Abstract
T-cell activation, proliferation and selection of the cell death pathway depend on the production of reactive oxygen intermediates (ROIs) and ATP synthesis, which are tightly regulated by the mitochondrial transmembrane potential (ΔΨm). Mitochondrial hyperpolarization (MHP) and ATP depletion represent early and reversible steps in T-cell activation and apoptosis. By contrast, T cells of patients with systemic lupus erythematosus (SLE) exhibit persistent MHP, cytoplasmic alkalinization, increased ROI production and depleted ATP, which mediate enhanced spontaneous and diminished activation-induced apoptosis and sensitize lupus T cells to necrosis. Necrotic, but not apoptotic, cell lysates activate dendritic cells and might account for increased interferon a production and inflammation in lupus patients. MHP is proposed as a key mechanism of SLE pathogenesis and is therefore a target for pharmacological intervention.
Innate and adaptive immune responses depend on controlled production of ATP and reactive oxygen intermediates (ROIs) in mitochondria. In response to antigenic stimulation, clonal expansion of T and B cells are continuously downsized and potentially autoreactive cells are eliminated by apoptosis. An array of signals through the T-cell receptor (TCR), co-stimulatory molecules, cell death receptors, lymphokines, and other circulating metabolites, such as ATP, NAD, cADPR, glucose, glutathione, nitric oxide (NO) and ROIs, determine the fate of T cells [1]. T-cell activation and death pathway selection depend on the production of ROIs and ATP synthesis, which are tightly regulated by the mitochondrial transmembrane potential (ΔΨm) (Box 1). Disruption of ΔΨm has been proposed as the point of no return in apoptotic signaling [2]. We recently discovered that an elevation of Dcm [or mitochondrial hyperpolarization (MHP)] occurs before activation of caspases, phosphatidylserine (PS) externalization and disruption of ΔΨmin Fas- [3] and H2O2-induced apoptosis of Jurkat human leukemia T cells and normal human peripheral blood lymphocytes (PBLs) [4]. These observations were confirmed and extended to p53 [5], tumor necrosis factor (TNF)-α [6], staurosporin [7], camptothecin [8] and NO-induced apoptosis [9] (Table 1). MHP is also triggered by activation of the CD3–CD28 complex [10] or stimulation with concanavalin A (ConA) [3], interleukin (IL)-10, IL-3, interferon (IFN)-γ or transforming growth factor (TGF)-β [11]. T-cell-activation-induced MHP is associated with transient inhibition of F0F1-ATPase, enhanced ROI production, ATP depletion and sensitization to necrosis [10], suggesting that ΔΨm elevation is a crucial checkpoint of T-cell fate decisions.
Box 1. Regulation of the mitochondrial transmembrane potential (ΔΨm).
The mitochondrial transmembrane potential (ΔΨm; negative inside and positive outside) is the result of an electrochemical gradient maintained by two transport systems – the electron transport chain and the F0F1-ATPase complex [44]. The electron transport chain catalyzes the flow of electrons from NADH to molecular oxygen and the translocation of protons across the inner mitochondrial membrane, thus creating a voltage gradient with negative charges inside the mitochondrial matrix [22]. A small fraction of electrons react directly with oxygen and form reactive oxygen intermediates (ROIs). Activity of the F0F1-ATPase complex has crucial roles in oxidative phosphorylation (i.e. conversion of ADP to ATP at the expense of the electrochemical gradient during oxidative phosphorylation) [22]. Mitochondrial membrane integrity is dependent on the oxidation–reduction equilibrium of ROI, pyridine nucleotides (NADH/NAD + NADPH/NADP) and reduced glutathione (GSH) levels [68]. Regeneration of GSH by glutathione reductase from its oxidized form, GSSG, depends on NADPH produced by the pentose phosphate pathway (PPP) [43]. ROI levels and ΔΨm are regulated by transaldolase through the supply of reducing equivalents from PPP [3,34], Ca2+ fluxing and NO production [23]. Whereas ROIs have been considered as toxic byproducts of aerobic existence, evidence is now accumulating that controlled levels of ROIs modulate various aspects of cellular function and are necessary for signal transduction pathways, including those mediating T-cell activation and apoptosis [1]. Mitochondrial hyperpolarization (MHP), an early event of T-cell activation and death, appears to be mediated through inhibition of F0F1-ATPase or dephosphorylation of cytochrome c oxidase [44]. Nitric oxide (NO), acting as a competitive antagonist of oxygen, can also reversibly inhibit cytochrome c oxidase and cause MHP [69]. Using the energy of ATP, F0F1-ATPase can pump protons out of the mitochondrial matrix into the intermembrane space, thus causing ΔΨm elevation. MHP leads to uncoupling of oxidative phosphorylation (i.e. continued ROI production in the absence of ATP synthesis), which disrupts ΔΨm and damages integrity of the inner mitochondrial membrane. Disruption of ΔΨm has been proposed as the point of no return in cell death signaling [2]. This releases cytochrome c and other cell-death-inducing factors from mitochondria into the cytosol. Intracellular ATP concentration is a key switch in the decision of the cell to die by apoptosis or necrosis [26]. Whereas apoptosis is energy dependent and persistent, MHP has been associated with ATP depletion and sensitization to necrosis in lupus T cells [10]. Thus, regulation of ΔΨm is a crucial checkpoint of T-cell fate decisions.
Table 1.
Signals eliciting MHP and ROI productiona
| Signal | Refs |
|---|---|
| Fas | [3] |
| TNF-α | [6] |
| ConA | [3] |
| HIV | [62,63] |
| H2O2 | [4,10,64] |
| Activation of the CD3–CD28 complex | [10,11] |
| IL-10 | [11] |
| IL-3 | [11] |
| TGF-β | [11] |
| IFN-γ | [11] |
| Staurosporin | [7] |
| Camptothecin | [8] |
| NO | [9,23] |
Abbreviations: ConA, concanavalin A; IFN-γ, interferon γ; IL, interleukin; MHP, mitochondrial hyperpolarization; NO, nitric oxide; ROI, reactive oxygen intermediate; TGF-β, transforming growth factor β; TNF-α, tumor necrosis factor α
T-cell activation is regulated by mitochondrial ROI production
ROIs modulate T-cell activation, cytokine production and proliferation at multiple levels [12]. The antigen-binding αβ or γδTCR is associated with a multimeric receptor module comprising the CD3γδε and TCRζ chains. The cytoplasmic domains of the CD3 and ζ chains contain a common motif, termed the immunoreceptor tyrosinebased activation motif (ITAM), which is crucial for the coupling of intracellular tyrosine kinases [13]. Expression of the TCRζ chain is suppressed by ROIs [14]. Binding of p56lck to CD4 or CD8 attracts this kinase to the TCRz–CD3 complex, leading to phosphorylation of ITAM. Phosphorylation of both tyrosines of each ITAMis required for Src-homology 2 (SH2)-mediated binding by ZAP-70 (ζ-chain-associated protein of 70 kDa) or the related SYK. ZAP-70 is activated through phosphorylation by p56lck. Activated ZAP-70 and SYK target two key adaptor proteins, LAT and SLP-76 [13]. Oxidative stress reduces phosphorylation and displacement of LAT from the cell membrane, thus causing T-cell hyporesponsiveness [15]. Phosphorylated LAT binds directly to phospholipase Cγ1, which controls hydrolysis of phosphatidylinositol (4,5)-bisphosphate [PtdIns(4,5)P2] to inositol (1,4,5)-trisphosphate [Ins(1,4,5)P3] and diacylglycerol (DAG). Phosphorylation of inositol lipid second messengers is mediated by phosphoinositide 3-kinase (PI3-K). The stimulatory effect of the TCR alone on PI3-K activity is small. Concurrent triggering of the CD28 co-stimulatory molecule by its ligands CD80 or CD86 is required for optimal PI3-K activation. Ins(1,4,5)P3 binds to its receptors in the endoplasmic reticulum, opening Ca2+ channels that release Ca2+ into the cytosol. Increased cytosolic Ca2+ concentration activates the phosphatase calcineurin, which dephosphorylates the transcription factor NFAT. Dephosphorylated NFAT can translocate to the nucleus, where it promotes transcription of IL-2 in concert with AP-1, NF-κB and Oct-1. Whereas activities of AP-1 and NF-κB are increased by oxidative stress [16], both thiol insufficiency and H2O2 treatment suppresses calcineurin-mediated activation of NFAT [17]. Thus, expression of cytokines can be selectively regulated by oxidative stress depending on the relative expression level of transcription factors involved (e.g. IL-2 is expressed through a promoter that has AP-1 and NFAT motifs, and IL-4 is expressed through an AP-1-less NFAT enhancer; Figure 1).
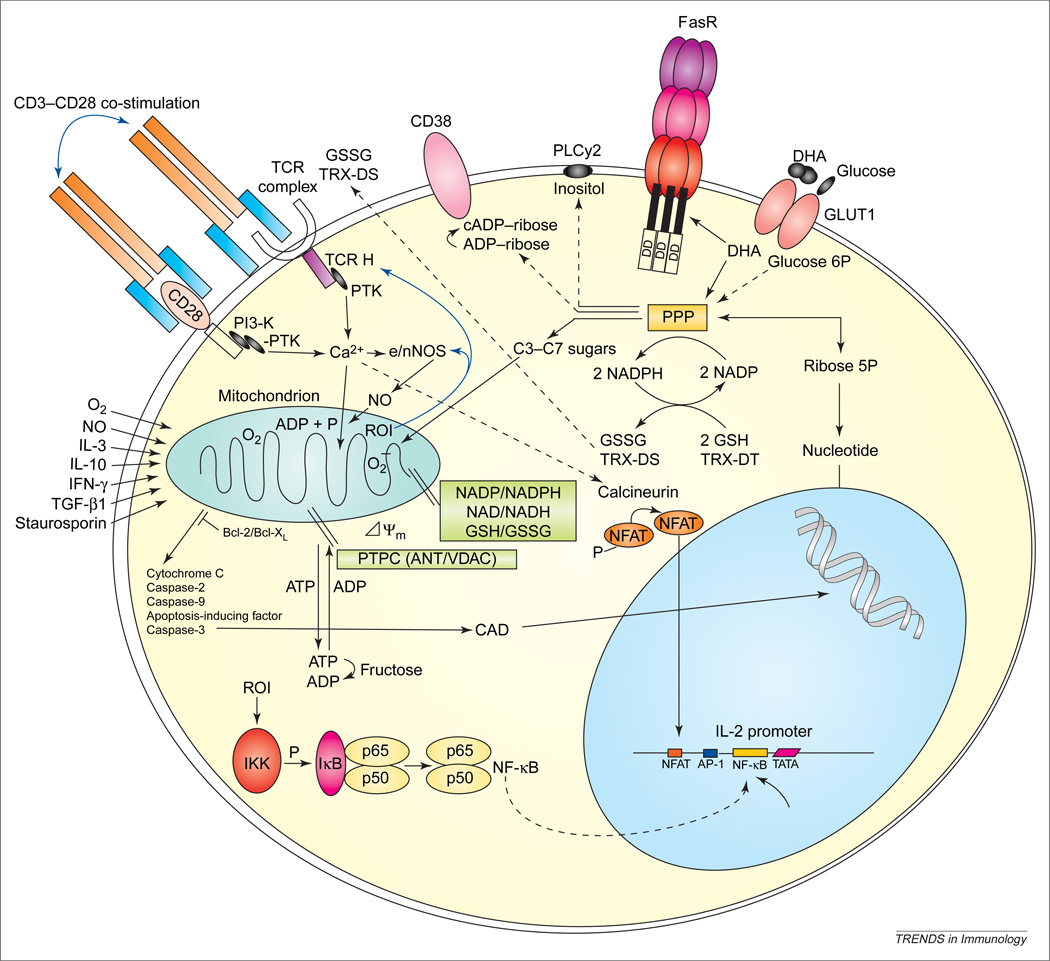
Figure 1.
Mitochondrial and metabolic checkpoints of T-cell activation and apoptosis signals. Antigen binding-initiated signaling through the T-cell receptor (TCR)–CD3–TCRζ-chain complex and the CD28 co-stimulatory molecule activate phosphoinositide 3-kinase (PI3-K) and protein tyrosine kinase (PTK). Increased cytosolic Ca2+; concentration activates the serine/threonine phosphatase calcineurin, which dephosphorylates the NFAT. Dephosphorylated NFAT can translocate to the nucleus, where it promotes transcription of interleukin-2 (IL-2) in concert with AP-1 and NF-κB. Ca2+ flux into mitochondria increases production of reactive oxygen intermediates (ROIs) and NF-κB activation [22]. Excess ROI production and disruption of ΔΨm lead to activation-induced cell death executed by caspase-3 (digesting vitally important proteins PARP, 70K U1RNP, lamin and actin) and caspase-activated DNase (CAD). Cleavage by caspase-3 is thought to expose cryptic epitopes and cause autoantigenicity of self-antigens [70]. Dehydroascorbate (DHA) is imported through glucose transporter 1 (GLUT1) and is metabolized through the pentose phosphate pathway (PPP), thereby enhancing reduced glutathione (GSH) levels. DHA also increases surface expression of Fas receptor (FasR) [64]. Glutathione reductase and TRX reductase synthesize GSH and TRX-DT at the expense of NADPH. Formulation of the PPP and its efficiency to provide NADPH is dependent on the expression of G6PD (glucose 6-phosphate dehydrogenase) and transaldose (TAL) [34,43]. Dcm is controlled by intracellular GSH/NADH/NADPH levels, integrity of the permeability transition pore complex (PTPC), largely composed of the adenine nucleotie translocator (ANT) (inner membrane) and voltage-dependent anion channel (VDAC) (outer membrane), and translocation and dimerization of proand anti-apoptotic Bcl-2 family members in the intermembrane space [21]. TCR-activation-induced mitochondrial hyperpolarization is mediated through nitric oxide (NO) production by endothelial/neuronal NO synthase (e/nNOS), which, in turn, is regulated by ROIs and Ca2+; [23]. Abbreviations: GSSG, oxidized glutathione; IFN-γ, interferon γ; P, phosphate; PLCγ2, phospholipase Cγ2; TGF-β1, transforming growth factor β1; TRX-DT, thioredoxin.
Redox control of apoptosis signal processing
Programmed cell death (PCD) or apoptosis is a physiological mechanism for elimination of autoreactive lymphocytes during development. Signaling through the Fas or structurally related TNF family of cell-surface death receptors has emerged as a major pathway in the elimination of unwanted cells under physiological and disease conditions [18]. Fas and TNF receptors mediate cell death through cytoplasmic death domains (DDs) shared by both receptors [19]. They trigger sequential activation of caspases, resulting in cleavage of cellular substrates and the characteristic morphological and biochemical changes of apoptosis [20].
Disruption of the mitochondrial membrane potential (ΔΨm) has been proposed as the point of no return in apoptotic signaling, leading to caspase activation and disassembly of the cell [2]. Interestingly, MHP and ROI production precede disruption of ΔΨm, activation of caspases and PS externalization in Fas- [3], TNF-α- [6] and H2O2-induced apoptosis of Jurkat human leukemia T cells and normal human PBLs [4]. Elevation of ΔΨm is independent from activation of caspases and represents an early event in apoptosis [3]. Pretreatment with caspase inhibitors completely abrogated Fas-induced PS externalization, indicating that activation of caspase-3, caspase-8, and related cysteine proteases were absolutely required for cell death [3]. ROI levels were partially inhibited in Jurkat cells treated with caspase inhibitor, suggesting that caspase-3 activation, perhaps through damage of mitochondrial membrane integrity, contributes to ROI production and serves as a positive-feedback loop at later stages of the apoptotic process. Cleavage of cytosolic Bid by caspase-8 generates a p15 carboxy-terminal fragment that translocates to mitochondria. This might increase mitochondrial membrane permeability and lead to secondary elevation of ROI levels in the Fas and TNF pathway [21].
Mitochondrial checkpoints of cell death pathway selection
MHP appears to be the earliest change associated with several apoptosis pathways (Table 1). Elevation of ΔΨm is also triggered by activation of the CD3–CD28 complex [10] or stimulation with ConA [3], IL-10, IL-3, IFN-γ or TGF-β [11]. Therefore, MHP represents an early but reversible switch not exclusively associated with apoptosis. MHP is a probable cause of increased ROI production [22] and might be ultimately responsible for increased susceptibility to apoptosis following T-cell activation [10].
MHP in T cells is associated with a dramatic increase, more than sixfold, of NO production lasting 24 hours after CD3–CD28 co-stimulation. Molecular ordering of T-cell-activation-induced NO production revealed crucial roles for ROI production and cytoplasmic and mitochondrial Ca2+ influx [23]. CD3–CD28 co-stimulation-induced ROI production, similar to H2O2, enhances expression of nitric oxide synthase (NOS) isoforms eNOS and nNOS, which require elevated Ca2+ levels for enzymatic activity. These results suggest that T-cell-activation-induced ROI and Ca2+ signals contribute to NO production, with the latter representing a final and dominant step in MHP (Figure 1).
Proteins of the Bcl-2 family are localized to membranes of distinct organelles including mitochondria [24]. Both the pro-apoptotic (Bax, Bad) and anti-apoptotic (Bcl-2, Bcl-XL) members of the family can form ion-conducting channels in lipid membranes [21]. Bax can create a channel in the outer mitochondrial membrane, thus releasing cytochrome c and other caspase-activating moieties into the cytosol. Bcl-2 and Bcl-XL inhibit this process through dimerization with Bad or Bax. Bcl-2 expression appears to be unaltered in lupus PBLs [25].
The mitochondrion is the site of ATP synthesis through oxidative phosphorylation. The synthesis of ATP is driven by an electrochemical gradient across the inner mitochondrial membrane maintained by an electron transport chain and the membrane potential. Caspase activity requires ATP to the extent that depletion of ATP by inhibition of F0F1-ATPase with oligomycin [26] or exhaustion of intracellular ATP stores by prior apoptosis signals, Fas stimulation [26] or H2O2 pretreatment leads to necrosis [27]. Thus, intracellular ATP concentration is a key switch in the decision of the cell to die by apoptosis or necrosis [26].
MHP, increased ROI production, cytoplasmic alkalinization and ATP depletion in lupus T cells
Current evidence suggests that the regulation of PCD is impaired in both human and murine systemic lupus erythematosus (SLE), and could contribute to disease pathogenesis [28]. In lpr and gld mice, defects in PCD signaling through the Fas pathway appear to predispose to autoimmunity [29]. Whereas mutations of the Fas receptor (FasR) or Fas ligand (FasL) have been associated with a lupus-like autoimmune syndrome in mice with the lpr or gld genetic background [29], Fas-mediated signaling appears to be intact in human SLE [30]. Lupus T cells demonstrate defective activation-induced cell death (AICD) [31]. By contrast, increased spontaneous apoptosis of PBLs has also been observed in SLE [32]. Thus, paradoxically, SLE T cells exhibit both defective AICD and enhanced spontaneous apoptosis (Table 2).
Table 2.
Signaling abnormalities of T-cell death in patients with SLEa
| Signal | Effect | Refs |
|---|---|---|
| ΔΨm↑ | ROI ↑, ATP ↓ | [10] |
| ROI↑ | Spontaneous apoptosis ↑, IL-10 production↑ | [10,11] |
| GSH ↓ | ROI ↑, spontaneous apoptosis ↑ | [10,34] |
| Spontaneous apoptosis ↑ | Compartmentalized autoantigen release, disease activity↑ | [32,65] |
| H2O2 | Apoptosis ↓, necrosis ↑ | [10] |
| Activation of the CD3–CD28 complex | AICD ↓, necrosis ↑ | [11] |
| ATP ↓ | Predisposes for necrosis | [10,26] |
| Necrosis ↑ | Inflammation ↑ | [10,48] |
| AICD ↓ | Persistence of autoreactive cells | [11,31] |
| FasR ↑ | Spontaneous apoptosis ↑ | [30] |
| FasL ↑ | Spontaneous apoptosis ↑ | [30] |
| IL-10 ↑ | Selective induction of apoptosis in SLE | [11,30,66] |
| NO ↑ | Disease activity ↑ | [67] |
| IL-10 blockade | Spontaneous apoptosis ↓, ROI ↓ | [11,30] |
| IL-12 | Spontaneous apoptosis ↓, ROI ↓ | [11] |
Abbreviations: AICD, activation-induced cell death; FasL, Fas ligand; FasR, Fas receptor; GSH, reduced glutathione; IL, interleukin; NO, nitric oxide; ROI, reactive oxygen intermediate; SLE, systemic lupus erythematosus; ↑, increase; ↓, decrease.
Coordinate MHP and ATP depletion play key roles in abnormal T-cell death in lupus patients [10]. ΔΨm and ROI levels, as well as cytoplamic pH, are elevated in patients with SLE in comparison with healthy or rheumatoid arthritis controls [10,11]. Baseline MHP and ROI levels correlated with diminished levels of reduced glutathione (GSH), suggesting increased utilization of reducing equivalents in patients with SLE. It is presently unclear whether synthesis of GSH or its regeneration from its oxidized form (GSSG) is deficient in lupus patients. GSH is also required for IL-2-dependent T-cell proliferation [33], as well as CD2- and CD3-mediated T-cell activation [12]. Thus, low GSH content might also inhibit CD3-induced H2O2 production. Nevertheless, GSH deficiency predisposes for ROI-induced cell death [4,34]. Diminished H2O2-induced apoptosis of cells with low baseline GSH levels indicates a severe dysfunction of redox signaling in patients with SLE [10].
Increased ROI production might lead to skewed expression of redox-sensitive surface receptors and lymphokines in SLE (Table 2). As examples, ROIs regulate gene transcription and release of TNF-α and IL-10 [35], both of which are elevated in sera [36] and freshly isolated PBLs of SLE patients [37]. Expression of the TCRζ chain is sensitive to oxidative stress [14] and thus increased ROI levels could explain, at least in part, the low expression of the TCRz chain in lupus T cells [38]. Cell-surface expression of FasR [39] and FasL is also redox sensitive [40]. Increased ROI levels might be related to increased IL-10 production, release of FasL and overexpression of FasR in SLE [30]. Elevated NO production might also contribute to increased spontaneous apoptosis [41]. Increased ROI levels confer sensitivity to H2O2, NO, TNF-α or Fas-induced cell death [34]. Therefore, persistent MHP, causing increased ROI production (a trigger of apoptosis) and depletion of ATP (required for AICD), might be responsible for the paradox of increased spontaneous apoptosis and diminished AICD in SLE.
MHP and ATP depletion predispose lupus T cells to necrosis
In response to treatment with exogenous H2O2, a precursor of ROIs, lupus T cells failed to undergo apoptosis, and cell death preferentially occurred by necrosis. Endogenous H2O2 is generated by superoxide dismutase from the ROIs or OH− in mitochondria [42]. In turn, H2O2 is scavenged by catalase and glutathione peroxidase [43]. Whereas H2O2 is freely diffusible, it has no unpaired electrons and, by itself, is not a ROI [42]. Induction of apoptosis by H2O2 requires mitochondrial transformation into an ROI (e.g. OH−) through the Fenton reaction. [22,42]. As previously noted [4], H2O2 triggered a rapid increase of ΔΨm and ROI production that was followed by apoptosis of PBLs in healthy subjects. By contrast, H2O2 failed to elevate ΔΨm, ROI production and apoptosis, but rather elicited necrosis of lupus T cells. Both CD3–CD28-induced H2O2 production and H2O2-induced apoptosis require mitochondrial ROI production. Therefore, diminished CD3–CD28-induced H2O2 production and H2O2-induced apoptosis, together with deficient elevation of ΔΨm and ROI levels, reveal deviations of key biochemical checkpoints in mitochondria of patients with SLE.
ΔΨm is dependent upon the electron transport chain transferring electrons from NADH to molecular oxygen and proton transport mediated by the F0F1-ATPase complex [22]. During oxidative phosphorylation, the F0F1-ATPase converts ADP to ATP by utilizing the energy stored in the electrochemical gradient. Alternatively, using the energy of ATP hydrolysis, F0F1-ATPase can pump protons out of the mitochondrial matrix into the intermembrane space, causing ΔΨm elevation. Thus, MHP can occur in several ways. First, deficiency of cellular ADP could cause diminished utilization of the electrochemical gradient, ATP depletion and MHP. However, ADP levels were not diminished but slightly elevated in lupus PBLs [10]. This suggested that ATP depletion and ΔΨm hyperpolarization were not caused by a lack of ADP in patients with SLE. Second, MHP might occur through calcium-activated dephosphorylation of cytochrome c oxidase [44]. Phosphorylation of cytochrome c oxidase is mediated by protein kinase A (PKA); thus, deficiency of PKA could also contribute to MHP in SLE [45]. Third, inhibition of the enzymatic activity of F0F1-ATPase would decrease utilization of the electrochemical gradient and cause ΔΨm hyperpolarization, ATP depletion and ADP accumulation. A similar mechanism might also be operational in patients with SLE given that blocking of F0F1-ATPase by oligomycin led to ΔΨm hyperpolarization and elevated ROI production, prevented H2O2- or CD3–CD28-induced elevation of ΔΨm in normal PBLs, and sensitized to H2O2-induced necrosis [10]. With ΔΨm hyperpolarization and extrusion of H+ ions from the mitochondrial matrix, the cytochromes within the electron transport chain become more reduced, which favors generation of ROIs [22]. Thus, MHP is a likely cause of increased ROI production and might be ultimately responsible for increased spontaneous apoptosis in patients with SLE.
A 28–32% increase of the 2200 mV ΔΨm might have a tremendous impact on mitochondrial energy coupling and ATP synthesis [22]. Both T-cell activation and apoptosis require the energy provided by ATP [46]. Intracellular ATP concentration is a key switch in the decision of the cell to die by apoptosis or necrosis [26] and, therefore, depletion of ATP might be responsible for defective H2O2-induced apoptosis and a shift to necrosis in patients with SLE. Apoptosis is a physiological process that results in nuclear condensation and break-up of the cell into membrane-enclosed apoptotic bodies suitable for phagocytosis by macrophages, thus preventing inflammation. By contrast, necrosis is a pathological process that results in cellular swelling, followed by lysis and release of proteases, oxidizing molecules, and other proinflammatory and chemotactic factors, resulting in inflammation and tissue damage [46]. Indeed, lymphocyte necrosis occurs in the bone marrow [47] and lymph nodes of lupus patients, and could significantly contribute to the inflammatory process [48].
Increased necrosis might initiate a proinflammatory state, activation of DCs and IFN production in SLE
Swollen lymph nodes of patients with SLE harbor increased numbers of necrotic T cells and dendritic cells (DCs) [49]. Necrotic, but not apoptotic, cell death generates inflammatory signals that are necessary for the activation and maturation of DCs, which are the most potent antigen-presenting cells [50–52]. High-mobility group B1 (HMGB1) protein, an abundant DNA-binding protein, remains immobilized on chromatin of apoptotic bodies, but is released from necrotic cells [53]. HMGB1 stimulates human monocytes to release TNF-α, IL-1α, IL-1β, IL-1 receptor antagonist (IL-1RA), IL-6, IL-8, macrophage inflammatory protein (MIP)-1α, and MIP-1β, but not IL-10 or IL-12 [54], and induces arthritis [55]. Necrotic, but not apoptotic, cells also release heat shock proteins (HSPs) HSPgp96, HSP90, HSP70 and calreticulin. HSPs stimulate macrophages to secrete cytokines, and induce expression of antigen-presenting and co-stimulatory molecules on the DC [51]. Mature DCs express high levels of the DC-restricted markers CD83 and lysosome-associated membrane glycoprotein (DC-LAMP), and the co-stimulatory molecules CD40 and CD86 [52], which might contribute to the altered intercellular signaling in SLE (Figure 2). CD14+ monocytes isolated from the blood of lupus patients, but not those from healthy individuals, act as DCs [56]. Their activation is driven by circulating IFN-α, which might be generated by one of the DC subsets [e.g. plasmacytoid dendritic cells (PDCs)] that infiltrate lupus skin lesions. Tissue lesions [57,58] and blood of patients with SLE harbor activated PDCs that might be responsible for increased production of IFN-α in SLE[56,59].
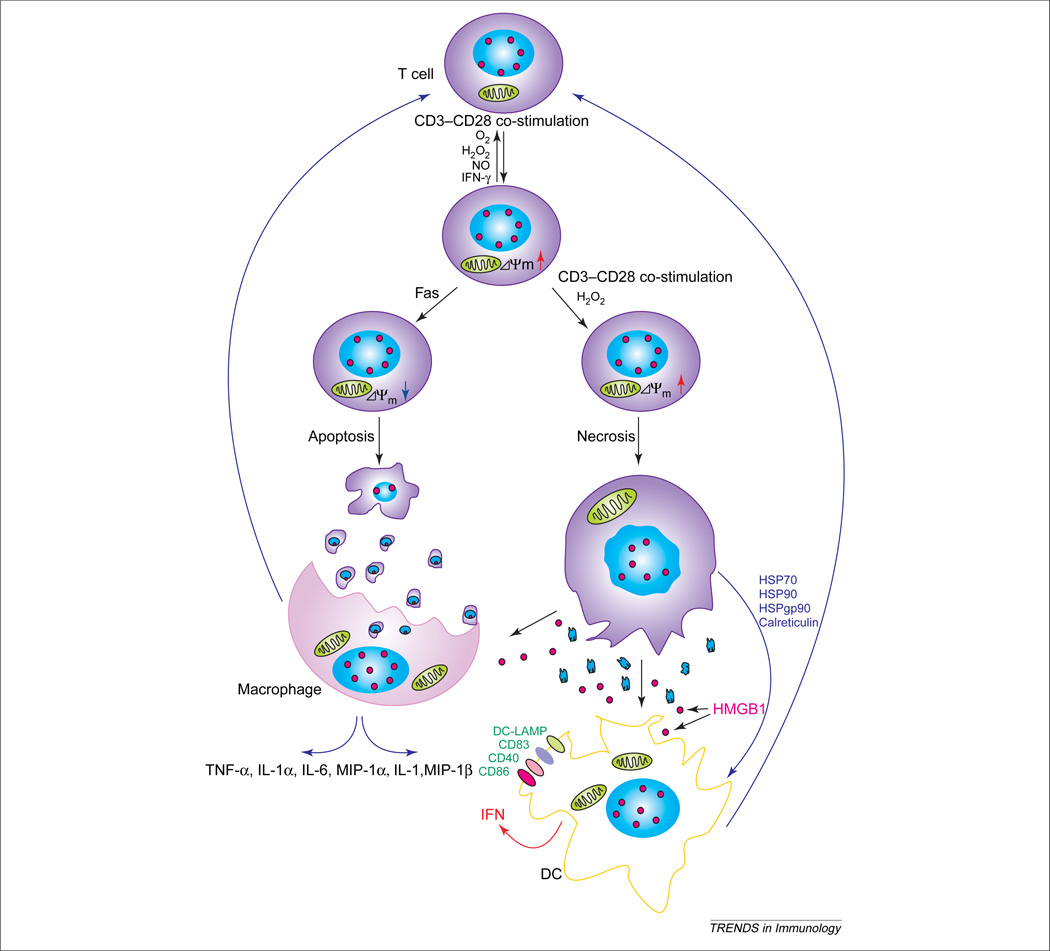
Figure 2.
Impact of ΔΨm on cell death pathway selection and activation of T cells by macrophages and dendritic cells (DCs). CD3–CD28 co-stimulation, reactive oxygen intermediate (ROI), H2O2, nitric oxide (NO) and cytokines, such as interferon-γ (IFN-γ), might elicit reversible elevation of ΔΨm [i.e. mitochondrial hyperpolarization (MHP)]. Transient MHP is an early event in Fas-mediated apoptosis, resulting in cellular shrinkage and formation of membrane-enclosed apoptotic bodies that are phagocytosed by macrophages. Repeated T-cell receptor (TCR) activation or exposure to ROIs induces necrosis instead of MHP, which, in turn, leads to cellular swelling, membrane breakdown and release of proinflammatory cell lysate. While high-mobility group B1 (HMGB1) protein remains immobilized on chromatin of apoptotic bodies, it is released from necrotic cells. HMGB1, heat-shock protein 70 (HSP70), HSP90, HSPgp95 and calreticulin induce differentiation of DCs and activate macrophages. DCs produce IFN-γ, whereas macrophages produce a series of proinflammatory cytokines. Both cell types increase expression of co-stimulation molecules, thus contributing to T-cell activation. These positive-feedback loops might play key roles in aberrant T-cell activation in patients with systemic lupus erythematosus (SLE). Abbreviations: DC-LAMP, lysosome-associated membrane glycoprotein; MIP-1γ, macrophage inflammatory protein 1γ; TNF-γ, tumor necrosis factor γ.
Outstanding questions and future directions
MHP represents an early but reversible checkpoint associated with activation and apoptosis of human T cells. Although Ca2+-, ROI- and NADPH-dependent production of NO appears to be a dominant factor in T-cell-activation-induced MHP, the relative impact and hierarchy of the metabolic and redox signaling pathways involved require further studies. The chemical composition of ROIs (i.e. OH−, , ONOO− and ONOOH), and their compartmentalization during T-cell activation and cell death, are unexplored. Selective targeting of ROIs might prove valuable in regulating T helper (Th)-cell differentiation and cytokine production, activation of cytotoxic T cells, and selection of the cell death pathway. Although MHP was not affected, IL-10 antibody or IL-12 was found to normalize ROI production and intracellular alkalinization in lupus PBLs [11]. Therefore, IL-10 antagonists might partially correct the redox signaling dysfunction in lupus. Bz-423, an experimental drug that binds to the benzodiazepine receptor in mitochondria, was found to reduce ΔΨm, induce selective death of autoreactive lymphocytes, and improve clinical outcome of lupus in two different murine models [60,61]. Precise delineation of the mechanism of MHP and ATP depletion could identify novel targets for pharmacological intervention in patients with SLE.
Acknowledgements
This work was supported in part by grants DK 49221 and AI 48079 from the National Institutes of Health and the Central New York Community Foundation. We thank P. Phillips for continued encouragement and support.
References
- 1.Perl A, et al. Metabolic switches of T-cell activation and apoptosis. Antioxid. Redox Signal. 2002;4:427–443. doi: 10.1089/15230860260196227. [DOI] [PubMed] [Google Scholar]
- 2.Susin SA, et al. The central executioner of apoptosis: multiple connections between protease activation and mitochondria in Fas/Apo-1/CD95- and ceramide-induced apoptosis. J. Exp. Med. 1997;186:25–37. doi: 10.1084/jem.186.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Banki K, et al. Elevation of mitochondrial transmembrane potential and reactive oxygen intermediate levels are early events and occur independently from activation of caspases in Fas signaling. J. Immunol. 1999;162:1466–1479. [PMC free article] [PubMed] [Google Scholar]
- 4.Puskas F, et al. Stimulation of the pentose phosphate pathway and glutathione levels by dehydroascorbate, the oxidized form of vitamin C. FASEB J. 2000;14:1352–1361. doi: 10.1096/fj.14.10.1352. [DOI] [PubMed] [Google Scholar]
- 5.Li P-F, et al. p53 regulates mitochondrial membrane potential through reactive oxygen species and induces cytochrome c-independent apoptosis blocked by bcl-2. EMBO J. 1999;18:6027–6036. doi: 10.1093/emboj/18.21.6027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gottlieb E, et al. Bcl-XL prevents the initial decrease in mitochondrial membrane potential and subsequent reactive oxygen species production during tumor necrosis factor alpha-induced apoptosis. Mol. Cell. Biol. 2000;20:5680–5689. doi: 10.1128/mcb.20.15.5680-5689.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Scarlett JL, et al. Changes in mitochondrial membrane potential during staurosporin-induced apoptosis in Jurkat cells. FEBS Lett. 2000;475:267–272. doi: 10.1016/s0014-5793(00)01681-1. [DOI] [PubMed] [Google Scholar]
- 8.Sanchez-Alcazar JA, et al. Increased mitochondrial cytochrome c levels and mitochondrial hyperpolarization precede camptothecin- induced apoptosis in Jurkat cells. Cell Death Differ. 2000;7:1090–1100. doi: 10.1038/sj.cdd.4400740. [DOI] [PubMed] [Google Scholar]
- 9.Almeida A, et al. Different responses of astrocytes and neurons to nitric oxide: the role of glycolytically generated ATP in astrocyte protection. Proc. Natl. Acad. Sci. U. S. A. 2001;98:15294–15299. doi: 10.1073/pnas.261560998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gergely PJ, et al. Mitochondrial hyperpolarization and ATP depletion in patients with systemic lupus erythematosus. Arthritis Rheum. 2002;46:175–190. doi: 10.1002/1529-0131(200201)46:1<175::AID-ART10015>3.0.CO;2-H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gergely PJ, et al. Persistent mitochondrial hyperpolarization, increased reactive oxygen intermediate production, and cytoplasmic alkalinization characterize altered IL-10 signaling in patients with systemic lupus erythematosus. J. Immunol. 2002;169:1092–1101. doi: 10.4049/jimmunol.169.2.1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Suthanthiran M, et al. Glutathione regulates activation-dependent DNA synthesis in highly purified normal human T lymphocytes stimulated via the CD2 and CD3 antigens. Proc. Natl. Acad. Sci. U. S. A. 1990;87:3343–3347. doi: 10.1073/pnas.87.9.3343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Koretzky GA, Boerth NJ. The role of adapter proteins in T cell activation. Cell. Mol. Life Sci. 1999;56:1048–1060. doi: 10.1007/s000180050492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Otsuji M, et al. Oxidative stress by tumor-derived macrophages suppresses the expression of CD3 zeta chain of T-cell receptor complex and antigen-specific T-cell responses. Proc. Natl. Acad. Sci. U. S. A. 1996;93:13119–13124. doi: 10.1073/pnas.93.23.13119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gringhuis SI, et al. Displacement of linker for activation of T cells from the plasma membrane due to redox balance alterations results in hyporesponsiveness of synovial fluid T lymphocytes in rheumatoid arthritis. J. Immunol. 2000;164:2170–2179. doi: 10.4049/jimmunol.164.4.2170. [DOI] [PubMed] [Google Scholar]
- 16.Beiqing L, et al. Sublethal levels of oxidative stress stimulate transcriptional activation of c-jun and suppress IL-2 promoter activation in Jurkat T cells. J. Immunol. 1996;157:160–169. [PubMed] [Google Scholar]
- 17.Furuke K, et al. Fas ligand induction in human NK cells is regulated by redox through a calcineurin-nuclear factors of activated T cell-dependent pathway. J. Immunol. 1999;162:1988–1993. [PubMed] [Google Scholar]
- 18.Nagata S. Apoptosis by death factor. Cell. 1997;88:355–365. doi: 10.1016/s0092-8674(00)81874-7. [DOI] [PubMed] [Google Scholar]
- 19.Itoh N, Nagata S. A novel protein domain required for apoptosis: mutational analysis of human Fas antigen. J. Biol. Chem. 1993;268:10932–10937. [PubMed] [Google Scholar]
- 20.Martin SJ, Green DR. Protease activation during apoptosis: death by a thousand cuts. Cell. 1995;82:349–352. doi: 10.1016/0092-8674(95)90422-0. [DOI] [PubMed] [Google Scholar]
- 21.Gross A, et al. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 1999;13:1899–1911. doi: 10.1101/gad.13.15.1899. [DOI] [PubMed] [Google Scholar]
- 22.Skulachev VP. Mitochondrial physiology and pathology; concepts of programmed death of organelles, cells and organisms. Mol. Aspects Med. 1999;20:139–140. doi: 10.1016/s0098-2997(99)00008-4. [DOI] [PubMed] [Google Scholar]
- 23.Nagy G, et al. T cell activation-induced mitochondrial hyperpolarization is mediated by Ca2+- and redox-dependent production of nitric oxide. J. Immunol. 2003;171:5188–5197. doi: 10.4049/jimmunol.171.10.5188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Green DR, Reed JC. Mitochondria and apoptosis. Science. 1998;281:1309–1312. doi: 10.1126/science.281.5381.1309. [DOI] [PubMed] [Google Scholar]
- 25.Rose LM, et al. Bcl-2 expression is unaltered in unfractionated peripheral blood mononuclear cells in patients with systemic lupus erythematosus. Br. J. Rheumatol. 1995;34:316–320. doi: 10.1093/rheumatology/34.4.316. [DOI] [PubMed] [Google Scholar]
- 26.Leist M, et al. Intracellular adenosine triphosphate (ATP) concentration: a switch in the decision between apoptosis and necrosis. J. Exp. Med. 1997;185:1481–1486. doi: 10.1084/jem.185.8.1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee Y, Shacter E. Oxidative stress inhibits apoptosis in human lymphoma cells. J. Biol. Chem. 1999;274:19792–19798. doi: 10.1074/jbc.274.28.19792. [DOI] [PubMed] [Google Scholar]
- 28.Perl A, Banki K. Molecular mimicry, altered apoptosis, and immunomodulation as mechanisms of viral pathogenesis in systemic lupus erythematosus. In: Kammer GM, Tsokos GC, editors. Lupus: Molecular and Cellular Pathogenesis. 1st edn. Humana Press; 1999. pp. 43–64. [Google Scholar]
- 29.Nagata S, Golstein P. The Fas death factor. Science. 1995;267:1449–1456. doi: 10.1126/science.7533326. [DOI] [PubMed] [Google Scholar]
- 30.Georgescu L, et al. Interleukin-10 promotes activation-induced cell death of SLE lymphocytes mediated by Fas ligand. J. Clin. Invest. 1997;100:2622–2633. doi: 10.1172/JCI119806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kovacs B, et al. Defective CD3-mediated cell death in activated T cells from patients with systemic lupus erythematosus: role of decreased intracellular TNF-alpha. Clin. Immunol. Immunopathol. 1996;81:293–302. doi: 10.1006/clin.1996.0192. [DOI] [PubMed] [Google Scholar]
- 32.Emlen W, et al. Accelerated in vitro apoptosis of lymphocytes from patients with systemic lupus erythematosus. J. Immunol. 1994;152:3685–3692. [PubMed] [Google Scholar]
- 33.Hamilos DL, Wedner HJ. The role of glutathione in lymphocyte activation. I. Comparison of inhibitory effects of buthionine sulfoximine and 2-cyclohexene-1-one by nuclear size transformation. J. Immunol. 1985;135:2740–2747. [PubMed] [Google Scholar]
- 34.Banki K, et al. Glutathione levels and sensitivity to apoptosis are regulated by changes in transaldolase expression. J. Biol. Chem. 1996;271:32994–33001. doi: 10.1074/jbc.271.51.32994. [DOI] [PubMed] [Google Scholar]
- 35.Le Moine O, et al. Role of reactive oxygen intermediates in interleukin 10 release after cold liver ischemia and reperfusion in mice. Gastroenterology. 1997;113:1701–1706. doi: 10.1053/gast.1997.v113.pm9352875. [DOI] [PubMed] [Google Scholar]
- 36.al Janadi M, et al. Cytokine profile in systemic lupus erythematosus, rheumatoid arthritis, and other rheumatic diseases. J. Clin. Immunol. 1993;13:58–67. doi: 10.1007/BF00920636. [DOI] [PubMed] [Google Scholar]
- 37.Swaak AJ, et al. Cytokine production (IL-6 and TNF alpha) in whole blood cell cultures of patients with systemic lupus erythematosus. Scand. J. Rheumatol. 1996;25:233–238. doi: 10.3109/03009749609069992. [DOI] [PubMed] [Google Scholar]
- 38.Liossis SN, et al. Altered pattern of TCR/CD3-mediated protein-tyrosyl phosphorylation in T cells from patients with systemic lupus erythematosus. Deficient expression of the T cell receptor zeta chain. J. Clin. Invest. 1998;101:1448–1457. doi: 10.1172/JCI1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li D, et al. Ox-LDL induces apoptosis in human coronary artery endothelial cells: role of PKC PTK, bcl-2, and Fas. Am. J. Physiol. 1998;275:H568–H576. doi: 10.1152/ajpheart.1998.275.2.H568. [DOI] [PubMed] [Google Scholar]
- 40.Kasibhatla S, et al. Regulation of Fas ligand expression during activation-induced cell death in T lymphocytes. J. Biol. Chem. 1999;274:987–992. doi: 10.1074/jbc.274.2.987. [DOI] [PubMed] [Google Scholar]
- 41.Cooper GS, et al. Hormonal, environmental, and infectious risk factors for developing systemic lupus erythematosus. Arthritis Rheum. 1998;41:1714–1724. doi: 10.1002/1529-0131(199810)41:10<1714::AID-ART3>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 42.Halliwell B, Gutteridge JM. Role of free radicals and catalytic metal ions in human disease: an overview. Methods Enzymol. 1990;186:1–85. doi: 10.1016/0076-6879(90)86093-b. [DOI] [PubMed] [Google Scholar]
- 43.Mayes PA. The pentose phosphate pathway and other pathways of hexose metabolism. In: Murray RK, editor. Harper’s Biochemistry. 23rd edn. Appleton & Lange: 1993. pp. 201–211. [Google Scholar]
- 44.Kadenbach B. Intrinsic and extrinsic uncoupling of oxidative phosphorylation. Biochim. Biophys. Acta. 2003;1604:77–94. doi: 10.1016/s0005-2728(03)00027-6. [DOI] [PubMed] [Google Scholar]
- 45.Kammer GM, et al. Abnormal T cell signal transduction in systemic lupus erythematosus. Arthritis Rheum. 2002;46:1139–1154. doi: 10.1002/art.10192. [DOI] [PubMed] [Google Scholar]
- 46.Fiers W, et al. More than one way to die: apoptosis, necrosis and reactive oxygen damage. Oncogene. 1999;18:7719–7730. doi: 10.1038/sj.onc.1203249. [DOI] [PubMed] [Google Scholar]
- 47.Lorand-Metze I, et al. Morphology of bone marrow in systemic lupus erythematosus. Pathologe. 1994;15:292–296. doi: 10.1007/s002920050057. [DOI] [PubMed] [Google Scholar]
- 48.Eisner MD, et al. Necrotizing lymphadenitis associated with systemic lupus erythematosus. Semin. Arthritis Rheum. 1996;26:477–482. doi: 10.1016/s0049-0172(96)80028-x. [DOI] [PubMed] [Google Scholar]
- 49.Kojima M, et al. Reactive follicular hyperplasia in the lymph node lesions from systemic lupus erythematosus patients: a clinico-pathological and immunohistological study of 21 cases. Pathol. Int. 2000;50:304–312. doi: 10.1046/j.1440-1827.2000.01052.x. [DOI] [PubMed] [Google Scholar]
- 50.Gallucci S, et al. Natural adjuvants: endogenous activators of dendritic cells. Nat. Med. 1999;5:1249–1255. doi: 10.1038/15200. [DOI] [PubMed] [Google Scholar]
- 51.Basu S, et al. Necrotic but not apoptotic cell death releases heat shock proteins, which deliver a partial maturation signal to dendritic cells and activate the NF-κB pathway. Int. Immunol. 2000;12:1539–1546. doi: 10.1093/intimm/12.11.1539. [DOI] [PubMed] [Google Scholar]
- 52.Sauter B, et al. Consequences of cell death: exposure to necrotic tumor cells, but not primary tissue cells or apoptotic cells, induces the maturation of immunostimulatory dendritic cells. J. Exp. Med. 2000;191:423–434. doi: 10.1084/jem.191.3.423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bustin M. At the crossroads of necrosis and apoptosis: signaling to multiple cellular targets by HMGB1. Sci. STKE. 2002:PE39. doi: 10.1126/stke.2002.151.pe39. 2002. [DOI] [PubMed] [Google Scholar]
- 54.Andersson U, et al. High mobility group 1 protein (HMG-1) stimulates proinflammatory cytokine synthesis in human monocytes. J. Exp. Med. 2000;192:565–570. doi: 10.1084/jem.192.4.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pullerits R, et al. High mobility group box chromosomal protein 1, a DNA binding cytokine, induces arthritis. Arthritis Rheum. 2003;48:1693–1700. doi: 10.1002/art.11028. [DOI] [PubMed] [Google Scholar]
- 56.Pascual V, et al. The central role of dendritic cells and interferon-alpha in SLE. Curr. Opin. Rheumatol. 2003;15:548–556. doi: 10.1097/00002281-200309000-00005. [DOI] [PubMed] [Google Scholar]
- 57.Mori M, et al. Dendritic cells in cutaneous lupus erythematosus: a clue to the pathogenesis of lesions. Histopathology. 1994;24:311–321. doi: 10.1111/j.1365-2559.1994.tb00531.x. [DOI] [PubMed] [Google Scholar]
- 58.Farkas L, et al. Plasmacytoid dendritic cells (natural interferon- alpha/beta-producing cells) accumulate in cutaneous lupus erythematosus lesions. Am. J. Pathol. 2001;159:237–243. doi: 10.1016/s0002-9440(10)61689-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ronnblom L, Alm GV. The natural interferon-alpha producing cells in systemic lupus erythematosus. Hum. Immunol. 2002;63:1181–1193. doi: 10.1016/s0198-8859(02)00757-7. [DOI] [PubMed] [Google Scholar]
- 60.Blatt NB, et al. Benzodiazepine-induced superoxide signals B cell apoptosis: mechanistic insight and potential therapeutic utility. J. Clin. Invest. 2002;110:1123–1132. doi: 10.1172/JCI16029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bednarski JJ, et al. Attenuation of autoimmune disease in Fas-deficient mice by treatment with a cytotoxic benzodiazepine. Arthritis Rheum. 2003;48:757–766. doi: 10.1002/art.10968. [DOI] [PubMed] [Google Scholar]
- 62.Banki K, et al. Molecular ordering in HIV-induced apoptosis: oxidative stress, activation of caspases, and cell survival are regulated by transaldolase. J. Biol. Chem. 1998;273:11944–11953. doi: 10.1074/jbc.273.19.11944. [DOI] [PubMed] [Google Scholar]
- 63.Matarrese P, et al. Mitochondrial membrane hyperpolarization hijacks activated T lymphocytes toward the apoptotic-prone phenotype: homeostatic mechanisms of HIV protease inhibitors. J. Immunol. 2003;170:6006–6015. doi: 10.4049/jimmunol.170.12.6006. [DOI] [PubMed] [Google Scholar]
- 64.Puskas F, et al. Differential regulation of hydrogen peroxide and Fas-dependent apoptosis pathways by dehydroascorbate, the oxidized form of vitamin C. Antioxid. Redox Signal. 2002;4:357–369. doi: 10.1089/15230860260196164. [DOI] [PubMed] [Google Scholar]
- 65.Casciola-Rosen LA, et al. Autoantigens targeted in systemic lupus erythematosus are clustered in two populations of surface structures on apoptotic keratinocytes. J. Exp. Med. 1994;179:1317–1330. doi: 10.1084/jem.179.4.1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Llorente L, et al. Role of interleukin 10 in the B lymphocyte hyperactivity and autoantibody production of human systemic lupus erythematosus. J. Exp. Med. 1995;181:839–844. doi: 10.1084/jem.181.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Oates JC, et al. Prospective measure of serum 3-nitrotyrosine levels in systemic lupus erythematosus: correlation with disease activity. Proc. Assoc. Am. Physicians. 1999;111:611–621. doi: 10.1046/j.1525-1381.1999.99110.x. [DOI] [PubMed] [Google Scholar]
- 68.Constantini P, et al. Modulation of the mitochondrial permeability transition pore by pyridine nucleotides and dithiol oxidation at two separate sites. J. Biol. Chem. 1996;271:6746–6751. doi: 10.1074/jbc.271.12.6746. [DOI] [PubMed] [Google Scholar]
- 69.Brown GC. Nitric oxide and mitochondrial respiration. Biochim. Biophys. Acta. 1999;1411:351–369. doi: 10.1016/s0005-2728(99)00025-0. [DOI] [PubMed] [Google Scholar]
- 70.Casciola-Rosen LA, et al. Specific cleavage of the 70 kDa protein component of the U1 small nuclear ribonucleoprotein is a characteristic biochemical feature of apoptotic cell death. J. Biol. Chem. 1994;269:30757–30760. [PubMed] [Google Scholar]