Abstract
Wolbachia are widespread maternally-transmitted intracellular bacteria that infect most insect species and are able to alter the reproduction of innumerous hosts. The cellular bases of these alterations remain largely unknown. Here we report that Drosophila mauritiana infected with a native Wolbachia wMau strain produces about four times more eggs than the non-infected counterpart. Wolbachia infection leads to an increase in the mitotic activity of germline stem cells (GSCs) as well as a decrease in programmed cell death in the germarium. Our results suggest that upregulation of GSCs division is mediated by a tropism of Wolbachia for the germline stem cell niche (GSCN), the cellular microenvironment that supports GSCs.
Wolbachia are maternally transmitted intracellular bacteria infecting a large number of invertebrates such as insects and parasitic worms (1). Many invertebrates that harbor these bacteria are either the vectors (e.g. mosquitoes) or the causative agent (e.g. filarial nematodes) of devastating human infectious diseases. By understanding the biology at the interface between Wolbachia and their hosts, advances in the treatment of filarial diseases and the control of disease vectors are made possible (2–7). Furthermore, Wolbachia can dramatically alter host reproduction, affecting the evolutionary history of numerous invertebrates (1). Therefore, understanding how Wolbachia affect their hosts is an important ecological, evolutionary and human health question.
To investigate the influence of Wolbachia on their hosts at the cellular level, we used the Drosophila gonad, a powerful experimental system. We have previously shown that in Drosophila melanogaster, Wolbachia target the somatic stem cell niche (SSCN, Fig. 1A) – the microenvironment that supports the somatic stem cells – in the female ovary (8). Further work shows that Wolbachia also target the somatic stem cell niche in the ovary of other insects (9, 10). Here we report two additional stem cell niches preferentially colonized (i.e., cell tropism) by Wolbachia: the female germline stem cell niche (GSCN, Fig. 1A), and the hub, at the apical tip of the testis (discussed below). In a D. mauritiana stock infected with Wolbachia wMau, we consistently noticed an intense accumulation of bacteria in the GSCN, the structure harboring the GSCs (infection frequency = 91% ± 5.7%, N = 958 germaria) (see Wolbachia, labeled green in Fig. 2A and B, Fig. 3A and fig. S1A). This GSCN accumulation was absent in D. melanogaster (GSCN infection frequency = 0%, N = 180 germaria, see fig. S1, B compared to A). Electron microscopy and three-dimensional reconstruction of confocal images show that the vast majority of the cytoplasmic volume of the GSCN is occupied by Wolbachia wMau [see Fig. 1B, the Wolbachia cells (a red asterisk indicates a single bacterial cell) occupy most of the GSCN (colored in green), compared to the non-infected control in fig. S1C; see also Sup. Movie]. Because GSCN function is essential for stem cell maintenance and activity (11), we hypothesized that the high levels of infection in the niche would impair its associated stem cells to a certain degree. An easy readout of GSC activity is egg production, since every egg produced originates from the division of a stem cell associated with the GSCN (Fig. 1A’). The total number of eggs laid per Wolbachia-infected female was 3.5 times higher than that observed in non-infected flies (herein referred to as “W-”; the genetic background of the W- flies was homogenized by successive backcrossing to infected males, as shown in fig. S2). This experiment was repeated under different temperature, humidity and age conditions (see SOM Methods and Table S1) (12). Under these different conditions, infected flies (referred to as “W+”) still produced approximately fourfold more eggs than the non-infected females (see Fig. 1C and table S1).
Fig. 1. Wolbachia target the GSCN and infection increases egg production.
(A) Drosophila ovariole with the germline shown in light blue and the somatic follicle cells in white. Egg chambers are formed in the germarium (left) and mature into the egg. The upward-pointing green arrow indicates germline stem cell (GSC, dark blue) division, which positively affects egg production [see inset A’: GSC divides asymmetrically and one daughter cell exits the germline stem cell niche (GSCN, green) and forms the egg’s germline (light blue)]. The downward-pointing red arrows indicate developmental points where the onset of programmed cell death (PCD) reduces egg production, either in the germarium or in previtellogenic egg chambers. (Lower left) A magnified view of the germarium shows both the somatic stem cell niche (SSCN, green arrowhead) and the GSCN (yellow bracket), formed by the terminal filament (light green) and the cap cells (dark green), which contact the GSCs (blue arrowhead). (B) Electron micrographs of a GSCN (green) and the GSC (blue) in infected D. mauritiana. Most of the cytoplasm of the cap cells (GSCN) is occupied by Wolbachia wMau (red asterisk, see also Sup. Movie). Scale bar = 1μm. The inset shows a magnified view of the GSCN, red asterisk highlighting a single Wolbachia wMau. (C) Fold change of total amount of eggs laid per infected female (W+, green) under different conditions normalized to non-infected (W-, yellow). Relative egg production was measured in triplicate for each condition: room temperature (RT, 20 days and 46 days, light green) or at 25ºC (20 days, dark green). Wolbachia significantly induced fecundity gains at all conditions (Student’s t-test, PRT 20 days = 6.5 × 10−4, PRT 46 days = 3.9 × 10−4 and P 25ºC 20 days = 1.7 × 10−2). See table S1 and (12).
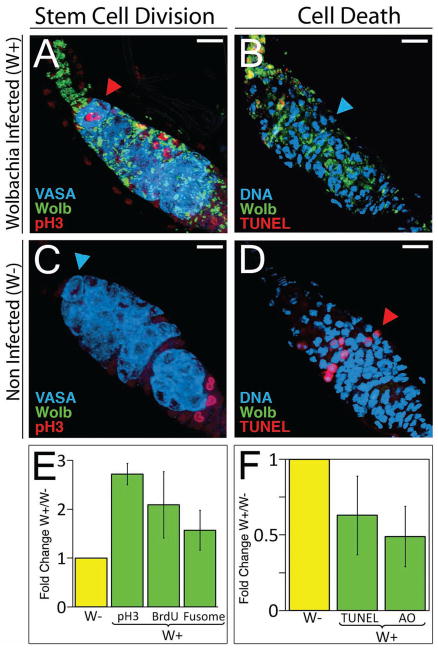
Fig. 2. Wolbachia infection increases GSC mitotic activity and suppresses PCD in the germarium.
Representative confocal images of D. mauritiana germaria infected [W+, Wolbachia shown in green (A and B)] and non-infected [W-, (C and D)]. Arrowheads indicate the presence (red arrowhead) or absence (blue arrowhead) of GSC division [pH3 (phospho-Histone H3), red in A and C] or PCD (TUNEL, red in B and D). Germline is labeled with anti-Vasa (blue), Scale bar = 10μm. (E and F). Average fold difference for each marker indicated below graphs, normalized to W- (mean of triplicates, 15 independent experiments total). Infection significantly affects GSC mitosis (E) and PCD (F) for all markers (Logistic regression, PpH3 = 5.4 × 10−3, N = 621; PBrdU = 2.0 × 10−2, N = 1061; PFusome = 4.3 × 10−3, N = 695; PTUNEL = 8.0 × 10−3, N = 802; PAcridine Orange = 1.2 × 10−7, N = 754, N = number of germaria,). See also tables S2, S4 and (12).
Fig. 3. High levels of Wolbachia at the GSCN upregulate GSC mitosis.
(A – B) Niches (yellow brackets) from infected flies are classified as highly infected (HN, A) and with low infection (LN, B). Fusome staining (red) shows GSC in the HN dividing (“!” morphology in A). Scale bar = 5 μm. (C) Frequency of HN (solid green) and LN (hatched green) in four independent experiments. The numbers in each category and the total number of germaria analyzed are indicated for each experiment. (D) For each germarium counted in graph D, the frequency of GSC division was determined by either fusome morphology (Exp. 1 and 2) or BrdU incorporation (Exp. 3 and 4). HN significantly increases GSC mitosis (logistic regression, P= 2.4 × 10−2). (E and F) In infected testes of D. mauritiana, Wolbachia also target the stem cell niche (aka hubs, yellow arrowhead) at high (HN, E) and low levels (LN, F). (F) pH3 staining (white) labels a dividing testis stem cell adjacent to an HN niche. Scale bar = 5 μm.
Given these levels of egg production, we reasoned that W+ ovaries contain GSCs that are more active. To test this possibility, we measured the frequency of GSC division in W+ and W- flies using three different markers for three distinct phases of the cell cycle. The initial assessment was performed using an anti-phospho-histone H3 antibody, that labels cells in mitosis (Fig. 2, A and C, fig. S3G) (12). The labeling of GSCs in W+ flies was on average 2.7 (± 0.23) fold higher than in W- flies (Fig. 2E and table S2). This increase could indicate either a higher GSC division in infected germaria or an arrest during the mitotic phase of the cell cycle.
We further investigated GSC proliferation using two additional markers: incorporation of the thymidine analog BrdU, an indicator of DNA synthesis during S phase (fig S3, A, D and G), and a particular fusome morphology characteristic of GSCs in G2 (fig. S3, B, E and H). The fusome is a germline-specific organelle that assumes the shape of an exclamation mark (!) during G2 (13, 14). Both markers corroborated a higher GSC proliferation rate in W+ (Fig 2E). The probability of stem cell division is significantly increased in Wolbachia infected flies (SOM Methods and table S2) (12). In nine independent experiments, utilizing three different methods, stem cell division in W+ flies was on average doubled (2.12 ± 0.66, table S2). Although significant, this amount by itself does not suffice to explain the fourfold increase in egg production in infected flies. An additional cellular event that could alter egg production in a Wolbachia-dependent manner could be cell death in the ovary. Programmed cell death (PCD) is a known key regulator of egg production in Drosophila melanogaster (15). Furthermore, previous studies in wasps and in human neutrophils have shown that the presence of Wolbachia or Wolbachia-derived proteins, respectively, inhibits host apoptosis (16, 17).
We quantified the influence of Wolbachia infection on two developmentally-regulated PCD events that modulate egg production in Drosophila, the first in the germarium (Fig.1A, left red arrow) and the second during the onset of vitellogenesis (Fig. 1A, right red arrow) (15, 18). In the parasitic wasp Asobara tabida, removal of Wolbachia causes sterility through massive cell death in mid-oogenesis, at the pre-vitellogenic stages (16). Therefore, we initially measured PCD at these stages. We found that the differences in PCD between W+ and W- previtellogenic egg chambers were highly variable and not significant regarding Wolbachia’s effects at this developmental point (fig. S4 and table S3) (12).
Accordingly, we measured the levels of PCD in the germarium. Using two different assays – DNA fragmentation in fixed tissue (TUNEL, Fig. 2, B and D) and visualization of dead cells via live imaging (Acridine Orange, fig. S3, C and F) – Wolbachia infection consistently decreased PCD in the germarium by approximately one half as compared to non-infected flies (Fig. 2F, and table S4) (12). Wolbachia-driven reduction of PCD in the germarium was statistically significant (Fig. 2 and table S4). Together, these results indicate that the increase in egg production in W+ D. mauritiana is due to both increased GSC mitosis and decreased PCD in the germarium.
Next, the mechanistic foundation for Wolbachia’s manipulation of GSC mitotic activity was examined. Considering that GSCN regulates stem cell physiology (19), we designed an experiment to test if levels of Wolbachia in the GSCN correlate with mitotic activity of the GSC (see fig. S5). During this assay we used only Wolbachia infected flies. Even though in these W+ flies most of the GSCNs were highly infected (91% ± 6.5%, N = 788) (Fig. 3, A and C), there is a small population of niches that have either very low or no Wolbachia present. These distinct types of niches were termed “LN” (low infection in the niche, Fig. 3B) and their infected counterparts “HN” (high infection in the niche, Fig. 3A; see also fig. S6, A compared to B, fig. S7). Because these distinct populations of GSCs are present inside the same infected flies, all of the environmental and systemic factors are exactly the same. In four independent experiments, the mitotic activity of GSCs residing in LN niches was substantially lower or absent in comparison to HN niches (Fig. 3, C and D). There is a statistically significant association of GSC mitosis with the high density of Wolbachia in the niche (P = 2.4 × 10−2, see table S5) (12). This observation favors a mechanism in which Wolbachia’s infection in the niche modulates stem cell activity, although it does not rule out a contribution from systemic or stem cell-intrinsic signals (see SOM text S1, fig. S8, S9).
We found that Wolbachia wMau also target the hub, a group of somatic cells that form the niche supporting germline and somatic stem cells of the testis (20). In males, both the targeting of the hub (64%, N = 77, Fig. 3, E and F) as well as the upregulation of GSC division did not occur to the same degree as in females (fig. S10, table S6). It is possible that phenotypic consequences of niche tropism are diverse in males. Wolbachia and other maternally inherited endosymbionts can evolve drastically different germline manipulation phenotypes between sexes (21).
The vast majority of insects have symbiotic associations with bacteria that are vertically transmitted through the egg cytoplasm (22). Because of maternal transmission, these host-bacteria partnerships evolve to favor the reproductive success of infected mothers (1, 23–25). In the Drosophila genus, there are several reports of Wolbachia-induced changes in fecundity, including cases of rapid evolution of both partners, changing from a parasitic to mutualistic association in 20 years (24, 26–29). There is little understanding of these dramatic and widespread interferences with host reproduction at the cellular and molecular level (30). Here, we have identified two cellular events that are manipulated by Wolbachia. The combination of Wolbachia-induced alterations of both PCD in the germarium and GSC mitosis results in higher egg production, which further promotes Wolbachia spreading through maternal transmission. These findings provide the cellular mechanisms for Wolbachia’s effects on host fecundity observed in this infected D. mauritiana strain over its non-infected counterpart (see SOM text S2).
Advancing our understanding of how endosymbionts subvert the cellular processes of insects will also be relevant to the growing efforts towards controlling human infectious diseases through symbiotic bacteria (3–7, 31).
Supplementary Material
Acknowledgments
We are grateful to K. McCall, G. Cooper, D. Waxman, C. Bradham and A. Boxer for valuable suggestions in the manuscript. We also thank E. Wieschaus, the McCall Lab, T. Blute, D. Gantz and M. Bisher for help and support with PCD and EM experiments, E. Wieschaus, T. Schüpbach, R. Lehmann, P. Lasko, D. Stern, V. Orgogozo and M. Ramos for fly stocks and reagents, members of the Frydman Lab for assistance and suggestions during the realization of this work, J. Li and D. Robson for help with MatLab software, A. Mahowald for sharing his unpublished results and encouraging us to analyze Wolbachia in the testis. Finally, we would like to thank the anonymous reviewers for their helpful comments. This work was supported by funds from Boston University and NIAID (1K22AI74909-01A1 to H.M.F.) The data described in this paper is available in the Supporting Online Material.
REFERENCES AND NOTES
- 1.Werren JH, Baldo L, Clark ME. Wolbachia: master manipulators of invertebrate biology. Nat Rev Microbiol. 2008 Oct;6:741. doi: 10.1038/nrmicro1969. [DOI] [PubMed] [Google Scholar]
- 2.Pfarr KM, Hoerauf AM. Antibiotics which target the Wolbachia endosymbionts of filarial parasites: a new strategy for control of filariasis and amelioration of pathology. Mini reviews in medicinal chemistry. 2006 Feb;6:203. doi: 10.2174/138955706775475984. [DOI] [PubMed] [Google Scholar]
- 3.Bourtzis K. Wolbachia-based technologies for insect pest population control. Advances in experimental medicine and biology. 2008;627:104. doi: 10.1007/978-0-387-78225-6_9. [DOI] [PubMed] [Google Scholar]
- 4.McMeniman CJ, et al. Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science. 2009 Jan 2;323:141. doi: 10.1126/science.1165326. [DOI] [PubMed] [Google Scholar]
- 5.Kambris Z, Cook PE, Phuc HK, Sinkins SP. Immune activation by life-shortening Wolbachia and reduced filarial competence in mosquitoes. Science. 2009 Oct 2;326:134. doi: 10.1126/science.1177531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hughes GL, Koga R, Xue P, Fukatsu T, Rasgon JL. Wolbachia infections are virulent and inhibit the human malaria parasite Plasmodium falciparum in anopheles gambiae. PLoS pathogens. 2011 May;7:e1002043. doi: 10.1371/journal.ppat.1002043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.O'Neill SL, et al. The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature. 2011 Aug 25;476:450. doi: 10.1038/nature10355. [DOI] [PubMed] [Google Scholar]
- 8.Frydman HM, Li JM, Robson DN, Wieschaus E. Somatic stem cell niche tropism in Wolbachia. Nature. 2006 May 25;441:509. doi: 10.1038/nature04756. [DOI] [PubMed] [Google Scholar]
- 9.Hosokawa T, Koga R, Kikuchi Y, Meng XY, Fukatsu T. Wolbachia as a bacteriocyte-associated nutritional mutualist. Proc Natl Acad Sci U S A. 2010 Jan 12;107769 doi: 10.1073/pnas.0911476107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sacchi L, et al. Bacteriocyte-like cells harbour Wolbachia in the ovary of Drosophila melanogaster (Insecta, Diptera), Zyginidia pullula (Insecta, Hemiptera) Tissue Cell. 2010 Oct;42:328. doi: 10.1016/j.tice.2010.07.009. [DOI] [PubMed] [Google Scholar]
- 11.Xie T, Spradling AC. A niche maintaining germ line stem cells in the Drosophila ovary. Science. 2000 Oct 13;290:328. doi: 10.1126/science.290.5490.328. [DOI] [PubMed] [Google Scholar]
- 12.Material and methods are available as supporting material on Science Online.
- 13.de Cuevas M, Spradling AC. Morphogenesis of the Drosophila fusome and its implications for oocyte specification. Development. 1998 Aug;125:2781. doi: 10.1242/dev.125.15.2781. [DOI] [PubMed] [Google Scholar]
- 14.Lin H, Yue L, Spradling AC. The Drosophila fusome, a germline-specific organelle, contains membrane skeletal proteins and functions in cyst formation. Development. 1994 Apr;120:947. doi: 10.1242/dev.120.4.947. [DOI] [PubMed] [Google Scholar]
- 15.Drummond-Barbosa D, Spradling AC. Stem cells and their progeny respond to nutritional changes during Drosophila oogenesis. Dev Biol. 2001 Mar 1;231:265. doi: 10.1006/dbio.2000.0135. [DOI] [PubMed] [Google Scholar]
- 16.Pannebakker BA, Loppin B, Elemans CP, Humblot L, Vavre F. Parasitic inhibition of cell death facilitates symbiosis. Proc Natl Acad Sci U S A. 2007 Jan 2;104:213. doi: 10.1073/pnas.0607845104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bazzocchi C, et al. Wolbachia surface protein (WSP) inhibits apoptosis in human neutrophils. Parasite Immunology. 2007 Feb;29:73. doi: 10.1111/j.1365-3024.2006.00915.x. [DOI] [PubMed] [Google Scholar]
- 18.Pritchett TL, Tanner EA, McCall K. Cracking open cell death in the Drosophila ovary. Apoptosis. 2009 Aug;14:969. doi: 10.1007/s10495-009-0369-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Morrison SJ, Spradling AC. Stem cells and niches: mechanisms that promote stem cell maintenance throughout life. Cell. 2008 Feb 22;132:598. doi: 10.1016/j.cell.2008.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Cuevas M, Matunis EL. The stem cell niche: lessons from the Drosophila testis. Development. 2011 Jul;138:2861. doi: 10.1242/dev.056242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Werren JH. Colloquium Paper: Selfish genetic elements, genetic conflict, and evolutionary innovation. P Natl Acad Sci USA. 2011 Jun 28;108(Suppl 2):10863. doi: 10.1073/pnas.1102343108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH. How many species are infected with Wolbachia?--A statistical analysis of current data. FEMS Microbiol Lett. 2008 Apr;281:215. doi: 10.1111/j.1574-6968.2008.01110.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Turelli M, Hoffmann AA. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature. 1991 Oct 3;353:440. doi: 10.1038/353440a0. [DOI] [PubMed] [Google Scholar]
- 24.Weeks AR, Turelli M, Harcombe WR, Reynolds KT, Hoffmann AA. From parasite to mutualist: rapid evolution of Wolbachia in natural populations of Drosophila. PLoS Biol. 2007 May;5:e114. doi: 10.1371/journal.pbio.0050114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Himler AG, et al. Rapid spread of a bacterial symbiont in an invasive whitefly is driven by fitness benefits and female bias. Science. 2011 Apr 8;332:254. doi: 10.1126/science.1199410. [DOI] [PubMed] [Google Scholar]
- 26.Hoffmann AA, Turelli M, Harshman LG. Factors affecting the distribution of cytoplasmic incompatibility in Drosophila simulans. Genetics. 1990;126:933. doi: 10.1093/genetics/126.4.933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fry AJ, Palmer MR, Rand DM. Variable fitness effects of Wolbachia infection in Drosophila melanogaster. Heredity. 2004 Oct;93:379. doi: 10.1038/sj.hdy.6800514. [DOI] [PubMed] [Google Scholar]
- 28.Reynolds KT, Thomson LJ, Hoffmann AA. The effects of host age, host nuclear background and temperature on phenotypic effects of the virulent Wolbachia strain popcorn in Drosophila melanogaster. Genetics. 2003 Jul;164:1027. doi: 10.1093/genetics/164.3.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Poinsot D, Mercot H. Wolbachia infection in Drosophila simulans: Does the female host bear a physiological cost? Evolution. 1997 Feb;51:180. doi: 10.1111/j.1558-5646.1997.tb02399.x. [DOI] [PubMed] [Google Scholar]
- 30.Serbus LR, Casper-Lindley C, Landmann F, Sullivan W. The genetics and cell biology of Wolbachia-host interactions. Annual review of genetics. 2008;42:683. doi: 10.1146/annurev.genet.41.110306.130354. [DOI] [PubMed] [Google Scholar]
- 31.Weiss B, Aksoy S. Microbiome influences on insect host vector competence. Trends in parasitology. 2011 doi: 10.1016/j.pt.2011.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.