroblast cells, smooth muscle proliferation, nerve elements, glandular hyperplasia, and cystic gland dilatation.
Figure 1.
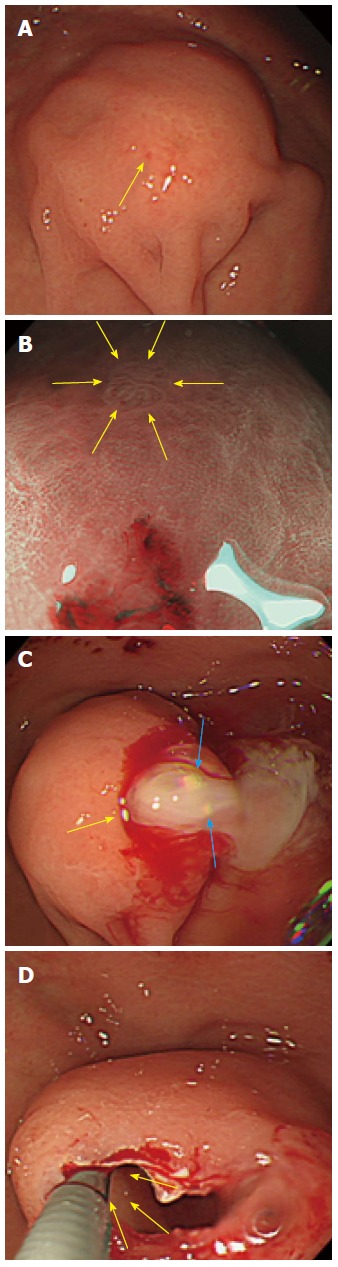
Esophagogastroduodenoscopy showed a submucosal tumor. A: The esophagogastroduodenoscopy (EGD) showed a gastric submucosal tumor (SMT) measuring 30 mm in diameter in the greater curvature of upper body with bridging folds and a tiny reddish spot on top (yellow arrow); B: Using NBI-ME, the tiny area was observed to be dilated with banded marginal crypt epithelium (yellow arrows); C: Milky white mucous and calcification structures (blue arrow) were exuding from the biopsy site (yellow arrow); D: The solid portion inside the wall of the SMT with small cystic changes were exposed using forceps (yellow arrow).
Figure 2.
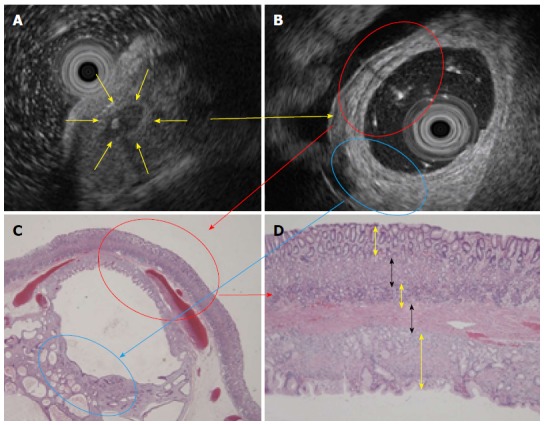
Endoscopic ultrasound before and after the biopsy and histology. A: An EUS before biopsy revealed a heterogeneous tumor with a cystic area and calcification spot (yellow arrow); B: An endoscopic ultrasound (EUS) from inside the submucosal tumor (SMT) filled with distilled water and an inserted EUS probe showed 5 specific layers (red circle); C: An HE stain (× 20) also showed the 5 structural layers. The hyperechoic portion showed proliferation of pseudo-pylorus glands and cystic gland dilatation without cytological atypia (blue circle); D: The 5 layers consisted of a normal mucosal layer (yellow arrow), immature fibroblasts (black arrow), pyloric glands (yellow arrow), muscularis mucosa (black arrow) and another normal mucosal layer (yellow arrow) (HE stain × 100).
A 65-year-old man was admitted to treat a SMT measuring 40 mm in diameter in the greater curvature of the middle body. The size of the SMT had increased from 20 mm in diameter, which had been reported 5 years previously; in addition, on the top of the SMT, there was a reddish and erosive nodular surface measuring 10 mm in diameter that had been previously reported to measure only a few mm in diameter (Figure 3A, B). An EUS (radial scan, 20 MHz) revealed a heterogeneous tumor with small spotty cystic areas, a large anechoic cystic part with papillary structures, and 5 specific structural layers (high-low-high-low-high) as its wall (Figure 3C, D). Considering the EGD and EUS results and the clinical progression of the SMT size, the GHIP may have been paracancerous. Therefore, an en bloc resection with ESD was performed to obtain an accurate diagnosis (Figure 4A). During the ESD (as shown in Figure 4B, C), milky white components similar to those in Case 1 were observed. The lesion was resected as a complete en bloc specimen with a sufficient safety margin from the tumor (Figure 4D).
Figure 3.
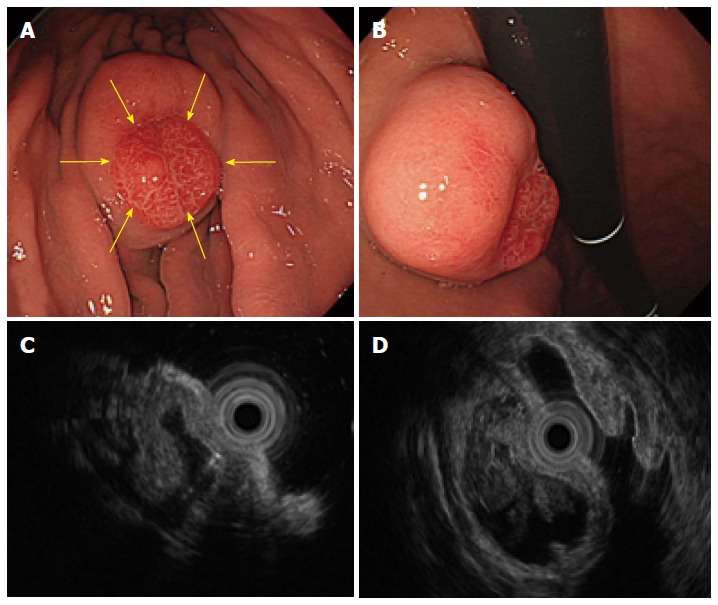
An submucosal tumor measuring 40 mm in diameter was found in the greater curvature of the middle body. A and B: The size of the submucosal tumor (SMT) had increased from the 20 mm in diameter that was reported 5 years before. In addition, a reddish erosive part 10 mm in diameter was found on the top of the SMT (yellow arrows). C and D: An endoscopic ultrasound revealed a heterogeneous tumor with small spotty cystic areas, a large anechoic cystic part, with papillary structures and 5 specific structural layers as its wall.
Figure 4.
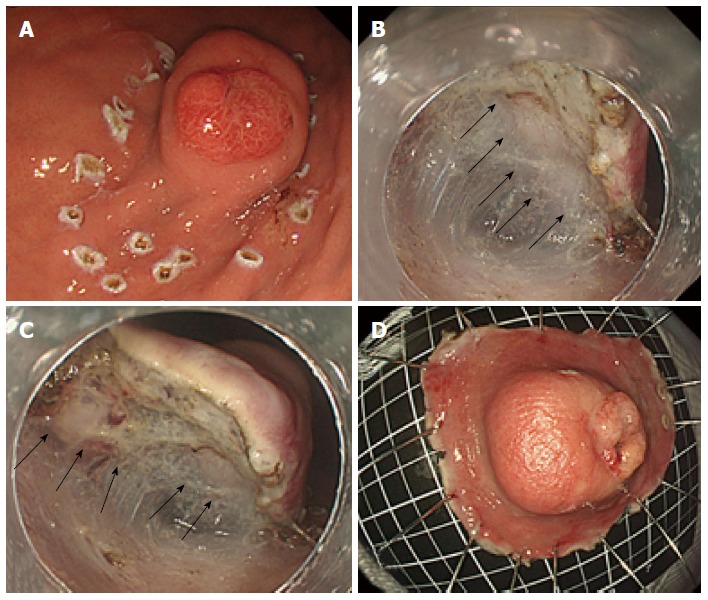
Endoscopic submucosal dissection to resect the submucosal tumor. A: An en bloc resection using endoscopic submucosal dissection (ESD) was performed to obtain an accurate diagnosis; B and C: Similar to Case 1, milky white components were observed during the ESD (black arrows); D: The lesion was completely resected with a sufficient safety margin.
Similar to Case 1, the 5 structural layers (high-low-high-low-high) shown in the EUS and HE stain (× 20) (Figure 5A, B, yellow circle) consisted of a normal gastric mucosal layer, immature fibroblast cell rich layer, pyloric gland rich layer, muscularis mucosa, and normal gastric mucosa. The high echo solid portion with tiny low echoic cystic spots (Figure 5A, B and C, blue circle) showed proliferation of pseudo-pylorus glands, cystic glands with cytological atypia, fibroblast cells, smooth muscle proliferation, nerve elements, glandular hyperplasia, and cystic gland dilatation (Figure 5D). This SMT was inverted into the submucosal layer, as highlighted by the red bold arrow (Figure 5B).
Figure 5.
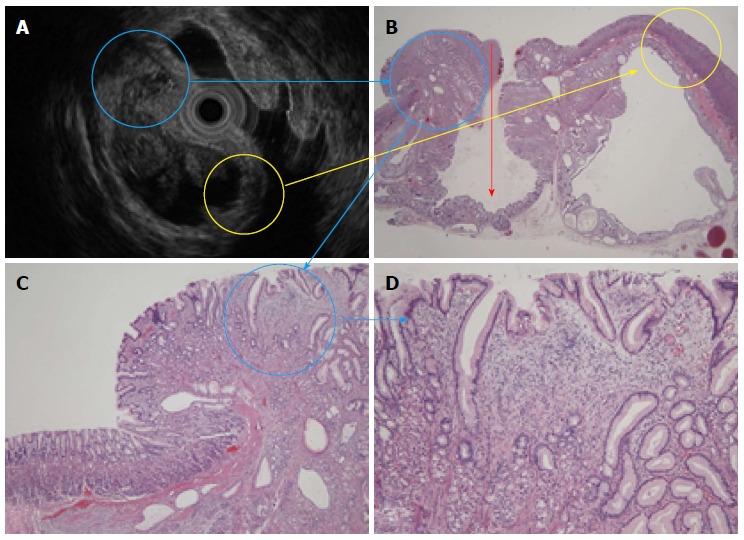
An endoscopic ultrasound and histological comparison. A: An endoscopic ultrasound (EUS) revealed a heterogeneous tumor; B: The 5 layers shown in the EUS and HE stain (× 20) (yellow circle) consisted of a normal mucosa layer, immature fibroblast cells, pyloric glands, muscularis mucosa, and another normal mucosa layer. The surface mucosa was inverted into the submucosal layer (red arrow); C and D: The hyperechoic solid portion had tiny low echoic cystic spots (blue circle) and showed the proliferation of pseudo-pylorus glands, cystic glands, fibroblast cells, smooth muscle, and nerve elements.
DISCUSSION
GHPs are pathologically defined by cystic dilated hypertrophic pseudo-pylorus gland proliferation and smooth muscle located in the submucosal layer with branching from the proliferation of smooth muscle bundles[7]. Although Peutz-Jeghers syndrome, juvenile polyposis and Cowden disease are hereditary, it is rare to encounter 2 cases of monostotic and asymptomatic gastric hamartomas[8-12]. GHIP is difficult to diagnose accurately without endoscopic resection and pathological investigation because of its inverted grown into the submucosal layer and the paucity of case reports[2]. However, GHIP may be identified by the exuding of milky mucous, calcifications from the biopsy site, and specific EUS findings. There have been reports that GHIP was diagnosed by EUS examination and the observation of hyper echoic lesions, including small cystic hypo echoic spots (a heterogeneous tumor), in the 3rd layer (submucosal layer)[3]. In Case 1, an EUS examination from inside the tumor cavity was performed by inserting an EUS probe after cutting a portion of the tumor surface; forceps were used to obtain additional detailed information about the wall structure (high-low-high-low-high). A previous Japanese case report mentioned that EUS-fine needle aspiration was useful for diagnosing GHIP with 82% sensitivity, 100% specificity, and an overall 92% success rate. In addition, a differential diagnosis of GHIP and an aberrant pancreas with cystic pancreatic ducts was reported to be particularly difficult[2]. Pathological findings of inverted ectopic gastric pseudo-pyloric glands in the submucosal layer is vital for diagnosing GHIP; therefore, an en bloc resection using an ESD method was a viable treatment option in our 2 reported cases.
The pathogeneses of hamartomatous inverted polyps (HIP) and inverted hyperplastic polyps (IHP) remain controversial because of the paucity of documented cases[13]. A previous report suggested that GHIP should be classified as IHP because the rupture, thinning, and frailty of muscularis mucosae is caused by repeated erosions reaching the muscularis mucosae and gastric glands that are inverted into the submucosal layer, and the obstruction of a drain outlet for produced mucous leads to cystic dilatation of ectopic gastric glands[3]. There are currently 3 hypotheses regarding the pathogenesis of complete gastric inverted polyps. One hypothesis contends that after a hyperplastic change occurs on the gastric mucosa surface, these components invert into the submucosal layer, and the submucosal glandular or cystic elements transform into an SMT-like tumor. Another hypothesis speculates that after a hamartomatous change occurs in the submucosal layer, some of these components are exposed to the gastric mucosa, thereby forming a hypertrophic lesion. A third hypothesis argues that a hyperplastic polyp is incidentally formed on the top of mucosa from the original submucosal hamartomatous change. Based on our experience with 2 rare cases of GHIP, we affirm that the second hypothesis is likely the basis for the GHIP pathogenesis. Case 1 supports this hypothesis because a tiny hypertrophic change was first detected on the top of the SMT using detailed NBI-ME. We found that a portion of the submucosal hamartomatous components were exposed to the surface of the SMT, with a tiny hypertrophic change. In Case 2, the SMT grew to 20 mm in diameter in the greater curvature of the middle body after it had been originally detected 5 years prior; in addition, a tiny reddish and erosive nodular spot in the hypertrophic mucosa on top of the SMT had grown to 10 mm in diameter. These data confirm the chronological changes in the hypertrophic mucosa on top of the SMT.
No report has confirmed the milky white mucous and calcification structures exuding from the biopsy site directly and confirmed indirectly during the ESD[14]. Although GHPs are benign tumors, approximately 20% coexist with paracancerous or cancerous areas[15]; therefore, it is important to diagnosis these polyps, and the recommended treatment is an en bloc resection using an ESD method. There was an atypical portion with high density nuclei in Case 2; thus, it was suggested that en bloc resection using an ESD method was pathologically more important than a piecemeal resection with EMR.
In conclusion, as submucosal hamartomatous changes of GHIP might occur prior to the development of secondary hypertrophic portions from our experiences of two rare cases, more cases are needed to be stored to evaluate the pathogenesis of GHIP. For treatment, an en bloc resection using an ESD method is recommended.
ACKNOWLEDGMENTS
We thank Dr. Makoto Oryu, Professor, for providing technical and editorial assistance.
Footnotes
P- Reviewers: Geng X, Hou YY, Shimi SM S- Editor: Wen LL L- Editor: A E- Editor: Wang CH
References
- 1.Lin JC, Huang TY, Shih YL, Chang YM, Chu HC, Chang WK, Hsieh TY, Chen RJ. Management of Symptomatic Hamartomatous Polyps in Stomach: Analysis of a Single Center Experience. J Med Sci. 2013;33:29–36. [Google Scholar]
- 2.Aoki M, Yoshida M, Saikawa Y, Otani Y, Kubota T, Kumai K, Wakabayashi G, Omori T, Mukai M, Kitajima M. Diagnosis and treatment of a gastric hamartomatous inverted polyp: report of a case. Surg Today. 2004;34:532–536. doi: 10.1007/s00595-004-2761-1. [DOI] [PubMed] [Google Scholar]
- 3.Iishi H, Tatsuta M, Okuda S. Clinicopathological features and natural history of gastric hamartomatous polyps. Dig Dis Sci. 1989;34:890–894. doi: 10.1007/BF01540275. [DOI] [PubMed] [Google Scholar]
- 4.Odashima M, Otaka M, Nanjo H, Jin M, Horikawa Y, Matsuhashi T, Ohba R, Koizumi S, Kinoshita N, Takahashi T, et al. Hamartomatous inverted polyp successfully treated by endoscopic submucosal dissection. Intern Med. 2008;47:259–262. doi: 10.2169/internalmedicine.47.0360. [DOI] [PubMed] [Google Scholar]
- 5.Carfagna G, Pilato FP, Bordi C, Barsotti P, Riva C. Solitary polypoid hamartoma of the oxyntic mucosa of the stomach. Pathol Res Pract. 1987;182:326–330. doi: 10.1016/S0344-0338(87)80067-5. [DOI] [PubMed] [Google Scholar]
- 6.Hanada M, Takami M, Hirata K, Kishi T, Nakajima T. Hyperplastic fundic gland polyp of the stomach. Acta Pathol Jpn. 1983;33:1269–1277. doi: 10.1111/j.1440-1827.1983.tb02172.x. [DOI] [PubMed] [Google Scholar]
- 7.Itoh K, Tsuchigame T, Matsukawa T, Takahashi M, Honma K, Ishimaru Y. Unusual gastric polyp showing submucosal proliferation of glands: case report and literature review. J Gastroenterol. 1998;33:720–723. doi: 10.1007/s005350050161. [DOI] [PubMed] [Google Scholar]
- 8.Grisendi A, Lonardo A. Solitary Peutz-Jeghers type polyp of the stomach. Endoscopy. 1990;22:153. doi: 10.1055/s-2007-1012826. [DOI] [PubMed] [Google Scholar]
- 9.Kuwano H, Takano H, Sugimachi K. Solitary Peutz-Jeghers type polyp of the stomach in the absence of familial polyposis coli in a teenage boy. Endoscopy. 1989;21:188–190. doi: 10.1055/s-2007-1012939. [DOI] [PubMed] [Google Scholar]
- 10.Tatsuta M, Okuda S, Tamura H, Taniguchi H. Gastric hamartomatous polyps in the absence of familial polyposis coli. Cancer. 1980;45:818–823. doi: 10.1002/1097-0142(19800215)45:4<818::aid-cncr2820450435>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 11.Sakadamis AK, Ballas KD, Fardellas JG, Papanikolaou A. A solitary gastric Peutz-Jeghers type polyp: report of a case. Surg Today. 2001;31:517–520. doi: 10.1007/s005950170113. [DOI] [PubMed] [Google Scholar]
- 12.Iida M, Yao T, Watanabe H, Itoh H, Iwashita A. Fundic gland polyposis in patients without familial adenomatosis coli: its incidence and clinical features. Gastroenterology. 1984;86:1437–1442. [PubMed] [Google Scholar]
- 13.Park do Y, Lauwers GY. Gastric polyps: classification and management. Arch Pathol Lab Med. 2008;132:633–640. doi: 10.5858/2008-132-633-GPCAM. [DOI] [PubMed] [Google Scholar]
- 14.Yamashita M, Hirokawa M, Nakasono M, Kiyoku H, Sano N, Fujii M, Koyama T, Yoshida S, Sano T. Gastric inverted hyperplastic polyp. Report of four cases and relation to gastritis cystica profunda. APMIS. 2002;110:717–723. doi: 10.1034/j.1600-0463.2002.1101005.x. [DOI] [PubMed] [Google Scholar]
- 15.Oh SJ, Oh CA, Kim DH, Choi MG, Noh JH, Sohn TS, Kim KM, Bae JM, Kim S. Adenocarcinoma derived from gastric hamartomatous polyps. J Korean Surg Soc. 2011;81:419–422. doi: 10.4174/jkss.2011.81.6.419. [DOI] [PMC free article] [PubMed] [Google Scholar]


