Abstract
Citric acid is a weak organic acid found in the greatest amounts in citrus fruits. This study examined the effect of citric acid on endotoxin-induced oxidative stress of the brain and liver. Mice were challenged with a single intraperitoneal dose of lipopolysaccharide (LPS; 200 μg/kg). Citric acid was given orally at 1, 2, or 4 g/kg at time of endotoxin injection and mice were euthanized 4 h later. LPS induced oxidative stress in the brain and liver tissue, resulting in marked increase in lipid peroxidation (malondialdehyde [MDA]) and nitrite, while significantly decreasing reduced glutathione, glutathione peroxidase (GPx), and paraoxonase 1 (PON1) activity. Tumor necrosis factor-alpha (TNF-α) showed a pronounced increase in brain tissue after endotoxin injection. The administration of citric acid (1–2 g/kg) attenuated LPS-induced elevations in brain MDA, nitrite, TNF-α, GPx, and PON1 activity. In the liver, nitrite was decreased by 1 g/kg citric acid. GPx activity was increased, while PON1 activity was decreased by citric acid. The LPS-induced liver injury, DNA fragmentation, serum transaminase elevations, caspase-3, and inducible nitric oxide synthase expression were attenuated by 1–2 g/kg citric acid. DNA fragmentation, however, increased after 4 g/kg citric acid. Thus in this model of systemic inflammation, citric acid (1–2 g/kg) decreased brain lipid peroxidation and inflammation, liver damage, and DNA fragmentation.
Key Words: : antioxidant activity, citric acid, cytokines, dietary supplementation, peripheral infection, systemic inflammation
Introduction
Oxidative stress is the term used to indicate the imbalance between reactive oxygen species and antioxidant defense mechanisms. Under physiological conditions, reactive oxygen species play integral roles in intracellular signaling, physiological immunological responses, and gene expression. Reactive oxygen metabolites can be generated in excess from many sources. The most important source is the leakage of electrons from the mitochondrial electron transport chain to generate superoxide radical (O2•−). Other sources are xanthine oxidase, NADPH oxidases, activated phagocytes, and nitric oxide synthases (NOSs). When excessively produced, however, these species could result in potential cellular and tissue damage. Being highly unstable molecules with unpaired electrons, reactive oxygen metabolites, such as superoxide radical and hydroxyl radical, react with the cellular membrane polyunsaturated fatty acids to form lipid peroxides, oxidize and cross-link proteins including enzymes, or oxidize DNA, with the potential to produce a harmful or even lethal event.1–3 Cellular defenses against free radicals and reactive oxygen species include enzymes, such as catalase, glutathione peroxidase (GPx), and superoxide dismutase, as well as nonenzymatic antioxidant mechanisms, for example, glutathione (GSH), ascorbic acid, carotenoids, and vitamin E.2,4 Oxidative stress occurs when redox homeostasis is tipped toward an overbalance of free radicals, due to either their overproduction or deficiencies in antioxidant defense.5 Oxidative stress has been implicated in the pathogenesis of numerous diseases, such as diabetes mellitus, cardiovascular disease, and neurodegenerative and psychiatric disorders.6,7 The brain is considered particularly vulnerable to oxidative damage because of its high oxygen utilization and hence generation of free radical byproducts, the high content of polyunsaturated lipids, the biomacromolecules most susceptible to oxidation, its modest antioxidant defenses, and the presence of redox-catalytic metals, such as iron and copper.7,8
Citric acid (2-hydroxy-1,2,3-propane-tricarboxylic acid) is a weak organic acid found in the greatest amounts in citrus fruits, such as lemon, grapefruit, tangerine, and orange. Lemon and lime juices are rich sources.9 It is used as a natural preservative and also to add an acidic (sour) taste to foods and soft drinks.10 Being a component of the tricarboxylic acid or Krebs cycle, citric acid is found in all animal tissues as an intermediary substance in oxidative metabolism. Studies indicated that citrate decreases lipid peroxidation and downregulates inflammation by reducing polymorphonuclear cell degranulation and attenuating the release of myeloperoxidase, elastase, interleukin (IL)-1β, and platelet factor 4.11–13 In vitro, citrate improved endothelial function by reducing the inflammatory markers and decreasing neutrophil diapedesis in hyperglycemia.14 Moreover, citric acid has been shown to reduce hepatocellular injury evoked in rats by carbon tetrachloride.15 Citric acid might thus prove of value in decreasing oxidative stress.
Thus, in view of the antioxidant and anti-inflammatory effects for citrate reported just now and since citrate anticoagulation has been employed in the critically ill patients, it looked pertinent to study the effect of citric acid administration on oxidative stress and tissue injury in a model of systemic inflammatory illness caused by intraperitoneal (i.p.) lipopolysaccharide (LPS) administration in mice. LPS is a constituent of the cell walls of gram-negative bacteria. When given systemically, LPS potently stimulates the immune cells in the periphery (through plasma membrane proteins, e.g., the toll like receptor 4 [TLR4] and CD14) to release proinflammatory cytokines, such as necrosis factor-alpha (TNF-α), IL-1β, and IL-6 in the periphery and brain. This results in the development of systemic and neuroinflammation.16–19 LPS-induced endotoxemia is a well-established model for infection with gram-negative bacteria and is widely used to study endotoxin effects on peripheral tissue/organs and the influence of systemic inflammation on the brain.
Materials and Methods
Animals
Swiss male albino mice that weigh 22–25 g (age 5–6 weeks) were used. Mice were obtained from animal house colony of the National Research Centre. Standard laboratory food and water were provided ad libitum. Animal procedures were performed in accordance with the Ethics Committee of the National Research Centre and followed the recommendations of the National Institutes of Health Guide for Care and Use of Laboratory Animals (Publication No. 85-23, revised 1985).
Drugs and chemicals
A purified, lyophilized Escherichia coli endotoxin (Serotype 055:B5; Sigma) was used; it was dissolved in sterile physiological saline, aliquoted, and frozen at −20°C. The same stock solutions were used for all experiments. Citric acid and all other chemicals were of analytical grade and were obtained from Sigma. The dose of LPS (200 μg/kg) and the time for tissue sampling were based on previous studies.20
Study design
Mice were randomly divided into five equal groups (six mice each). Mice were treated with either 0.2mL of: sterile physiological saline (group 1) or citric acid at doses of 1, 2, and 4 g/kg, orally (groups 2–4). Treatments were given just prior to endotoxin administration (LPS: 200 lg/kg, injected intraperitoneally, 0.1 mL). The fifth group received just the vehicle, no LPS (negative control). Mice were euthanized after 4 h of LPS or vehicle injection by decapitation under ether anesthesia, where the brain and liver of each mouse were then removed, washed with ice-cold phosphate-buffered saline (PBS; pH 7.4), weighed, and stored at −80°C until the biochemical analyses. The tissues were homogenized with 0.1 M PBS at pH 7.4, to give a final concentration of 0.1g/mL for the biochemical assays. Reduced GSH, malondialdehyde (MDA), nitric oxide (nitrite), GPx, and paraoxonase 1 (PON1) activity was determined in brain and liver tissues. TNF-α was measured in brain tissue. Alanine aminotransferase (ALT), aspartate aminotransferase (AST), and DNA fragmentation were measured in liver tissue.
Determination of lipid peroxidation, reduced GSH, and nitrite levels
Lipid peroxidation was assayed by measuring the level of MDA in brain tissue using the method of Ruiz-Larrea et al.21 Reduced GSH was determined in tissue by Ellman's method.22 Nitric oxide measured as nitrite was determined by using Griess reagent, according to the method of Moshage et al.23
Determination of GPx activity
GPx activity in supernatants was determined spectrophotometrically at 340 nm by the analysis of NADPH oxidation using glutathione peroxidase kit (Biodiagnostics).24 One unit of GPx activity is defined as the amount of protein that oxidized 1 mM NADPH per minute. The activity of GPx is expressed as mU/mL.
Determination of paraoxonase activity
Arylesterase activity of paraoxonase was measured spectrophotometrically in supernatants using phenyl acetate as a substrate.25,26 In this assay, arylesterase/paraoxonase catalyzes the cleavage of phenyl acetate resulting in phenol formation. The rate of formation of phenol is measured by monitoring the increase in absorbance at 270 nm at 25°C. The working reagent consisted of 20 mM Tris/HCl buffer (pH 8.0) containing 1 mM calcium chloride and 4 mM phenyl acetate as the substrate. Samples diluted 1:3 in buffer are added and the change in absorbance is recorded following a 20 s lag time. Absorbance at 270 nm was taken every 15 s for 120 s using a UV-Vis Recording Spectrophotometer (Shimadzu Corporation). One unit of arylesterase activity is equal to 1 μM of phenol formed per minute. The activity is expressed in kU/L, based on the extinction coefficient of phenol of 1310 M/cm at 270 nm, pH 8.0, and 25°C. Blank samples containing water are used to correct for the spontaneous hydrolysis of phenyl acetate.
Determination of TNF-α, DNA fragmentation, and liver enzymes
Tissue TNF-α was determined in brain tissue according to Chen et al.27 by enzyme-linked immunosorbent assay using TNF-α kits (Biosource International) and microtiter plate reader (Fisher Biotech). Quantitation of DNA fragmentation in liver tissue was done according to the method described by Gercel-Taylor.28 ALT and AST activities in liver were measured using commercially available kits (BioMérieux).29,30
Histological assessment of liver injury
The liver from each mouse was rapidly removed and fixed in freshly prepared 10% neutral buffered formalin, processed routinely, and embedded in paraffin. Sections of 5-μm thick were cut and stained by hematoxylin and eosin (H&E) for histopathological examination. All sections were investigated by the light microscope.
Immunohistochemistry for caspase-3 and inducible nitric oxide synthase
Paraffin-embedded liver sections were deparaffinized, and hydrated. Immunohistochemistry was performed with a mouse monoclonal caspase-3 and inducible nitric oxide synthase (iNOS) for detection of the caspase cleavage and iNOS activity. The paraffin sections were heated in a microwave oven (25 min at 720 W) for antigen retrieval and incubated with either anti-caspase or iNOS antibodies (1:50 dilution) overnight at 4°C. After washing with PBS, followed by incubation with biotinylated goat-anti-rabbit-immunoglobulin G secondary antibodies (1:200 dilution; Dako Corp.) and streptavidin/alkaline phosphatase complex (1:200 dilution; Dako) for 30 min at room temperature, the binding sites of antibody were visualized with DAB (Sigma). After washing with PBS, the samples were counterstained with H&E for 2–3 min, and dehydrated by transferring them through increasing ethanol solutions (30%, 50%, 70%, 80%, 95%, and 100% ethanol). Following dehydration, the slices were soaked twice in xylen at room temperature for 5 min, mounted, examined, and evaluated by high-power light microscope.31
Statistical analysis
Data are expressed as mean±standard error. Data were analyzed by one-way analysis of variance, followed by Duncan's multiple-range test for post hoc comparison of group means. Effects with a probability of P<.05 were considered to be significant.
Results
Effect of citric acid on LPS-induced oxidative stress
Lipid peroxidation
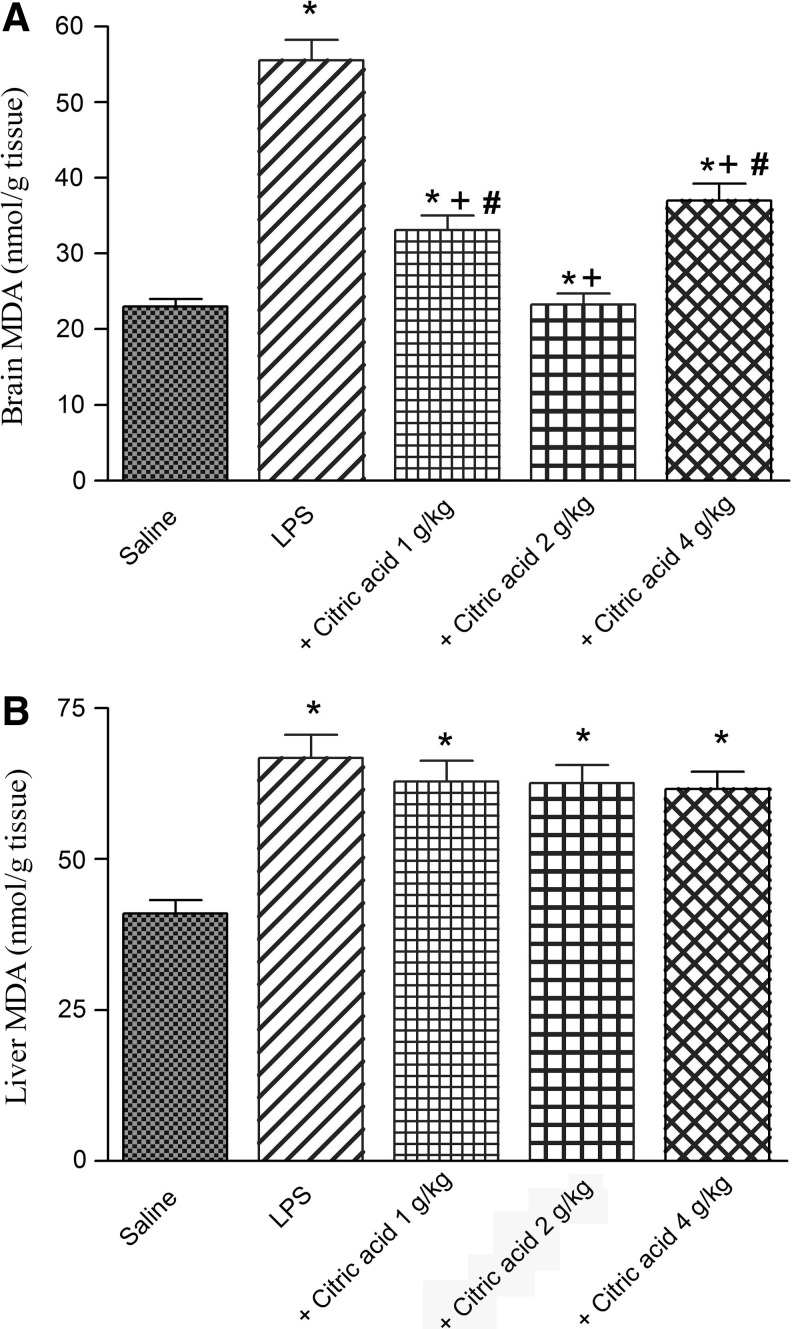
The administration of LPS resulted in a significant increase in the level of MDA in brain and liver tissues by 140.3% (23.1±1.0 vs. 55.5±2.7 nmol/g tissue) and 62.9% (66.8±3.8 vs. 41.0±2.2 nmol/g tissue), respectively, compared with the saline control group (Fig. 1A, B).
FIG. 1.
(A) Brain and (B) liver tissue concentrations of malondialdehyde (MDA: nmol/g tissue) in mice given lipopolysaccharide (LPS) or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control. +P<.05 versus LPS control group. #P<.05 versus LPS+2 g/kg of citric acid. p.o., per os.
Brain MDA was significantly decreased by 40.4% and 58% after treatment with 1 and 2 g/kg citric acid, respectively, compared with the LPS control group (33.1±1.9 and 23.3±1.4 vs. 55.5±2.7 nmol/g tissue). The higher dose of citric acid (4 g/kg) resulted in 33.3% inhibition of brain MDA (Fig. 1A).
In contrast, no significant effect on liver MDA has been observed after treatment with citric acid (1–4 g/kg; Fig. 1B).
Reduced GSH
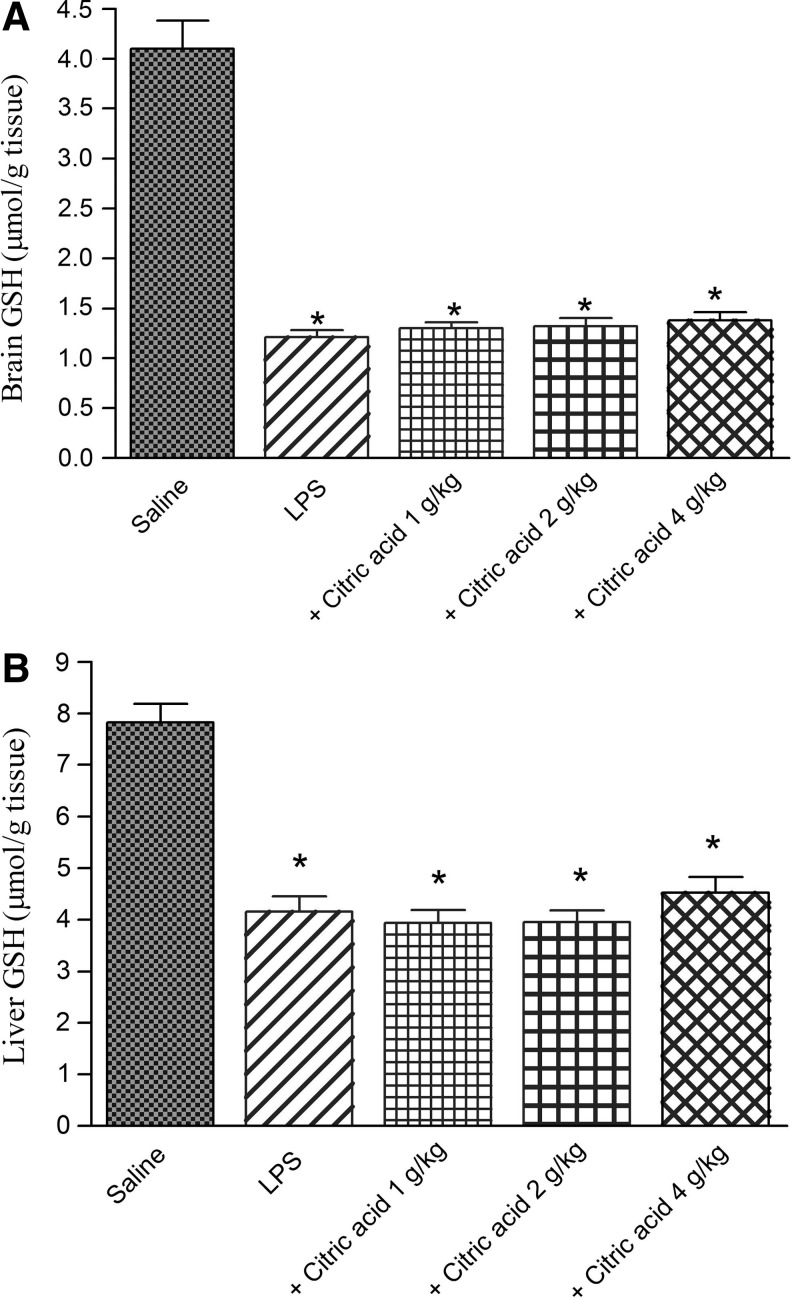
Following LPS challenge, the level of GSH decreased in brain and liver tissues by 72.1% (1.21±0.07 vs. 4.1±0.28 μmol/g tissue) and 46.9% (4.16±0.29 vs. 7.83±0.36 μmol/g tissue), respectively. Treatment with citric acid (1–4 g/kg) had no significant effect on brain or liver GSH (Fig. 2A, B).
FIG. 2.
(A) Brain and (B) liver tissue concentrations of reduced glutathione (GSH: μmol/g tissue) in mice given LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control.
Nitrite
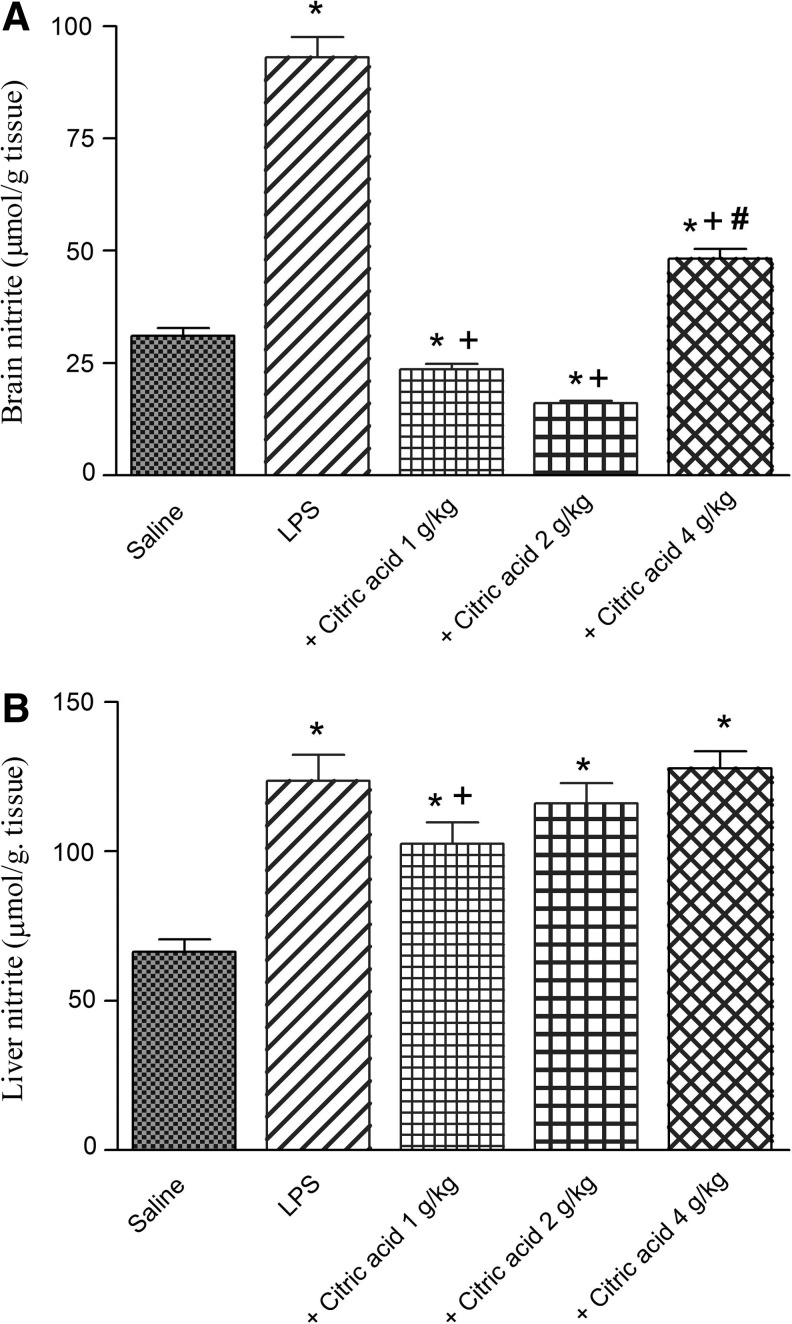
Marked and significant increase in brain nitrite was observed after treatment with LPS compared with the vehicle-treated group (93.0±4.6 vs. 31.0±1.8 μmol/g tissue; Fig. 3A). Similarly, the level of liver nitrite was significantly increased by 86.3% after LPS administration compared with vehicle-treated group (123.7±8.6 vs. 66.4±4.1 μmol/g tissue; Fig. 3B).
FIG. 3.
(A) Brain and (B) liver tissue concentrations of nitrite (μmol/g tissue) in mice after treatment with LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control. +P<.05 versus LPS control group. #P<.05 versus LPS+2 g/kg of citric acid.
In LPS-treated mice, the level of nitrite in brain tissue was markedly inhibited by 74.6% and 82.8% by citric acid at 1–2 g/kg (23.6±1.2 and 16.0±0.63 vs. 93.0±4.6 μmol/g tissue). Nitric oxide decreased by 48.1% after citric acid at 4 g/kg, compared with the LPS-only group (Fig. 3A).
In the liver, nitrite decreased significantly by 17% by citric acid given at 1 g/kg compared with the LPS control group. The higher doses of citric acid, however, failed to significantly alter nitrite in liver tissue (Fig. 3B).
GPx activity
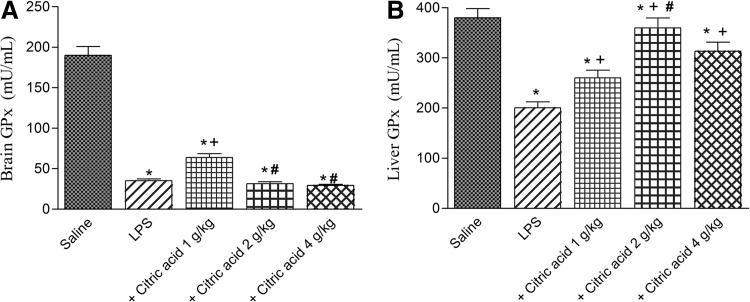
GPx activity showed a significant decrease in brain (by 81.6%) and liver tissues (by 47.3%) after LPS challenge compared with the vehicle-treated group. Brain GPx activity increased by 82.6% after treatment with 1 g/kg citric acid (P<.05) compared with the LPS control group (0.80±0.052 vs. 1.015±0.061 U/g tissue). No significant effect was observed in brain GPx activity after treatment with citric acid at 2 or 4 g/kg (Fig. 4A). On the other hand, liver GPx activity significantly increased by 29.7%, 79.6%, and 56.5% after treatment with 1, 2, and 4 g/kg of citric acid, respectively (Fig. 4B).
FIG. 4.
Glutathione peroxidase (GPx) activity in (A) brain and (B) liver of mice after LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control. +P<.05 versus LPS control group. #P<.05 versus LPS+1 g/kg of citric acid.
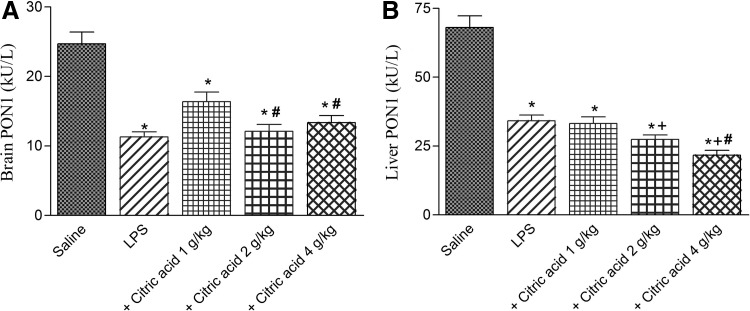
Paraoxonase activity
Paraoxonase activity significantly decreased in brain and liver tissues by 54.2% (11.3±0.7 vs. 24.7±1.8 kU/L) and 49.8% (34.2±2.1 vs. 68.1±4.2 kU/L), respectively, after LPS challenge (Fig. 5A, B). Brain PON1 activity increased by 44.9% following treatment with citric acid at 1 g/kg. Higher doses, however, failed to significantly alter PON1 activity (Fig. 5A). On the other hand, liver PON1 activity significantly decreased by 19.7% and 36.6% after treatment with citric acid at 2 and 4 g/kg, respectively, compared with the LPS control group (Fig. 5B).
FIG. 5.
Paraoxonase 1 (PON1) activity in mice (A) brain and (B) liver after treatment with LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control. +P<.05 versus LPS control group. #P<.05 versus LPS+1 g/kg of citric acid.
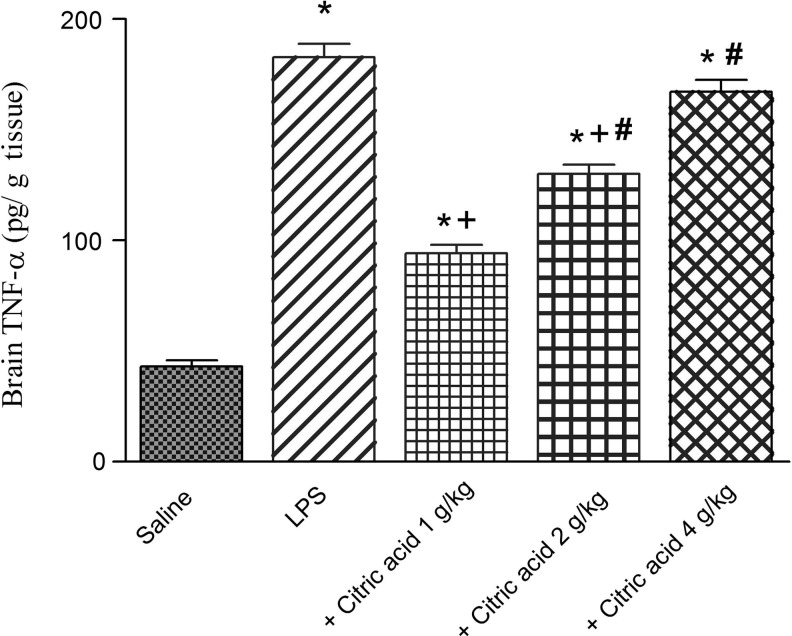
TNF-α in brain tissue
A pronounced increase in TNF-α in mice brain was observed following i.p. injection of LPS (324.9% increase: 182.7±6.2 vs. saline control value of 43.0±2.7 pg/g tissue). TNF-α showed a significant decrease by 48.4% and 28.8% after treatment with citric acid at 1 and 2 g/kg (93.3±3.8 and 130.0±4.3 vs. LPS control value of 182.7±6.2 pg/g tissue). The administration of citric acid at 4 g/kg failed to decrease the level of TNF-α (Fig. 6).
FIG. 6.
Brain tissue tumor necrosis factor-alpha (TNF-α; pg/g tissue) in mice given LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus the saline control. +P<.05 versus LPS control group. #P<.05 versus the LPS+1 g/kg of citric acid.
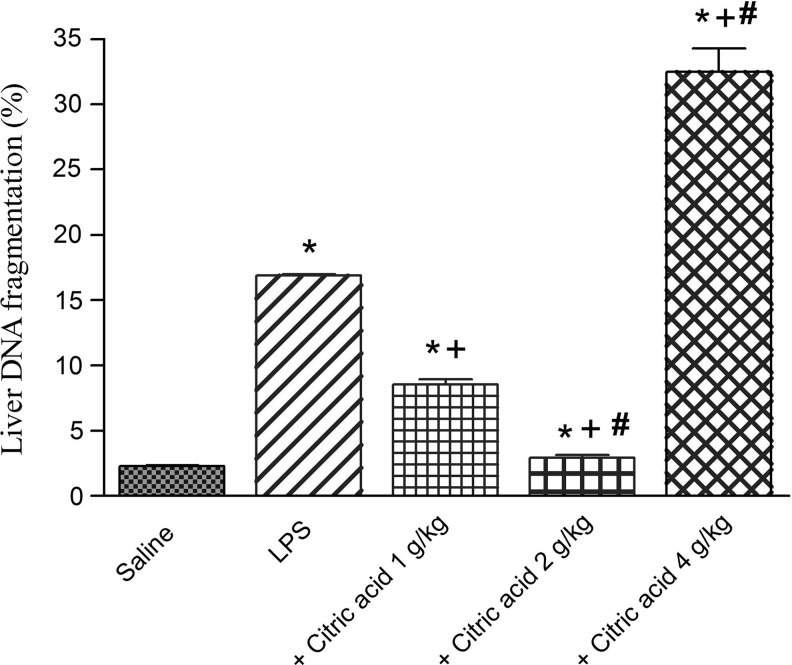
DNA fragmentation in the liver
DNA fragmentation in the liver was significantly and markedly increased by 633.5% after LPS injection compared with the vehicle-treated mice. It showed a 49.4% and 82.6% decrease after treatment with citric acid at 1 and 2 g/kg, respectively, compared with the LPS control value. However, a 92.5% increment in DNA fragmentation was observed after the highest dose of citric acid (4 g/kg; Fig. 7).
FIG. 7.
DNA fragmentation (%) in mice liver after LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control. +P<.05 versus LPS control group. #P<.05 versus LPS+1 g/kg of citric acid.
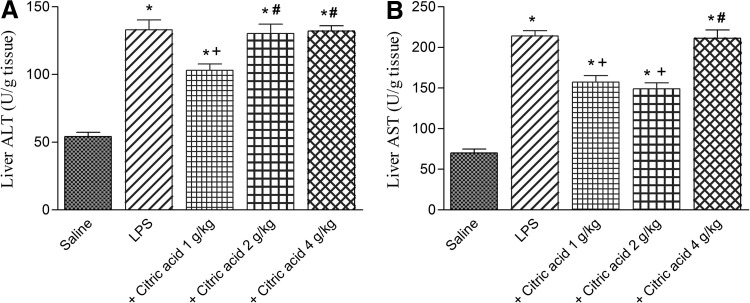
Liver transaminases
In LPS-treated mice liver, ALT and AST significantly increased by 145.4% and 204.8% compared with the saline-treated group. ALT significantly decreased by 22.5% after treatment with 1 g/kg of citric acid. The higher doses of citric acid, however, failed to significantly alter ALT in the liver of LPS-treated mice. Meanwhile, AST significantly decreased by 26.5% and 30.4% after treatment with 1 and 2 g/kg citric acid, respectively. The highest dose of citric acid, however, had no significant effect on liver AST in LPS-treated mice (Fig. 8A, B).
FIG. 8.
(A) Alanine aminotransferase (ALT) and (B) aspartate aminotransferase (AST) activities in mice liver after LPS or LPS+citric acid (1–4 g/kg, p.o.). *P<.05 versus saline control. +P<.05 versus LPS control group. #P<.05 versus LPS+1 g/kg of citric acid.
Histological results
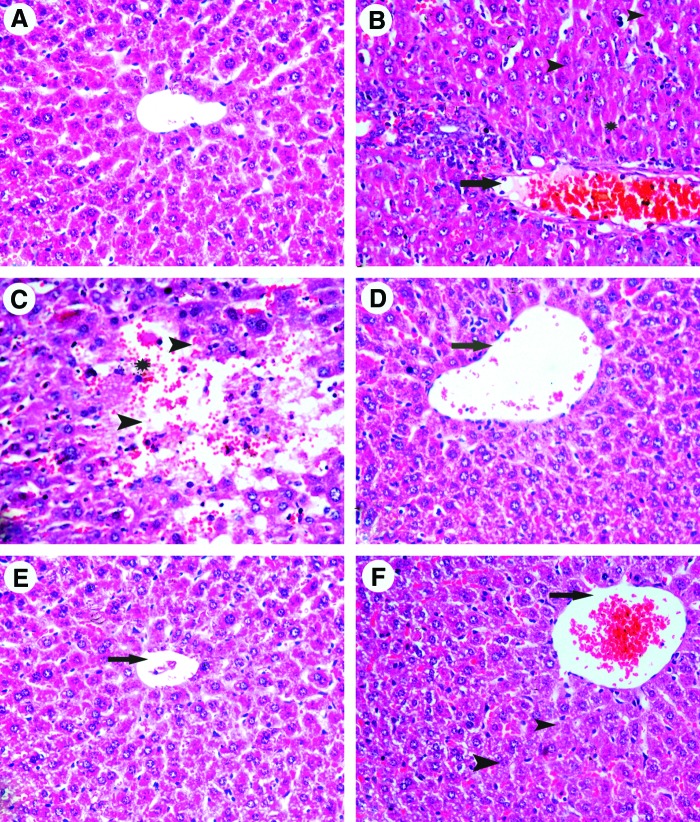
The control livers showed normal hepatic architecture with distinct hepatic cells, sinusoidal spaces, and a central vein (Fig. 9A).
FIG. 9.
Hematoxylin and eosin (H&E)–stained liver sections from mice treated with (A) saline (control). (B) LPS: inflammatory leukocytic cell infiltration around portal area (long arrow), necrosis (arrow head), dilated and congested blood sinusoids, and marinated nuclear chromatin (star). (C) LPS: focal necrotic area (arrow head), activated Kupffer cells, dilated and congested blood sinusoids, and pyknotic nuclei (star). (D) LPS+citric acid 1 g/kg: congestion of central vein (long arrow), dilated blood sinusoids, and few necrotic cells (arrowhead). (E) LPS+citric acid 2 g/kg: normal central vein (long arrow), minimally dilated blood sinusoids, and few Kupffer cells. (F) LPS+4 g/kg of citric acid: congestion of central vein (long arrow), signs of degeneration of hepatocytes, dilated congested blood sinusoids, and few Kupffer cells (H&E×400). Color images available online at www.liebertpub.com/jmf
Histological examination of the liver from LPS-treated mice revealed mononuclear cell infiltrations, bile duct proliferation in the periportal areas, and minimal enlargement in the periportal areas. In the LPS group we also observed dilatation and congestion of the central vein and blood sinusoids that showed numerous Kupffer cells. Hepatocytes exhibited necrotic changes in the form of small pyknotic nuclei with condensed or marginated chromatin, lack of nucleoli, and strongly acidophilic cytoplasm (Fig. 9B, C).
On the other hand, histological examination of liver sections from mice treated with LPS+citric acid at 1 g/kg showed nearly normal hepatic architecture. The hepatic lobules appeared with prominent central vein with less sinusoidal dilatation and decreased number of Kupffer cells compared with the LPS-only-treated group (Fig. 9D).
The improvement in histological appearance was more pronounced after treatment with citric acid at 2 g/kg, evidenced in normal appearance of liver lobules with strains of hepatocytes compared with section of LPS groups (Fig. 9E).
In contrast, sections from mice treated with LPS and 4 g/kg of citric acid showed mild improvement with dilated portal areas. The hepatocytes exhibited some degree of histological regeneration with less sinusoidal dilatation and with decreased number of Kupffer cells and less necrotic cells (Fig. 9F).
Caspase-3 and iNOS immunoreactivity
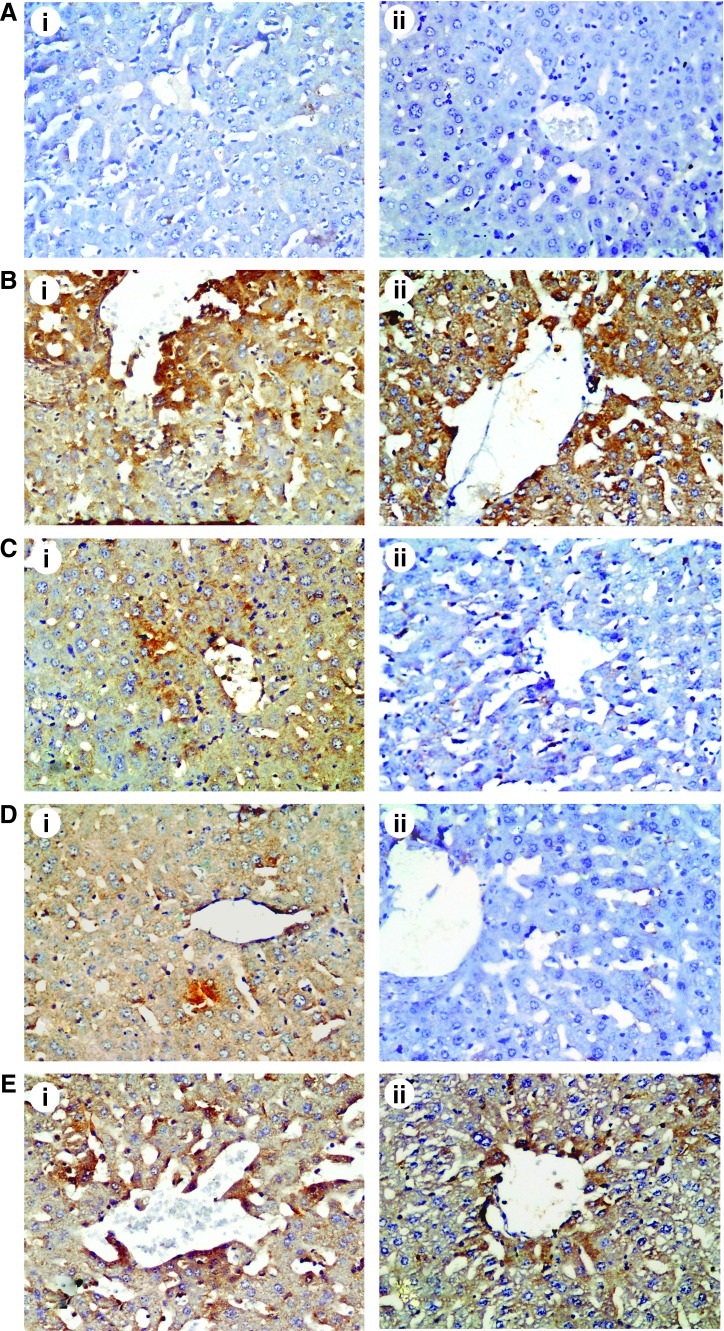
Activated caspase-3 labeling was specific in delineating morphologically apoptotic cells. Caspase-3 and iNOS expression was localized in the cytoplasm of hepatocytes. There was negligible caspase-3 (Fig. 10A-i) and iNOS (Fig. 10A-ii) immunopositivity in the livers of vehicle-treated mice. After treatment with LPS strong expression of caspase-3 (Fig. 10B-i) and iNOS (Fig. 10B-ii) was observed compared with the vehicle control group. In these sections, caspase-3 and iNOS immunoreactivity was observed mainly around central vein.
FIG. 10.
The effect of LPS and citric acid treatment on hepatic caspase-3 (i) and inducible nitric oxide synthase (iNOS) (ii) immunostaining: (A) control liver; (B) LPS; (C) LPS+citric acid 1 g/kg; (D) LPS+citric acid 2 g/kg; (E) citric acid 4 g/kg (caspase-3 and iNOS immunohistochemistry, hematoxylin counterstain ×400). Color images available online at www.liebertpub.com/jmf
Caspase-3 and iNOS immunopositivity decreased in the livers of LPS-intoxicated mice treated with 1 g/kg of citric acid (Fig. 10C) and 2 g/kg of citric acid (Fig. 10D), respectively. In contrast, citric acid in the high dose of 4 g/kg was not effective in reducing caspase-3 (Fig. 10E-i) and iNOS expression (Fig. 10E-ii).
Discussion
In the present model of mild systemic inflammation caused by a subseptic dose of LPS endotoxin and associated with increased oxidative stress in brain and liver tissues, citric acid exerted important pharmacological effects. A significant and marked decrease in lipid peroxidation (measured as MDA) was observed in brain tissue after treatment with citric acid, thereby suggesting decreased free radical attack on polyunsaturated fatty acids. In contrast, no significant effect on liver MDA has been observed after treatment citric acid. In both the brain and liver, however, citric acid displayed marked inhibitory effect on nitric oxide. Under physiological conditions, this free radical gas synthesized from the amino acid l-arginine by the enzyme NOS is important in neurotransmission, maintaining vascular tone, immune regulation, synaptic plasticity, and many other functions.32,33 Increased levels of nitric oxide generated by glial cells, including astrocytes and microglia, due to action of inducible NOS, however, contributes to neuronal cell death in inflammatory, infectious, ischemic, and neurodegenerative diseases.34 This is due to the ability of nitric oxide to react with other free radicals, especially with the oxygen radical superoxide (O2−), to form peroxynitrite (ONOO−), decomposing to form the powerful and cytotoxic oxidants hydroxyl radical and nitrogen dioxide.35,36
In face of increased free radicals and reactive oxygen species, cells are equipped with a number of antioxidant mechanisms, such as catalases, GPxs, glutathione transferase, superoxide dismutase, and GSH.3 The administration of LPS was associated with an increase in lipid peroxidation and a drop in GSH level and GPx activity in brain and liver tissues, which indicates increased generation of free radicals. In LPS-treated mice, brain and liver GSH were not altered by citric acid. Meanwhile, treatment with citric acid at 1 g/kg was associated with increased GPx activities in brain and liver tissues, possibly due to an antioxidant effect of citric acid. In the current study, decreased brain and liver PON1 activity was observed after the administration of LPS. PON1 enzyme that plays an important role in the metabolism of many xenobiotic compounds has recently drawn attention, for a possible role in protecting cellular membranes against lipid peroxidation.25,26 In brain tissue, PON1 activity was improved by citric acid given at 1 g/kg. PON1 activity in liver tissue, however, decreased following higher doses of citric acid, possibly reflecting consumption or inactivation of the enzyme by increased free radicals with high concentration of citric acid.
One potent proinflammatory cytokine is TNF-α, which is produced in the brain by glial cells in response to various stimuli and induces astrocytes and microglial cells to secrete several inflammatory mediators, such as chemokines, lipid mediators, nitric oxide, and other free radicals. TNF-α has been demonstrated to play an important role in central nervous system neuroinflammation-mediated cell death in various neurodegenerative conditions.37,38 In the present study, the cytokine was markedly increased in brain tissue after LPS administration. Here we demonstrate that citric acid treatment was associated with marked inhibitory effect on TNF-α production within brain tissue after LPS challenge. This ability of citric acid to decrease pathological TNF-α production in the brain might be of value in relevance to neurodegenerative diseases. TNF-α expression appears to be upregulated in several neurodegenerative disorders, such as Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis, and pharmacological manipulation of TNF-α within the brain has been proposed as one potential target in the treatment of these conditions and may represent a valuable target for intervention.39–41
The present data indicate that citric acid can act directly on brain cells to inhibit their production of TNF-α and nitrite. LPS acts on TLR4 receptors on macrophages, dendritic cells, and other immune cells to release proinflammatory cytokines, such as TNF-α and IL-1β, which might gain direct access to the brain via the blood–brain barrier or signal to the brain via the vagus nerve, the so-called gut–brain immune communication.42,43 Thus it is also possible that the effects of citric acid on brain are accounted for by modulating the release of inflammatory mediators from leucocytes in the periphery.
Differences in results for the same markers in liver and brain tissues were observed. Thus, in contrast to the effects of citric acid in decreasing lipid peroxidation in the brain, no significant effect on liver MDA was observed. Moreover, PON1 activity in liver tissue decreased following citric acid at high doses. This might be due to the particular metabolic pathway interrelationships within each so different organ. Nevertheless, DNA fragmentation, serum transaminase elevations, caspase-3 and iNOS expression, and histological damage were all attenuated by 1–2 g/kg of citric acid. These data clearly indicated a protective effect for citric acid administration within this dose range on hepatic damage during endotoxemia. Citric acid intake, therefore, is likely to have a beneficial effect on the liver under toxic and inflammatory conditions. Citric acid might prevent liver injury through (1) reducing polymorphonuclear cell degranulation and attenuating the release of myeloperoxidase, elastase, IL-1β, and platelet factor; (2) stimulation of glycolysis and the tricarboxylic acid cycle; (3) increased production of bicarbonate with improvement of tissue acidosis in inflammatory conditions and therefore maintains tissue and cellular integrity; and (4) stimulation of vagal sensory afferents involved in signaling hepatic protection.11–13,15,44,45
It should be noted, however, that some of the beneficial effects observed for citric acid in brain and liver tissues were only in the dose range of 1–2 g/kg. This protective effect is lost when the dose is increased to 4 g/kg; for example, GPX and PON1 activities were increased only with 1 g/kg and TNF was decreased only by 1–2 g/kg of citric acid; the doses that were most effective in inhibiting brain nitrite. The protective effects on the liver were also lost with the higher dose of 4 g/kg, which also increased liver DNA fragmentation. Since citric acid is found in all animal tissues as an intermediate in the Krebs cycle, no limit has been set on the acceptable daily intake for humans for either the acid or salt.46 It is possible, however, that at higher concentrations, citric acid acts as a pro-oxidant. Several antioxidants show pro-oxidant effects at higher doses/concentrations, for example, carotenoids,47 vitamin E, and vitamin C.48,49 Natural compounds also display double-edged effects on inflammatory reactions, depending potentially on their concentrations: physiologic doses leading to beneficial effects whereas high doses may result in harmful effects.50
In summary, the present data suggest an antioxidant and anti-inflammatory effect for orally given citric acid at 1–2 g/kg in brain tissue. Citric acid also demonstrated a beneficial hepatic protective effect at this dose range. Given that both increased brain oxidative stress and chronic inflammation have been linked to the development of neurodegenerative diseases, citric acid might thus prove of clinical benefit in such conditions. The present study suggests that citric acid might find utility in treatment of toxic and inflammatory conditions of the brain and liver tissues. This can take the form of supplementation as nutraceutical citric acid. Meanwhile, citric acid is naturally concentrated in citrus fruits with lemon juice and lime juice being rich sources of citric acid and intake of these has been suggested as an effective means of treating oxalate stones.1,51,52 These studies have addressed the utility of dietary intervention with fruits and fruit juices with high citrate content (orange juice and lemonade) as an alternative to potassium citrate in increasing urinary pH and citrate, but the combination of citrate supplementation and fruit juices was not evaluated. This latter approach might prove a useful one combining the advantages of both classes of food additives. The presence of flavonoids and vitamin C in citrus fruits and juices makes the latter option an attractive one.
Author Disclosure Statement
The authors declare that there are no conflicts of interest.
References
- 1.Halliwell B: Reactive oxygen species and the central nervous system. J Neurochem 1992;59:1609–1623 [DOI] [PubMed] [Google Scholar]
- 2.Halliwell B: Biochemistry of oxidative stress. Biochem Soc Trans 2007;35:1147–1150 [DOI] [PubMed] [Google Scholar]
- 3.Wickens AP: Ageing and the free radical theory. Respir Physiol 2001;128:379–391 [DOI] [PubMed] [Google Scholar]
- 4.Gutteridge JMC: Lipid peroxidation and antioxidants as biomarkers of tissue damage. Clin Chem 1995;41:1819–1828 [PubMed] [Google Scholar]
- 5.Sies H: Oxidative stress: oxidants and antioxidants. Exp Physiol 1997;82:291–295 [DOI] [PubMed] [Google Scholar]
- 6.Halliwell N: Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging 2001;18:685–716 [DOI] [PubMed] [Google Scholar]
- 7.Valko M, Leibfritz D, Moncol J, et al. : Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 2007;39:44–84 [DOI] [PubMed] [Google Scholar]
- 8.Sayre LM, Perry G, Smith MA: Oxidative stress and neurotoxicity. Chem Res Toxicol 2008;21:172–188 [DOI] [PubMed] [Google Scholar]
- 9.Penniston KL, Nakada SY, Holmes RP, et al. : Quantitative assessment of citric acid in lemon juice, lime juice, and commercially-available fruit juice products. J Endourol 2008;22:567–570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grigor JMV, Johnson WS, Salminen S: Food additives for special dietary purposes. In: Food Additives, 2nd edition. (Branen AL, Davidson PM, Salminen S, Thorngate JH, 3rd, eds.) Marcel Dekker, Inc., Basel, New York, 2002, pp. 341 [Google Scholar]
- 11.Gabutti L, Ferrari N, Mombelli G, et al. : The favorable effect of regional citrate anticoagulation on interleukin-1beta release is dissociated from both coagulation and complement activation. J Nephrol 2004;17:819–825 [PubMed] [Google Scholar]
- 12.Gritters M, Grooteman MP, Schoorl M, et al. : Citrate anticoagulation abolishes degranulation of polymorphonuclear cells and platelets and reduces oxidative stress during haemodialysis. Nephrol Dial Transplant 2006;21:153–159 [DOI] [PubMed] [Google Scholar]
- 13.Tiranathanagul K, Jearnsujitwimol O, Susantitaphong P, et al. : Regional citrate anticoagulation reduces polymorphonuclear cell degranulation in critically ill patients treated with continuous venovenous hemofiltration. Ther Apher Dial 2011;15:556–564 [DOI] [PubMed] [Google Scholar]
- 14.Bryland A, Wieslander A, Carlsson O, et al. : Citrate treatment reduces endothelial death and inflammation under hyperglycaemic conditions. Diab Vasc Dis Res 2012;9:42–51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Abdel Salam OME, Sleem AA, Shaffie NM: Hepatoprotective effects of citric acid and aspartame on carbon tetrachloride-induced hepatic damage in rats. EXCLI J 2009;8:41–49 [Google Scholar]
- 16.Raetz CR, Whitfield C: Lipopolysaccharide endotoxin. Annu Rev Biochem 2002;71:635–700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Qin L, Wu X, Block ML, et al. : Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 2007;55:453–462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Buttini M, Mir A, Appel K, et al. : Lipopolysaccharide induces expression of tumour necrosis factor alpha in rat brain: Inhibition by methylprednisolone and by rolipram. Br J Pharmacol 1997;122:1483–1489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Turrin NP, Gayle D, Ilyin SE, et al. : Pro-inflammatory and anti-inflammatory cytokine mRNA induction in the periphery and brain following intraperitoneal administration of bacterial lipopolysaccharide. Brain Res Bull 2001;54:443–453 [DOI] [PubMed] [Google Scholar]
- 20.Fiorucci S, Mencarelli A, Meneguzzi A, et al. : NCX-4016 (NO-Aspirin) inhibits lipopolysaccharide-induced tissue factor expression in vivo. Role of nitric oxide. Circulation 2002;106:3120–3125 [DOI] [PubMed] [Google Scholar]
- 21.Ruiz-Larrea MB, Leal AM, Liza M, et al. : Antioxidant effects of estradiol and 2-hydroxyestradiol on iron-induced lipid peroxidation of rat liver microsomes. Steroids 1994;59:383–388 [DOI] [PubMed] [Google Scholar]
- 22.Ellman GL: Tissue sulfhydryl groups. Arch Biochem 1959;82:70–77 [DOI] [PubMed] [Google Scholar]
- 23.Moshage H, Kok B, Huizenga JR: Nitrite and nitrate determination in plasma: a critical evaluation. Clin Chem 1995;41:892–896 [PubMed] [Google Scholar]
- 24.Paglia DE, Valentine WN: Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 1967;70:158–169 [PubMed] [Google Scholar]
- 25.Higashino K, Takahashi Y, Yamamura Y: Release of phenyl acetate esterase from liver microsomes by carbon tetrachloride. Clin Chim Acta 1972;41:313–320 [DOI] [PubMed] [Google Scholar]
- 26.Watson AD, Berliner JA, Hama SY, et al. : Protective effect of high density lipoprotein associated paraoxonase. Inhibition of the biological activity of minimally oxidized low density lipoprotein. J Clin Invest 1995;96:2882–2891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen W, Jin W, Cook M, et al. : Oral delivery of group a streptococcal cell walls augments circulating TGF-beta and suppresses streptococcal cell wall arthritis. J Immunol 1998;161:6297–6304 [PubMed] [Google Scholar]
- 28.Gercel-Taylor C: Diphenylamine assay of DNA fragmentation for chemosensitivity testing. Methods Mol Med 2005;111:79–82 [DOI] [PubMed] [Google Scholar]
- 29.Crowley LV: The Reitman-Frankel colorimetric transaminase procedure in suspected myocardial infarction. Clin Chem 1967;13:482–487 [PubMed] [Google Scholar]
- 30.Belfield A, Goldberg DM: Revised assay for serum phenyl phosphatase activity using 4-amino-antipyrine. Enzyme 1971;12:561–573 [DOI] [PubMed] [Google Scholar]
- 31.Gown AM, Willingham MC: Improved detection of apoptotic cells in archival paraffin sections: immunohistochemistry using antibodies to cleaved caspase 3. J Histochem Cytochem 2002;50:449–454 [DOI] [PubMed] [Google Scholar]
- 32.Dawson TM, Snyder SH: Gases as biological messengers: Nitric oxide and carbon monoxide in the brain. J Neurosci 1994;14:5147–5159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Förstermann U, Sessa WC: Nitric oxide synthases: Regulation and function. Eur Heart J 2012;33:829–837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bal-Price A, Brown GC: Inflammatory neurodegeneration mediated by nitric oxide from activated glia-inhibiting neuronal respiration, causing glutamate release and excitotoxicity. J Neurosci 2001;21:6480–6491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Beckman JS: The double-edged role of nitric oxide in brain function and superoxide-mediated injury. J Dev Physiol 1991;15:53–59 [PubMed] [Google Scholar]
- 36.Moncada S, Bolanos JP: Nitric oxide, cell bioenergetics and neurodegeneration. J Neurochem 2006;97:1676–1689 [DOI] [PubMed] [Google Scholar]
- 37.Feueurstein G, Liu T, Barone F: Cytokines, inflammation and brain injury: role of TNF alpha. Cerebrovasc Brain Metab Rev 1994;6:341–360 [PubMed] [Google Scholar]
- 38.Tansey MG, Wyss-Coray T: Cytokines in CNS inflammation and disease. In: Central Nervous System Diseases and Inflammation. (Lane TE, Carson M, Bergmann C, Wyss-Coray T, eds.) Springer, New York, 2008, pp. 59–106 [Google Scholar]
- 39.Tweedie D, Sambamurti K, Greig NH: TNF-alpha inhibition as a treatment strategy for neurodegenerative disorders: new drug candidates and targets. Curr Alzheimer Res 2007;4:378–385 [DOI] [PubMed] [Google Scholar]
- 40.McCoy MK, Tansey MG: TNF signaling inhibition in the CNS: implications for normal brain function and neurodegenerative disease. J Neuroinflamm 2008;5:45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Frankola KA, Greig NH, Luo W, et al. : Targeting TNF-α to elucidate and ameliorate neuroinflammation in neurodegenerative diseases. CNS Neurol Disord Drug Targets 2011;10:391–403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Goehler LE, Gaykema RP, Hansen MK, et al. : Vagal immune-to-brain communication: a visceral chemosensory pathway. Auton Neurosci 2000;85:49–59 [DOI] [PubMed] [Google Scholar]
- 43.Romanovsky AA: Signaling the brain in the early sickness syndrome: are sensory nerves involved? Front Biosci 2004;9:494–504 [DOI] [PubMed] [Google Scholar]
- 44.Bjarnason I, Smethurst P, Macpherson A, et al. : Glucose and citrate reduce the permeability changes caused by indomethacin in humans. Gastroenterology 1992;102:1546–1550 [DOI] [PubMed] [Google Scholar]
- 45.Caudarella R, Vescini F, Buffa A, et al. : Citrate and mineral metabolism: kidney stones and bone disease. Front Biosci 2003;8:s1084–s1106 [DOI] [PubMed] [Google Scholar]
- 46.German JB: Antioxidants. In: Food Additives, 2nd edition. (Branen AL, Davidson PM, Salminen S, Thorngate JH, 3rd, eds.) Marcel Dekker, Inc., Basel, New York, 2002, pp. 538 [Google Scholar]
- 47.Palozza P: Evidence for pro-oxidant effects of carotenoids in vitro and in vivo implications in health and disease. In: Carotenoids in Health and Disease. (Mayne ST, Sies H, Krinsky NI, eds.) CRC Press, Marcel Dekker AG, New York, 2004, pp. 127–149 [Google Scholar]
- 48.Bowry VW, Stocker R: Tocopherol-mediated peroxidation. The prooxidant effect of vitamin E on the radical-initiated oxidation of human low-density lipoprotein. J Am Chem Soc 1993;115:6029–6044 [Google Scholar]
- 49.Podmore ID, Griffiths HR, Herbert KE, et al. : Vitamin C exhibits pro-oxidant properties. Nature 1998;392:559. [DOI] [PubMed] [Google Scholar]
- 50.Bouayed J, Bohn T: Exogenous antioxidants—double-edged swords in cellular redox state. Health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid Med Cell Longev 2010;3:228–237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kang DE, Sur RL, Haleblian GE, et al. : Long-term lemonade based dietary manipulation in patients with hypocitraturic nephrolithiasis. J Urol 2007;177:1358–1362 [DOI] [PubMed] [Google Scholar]
- 52.Haleblian GE, Leitao VA, Pierre SA, et al. : Assessment of citrate concentrations in citrus fruit-based juices and beverages: Implications for management of hypocitraturic nephrolithiasis. J Endourol 2008;22:1359–1366 [DOI] [PubMed] [Google Scholar]