| Patent Application Title: | Novel compounds that are ERK inhibitors | ||
| Patent Application Number: | WO 2013/063214 A1 | Publication date: | 2 May 2013 |
| Priority Application: | US 61/552194 | Priority date: | 27 October 2011 |
| Inventors: | Lim, J.; Kelley, E. H.; Methot, J. L.; Zhou, H.; Petrocchi, A.; Mansoor, U. F.; Fischer, C.; O’Boyle, B. M.; Guerin, D. J.; Bienstock, C. E.; Boyce, C. W.; Daniels, M. H.; Falcone, D.; Ferguson, R. D. II; Fevrier, S.; Huang, X.; Lipford, K. A.; Sloman, D. L.; Wilson, K.; Zhou, W.; Witter, D.; Maletic, M. M.; Siliphaivanh, P. | ||
| Assignee Company: | Merck Sharp and Dohme Corp.; 126 East Lincoln Avenue, Rahway, New Jersey 07065–0907, USA | ||
| Disease Area: | Cancer | Biological Target: | Extracellular-signal-regulated kinase 2 (ERK2) |
| Summary: | The invention in this patent application introduces compounds represented by formula I that can inhibit extracellular signal-regulated kinase 2 (ERK2). These compounds may potentially be useful in treatment of a broad spectrum of cancers. | ||
| Extracellular-signal-regulated kinase 2 (ERK2), also known as Mitogen-activated protein kinase 1 (MAPK1), is a serine/threonine kinase that is expressed in most mammalian tissues. It is part of the Ras-Raf-ERK signal transduction cascade, which is activated through a cascade of phosphorylation events and influences many cellular processes such as proliferation, differentiation, and survival. Mutations of Ras and BRAF (a serine-threonine kinase of the Raf family) and frequent activation of the ERK/MAPK pathway were identified in many cancerous tumors. Therefore, inhibition of the ERK signaling pathway can be an attractive target for anticancer treatments in a broad spectrum of human tumors. The compounds described in this patent application that inhibit ERK2 activity may potentially provide useful treatments of several forms of cancers including melanoma, pancreatic cancer, thyroid cancer, colorectal cancer, lung cancer, breast cancer, and ovarian cancer. | |||
| Important Compound Classes: | 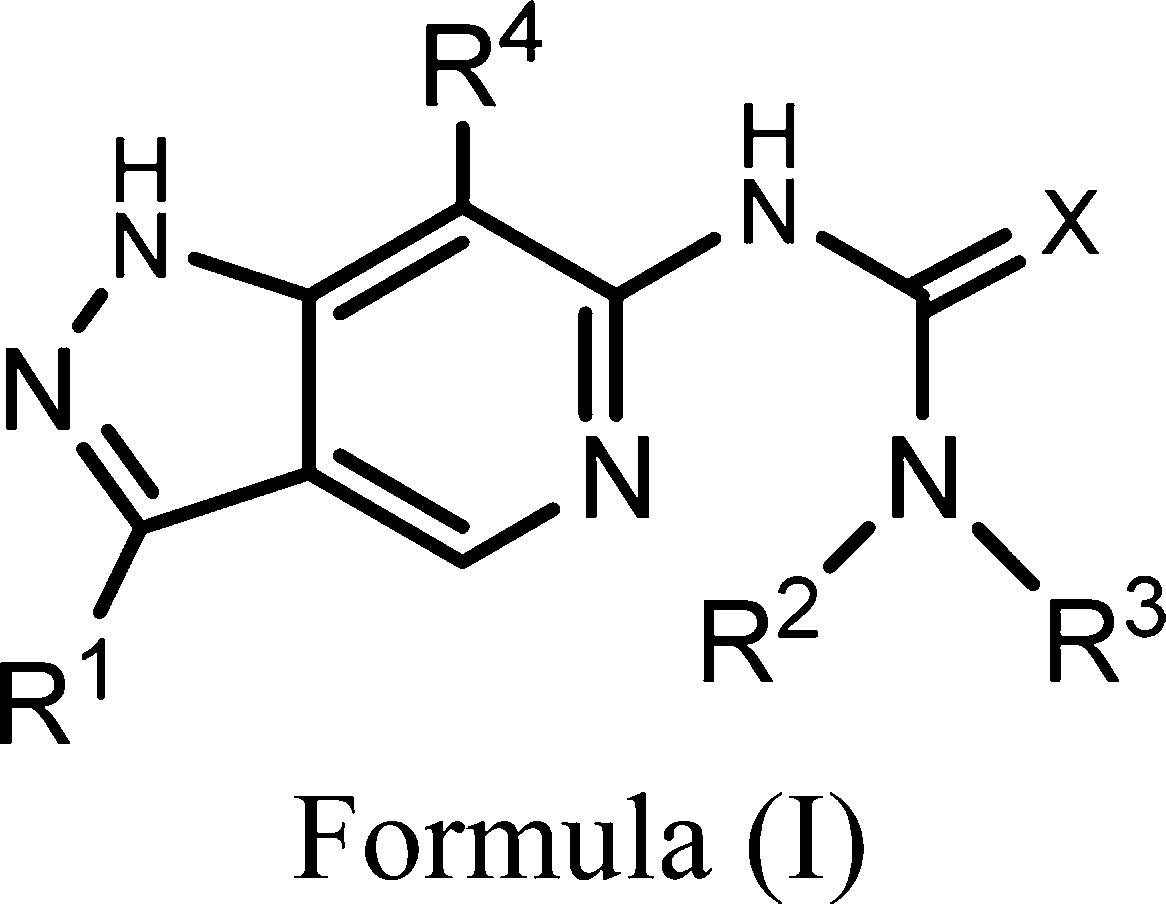 |
||
| Key Structures: | The inventors described the synthesis
of 701 examples of formula I; three of these examples are shown below: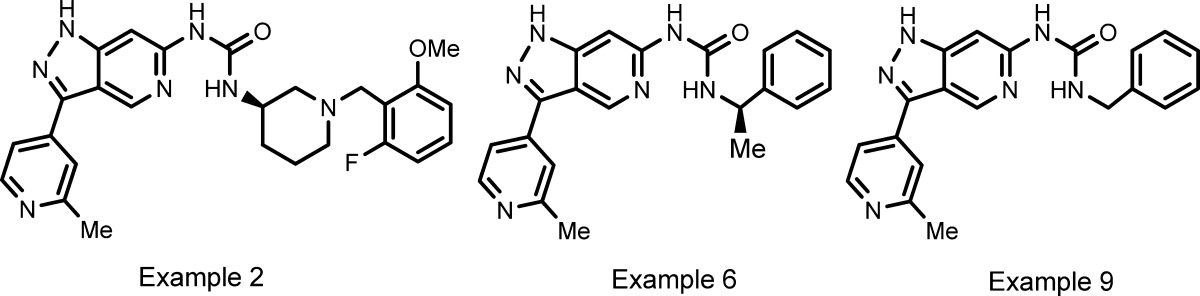
|
||
| Biological Assay: | Active human ERK2 (hERK2) Activity Assay | ||
| Active mouse ERK2 (mERK2) Activity Assay | |||
| Biological Data: | The IC50 data for the
above examples are listed in the following table: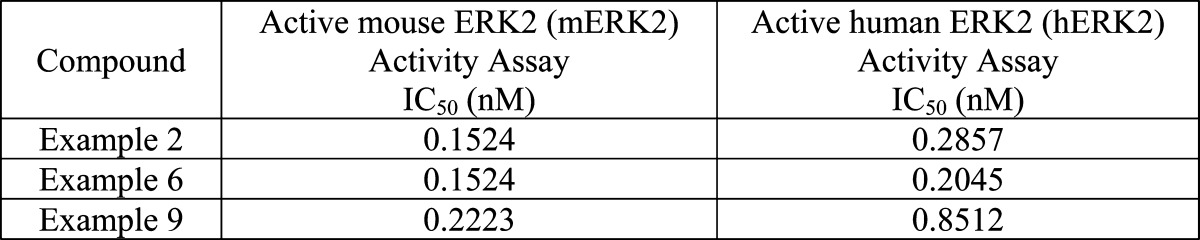
|
||
| Claims: | Claims 1–15: composition of matter, variations of formula I | ||
| Claim 15–19: composition of matter, selections specific examples of the compounds of formula I listed by example numbers as described in the application | |||
| Claim 20: pharmaceutical composition | |||
| Claims 21–23: methods of treatment of cancer with one of the invention compounds, alone or in combination with at least one chemotherapeutic agent | |||
| Recent Review Articles: | 1. Efferth T.Curr. Med. Chem. 2012, 19, 5735–5744. | ||
| 2. Kyriakis J. M.; Avruch J.. Physiol. Rev. 2012, 92, 689–737. | |||
The authors declare no competing financial interest.


