Abstract
Background
The diagnosis of prostate cancer involves invasive, sometimes harmful, procedures that can entail negative quality-of-life implications to individuals and high additional costs to the US healthcare system when these procedures result in retesting and iatrogenic harms. It is estimated that $1.86 billion is spent annually on prostate-specific antigen (PSA) testing alone. An advanced epigenetic molecular diagnostic test that uses methylation-specific polymerase chain reaction to assess the DNA methylation status of GSTP1, APC, and RASSF1 genes associated with oncogenesis enables a higher degree of accuracy (previously unattainable through prostate biopsy procedures alone) and produces clinical, financial, and health benefits by reducing the number of medically unnecessary and costly repeated biopsies that are part of today's standard of care.
Objectives
The purpose of this study is to quantify, using a budget impact model, the effect of a relatively new epigenetic assay on healthcare costs for commercial health plans that reimburse for the assay, by avoiding unnecessary repeated prostate biopsy procedures.
Methods
A budget impact model was developed to test the hypothesis that the epigenetic assay can produce cost-saving benefits to health plans, as well as clinical benefits to urologists and patients with prostate cancer, by providing guidance on how to offer patients more appropriate, and less costly, care. The budget impact model is presented from the perspective of a hypothetical commercial health plan, and direct costs are calculated over a 1-year time horizon, using 2013 Medicare fee-for-service rates. Using a plan of 1 million members, the model compares 1-year costs in a “reference scenario,” in which the epigenetic assay is not used for the screening and diagnosis of prostate cancer, to costs in a “new scenario,” in which the epigenetic assay is used to distinguish true-negative prostate biopsy results from false-negative biopsy results.
Results
Based on this analysis, administering the epigenetic assay to patients with histopathologically negative biopsies would result in a reduction of 1106 unnecessary biopsies for a health plan with 1 million members. The total 1-year cost of repeated prostate cancer biopsies to the health plan was found to be $2,864,142 in the reference scenario and $2,333,341 in the new scenario. This translates to a total budget impact, or an annual savings, of $530,801 to the plan. The total diagnostic cost was calculated to be $2584 per patient in the new scenario (using the genetic assay) compared with $3172 per patient in the reference scenario (that did not use the assay), resulting in a savings of $588 per patient management.
Conclusion
This analysis shows that the net cost to a commercial health plan with 1 million members would be reduced by approximately $500,000 if patients with histopathologically negative biopsies were managed with the use of the epigenetic assay to differentiate patients who should undergo repeated biopsy and those who should not. Using this genetic-based assay can save costs to health plans and to the US healthcare and improve the clinical management of patients with elevated PSA levels.
Prostate cancer is the most frequently detected cancer in men, and 1 of 6 men will be diagnosed with prostate cancer during their lifetime based on Medicare enrollment data.1 In the United States, approximately 19 million men annually are screened by prostate-specific antigen (PSA) testing,2 resulting in approximately 4.7 million abnormal PSA test results (≥4.0 ng/mL)3 leading to approximately 1.3 million biopsy procedures.4 According to the National Cancer Institute, 241,740 men are diagnosed with prostate cancer annually, and 28,170 prostate cancer–related deaths were reported in 2012.5 Although some forms of prostate cancer are deadly, many forms are low grade and can be managed by active surveillance. Aggressive variants of prostate cancer can be one of the deadliest cancers in men, and accurate diagnosis and follow-up remain a challenge and come at a considerable cost to the US healthcare system.
Clinical Burden
Despite the recent controversy that was raised by the US Preventive Services Task Force (USPSTF) findings on PSA testing, leading to their recommendations to stop routine PSA-based screening,6 the American Urological Association (AUA) continues to recommend the PSA blood test, along with digital rectal examination (DRE), for screening men at risk for prostate cancer. Screening has led to a shift of detecting earlier-stage disease, resulting in an increased likelihood for curative treatment. If screening is eliminated, urologists fear an increased incidence of advanced cancers and an increase in healthcare costs to effectively treat these patients.7 Today, urologists typically perform a biopsy for high-risk patients with a rising PSA and for patients with a PSA level ≥4.0 ng/mL, obtaining approximately 10 to 12 needle-core tissue samples according to the current standard of care.8,9
Of note, an abnormal PSA result can often be caused by factors other than cancer, including infection, inflammation, or other benign conditions, such as benign prostatic hyperplasia. This leads to the inclusion of many men with no cancer among those who are being subjected to prostate biopsies (ie, false-positive PSA screening). The rate of cancer detection in men undergoing prostate biopsies is approximately 30%, and approximately 75% of men who have undergone biopsies have negative prostate biopsy results.2,3
An elevated PSA and/or abnormal DRE identify men at high risk for prostate cancer, and, as a result, many of these men will undergo a biopsy procedure. However, because of the nature of random and limited sampling of the prostate, many cancers are undetected by histopathologic review. Studies by urology and pathology opinion leaders report that initial prostate biopsy histopathology has a 20% to 30% false-negative rate.8,10,11 Given these reported false-negative rates for histology, many patients with persistently elevated PSA values are at risk for occult cancer. This uncertainty poses a diagnostic dilemma, resulting in many men receiving 2, 3, and sometimes 4 repeated biopsy procedures.12–14
KEY POINTS
-
▸
The diagnosis of prostate cancer involves invasive procedures that can have negative clinical implications for patients and add costs to health plans and the US healthcare system.
-
▸
The appropriate use of PSA testing remains controversial, because elevated PSA levels can be caused by conditions unrelated to prostate cancer.
-
▸
Nevertheless, many men with benign conditions and elevated PSA levels undergo repeated prostate biopsies, which may be unnecessary, because of low risk of prostate cancer.
-
▸
A recent genetic-based assay has shown 90% negative predictive value in differentiating men with elevated PSA levels who should undergo a biopsy and those who should not.
-
▸
Applying a budget impact analysis to a hypothetical 1-million-member health plan based on current evidence-based data, the use of this assay would reduce the number of unnecessary biopsies in this plan by 1106 annually and save the plan $530,142.
-
▸
In line with the increasing focus on personalized medicine in oncology, using a genetic-based test to select appropriate candidates for prostate cancer biopsies can limit the number of unnecessary biopsies performed and save costs to payers and to the US healthcare system.
Repeated biopsies expose patients to the discomfort and risk of complications associated with this invasive procedure. Complications include infections, prostatitis, cystitis, sepsis, endocarditis, hypotension, gastrointestinal hemorrhage, hematuria, and urinary symptoms. Recently, antibiotic resistance has also been cited as a growing concern.1
Economic Burden
Repeated biopsies also generate high medical costs. Approximately $1.86 billion is spent annually on PSA tests alone.2,15 More than $4 billion is spent annually on therapies for prostate cancer, and this amount is expected to increase to $8.7 billion by the year 2019.16
Molecular Testing
With such high costs to the US healthcare system, as well as negative quality-of-life implications to patients, the AUA has called for new biomarkers for the improved diagnosis and treatment of prostate cancer.17 With the growing movement toward personalized medicine, the application of molecular testing to improve cancer detection and the management of patients represents an evolution in oncology.2,18
Epigenetic assays, in particular, have been reported to improve on the accuracy of prostate biopsies and histopathologic review.2,8 (Epigenetic refers to gene regulatory mechanisms; epigenetic assays are based on the differences in the chromatin structure of actively and silent genes.) In a recent multicenter, blinded study, such an epigenetic assay (ie, ConfirmMDx for Prostate Cancer) was used to detect occult cancer in histopathologically negative prostate biopsies.8 The assay was performed using multiplex methylation-specific polymerase chain reaction to assess the DNA methylation status of the GSTP1, APC, and RASSF1 genes associated with the presence of cancer in residual negative prostate biopsy tissue samples.3,8,10 Using this advanced molecular diagnostic test allows for a higher degree of accuracy that was previously unattainable through prostate biopsy procedures alone.
By detecting epigenetic changes that are known to be associated with oncogenesis progression, in cancer lesions or in neighboring cells (ie, field effect), these biomarkers aid in the identification of occult prostate cancer. This field effect accounts for improved detection in adjacent, benign-looking tissue, providing a higher negative predictive value than standard histopathology alone.19,20 The test results of this new epigenetic assay can guide urologists in decisions regarding the need to repeat a biopsy in patients with a previously negative biopsy who are still considered at risk for prostate cancer.8
A Budget Impact Analysis
A budget impact model was developed to assess whether the epigenetic assay can also produce financial benefits, beyond the reported clinical and health benefits. The model was designed to quantify the impact on the costs associated with repeated biopsies used for the screening and diagnosis of prostate cancer, and to test the hypothesis that the epigenetic assay produces a beneficial reduction in costs to commercial health plans, while providing improved guidance for patient management that leads to less invasive and less costly care. This type of analysis is an essential tool for healthcare managers and policymakers budgeting and instituting coverage decisions for prostate cancer diagnostics.
Methods
Study Design
The budget impact model is presented from the perspective of a hypothetical commercial health plan, and direct costs are calculated over a 1-year time horizon, using Medicare fee-for-service (FFS) rates.21,22 The membership of this health plan is based on an assumed size of 1 million members, half of whom are males. The membership is distributed among age categories according to US population data.23
The model's base-case analysis was conducted for a hypothetical plan using patient age-groups between ages 40 and 64 years (similar to those in commercial health plans). An additional sensitivity analysis was conducted for a hypothetical plan consisting of patients aged ≥65 years (similar to a Medicare health plan), using methods identical to the base case, with the exception of the patient ages and corresponding PSA rates. Biopsies and costs for patients younger than age 40 years or older than age 74 years were not included in the analysis.
The model screens the population to select patients who may receive a biopsy to test for the presence of prostate cancer. It then selects a specific subpopulation of those men, which are individuals who may receive a repeat biopsy.
For men at risk of undergoing a repeated biopsy, the model allows for the simulation of the current (reference scenario) and a counterfactual reality (new scenario). In the reference scenario, the model uses current clinical patterns of care to simulate the treatment of men at risk of prostate cancer in the reference scenario; a molecular assay was not utilized for prostate cancer detection. In the new scenario, men at risk for repeated biopsy are evaluated with the epigenetic assay, and those with a negative DNA methylation test result are spared a repeat of the biopsy, thereby reducing the number of unnecessary procedures.
The cost analysis was based on total costs of 1 year in the course of prostate cancer screening and evaluation through prostate biopsy. Costs to the health plan are assumed to be equal to the Medicare FFS rates, which provide a conservative benchmark for reimbursement rates paid for by other health plans. In the reference scenario, the health plan incurs the costs of 1 or several repeated biopsies and the associated iatrogenic costs. In the new scenario, the health plan incurs the cost of an additional diagnostic test performed on the residual prostate tissue from the original sample plus the cost for repeated biopsies and associated iatrogenic costs on patients who had positive test results.
The model assumes that the epigenetic assay would be used for all men meeting the assay's eligibility requirements, including an abnormal DRE, an elevated PSA level, and a negative prostate biospy. Both scenarios calculate a total cost and a plan budget impact, expressed on a per-member per-month (PMPM) basis, as well as the aggregate annual cost to the plan.
Data Sources
The PubMed database was searched for published clinical and pharmacoeconomic studies to assign values to the clinical and cost parameters used in the model. Studies were identified in PubMed that reflect current practice patterns of 10 to 12 core prostate biopsies in contrast to older studies that were based on sextant biopsy practice.
For cost parameters, a combination of published literature cross-referenced to Medicare payment rates was used. Parameters pertaining to the accuracy and outcomes of the assay (including the assay's sensitivity and specificity, and the positive and negative predictive values) were cited from the MATLOC clinical trial.8
Sample Selection
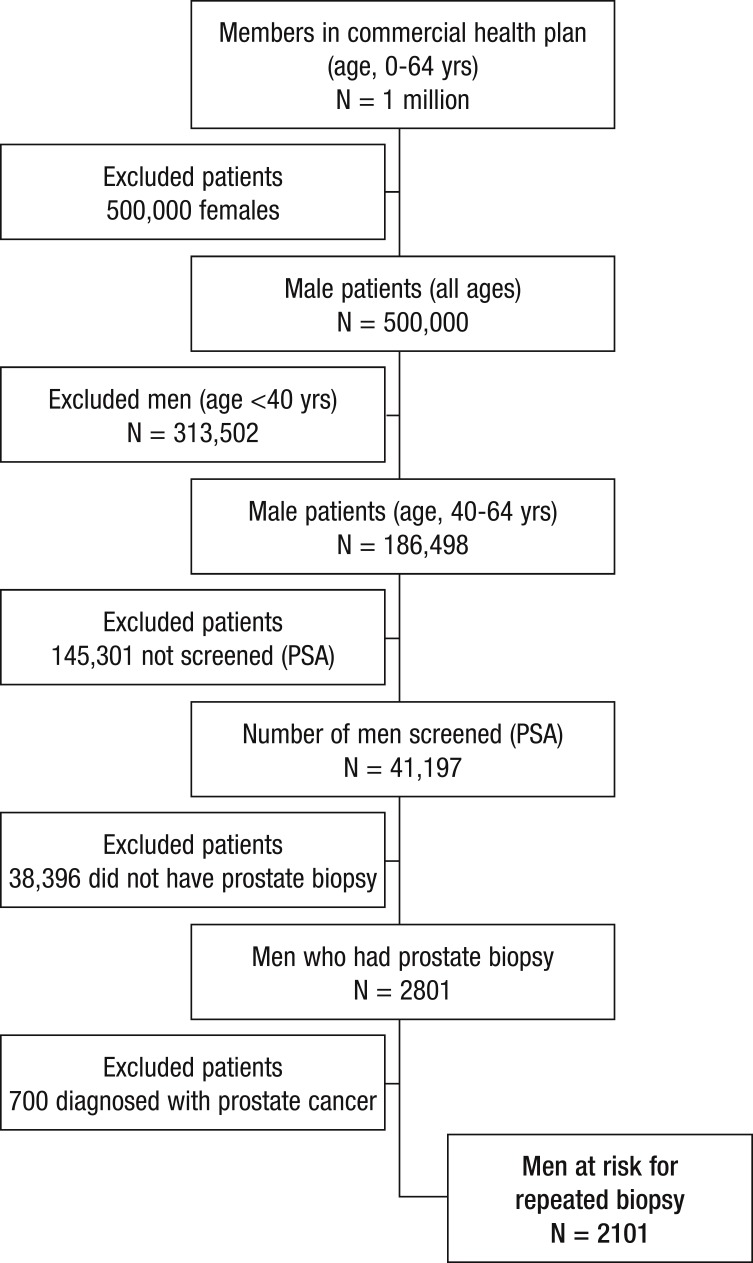
PSA screening rates in US males vary by age, ranging from approximately 8% to almost 50%.24 Of all men screened for PSA, 6.8% are assumed to undergo biopsy based on a PSA of at least 4 ng/mL.25 These PSA and biopsy rates were applied to the hypothetical commercial health plan. Of the hypothetical plan, patients aged 40 to 64 years were selected, using demographic data from the US Census Bureau 2010, for inclusion in the model. Applying the national PSA screening and biopsy rates to the hypothetical commercial population yields a total of 2801 men undergoing a prostate biopsy. A total of 2101 men were deemed at risk for repeated biopsy based on a prostate cancer detection rate of 25% (Figure).25
Figure. Potential Patients for Epigenetic Assay in a 1-Million-Member Health Plan.
PSA indicates prostate-specific antigen.
Total Men at Risk for a Repeated Biopsy
This budget impact analysis compares the proportion of the 2101 men in the cohort who are at risk for a repeated biopsy in 2 potential scenarios—the reference scenario and the new scenario, which is using the epigenetic assay.
In the reference scenario, many patients with rising or elevated PSA levels will be seen again for DRE or PSA testing, and be considered for a repeated biopsy. In this standard of care, 43% (903) of the patients with a histopathologically negative biopsy are referred for a repeated biopsy based on persistent clinical risk factors.13
In the new scenario, these same patients are triaged with the epigenetic assay. In the new scenario, 3% of patients would not be eligible for the epigenetic assay, because of atypical small acinar proliferation (ASAP) found in their previous biopsy tissue.12 The model assumes that patients with ASAP will receive a repeated biopsy, given the high risk of prostate cancer associated with this histopathologic finding. Approximately 99.9% of all cases would have sufficient tissue for this epigenetic assay, leaving 875 evaluable cases (based on 2 quality-not-sufficient cases of 749 in laboratory experience, through November 30, 2012, when the data were collected).
The Epigenetic Assay
The multiplex DNA methylation epigenetic assay became available in the United States in May 2012 (through MDxHealth's CLIA-accredited, CAP-certified laboratory in Irvine, CA). The performance characteristics of this assay (Table 1) were described in the MATLOC clinical trial, which investigated the clinical utility of this assay.8 These characteristics were used in our budget impact model to determine the anticipated number of patients who would be identified as positive or negative for methylation markers.
Table 1.
Diagnostic Accuracy of the Epigenetic Assay
| Variable | Rate |
|---|---|
| Initial biopsy false-negative rate | 0.18 |
| Multiplex DNA Methylation Epigenetic Assay | |
| Sensitivity | 0.68 |
| Specificity | 0.64 |
| Negative predictive value | 0.90 |
| Positive predictive value | 0.29 |
Source: Reference 10.
The test is designed such that its high (90%) negative predictive value accurately distinguishes the patients with negative prostate biopsies from patients who may have occult cancer.8 In the budget impact model, under the reference scenario, 43% (903) of men at risk for repeated biopsy were referred for repeated biopsy. In the new scenario, testing the high-risk patients with the epigenetic assay significantly reduced the number of repeated biopsies by confirming the histopathologically negative biopsy results for 510 men. The epigenetic assay identified 365 men with positive DNA methylation results who would be referred for repeated biopsy (Table 2).
Table 2.
Men at Risk for a Repeated Prostate Cancer Biopsy
| Variable | Reference scenario (standard of care) | New scenario (epigenetic assay) |
|---|---|---|
| Men at risk of repeated biopsy, N | 2101 | 2101 |
| Men referred for repeated biopsy or methylation test, % | 43 | 43 |
| Number of men referred for repeat biopsy or methylation test, N | 903 | 903 |
| Number of men with ASAP, % | 3 | 3 |
| Cases with sufficient tissue for methylation test, % | N/A | 99.9% |
| Risk stratification based on methylation markers | ||
| Evaluable cases, N | N/A | 875 |
| Cases with positive methylation markers, N | N/A | 365 |
| Cases with negative methylation markers, N | N/A | 510 |
| Total men referred for repeated biopsy, N | 903 | 365 |
ASAP indicates atypical small acinar proliferation; N/A, not applicable.
Model Variables and Assumptions
Conservative assumptions were made as to the number of repeated biopsies based on reported rates. In the standard of care, 43% of men are referred to have 1 repeated biopsy, 44% of which have a second biopsy, and 43% of these have a third biopsy. The 100% of men tested with the epigenetic assay who are methylation-positive were assumed to receive a repeated biopsy. A total of 1472 repeated biopsies are expected to be performed in the reference scenario compared with only 365 repeated biopsies with the epigenetic assay in the new scenario.
The average cost of a prostate biopsy procedure is $1946, which is a conservative estimate, based on decreased interim 2013 Medicare Physician Fee Schedule rates; this does not take into account prophylactic antibiotic, pain, or other concomitant medication costs.25 The total expected complication costs per patient for an initial or repeated biopsy were calculated using Surveillance, Epidemiology and End Results (SEER)-Medicare cancer registries' reported incidence of infectious and noninfectious complications and the associated mean payment for the Medicare Severity-Diagnosis Related Groups (MS-DRGs) from the 2012 Centers for Medicare & Medicaid Services MS-DRG payment schedule.22
Table 3 shows the calculations for the average cost ($392) of complications per patient undergoing repeated prostate cancer biopsy. The total weighted cost of a fully burdened biopsy is $1946—the sum of the procedural cost and the cost of complications weighted by incidence. The retail price for the epigenetic assay is $206 per individual core, or $2061 for a 10-core biopsy (pricing is based on the cost of the ConfirmMDx for Prostate Cancer Test, as provided by MDxHealth, the manufacturer of this test). The model assesses the health plan's costs compared with billed charges; therefore, the cost of this assay is discounted by 10%, to conservatively reflect payer costs, at $1855.02 per test.
Table 3.
Per-Patient Costs Associated with Biopsy Complications
| Infectious complications | DRG payment, $ | Incidence (SEER-Medicare) | Per-patient cost, $ |
|---|---|---|---|
| Kidney infection | 5594 | ||
| Urinary tract infection | 5594 | ||
| Prostatitis | 6220 | ||
| Cystitis | 5594 | ||
| Sepsis/bacteremia | 16,662 | ||
| Endocarditis | 10,943 | ||
| Hypotension | 5801 | ||
| Average payment for infectious complications | 8058 | 0.04 | 322 |
| Noninfectious complications | DRG payment, $ | Incidence (SEER-Medicare) | Per-patient cost, $ |
|---|---|---|---|
| Gastrointestinal hemorrhage | 6559 | ||
| Hematuria | 5366 | ||
| Acute posthemorrhagic anemia | 5660 | ||
| Urinary symptoms/retention | 5594 | ||
| Average payment for noninfectious complications | 5795 | 0.012 | 70 |
| Total cost | 392 |
DRG indicates diagnosis-related group; SEER, Surveillance, Epidemiology and End Results.
Results
Costs
This budget impact analysis demonstrates that the net cost to a commercial plan is lower if patients undergoing prostate cancer biopsies are managed using the assay. Although this involves an additional cost for the acquisition of the assay, using the assay results in a reduction of 1106 unnecessary biopsies for a health plan with 1 million members (Table 4).
Table 4.
Total Resource Use: Repeated Prostate Biopsies
| Resource | Reference scenario (standard of care) | New scenario (epigenetic assay) |
|---|---|---|
| Total men referred for first repeated biopsy, N | 903 | 903 |
| Total men referred for second repeated biopsy, N | 397 | |
| Total men referred for third repeated biopsy, N | 171 | |
| Total epigenetic tests, N | 0 | 875 |
| Total repeated biopsies, N | 1472 | 365 |
| Total repeated biopsies avoided, N | 1106 | |
The total cost of repeated biopsies avoided is $2,152,276 (1106 biopsies avoided × $1946 per biopsy). The total cost to the health plan in 1 year was calculated to be $2,864,142 in the reference scenario versus $2,333,341 with the epigenetic assay in the new scenario. To calculate the total diagnostic cost per patient in the reference scenario, the cost of a prostate biopsy ($1946) was applied and weighted to a repeated biopsy distribution rate for the percentage of men who receive first, second, and third repeated biopsies of 43%, 44%, and 43%, respectively.13
For the new scenario, the total diagnostic cost per patient includes the cost of the assay plus the weighted biopsy cost, applied and weighted to 43% of men who have positive results based on the epigenetic assay (based on the sensitivity, specificity, and negative and positive predictive values).8 The total diagnostic cost per patient was $3172 in the reference scenario compared with $2584 in the new scenario, resulting in a savings of $588 per patient managed. This results in a total savings of $530,801 annually to the health plan, or –$0.0442 PMPM (Table 5).
Table 5.
Total Annual Costs and Budget Impact: Reference Scenario versus New Scenario
| Reference scenario (standard of care) | New scenario (epigenetic assay) | |||
|---|---|---|---|---|
| Cost | Total annual cost, $ | PMPM cost, $ | Total annual cost, $ | PMPM cost, $ |
| Total cost of epigenetic assay | 0 | 0 | 1,623,143 | 0.14 |
| Total cost of repeated biopsies | 2,864,142 | 0.33 | 710,198 | 0.06 |
| Total cost of repeated biopsies avoided | 0 | 0 | 2,152,276 | 0.18 |
| Total diagnostic cost to health plan | 2,864,142 | 0.24 | 2,333,341 | 0.19 |
| Total diagnostic cost per patient | 3172 | 0.24 | 2584 | 0.19 |
| Total budget impact to plan | −530,801 | |||
| Total budget impact to plan PMPM | −0.442 | |||
PMPM indicates per member per month.
Sensitivity Analysis
To test for uncertainty among the model parameters, all calculations were repeated for a patient population of men aged ≥65 years, representative of the Medicare population. Patients older than age 74 years were excluded from the model, resulting in an at-risk cohort of 6917 men. Testing these patients with the assay resulted in a reduction of 3642 unnecessary biopsies.
The total cost of repeated biopsies avoided was $7,086,416. The total cost to the plan in 1 year was $9,429,097 in the reference scenario and $7,688,849 in the new scenario. This resulted in a total budget impact of –$1,740,248 to the plan, or –$0.1450 PMPM. The total diagnostic cost per patient was $3172 in the reference scenario compared with $2584 in the new scenario (Table 6), resulting in a savings of $588 per patient managed.
Table 6.
Sensitivity Analysis: Total Costs and Budget Impact
| Reference scenario (standard of care) | New scenario (epigenetic assay) | |||
|---|---|---|---|---|
| Cost | Total annual cost, $ | PMPM cost, $ | Total annual cost, $ | PMPM cost, $ |
| Total cost of epigenetic assay | 0 | 0.00 | 5,346,168 | 0.53 |
| Total cost of repeated biopsies | 9,429,097 | 1.09 | 2,342,681 | 0.27 |
| Total cost of repeated biopsies avoided | 0 | 0.00 | 7,086,416 | 0.82 |
| Total diagnostic cost to health plan | 9,429,097 | 1.09 | 7,688,849 | 0.81 |
| Total diagnostic cost per patient | 3172 | 0.33 | 2584 | 0.24 |
| Total budget impact to plan | −1,740,248 | |||
| Total budget impact to plan PMPM | −0.1450 | |||
PMPM indicates per member per month.
Discussion
The budget impact model was developed to evaluate the clinical and financial benefits of payer coverage for the epigenetic assay, also considering the clinical benefits, based on well-founded and conservative assumptions from existing evidence and current standards of care for patients considered at risk for prostate cancer.
The analysis demonstrates that a commercial health plan would realize cost-savings with the coverage of the epigenetic assay. The upfront cost of the epigenetic assay will be recovered based on the savings associated with avoided biopsy procedures and associated complications of biopsies. Given these assumptions and the costs associated with the current standard of care, the inclusion of the epigenetic assay into the management of men who are screened for prostate cancer would result in a net cost-savings of –$530,801 in the first year after the assay became available in the United States in a health plan with 1 million members.
The epigenetic assay provides clear and actionable results that aid the urologist in treatment decision-making, improving patient care, and yielding significant healthcare savings. A key assumption is that a health plan inclusion of the epigenetic assay in medical policy and coverage decisions will motivate a change in the behavior of urologists, resulting in a reduction of repeated biopsies. Policy tools that promote appropriate patient management according to evidence-based guidelines, such as value-based payment (eg, financial incentives for choosing evidence-based interventions) or coverage restrictions for repeated biopsies, may further enhance such outcomes.
Limitations
The results of this budget impact analysis are based on a hypothetical cohort modeled on the basis of values from the published literature. The use of national averages may not reflect the true variety in clinical practice. Initial prostate biopsy and repeated biopsy rates in specific geographic regions may be higher or lower than the reported national averages. Costs and resource utilization may also vary between practices and between geographic regions.
Another potential limitation to the application of this model is that the future rates of screening for prostate cancer may vary, given the recent recommendation of the USPSTF to stop routine PSA-based prostate cancer screening.6 The recommendation suggests that physicians discuss the benefit-risk ratio with their patients and decide if PSA testing is appropriate based on risk factors such as race or family history.6 How this will affect screening rates is not yet known and is not explored in this analysis.
This study is intended to address the financial impact of the epigenetic assay on the costs to commercial health plans of repeated biopsies. Because the cost impact is associated with a reduction in complications from the biopsy, the study provides some perspective on the impact of the assay on clinical outcomes. However, clinical outcomes were not evaluated in the design of the present study. A cost-effectiveness analysis would be the suitable approach to investigate the cost and the clinical outcomes associated with the use of the assay.
In addition, the assay's impact on the rates of prostate cancer diagnosis and earlier case identification were outside the scope of the study and were not methodically investigated.
Subgroup analyses were not performed for modestly elevated PSA patients versus those with markedly elevated PSA, because these patients are not managed differently in clinical practice.
Because of the negative predictive value of 90% of the epigenetic assay, 10% of patients testing negative with the assay could have a diagnosis of prostate cancer identified through recommended return screening. The clinical impact of this is not evaluated by the design of this model. The test has only been commercially available in the United States since May 2012.
Conclusion
Approximately $1.8 billion is spent annually on PSA testing alone, and more than $4 billion is spent on prostate cancer therapies, leading the AUA to call for new biomarkers to improve accurate diagnosis and reduce the cost burden. Epigenetic assays have been reported to improve the accuracy of prostate biopsies and help to prevent repeated biopsies, the majority of which show negative results. Based on a recent analysis, the results of a new epigenetic assay can guide urologists in decisions regarding the need to repeat a biopsy in patients with a previously negative biopsy who are still considered at risk for prostate cancer.
A budget impact analysis calculated whether this assay can also produce financial benefits, beyond the previously reported clinical benefits. Using a hypothetical health plan with 1 million members, this analysis shows that the total annual cost to the health plan would be reduced if patients with histopathologically negative biopsies would be managed with the epigenetic assay.
Specifically, the use of the assay would reduce the number of repeated biopsies from 1472 to 365, thereby preventing 1106 unnecessary biopsies and reducing the annual costs by approximately $500,000 to the health plan, based on the current standard of care. This test has only been available for a short time. Research to investigate the clinical impact of this essay based on real-world data will be appropriate.
Disclaimer
At the time this study was submitted for publication, prostate biopsy costs incorporated maximum payment reductions; conversion factor $25.0008 per the 2013 Medicare Physician Fee Schedule published December 5, 2012. If changes to the sustainable growth rate patch and sequestration cuts occur, savings for payers reimbursing the epigenetic assay may be greater than reported.
Author Disclosure Statement
Dr Aubry is a Consultant to MDxHealth. Dr Lieberthal receives research/grant support from Abbott Molecular, Genomic Health, and MDxHealth. Dr A. Willis is a Consultant to ConfirmMDx and 21st Century Oncology, and is on the Speaker's Bureau for Astellas, Clinlogix, GlaxoSmithKline, Eli Lilly, and sanofi aventis. Dr Bagley is a Consultant/Advisor to HillCo HEALTH. Mr Layton is a Consultant to ConfirmMDx. Mr S.M. Willis has nothing to disclose.
Contributor Information
Wade Aubry, Associate Clinical Professor of Medicine, Philip R. Lee Institute for Health Policy Studies, University of California, San Francisco, and Senior Medical Director, Quorum Consulting, San Francisco, CA.
Robert Lieberthal, Assistant Professor, Jefferson School of Population Health, Thomas Jefferson University, Philadelphia, PA.
Arnold Willis, Associate Dean of Clinical Sciences and Professor of Urology, Aureus University School of Medicine, Oranjestad, Aruba.
Grant Bagley, Senior Advisor, HillCo HEALTH, Washington, DC.
Simon M. Willis, III, Third-year medical student, Aureus University School of Medicine, Oranjestad, Aruba.
Andrew Layton, Director, Information Technology, Quorum Consulting, San Francisco, CA.
References
- 1.Loeb S, Carter HB, Berndt SI, et al. Complications after prostate biopsy: data from SEER-Medicare. J Urol. 2011; 186: 1830–1834 Epub 2011 Sep 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ekwueme DU, Stroud LA, Chen Y. Cost analysis of screening for, diagnosing, and staging prostate cancer based on a systematic review of published studies. Prev Chronic Dis. 2007; 4: A100. [PMC free article] [PubMed] [Google Scholar]
- 3.Van Neste L, Herman JG, Otto G, et al. The epigenetic promise for prostate cancer diagnosis. Prostate. 2012; 72: 1248–1261 [DOI] [PubMed] [Google Scholar]
- 4.Mosquera J-M, Mehra R, Regan MM, et al. Prevalence of TMPRSS2-ERG fusion prostate cancer among men undergoing prostate biopsy in the United States. Clin Cancer Res. 2009; 15: 4706–4711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.National Cancer Institute. Surveillance, Epidemiology and End Results. 2012. http://seer.cancer.gov/csr/1975_2009_pops09/results_single/sect_01_table.01.pdf Accessed November 1, 2012.
- 6.US Preventive Services Task Force. Screening for Prostate Cancer. May 2012. www.uspreventiveservicestaskforce.org/prostatecancerscreening/prostatefinalrs.htm Accessed September 20, 2012.
- 7.Crawford D, Grubb R, Pinsky P, Black A. Factors influencing initial treatment decisions in PLCO: comparison of screening and control arms. Abstract LBA4. Proceedings of the AUA Annual Meeting, May21, 2012; Atlanta, GA.
- 8.Stewart G, Van Neste L, Delvenne P, et al. Clinical utility of a multiplexed epigenetic gene assay to detect cancer in histopathologically negative prostate biopsies: results of the multicenter MATLOC study. J Urol. (2012), doi: 10.1016/j.juro.2012.08.219. [DOI] [PubMed]
- 9.Kehinde EO, Al-Maghrebi M, Sheikh M, Anim JT. Combined ciprofloxacin and amikacin prophylaxis in the prevention of septicemia after transrectal ultrasound-guided biopsy of the prostate. J Uro. 2012. September 23. pii: S0022–5347(12) 04945–2. doi: 10.1016/j.juro.2012.08.237. [DOI] [PubMed]
- 10.Trock B, Brotzman MJ, Mangold LA, et al. Evaluation of GSTP1 and APC methylation as indicators for repeat biopsy in a high-risk cohort of men with negative initial prostate biopsies. BJU Int. 2012; 110: 56–62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rabets JC, Jones JS, Patel A, Zippe CD. Prostate cancer detection with office based saturation biopsy in a repeat biopsy population. J Urol. 2004; 172: 94–97 [DOI] [PubMed] [Google Scholar]
- 12.Laurila M, van der Kwast T, Bubendorf L, et al. Detection rates of cancer, high grade PIN and atypical lesions suspicious for cancer in the European Randomized Study of Screening for Prostate Cancer. Eur J Cancer. 2010; 46: 3068–3072 [DOI] [PubMed] [Google Scholar]
- 13.Pinsky PF, Crawford ED, Kramer BS, et al. Repeat prostate biopsy in the prostate, lung, colorectal and ovarian cancer screening trial. BJU Int. 2007; 4: 775–779 [DOI] [PubMed] [Google Scholar]
- 14.Resnick MJ, Lee DJ, Magerfleisch L, et al. Repeat prostate biopsy and the incremental risk of clinically insignificant prostate cancer. Urology. 2011; 77: 548–552 [DOI] [PubMed] [Google Scholar]
- 15.National Cancer Institute Trends Progress Report-2009/2010 Updated. www.cancer.gov/newscenter/newsfromnci/2010/ProgressReport2010 Accessed November 1, 2012.
- 16.Merril J. Prostate cancer market snapshot: more than provenge. The Pink Sheet. November 22, 2010. Elsevier Business Intelligence Publications and Products. www.elsevierbi.com/Publications/The-Pink-Sheet/72/47/Prostate-Cancer-Market-Snapshot-More-Than-Provenge?result=2&total=2&searchquery=%253fq%253dProstate%252520Cancer%252520Market%252520Snapshot%2526date%253don%25253a11%25252f22%25252f2010 Accessed January 29, 2013.
- 17.Thompson I, Thrasher JB, Aus G, et al. Guideline for the management of clinically localized prostate cancer: 2007 update. J Urol. 2007; 177: 2106–2131 [DOI] [PubMed] [Google Scholar]
- 18.Stricker S. The era of personalized medicine in oncology: novel biomarkers ushering in new approaches to cancer therapy. Am Health Drug Benefits. 2011; 4: 387–388 [PMC free article] [PubMed] [Google Scholar]
- 19.Mehrotra J, Varde S, Wang H, et al. A quantitative, spatial resolution of the epigenetic field effect in prostate cancer. Prostate. 2008; 68: 152–160 [DOI] [PubMed] [Google Scholar]
- 20.Trujillo KA, Jones AC, Griffith JK, Bisoffi M. Markers of field cancerization: proposed clinical applications in prostate biopsies. Prostate Cancer. 2012; 2012: 302894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Centers for Medicare & Medicaid Services. Medicare Physician Fee Schedule 2013. www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/index.html?redirect=/physicianfeesched/ Accessed December 5, 2012.
- 22.Centers for Medicare & Medicaid Services. Medicare Inpatient Prospective Payment System 2012. www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html Accessed December 5, 2012.
- 23.US Census Bureau. Age and sex composition: 2010. 2010 census briefs. www.census.gov/prod/cen2010/briefs/c2010br-03.pdf Accessed November 1, 2012.
- 24.Drazer MW, Huo D, Schonberg MA, et al. Population-based patterns and predictors of prostate-specific antigen screening among older men in the United States. J Clin Oncol. 2011; 29: 1736–1743 Epub 2011 Mar 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nichol MB, Wu J, An JJ, et al. Budget impact analysis of a new prostate cancer risk index for prostate cancer detection. Prostate Cancer Prostatic Dis. 2011; 14: 253–261 [DOI] [PubMed] [Google Scholar]