Abstract
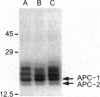
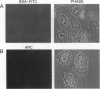
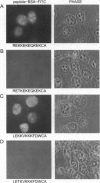
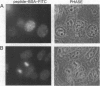
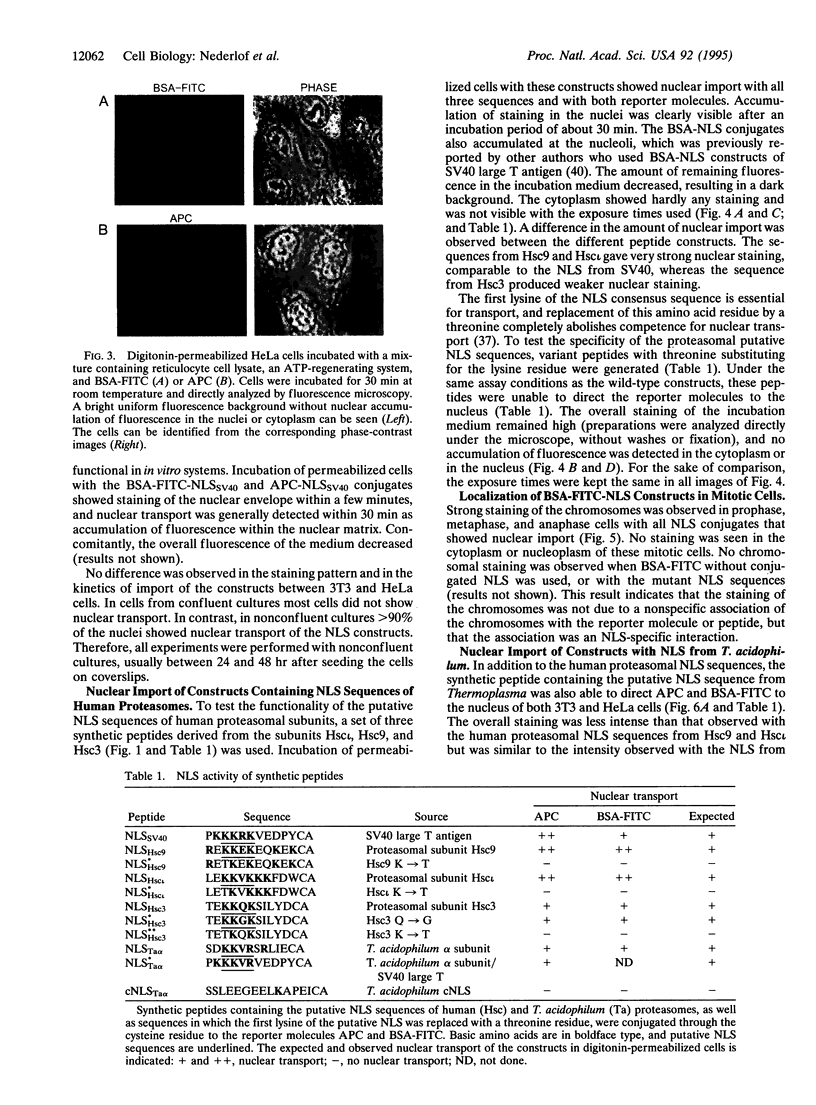
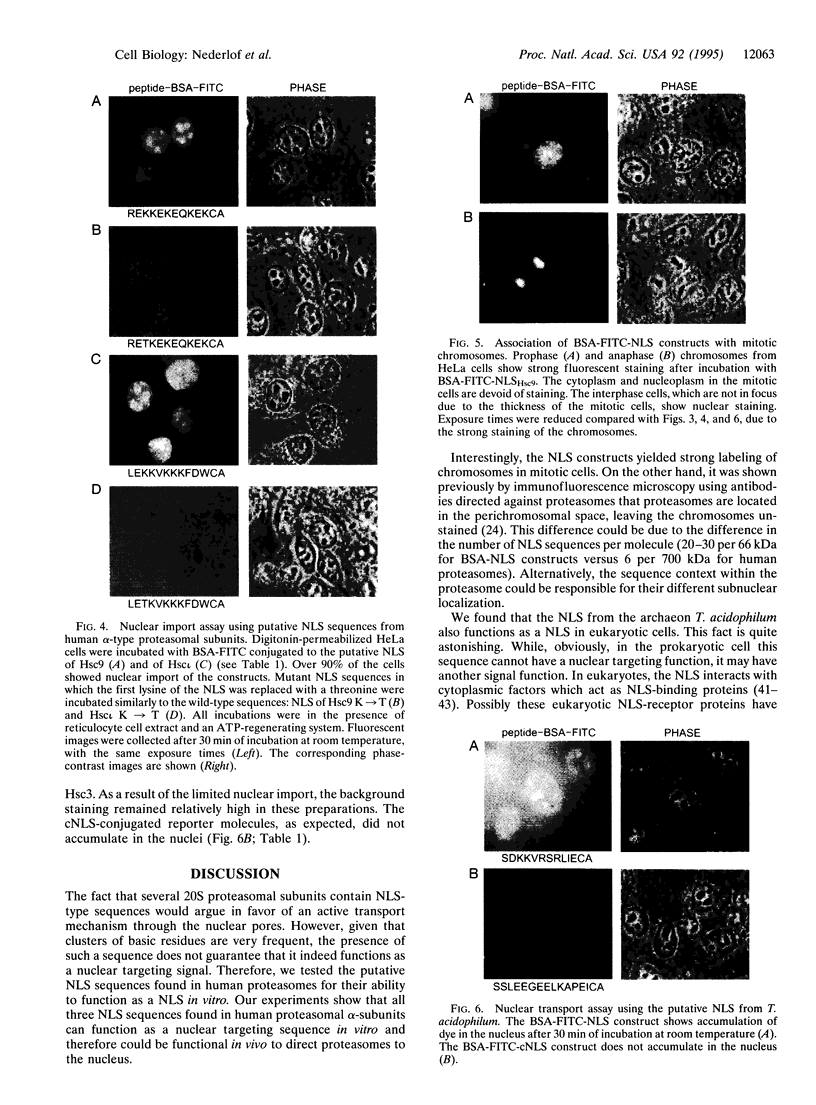
Proteasomes are located both in the nuclei and in the cytoplasm of eukaryotic cells. Active transport of these complexes through the nuclear pores has been proposed to be mediated by nuclear localization signals (NLS), which have been found in several of the alpha-type proteasomal subunits. We have tested three different putative NLS sequences from human alpha-type proteasomal subunits (Hsc iota, Hsc9, and Hsc3), as well as a putative NLS-type sequence from the archaeon Thermoplasma acidophilum, for their ability to direct non-nuclear proteins to the nucleus. Synthetic peptides containing these putative NLS sequences were generated and conjugated to large fluorescent reporter molecules: allophycocyanin or fluorescein-labeled bovine serum albumin. The conjugates were introduced into digitonin-permeabilized HeLa and 3T3 cells in the presence of cell lysate and ATP, and nuclear import was monitored by fluorescence microscopy. All three putative NLS sequences from human proteasomal subunits were able to direct the reporter molecules to the nucleus in both cell types, although differences in efficiency were observed. Substitution of threonine for the first lysine residue of the eukaryotic NLS motifs inhibited nuclear import completely. Interestingly, the putative NLS sequence found in T. acidophilum was also functional as a nuclear targeting sequence.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adam S. A., Gerace L. Cytosolic proteins that specifically bind nuclear location signals are receptors for nuclear import. Cell. 1991 Sep 6;66(5):837–847. doi: 10.1016/0092-8674(91)90431-w. [DOI] [PubMed] [Google Scholar]
- Adam S. A., Lobl T. J., Mitchell M. A., Gerace L. Identification of specific binding proteins for a nuclear location sequence. Nature. 1989 Jan 19;337(6204):276–279. doi: 10.1038/337276a0. [DOI] [PubMed] [Google Scholar]
- Amsterdam A., Pitzer F., Baumeister W. Changes in intracellular localization of proteasomes in immortalized ovarian granulosa cells during mitosis associated with a role in cell cycle control. Proc Natl Acad Sci U S A. 1993 Jan 1;90(1):99–103. doi: 10.1073/pnas.90.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahlmann B., Kopp F., Kuehn L., Niedel B., Pfeifer G., Hegerl R., Baumeister W. The multicatalytic proteinase (prosome) is ubiquitous from eukaryotes to archaebacteria. FEBS Lett. 1989 Jul 17;251(1-2):125–131. doi: 10.1016/0014-5793(89)81441-3. [DOI] [PubMed] [Google Scholar]
- Dworetzky S. I., Lanford R. E., Feldherr C. M. The effects of variations in the number and sequence of targeting signals on nuclear uptake. J Cell Biol. 1988 Oct;107(4):1279–1287. doi: 10.1083/jcb.107.4.1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldfarb D. S., Gariépy J., Schoolnik G., Kornberg R. D. Synthetic peptides as nuclear localization signals. Nature. 1986 Aug 14;322(6080):641–644. doi: 10.1038/322641a0. [DOI] [PubMed] [Google Scholar]
- Grziwa A., Baumeister W., Dahlmann B., Kopp F. Localization of subunits in proteasomes from Thermoplasma acidophilum by immunoelectron microscopy. FEBS Lett. 1991 Sep 23;290(1-2):186–190. doi: 10.1016/0014-5793(91)81256-8. [DOI] [PubMed] [Google Scholar]
- Görlich D., Prehn S., Laskey R. A., Hartmann E. Isolation of a protein that is essential for the first step of nuclear protein import. Cell. 1994 Dec 2;79(5):767–778. doi: 10.1016/0092-8674(94)90067-1. [DOI] [PubMed] [Google Scholar]
- Hegerl R., Pfeifer G., Pühler G., Dahlmann B., Baumeister W. The three-dimensional structure of proteasomes from Thermoplasma acidophilum as determined by electron microscopy using random conical tilting. FEBS Lett. 1991 May 20;283(1):117–121. doi: 10.1016/0014-5793(91)80567-m. [DOI] [PubMed] [Google Scholar]
- Hennekes H., Peter M., Weber K., Nigg E. A. Phosphorylation on protein kinase C sites inhibits nuclear import of lamin B2. J Cell Biol. 1993 Mar;120(6):1293–1304. doi: 10.1083/jcb.120.6.1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilt W., Heinemeyer W., Wolf D. H. Studies on the yeast proteasome uncover its basic structural features and multiple in vivo functions. Enzyme Protein. 1993;47(4-6):189–201. doi: 10.1159/000468678. [DOI] [PubMed] [Google Scholar]
- Hügle B., Kleinschmidt J. A., Franke W. W. The 22 S cylinder particles of Xenopus laevis. II. Immunological characterization and localization of their proteins in tissues and cultured cells. Eur J Cell Biol. 1983 Nov;32(1):157–163. [PubMed] [Google Scholar]
- Kalderon D., Richardson W. D., Markham A. F., Smith A. E. Sequence requirements for nuclear location of simian virus 40 large-T antigen. Nature. 1984 Sep 6;311(5981):33–38. doi: 10.1038/311033a0. [DOI] [PubMed] [Google Scholar]
- Lupas A., Koster A. J., Baumeister W. Structural features of 26S and 20S proteasomes. Enzyme Protein. 1993;47(4-6):252–273. doi: 10.1159/000468684. [DOI] [PubMed] [Google Scholar]
- Lupas A., Zwickl P., Baumeister W. Proteasome sequences in eubacteria. Trends Biochem Sci. 1994 Dec;19(12):533–534. doi: 10.1016/0968-0004(94)90054-x. [DOI] [PubMed] [Google Scholar]
- Löwe J., Stock D., Jap B., Zwickl P., Baumeister W., Huber R. Crystal structure of the 20S proteasome from the archaeon T. acidophilum at 3.4 A resolution. Science. 1995 Apr 28;268(5210):533–539. doi: 10.1126/science.7725097. [DOI] [PubMed] [Google Scholar]
- Moll T., Tebb G., Surana U., Robitsch H., Nasmyth K. The role of phosphorylation and the CDC28 protein kinase in cell cycle-regulated nuclear import of the S. cerevisiae transcription factor SWI5. Cell. 1991 Aug 23;66(4):743–758. doi: 10.1016/0092-8674(91)90118-i. [DOI] [PubMed] [Google Scholar]
- Moore M. S., Blobel G. The two steps of nuclear import, targeting to the nuclear envelope and translocation through the nuclear pore, require different cytosolic factors. Cell. 1992 Jun 12;69(6):939–950. doi: 10.1016/0092-8674(92)90613-h. [DOI] [PubMed] [Google Scholar]
- Muller M., Renkawitz R. The glucocorticoid receptor. Biochim Biophys Acta. 1991 Feb 16;1088(2):171–182. doi: 10.1016/0167-4781(91)90052-n. [DOI] [PubMed] [Google Scholar]
- Palmer A., Mason G. G., Paramio J. M., Knecht E., Rivett A. J. Changes in proteasome localization during the cell cycle. Eur J Cell Biol. 1994 Jun;64(1):163–175. [PubMed] [Google Scholar]
- Peters J. M., Cejka Z., Harris J. R., Kleinschmidt J. A., Baumeister W. Structural features of the 26 S proteasome complex. J Mol Biol. 1993 Dec 20;234(4):932–937. doi: 10.1006/jmbi.1993.1646. [DOI] [PubMed] [Google Scholar]
- Peters J. M., Franke W. W., Kleinschmidt J. A. Distinct 19 S and 20 S subcomplexes of the 26 S proteasome and their distribution in the nucleus and the cytoplasm. J Biol Chem. 1994 Mar 11;269(10):7709–7718. [PubMed] [Google Scholar]
- Peters J. M. Proteasomes: protein degradation machines of the cell. Trends Biochem Sci. 1994 Sep;19(9):377–382. doi: 10.1016/0968-0004(94)90115-5. [DOI] [PubMed] [Google Scholar]
- Pühler G., Weinkauf S., Bachmann L., Müller S., Engel A., Hegerl R., Baumeister W. Subunit stoichiometry and three-dimensional arrangement in proteasomes from Thermoplasma acidophilum. EMBO J. 1992 Apr;11(4):1607–1616. doi: 10.1002/j.1460-2075.1992.tb05206.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rihs H. P., Jans D. A., Fan H., Peters R. The rate of nuclear cytoplasmic protein transport is determined by the casein kinase II site flanking the nuclear localization sequence of the SV40 T-antigen. EMBO J. 1991 Mar;10(3):633–639. doi: 10.1002/j.1460-2075.1991.tb07991.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivett A. J. High molecular mass intracellular proteases. Biochem J. 1989 Nov 1;263(3):625–633. doi: 10.1042/bj2630625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivett A. J., Palmer A., Knecht E. Electron microscopic localization of the multicatalytic proteinase complex in rat liver and in cultured cells. J Histochem Cytochem. 1992 Aug;40(8):1165–1172. doi: 10.1177/40.8.1619280. [DOI] [PubMed] [Google Scholar]
- Schmitz M. L., Henkel T., Baeuerle P. A. Proteins controlling the nuclear uptake of NF-kappa B, Rel and dorsal. Trends Cell Biol. 1991 Nov;1(5):130–137. doi: 10.1016/0962-8924(91)90118-s. [DOI] [PubMed] [Google Scholar]
- Seemüller E., Lupas A., Stock D., Löwe J., Huber R., Baumeister W. Proteasome from Thermoplasma acidophilum: a threonine protease. Science. 1995 Apr 28;268(5210):579–582. doi: 10.1126/science.7725107. [DOI] [PubMed] [Google Scholar]
- Tanaka K., Tamura T., Yoshimura T., Ichihara A. Proteasomes: protein and gene structures. New Biol. 1992 Mar;4(3):173–187. [PubMed] [Google Scholar]
- Wolin S. L. From the elephant to E. coli: SRP-dependent protein targeting. Cell. 1994 Jun 17;77(6):787–790. doi: 10.1016/0092-8674(94)90124-4. [DOI] [PubMed] [Google Scholar]
- Yoshimura T., Kameyama K., Takagi T., Ikai A., Tokunaga F., Koide T., Tanahashi N., Tamura T., Cejka Z., Baumeister W. Molecular characterization of the "26S" proteasome complex from rat liver. J Struct Biol. 1993 Nov-Dec;111(3):200–211. doi: 10.1006/jsbi.1993.1050. [DOI] [PubMed] [Google Scholar]
- Zwickl P., Kleinz J., Baumeister W. Critical elements in proteasome assembly. Nat Struct Biol. 1994 Nov;1(11):765–770. doi: 10.1038/nsb1194-765. [DOI] [PubMed] [Google Scholar]
- Zwickl P., Lottspeich F., Dahlmann B., Baumeister W. Cloning and sequencing of the gene encoding the large (alpha-) subunit of the proteasome from Thermoplasma acidophilum. FEBS Lett. 1991 Jan 28;278(2):217–221. doi: 10.1016/0014-5793(91)80120-r. [DOI] [PubMed] [Google Scholar]