Abstract
Multidrug-resistant (MDR) Acinetobacter baumannii infections are difficult to treat owing to the extremely limited armamentarium. Expectations about antimicrobial peptides' use as new powerful antibacterial agents have been raised on the basis of their unique mechanism of action. Musca domestica cecropin (Mdc), a novel antimicrobial peptide from the larvae of Housefly (Musca domestica), has potently active against Gram-positive and Gram-negative bacteria standard strain. Here we evaluated the antibacterial activity of Mdc against clinical isolates of MDR-A. baumannii and elucidate the related antibacterial mechanisms. The minimal inhibitory concentration (MIC) of Mdc was 4 μg/mL. Bactericidal kinetics of Mdc revealed rapid killing of A. baumannii (30 min). Flow cytometry using viability stain demonstrated that Mdc causes A. baumannii membrane permeabilization in a concentration- and time-dependent process, which correlates with the bactericidal action. Moreover, transmission electron microscopic (TEM) examination showed that Mdc is capable of disrupting the membrane of bacterial cells, resulting in efflux of essential cytoplasmic components. Overall, Mdc could be a promising antibacterial agent for MDR-A. baumannii infections.
1. Introduction
Acinetobacter baumannii (A. baumannii) has been recognized as one of the most problematic pathogen threat to public health in recent years [1, 2]. Particularly, many strains of A. baumannii have become resistant to common antibiotics, including aminoglycosides, fluoroquinolones, tetracyclines, carbapenems, and other extended-spectrum β-lactams, through a variety of mechanisms [3–6]. Apart from its intrinsic resistance mainly due to constitutive expression of certain efflux pumps as well as the low permeability of the outer membrane to certain antibiotics, A. baumannii is able to easily acquire and incorporate genetic elements such as transposons, integrons, and plasmids [3, 7–9]. Nosocomial infections, particularly in intensive care units, due to multidrug-resistant (MDR) isolates of A. baumannii are associated with increased morbidity and mortality. Therefore, an increased effort to search for new antimicrobial agents with antibacterial mechanisms that differ from common antibiotics is required.
Antimicrobial peptides (AMPs) have been isolated from a wide range of insects, bacteria, vertebrates, and plants [10]. They play one of the most important roles against pathogenic microorganisms in host defense system [11, 12]. In contrast with most antibiotics, AMPs generally exert their antimicrobial effect through physical interactions with cell membrane of target organisms [13]. This unique mechanism of action may reduce likelihood of emergence of resistance, thus raising the expectations about antimicrobial peptides' use as new powerful antimicrobial agents. A significant number of AMPs have been investigated against multidrug- resistant isolates both in vitro and in vivo [14, 15]. Many of these belong to the family of cecropin peptide [16]. Previous studies in cecropin and its related peptides demonstrate that formation of membrane-spanning pores that disrupt the cell membrane of the bacteria seems to be the most likely mechanism [17, 18]. However, the details surrounding the mechanism of bactericidal still need to be elucidated.
Musca domestica cecropin (Mdc) has been identified and characterized from the larvae of Housefly (Musca domestica), which has been used clinically to cure osteomyelitis, decubital necrosis, lip boil, and ecthyma and others in China [19]. The peptide exerts a potent antibacterial effect against Gram-negative and Gram-positive bacteria standard strain [20]. A limitation for the transition of cecropin to therapeutic purpose is the hemolytic or cytotoxicity properties [21]. Of important note, Mdc did not show a perceptible cytotoxic effect on human red blood cells [22]. So, it is a subject for further studies whether Mdc will be a novel and effective clinical alternative against the increasing threat of a widespread dissemination of multidrug-resistant A. baumannii. The aim of the present study was to evaluate the antibacterial activity of Mdc against clinical isolates of multidrug-resistant nosocomial isolates of A. baumannii. We also attempted to acquire the details surrounding the antibacterial mechanism by flow cytometry and transmission electron microscopy.
2. Materials and Methods
2.1. Reagents
Musca domestica cecropin (GWLKKIGKKIERVGQHTRDATIQTIGVAQQAANVAATLKG-NH2) was prepared by conventional Fmoc solid-phase synthetic method with a 431 peptide synthesizer (Applied Biosystems Inc., Foster City, CA). The synthesized peptide was purified to near homogeneity (>95%) by preparative reversed phase-high performance liquid chromatography (RP-HPLC) (Waters Delta-Pak C18, 15 μm, 300 Å, 25 × 100 mm) and was further characterized by analytical RP-HPLC (Waters Symmetry C18, 3.5 μm, 100 Å, 4.6 × 150 mm) and mass determination of the eluate with an API electrospray ionization mass spectrometer (Perkin Elmer SCIEX). All other chemicals used were of analytical grade.
2.2. Microorganisms and Medium
Multidrug-resistant clinical isolate of A. baumannii GIM1.650 was obtained from the Center of Medical Laboratory of the First Affiliated Hospital of Guangdong Pharmaceutical University, Guangzhou, China. This strain is resistant to most tested antibiotics including ampicillin (>16 μg/mL), cefazolin (>16 μg/mL), ceftriaxone (>32 μg/mL), ceftazidime (>16 μg/mL), piperacillin (>64 μg/mL), cefepime (>16 μg/mL), aztreonam (>16 μg/mL), ampicillin/sulbactam (>16 μg/mL), amoxicillin/clavulanic acid (>16 μg/mL), piperacillin/tazobactam (>64 μg/mL), gentamicin (>8 μg/mL), ciprofloxacin (>2 μg/mL), amikacin (>32 μg/mL), levofloxacin (>32 μg/mL), imipenem (>8 μg/mL), meropenem (>8 μg/mL), Tetracycline (>8 μg/mL), bactrim (>2 μg/mL), and levofloxacin (>8 μg/mL) with the exception of polymyxin E (⩽0.5 μg/mL). The control strains A. baumannii ATCC 19606 were obtained from American Type Culture Collection (ATCC). The compositions of medium in this work are as follows: Luria-Bertani (LB) medium (w/v): 1% NaCl, 1% tryptone, 0.5% yeast extract and Mueller-Hutton (MH) medium (w/v): 1.75% casamino acid, 0.5% beef extract, and 0.15% starch. LB medium was used for preincubation of the test bacteria. MH medium was employed for antibacterial assays.
2.3. Antimicrobial Activity Assay
For determination of minimal inhibitory concentration (MIC), the broth microdilution method for Mdc was carried out according to the procedures outlined by the Clinical and Laboratory Standards Institute. Briefly, test bacteria strains were inoculated to 5 mL of MH broth (MHB), then grown overnight to OD600 nm of 0.8 with shaking (200 rpm), and diluted to 2 × 106 colony forming units (CFUs) per mL with MHB medium. A 100 μL cell suspension was dispensed in each well of sterile 96-well polypropylene microtiter plate (Sigma-Aldrich), and 100 μL of MHB without bacterial suspension was served as negative controls. A 100 μL of each 2-fold serial dilutions in phosphate-buffered saline (PBS) solution was prepared from a stock solution of the peptide (128 μg/mL), and PBS was used as positive controls. The plate was incubated for 18–24 h at 37°C, and the MIC was defined as the lowest concentration of antimicrobial agent that completely inhibits growth of the organism in the microdilution wells measured by turbidimetry at 600 nm. The results were present as mean values of three independent experiments. A. baumannii ATCC 19606, a reference strain that is susceptible to colistin sulfate, was used as a control. Minimal bactericidal concentration (MBC) was recorded as the lowest concentration of peptide that killed 99.9% of the test inoculum by viable counting assay. To identify the growth inhibitory activity kinetic, OD600 nm of 96-well plate prepared as above was monitored over time until 16 h stationary incubation at 37°C.
2.4. Kinetics of Bacterial Killing
Kinetics of bacterial killing was determined according to the method described in Lu et al. [22]. Log-phase A. baumannii strains GIM1.650 (106 CFU/mL) were incubated with culture medium containing zero or MIC, 1/4 MIC Mdc for 120 min. Aliquots of the mix were removed at fixed intervals, serially diluted 10-fold in PBS, plated on LB agar, and incubated 16–24 h at 37°C. CFU was counted to determine cell viability. PBS solution was used as the control. The experiments were carried out in duplicate.
2.5. Membrane Permeabilization by Flow Cytometry Analysis
Membrane permeabilization of Mdc on bacterial was investigated by flow cytometry using the DNA intercalating dye propidium iodide (PI). A. baumannii strains GIM1.650 strains were prepared as in Section 2.3. A suspension of approximately 2 × 106 cells per mL at log phase was treated with Mdc (0–2 × MIC) Mdc and incubated at 37°C for 0–120 min. The bacterial cells were harvested by centrifugation and stained with PI (Invitrogen Ltd, Paisley, UK) (final concentration 5 mg/mL) at room temperature in the dark for 30 min. Flow cytometry was performed using a FACScan (BD Biosciences, NJ, USA). Bacteria were initially gated using forward scatter (FS) and then analyzed for red fluorescence. All experiments were conducted in triplicate and for each sample 10 000 stained bacteria were recorded. Heat-killed cells (at 70°C for 30 min) were used as a positive control of PI and bacteria without Mdc were used as a viability control.
2.6. Transmission Electron Microscopy
Transmission electron microscopy (TEM) was used to evaluate the morphological changes in A. baumannii cells after treatment with Mdc. A. baumannii cells in log phase being collected and incubated for 60 min at 37°C in the presence and absence of the Mdc at a concentration of 8 μg/mL. Upon incubation, the pellet obtained after centrifugation was fixed in 0.1 M phosphate buffer (containing 3% of glutaraldehyde and 1.5% paraformaldehyde) for 2.5 h at 4°C. To postfix the cells, 1% osmium tetroxide and 1.5% potassium ferrocyanide were added and the samples were left at 4°C for 1.5 h. The samples were dehydrated in an ethanol series and embedded in epoxy resin. Thin slices of the pellet were made and stained with 2% uranyl acetate for 1 h. The sections were examined in a JEM1400 (Jeol, Tokyo, Japan) transmission electron microscopy and microphotographs were taken with a digital camera Bioscan 792 (Gatan Inc., Pleasanton, USA).
3. Results
3.1. Antimicrobial Activity
To test for antimicrobial activity of Mdc, MIC and MBC were determined by broth microdilution method. The results of MIC (4 μg/mL) and MBC (8 μg/mL) did not show significant differences among the control strain ATCC 19606 and the multidrug-resistant clinical isolate of A. baumannii (Table 1).
Table 1.
Minimal inhibition concentration (MIC) and minimum bactericidal concentration (MBC) of Mdc.
| MIC (μg/mL) | MBC (μg/mL) | |
|---|---|---|
| ATCC 19606 | 4 | 8 |
| GIM 1.650 | 4 | 8 |
3.2. Kinetics of Bacterial Killing
The growth inhibitory activity kinetic was investigated and the kinetic plot is shown in Figure 1. The MIC (4 μg/mL) completely inhibited A. baumannii GIM1.650 growth. Below the MIC (1 μg/mL), the growth lag was greater than the zero peptide control. To further explore its antimicrobial potency of Mdc, kinetics of bacterial killing were conducted to determine the rate of lysis. The results showed that Mdc reduced the A. baumannii GIM1.650 CFU at concentrations of MIC (4 μg/mL), 1/4 MIC (1 μg/mL) after 10 min incubation compared with negative controls. At concentrations of MIC (4 μg/mL), killing by Mdc was complete after a 30 min exposure period (Figure 2).
Figure 1.
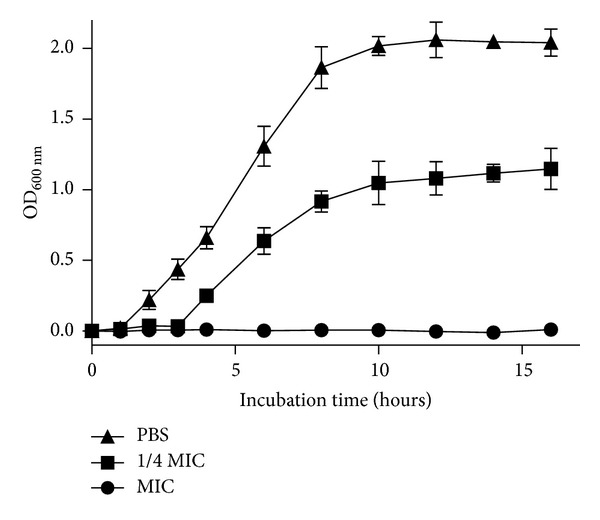
Growth inhibitory activity of Mdc against A. baumannii GIM1.650. The experiment was carried out in triplicate, and plots represent the average values. Error bars refer to the standard deviations.
Figure 2.
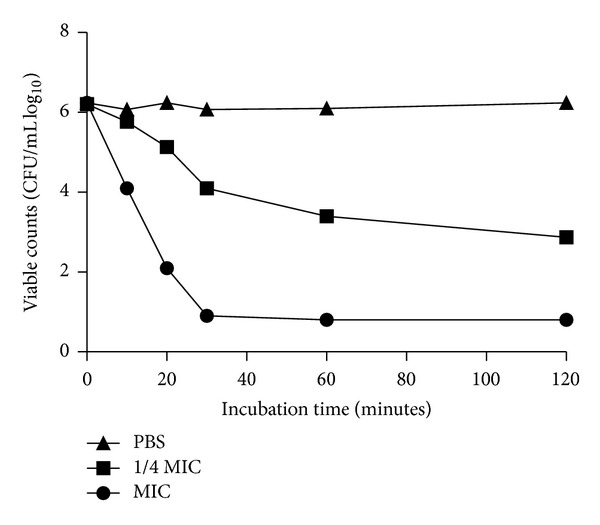
Bactericidal kinetics of Mdc against A. baumannii GIM1.650 after incubation with different concentration of Mdc. A 5-log reduction occurs after 30 min at the MIC (4 μg/mL). The experiment was carried out in triplicate, and average values are reported (SD ⩽10%).
3.3. Membrane Permeabilization by Flow Cytometry Analysis
To determine whether Mdc has an effect on the integrity of bacterial membrane, we tested its effect on bacterial cells by PI-based flow cytometry analysis. Flow cytometric analysis of A. baumannii viability control showed that the bacteria were 98.89% viable (Figure 3(a)). The positive control showed that a total of 86.87% of heat treated cells were permeable to PI (Figure 3(c)). It was interestingly found that following incubation with Mdc at MIC result in 99.78% of A. baumannii cells were PI-positive cells (Figure 3(b)). Kinetics of Mdc membrane permeability against A. baumannii was shown to be a time- and concentration-dependent process (Figure 3(e)).
Figure 3.
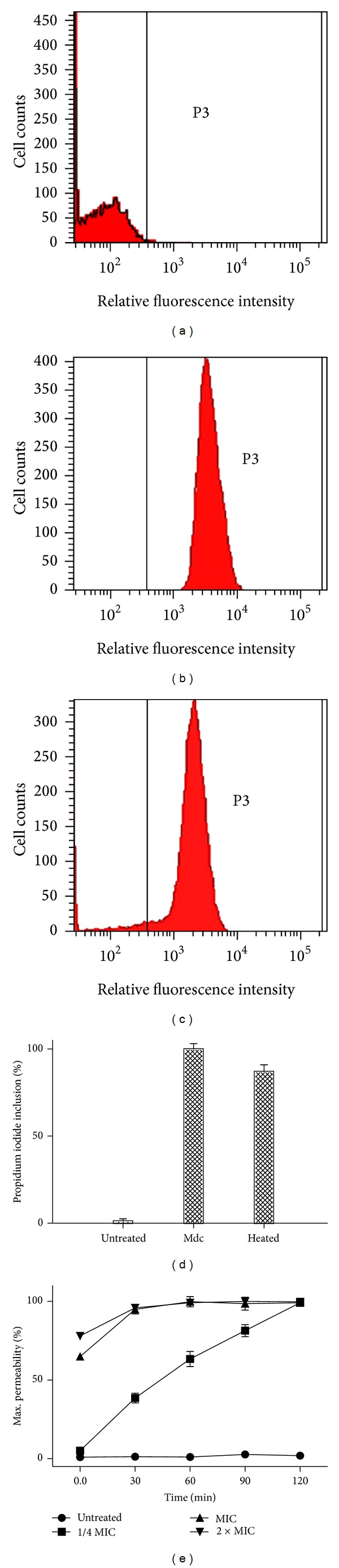
Effect of Mdc on the membrane permeability of A. baumannii GIM1.650 analyzed by the flow cytometer. The relative fluorescence intensities within the P3 regions were taken as PI-positive cells. (a) Untreated cells control, (b) cells treated with 4 μg/mL Mdc at 37°C for 1 hour, (c) dead control (cells heated at 70°C), (d) percentage of cells stained with propidium iodide (PI), and (e) kinetics of Mdc membrane permeability against A. baumannii.
3.4. Transmission Electron Microscopy (TEM)
To investigate the morphological changes of A. baumannii cells after being treated with Mdc, transmission electron microscopy was used. The intact (control) cells of A. baumannii were shown in Figure 4(a), in which the inner and outer membranes of the A. baumannii envelope were smooth. The cytoplasmic content of bacterial cell was evenly distributed and the cell aggregation was not detected. Compared with control cells, significant morphological changes were observed in Mdc treated A. baumannii cells (Figure 4(b)). A great number of cells appeared with their cell membrane damaging and the cytoplasm content leaked to the extracellular medium. The outer membrane of the bacteria cell is distended and displaced in relation to the plasma membrane. Furthermore, coagulated material in cytoplasm was observed.
Figure 4.
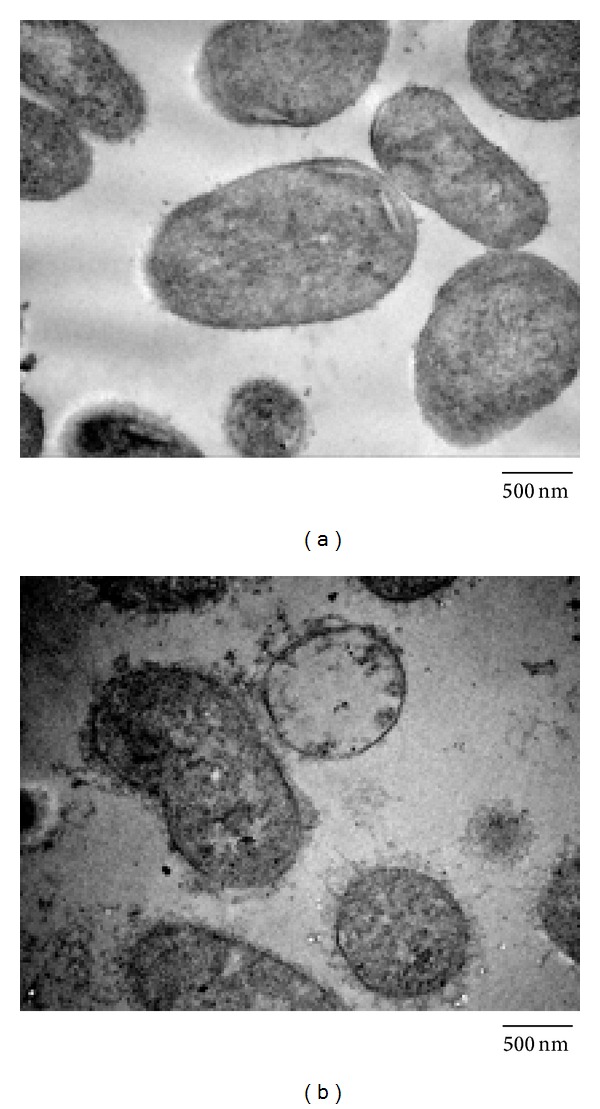
Ultrastructural damages in A. baumannii GIM1.650 treated with Mdc. Bacteria were untreated (a) or treated with Mdc (b) at a concentration of 8 μg/mL. Cells were processed to be analyzed under TEM.
4. Discussion
A. baumannii acts as an opportunistic pathogen, causing severe nosocomial infections with high mortality rates [2]. The outstanding capacity of A. baumannii to develop resistance against the common antibacterial agents [23] makes this pathogen as important target for bactericidal activity evaluation of Mdc. In the present study, the same MIC and MBC value of Mdc were found in control strain ATCC 19606 and multidrug-resistant clinical isolate GIM1.650. The results indicate that Mdc has potent antibacterial activity against A. baumannii, and its mechanism of antimicrobial action is different from the common antibacterial agents. The lag time and an obvious slower growth rate shown in Figure 1 indicating cell growth have been inhibited by Mdc initial at 1 μg/mL, but the interaction was not sufficient to kill the cells. Killing kinetic analysis was conducted to further explore the antimicrobial action of Mdc at the MIC, 1/4 MIC concentration. The results revealed that Mdc can mediate rapid killing of MDR-A. baumannii in 30 min and resulted in log orders of cell complete lysis at a concentration as low as 4 μg/mL (Figure 2). These observations were in agreement with previous results obtained in other multidrug-resistant strains of bacteria with antimicrobial peptides [17].
A number of techniques are available for studying the mechanisms of antimicrobial action on bacteria [13, 24, 25]. In the present work, we set up a study using different tools (flow cytometry and transmission electron microscopy) to address the same hypothesis. At first, to investigate the membrane permeability of MDR-A. baumannii, PI-based flow cytometry assay was performed to determine bacterial viability. PI is a specific nucleic acid-binding dye that is fully excluded from intact bacterial cells. When the membrane of the bacteria is compromised, the dye penetrates the cell and binds readily to nucleic acids, thus selectively labeling dead cells. The results of flow cytometry assay (Figures 3(b) and 3(d)) indicate that the bacterial membrane is permeabilized by Mdc. To explore a more definitive correlation of membrane permeability and bactericidal action, the kinetics of membrane permeability was assayed (Figure 3(e)). The results showed that membrane permeability of A. baumannii by Mdc is a time- and concentration-dependent manner. At the MIC of 4 μg/mL, the maximum permeability is achieved at 30 min. Above the MIC, the extent of permeability did not change. Below the MIC (1 μg/mL), it takes 2 h to approach the maximum permeability. The rate of permeability for 4 μg/mL and 8 μg/mL is similar, whereas 1 μg/mL is slightly faster. Bactericidal kinetics showed complete killing of A. baumannii in 30 min by Mdc at MIC (Figure 1), which directly correlates with the permeabilization. This suggests that, at and above the MIC, the immediate permeabilization caused damage too significant for the cell to recover and resume growth. Blow the MIC (1 μg/mL), Mdc appears capable of causing disruptions to the membrane, resulting in damaged cells that subsequently recover.
Second, in order to further investigate the effect of Mdc on cell membrane, we observed the Mdc treated cells using TEM. The results showed that Mdc is capable of disrupting the membrane of sensitive cells, resulting in efflux of essential cytoplasmic components. Moreover, the outer membrane is displaced and distended with the inner membrane, possibly due to the disruption of contact sites between outer membrane and peptidoglycan or the increasing number of Mdc-induced pores that cause the increasing hydrostatic pressure in the periplasmic space.
In conclusion, the results of the present study demonstrated that Mdc has potent antibacterial activity against multidrug-resistant nosocomial isolates of A. baumannii. Bactericidal kinetics, flow cytometry, and transmission electron microscopic assays indicated that Mdc could alter the permeability properties to mediate the disruption of A. baumannii membrane, which then leads to the death of bacterial cells. Overall, Mdc could be a novel and promising clinical alternative against the increasing threat of a widespread dissemination of multidrug-resistant A. baumannii.
Acknowledgment
This work was supported by a Grant from Science and Technology Innovation Commission of Shenzhen Municipality Program (no. JCYJ 20120830113244693).
Conflict of Interests
The authors hereby would like to declare that none of the contributing authors have any conflict of interests, including specific financial interests and relationships and affiliations to the subject matter or materials discussed in this paper.
Authors' Contribution
Shuiqing Gui and Rongjiang Li contributed equally to this paper.
References
- 1.Dijkshoorn L, Nemec A, Seifert H. An increasing threat in hospitals: multidrug-resistant Acinetobacter baumannii . Nature Reviews Microbiology. 2007;5(12):939–951. doi: 10.1038/nrmicro1789. [DOI] [PubMed] [Google Scholar]
- 2.Abbott I, Cerqueira GM, Bhuiyan S, Peleg AY. Carbapenem resistance in Acinetobacter baumannii: laboratory challenges, mechanistic insights and therapeutic strategies. Expert Review of Anti-Infective Therapy. 2013;11(4):395–409. doi: 10.1586/eri.13.21. [DOI] [PubMed] [Google Scholar]
- 3.Neonakis IK, Spandidos DA, Petinaki E. Confronting multidrug-resistant Acinetobacter baumannii: a review. International Journal of Antimicrobial Agents. 2011;37(2):102–109. doi: 10.1016/j.ijantimicag.2010.10.014. [DOI] [PubMed] [Google Scholar]
- 4.Bourigault C, Corvec S, Bretonniere C, et al. Investigation and management of multidrug-resistant Acinetobacter baumannii spread in a French medical intensive care unit: one outbreak may hide another. American Journal of Infection Control. 2012;41(7):652–653. doi: 10.1016/j.ajic.2012.08.003. [DOI] [PubMed] [Google Scholar]
- 5.Suzuki M, Matsui M, Suzuki S, et al. Genome sequences of multidrug-resistant Acinetobacter baumannii strains from nosocomial outbreaks in Japan. Genome Announcements. 2013;1(4) doi: 10.1128/genomeA.00476-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mishra SK, Rijal BP, Pokhrel BM. Emerging threat of multidrug resistant bugs—Acinetobacter calcoaceticus baumannii complex and methicillin resistant Staphylococcus aureus . BMC Research Notes. 2013:98–103. doi: 10.1186/1756-0500-6-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang Y, Chua KL. Assessment of the effect of efflux pump inhibitors on in vitro antimicrobial susceptibility of multidrug-resistant Acinetobacter baumannii . International Journal of Antimicrobial Agents. 2013;42(3):283–284. doi: 10.1016/j.ijantimicag.2013.05.011. [DOI] [PubMed] [Google Scholar]
- 8.Jiang W, Liu H, Zhong M, Yang YC, Xiao DW, Huang WF. Study on the resistant genes to carbapenems and epidemiological characterization of multidrug-resistant Acinetobacter baumannii isolates. Microbial Drug Resistance. 2013;19(2):117–123. doi: 10.1089/mdr.2012.0049. [DOI] [PubMed] [Google Scholar]
- 9.Zhao W-H, Hu Z-Q. Acinetobacter: a potential reservoir and dispenser for β-lactamases. Critical Reviews in Microbiology. 2012;38(1):30–51. doi: 10.3109/1040841X.2011.621064. [DOI] [PubMed] [Google Scholar]
- 10.Guerreiro CIPD, Fontes CMGA, Gama M, Domingues L. Escherichia coli expression and purification of four antimicrobial peptides fused to a family 3 carbohydrate-binding module (CBM) from Clostridium thermocellum. Protein Expression and Purification. 2008;59(1):161–168. doi: 10.1016/j.pep.2008.01.018. [DOI] [PubMed] [Google Scholar]
- 11.Guaní-Guerra E, Santos-Mendoza T, Lugo-Reyes SO, Terán LM. Antimicrobial peptides: general overview and clinical implications in human health and disease. Clinical Immunology. 2010;135(1):1–11. doi: 10.1016/j.clim.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 12.Huang L, Leong SSJ, Jiang R. Soluble fusion expression and characterization of bioactive human beta-defensin 26 and 27. Applied Microbiology and Biotechnology. 2009;84(2):301–308. doi: 10.1007/s00253-009-1982-z. [DOI] [PubMed] [Google Scholar]
- 13.Fernandez DI, Le Brun AP, Whitwell TC, Sani MA, James M, Separovic F. The antimicrobial peptide aurein 1.2 disrupts model membranes via the carpet mechanism. Physical Chemistry Chemical Physics. 2012;14(45):15739–15751. doi: 10.1039/c2cp43099a. [DOI] [PubMed] [Google Scholar]
- 14.Feng X, Sambanthamoorthy K, Palys T, Paranavitana C. The human antimicrobial peptide LL-37 and its fragments possess both antimicrobial and antibiofilm activities against multidrug-resistant Acinetobacter baumannii . Peptides. 2013;49(11):131–137. doi: 10.1016/j.peptides.2013.09.007. [DOI] [PubMed] [Google Scholar]
- 15.Mishra B, Leishangthem GD, Gill K, et al. A novel antimicrobial peptide derived from modified N-terminal domain of bovine lactoferrin: design, synthesis, activity against multidrug-resistant bacteria and Candida . Biochimica Et Biophysica Acta. 2013;1828(2):677–686. doi: 10.1016/j.bbamem.2012.09.021. [DOI] [PubMed] [Google Scholar]
- 16.Giacometti A, Cirioni O, Kamysz W, et al. In vitro activity and killing effect of the synthetic hybrid cecropin A-melittin peptide CA(1-7)M(2-9)NH2 on methicillin-resistant nosocomial isolates of Staphylococcus aureus and interactions with clinically used antibiotics. Diagnostic Microbiology and Infectious Disease. 2004;49(3):197–200. doi: 10.1016/j.diagmicrobio.2004.02.008. [DOI] [PubMed] [Google Scholar]
- 17.Arcidiacono S, Soares JW, Meehan AM, Marek P, Kirby R. Membrane permeability and antimicrobial kinetics of cecropin P1 against Escherichia coli . Journal of Peptide Science. 2009;15(6):398–403. doi: 10.1002/psc.1125. [DOI] [PubMed] [Google Scholar]
- 18.Schmitt P, Mercado L, Díaz M, Guzmán F, Arenas G, Marshall SH. Characterization and functional recovery of a novel antimicrobial peptide (CECdir-CECret) from inclusion bodies after expression in Escherichia coli . Peptides. 2008;29(4):512–519. doi: 10.1016/j.peptides.2007.12.012. [DOI] [PubMed] [Google Scholar]
- 19.Hou L, Shi Y, Zhai P, Le G. Antibacterial activity and in vitro anti-tumor activity of the extract of the larvae of the housefly (Musca domestica) Journal of Ethnopharmacology. 2007;111(2):227–231. doi: 10.1016/j.jep.2006.11.015. [DOI] [PubMed] [Google Scholar]
- 20.Lu X-M, Jin X-B, Zhu J-Y, et al. Expression of the antimicrobial peptide cecropin fused with human lysozyme in Escherichia coli . Applied Microbiology and Biotechnology. 2010;87(6):2169–2176. doi: 10.1007/s00253-010-2606-3. [DOI] [PubMed] [Google Scholar]
- 21.Mihajlovic M, Lazaridis T. Antimicrobial peptides bind more strongly to membrane pores. Biochimica et Biophysica Acta, Biomembranes. 2010;1798(8):1494–1502. doi: 10.1016/j.bbamem.2010.02.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lu X, Shen J, Jin X, et al. Bactericidal activity of Musca domestica cecropin (Mdc) on multidrug-resistant clinical isolate of Escherichia coli . Applied Microbiology and Biotechnology. 2012;95:939–945. doi: 10.1007/s00253-011-3793-2. [DOI] [PubMed] [Google Scholar]
- 23.Rodríguez-Hernández MJ, Saugar J, Docobo-Pérez F, et al. Studies on the antimicrobial activity of cecropin A-melittin hybrid peptides in colistin-resistant clinical isolates of Acinetobacter baumannii . Journal of Antimicrobial Chemotherapy. 2006;58(1):95–100. doi: 10.1093/jac/dkl145. [DOI] [PubMed] [Google Scholar]
- 24.Bouhdid S, Abrini J, Zhiri A, Espuny MJ, Manresa A. Investigation of functional and morphological changes in Pseudomonas aeruginosa and Staphylococcus aureus cells induced by origanum compactum essential oil. Journal of Applied Microbiology. 2009;106(5):1558–1568. doi: 10.1111/j.1365-2672.2008.04124.x. [DOI] [PubMed] [Google Scholar]
- 25.Park Y, Lee DG, Jang S-H, et al. A Leu-Lys-rich antimicrobial peptide: activity and mechanism. Biochimica et Biophysica Acta, Proteins and Proteomics. 2003;1645(2):172–182. doi: 10.1016/s1570-9639(02)00541-1. [DOI] [PubMed] [Google Scholar]


