Significance
Associations of plants and herbivores are regarded as the result of coevolution, which has produced an astonishing diversity of plant defenses and corresponding insect counteradaptations. We focus on the leaf beetle Phyllotreta striolata, which is adapted to the glucosinolate-myrosinase system present in its cruciferous host plants. We show that P. striolata adults not only selectively sequester intact glucosinolates from their host plants but also express their own myrosinase, a member of the β-glucosidase family capable of hydrolyzing glucosinolates to form toxic degradation products. Our results reveal the convergent evolution of a glucosinolate-myrosinase system in P. striolata that enables this herbivore to use glucosinolate hydrolysis products for its own purposes.
Keywords: convergent evolution, host plant specialization
Abstract
The ability of a specialized herbivore to overcome the chemical defense of a particular plant taxon not only makes it accessible as a food source but may also provide metabolites to be exploited for communication or chemical defense. Phyllotreta flea beetles are adapted to crucifer plants (Brassicales) that are defended by the glucosinolate-myrosinase system, the so-called “mustard-oil bomb.” Tissue damage caused by insect feeding brings glucosinolates into contact with the plant enzyme myrosinase, which hydrolyzes them to form toxic compounds, such as isothiocyanates. However, we previously observed that Phyllotreta striolata beetles themselves produce volatile glucosinolate hydrolysis products. Here, we show that P. striolata adults selectively accumulate glucosinolates from their food plants to up to 1.75% of their body weight and express their own myrosinase. By combining proteomics and transcriptomics, a gene responsible for myrosinase activity in P. striolata was identified. The major substrates of the heterologously expressed myrosinase were aliphatic glucosinolates, which were hydrolyzed with at least fourfold higher efficiency than aromatic and indolic glucosinolates, and β-O-glucosides. The identified beetle myrosinase belongs to the glycoside hydrolase family 1 and has up to 76% sequence similarity to other β-glucosidases. Phylogenetic analyses suggest species-specific diversification of this gene family in insects and an independent evolution of the beetle myrosinase from other insect β-glucosidases.
Phytophagous insects are confronted with an arsenal of constitutive and inducible chemical plant defenses (1). To avoid deleterious effects from plant toxins, herbivores require appropriate biochemical adaptations allowing them to deal with these metabolites, for example, by developing insensitive target sites or by enzymatic detoxification. Many specialized insects are able to sequester plant defense compounds and exploit them for their own protection against natural enemies and/or as semiochemicals in interspecific communication (2, 3).
The glucosinolate-myrosinase system characteristic of plants in the order Brassicales is one of the best-studied activated plant defense systems (4–6), and several different strategies for avoiding glucosinolate toxicity have been described in specialized insect herbivores of the orders Lepidoptera, Hemiptera, and Hymenoptera (7). Glucosinolates are a structurally diverse group of β-thioglucoside-N-hydroxysulfates that are classified according to their precursor amino acid as aliphatic, aromatic, or indolic glucosinolates (6, 8). Plant myrosinases, ascorbate-dependent β-thioglucosidases (EC 3.2.1.147) belonging to glycoside hydrolase family 1 (GH1), are stored separately from the glucosinolates and only come into contact with their substrate when the spatial compartmentalization is destroyed, for example, by herbivore feeding (6). The profile of hydrolysis products then depends on glucosinolate structure, cellular conditions, and the presence of so-called “specifier proteins” leading to the formation of isothiocyanates (ITCs), thiocyanates, nitriles, and epithionitriles (6). Due to their chemical properties, ITCs are the most reactive hydrolysis products of glucosinolates (6, 9). Although several generalist lepidopterans detoxify ITCs via conjugation with glutathione (10), the specialists investigated so far circumvent ITC formation (7, 11, 12). Several adapted herbivores sequester intact host plant glucosinolates (13–16), and two aphid species, Brevicoryne brassicae and Lipaphis erysimi, not only sequester glucosinolates but also convert these glucosinolates to toxic ITCs by using their own myrosinase, encoded in the aphid genome (17–20).
Flea beetles of the genus Phyllotreta (Coleoptera: Chrysomelidae) are closely associated with glucosinolate-containing plants in the Brassicaceae, Resedaceae, Tropaeolaceae, and Capparaceae on which they feed throughout their life cycle (21–23). In fact, the only confirmed exception is Phyllotreta vittula, which prefers Poaceae but also accepts crucifers as food plants (24). As specialized herbivores, Phyllotreta flea beetles are expected to be adapted to the glucosinolate-myrosinase system, but the mechanism for such tolerance is not known for any crucifer-feeding Coleoptera to date. Intact glucosinolates stimulate feeding of Phyllotreta spp., and thus play a role in host acceptance (22), but genetically increased myrosinase activity in Brassica rapa plants enhanced resistance to Phyllotreta cruciferae (25). However, ITCs are suggested to play a role in interspecific communication of Phyllotreta spp. because they were shown to enhance or synergize the response to the male-produced aggregation pheromone in several species (26–29).
In previous studies with Phyllotreta striolata (Coleoptera: Chrysomelidae), we detected several unexpected ITCs in the volatile emission of adults feeding on different Brassica host plants (30). For example, we identified alkenyl ITCs to be present in the headspace of adult P. striolata feeding on Brassica oleracea var. italica, although this plant was shown to accumulate methylsulfinylalkyl and indolic glucosinolates but no alkenyl glucosinolates (Fig. S1A). We thus hypothesized that the beetles themselves are able to emit these unexpected ITCs. Because the putative emission of volatile ITCs from feeding adult P. striolata was independent of the glucosinolate profile of the current food plant (30) (Fig. S1B), we investigated how these metabolites could be produced in beetles.
Here, we demonstrate that P. striolata adults selectively sequester glucosinolates during feeding on host plants, and probably are able to retain these previously accumulated glucosinolates when feeding on a plant with a distinct glucosinolate profile. Hence, by using their own endogenous myrosinase, the adults may be able to release bioactive hydrolysis products independent of the current host plants’ glucosinolate profile. A corresponding gene was cloned and heterologously expressed, and the enzyme was analyzed for its activity toward different glucosinolate structures. We further used phylogenetic analyses to trace the evolution of this myrosinase in P. striolata and to establish that its origin is independent of the only other known insect myrosinase, that from aphids.
Results
Host Plant Glucosinolates Are Selectively Sequestered by P. striolata Adults.
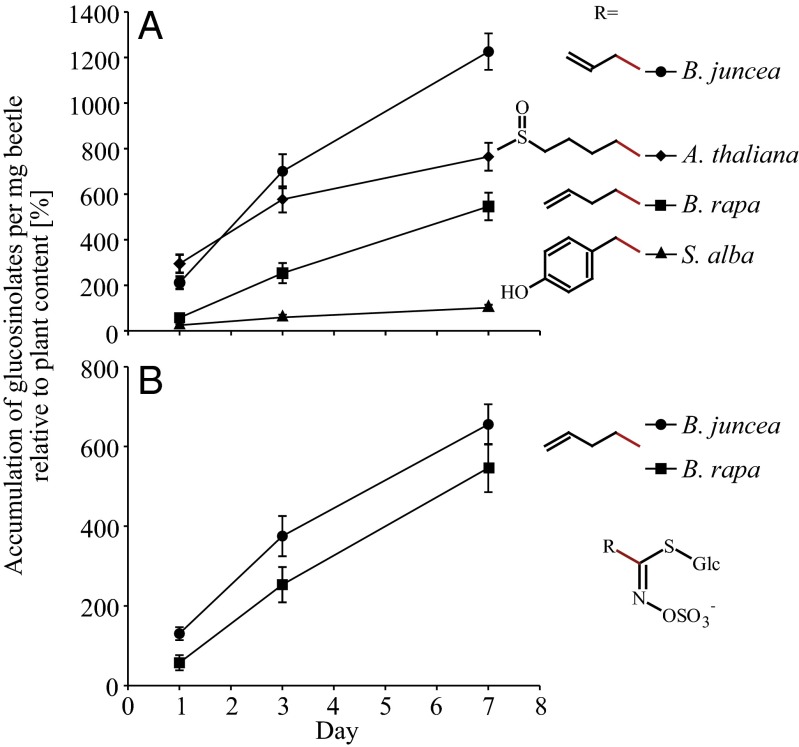
Analyses of a field-collected P. striolata population that had been maintained for several weeks on the Arabidopsis thaliana double-mutant myb28myb29, which only produces indolic glucosinolates, revealed high amounts of aliphatic and indolic glucosinolates (Table S1). The aliphatic glucosinolates probably accumulated from host plants before collection of P. striolata adults in the field. This glucosinolate profile was then compared with the profiles of beetles that had been transferred to the new host plants B. rapa, Brassica juncea, Sinapis alba, and A. thaliana Columbia (Col-0). Over 7 d, most of the glucosinolates of the new host plants accumulated in beetles, whereas concentrations of previously detected compounds had decreased (Fig. S2 and Dataset S1). To compare sequestration patterns, the beetle glucosinolate content (in picomoles per milligram per beetle) was expressed relative to the corresponding feeding-induced glucosinolate concentration in the plant (picomoles per milligram per plant fresh weight) (Table S2). Focusing on the major compounds, which account for 40–95% of the total glucosinolate content in each respective plant, we found that P. striolata adults could take up all of the major glucosinolates but that there were significant differences regarding the amount and rate of accumulation of individual compounds in the beetles [Fig. 1A; n = 7–12; analysis of covariance (ANCOVA), day: F = 231.81, P < 0.001; compound: F = 126.47, P < 0.001; day × compound: F = 12.06, P < 0.001]. It also depended on the plant species as to how strongly a certain glucosinolate accumulated (Fig. 1B; n = 11–12; ANCOVA, day: F = 174.21, P < 0.001; plant species: F = 14.15, P < 0.001; day × plant species: F = 0.732, P = 0.395).
Fig. 1.
P. striolata adults sequester a variety of glucosinolates from their host plants. Values are given as the amount per insect expressed relative to plant glucosinolate content (set to 100%) after 1, 3, and 7 d of feeding on B. juncea, B. rapa, A. thaliana, and S. alba. All values are given as mean ± SEM. (A) Sequestration of the major glucosinolates from various host plants: allyl glucosinolate (B. juncea, n = 12), 4MSOB glucosinolate (A. thaliana, n = 11–12), 3-butenyl glucosinolate (B. rapa, n = 11–12), and 4-hydroxybenzyl glucosinolate (S. alba, n = 7–12). (B) Accumulation of 3-butenyl glucosinolate from B. juncea and B. rapa (n = 11–12) in P. striolata.
The glucosinolates of different classes accumulated at divergent rates. Nearly all aliphatic glucosinolates accumulated in high amounts from B. juncea, B. rapa, and A. thaliana, although there were differences among individual compounds depending on structure and plant source (Fig. S2). In contrast, aromatic and indolic glucosinolates were detected in low amounts or not at all. For example, benzyl glucosinolate, a minor aromatic glucosinolate found constitutively in S. alba and induced after feeding in B. juncea and B. rapa, was detected in traces below the quantification limit and only in beetles that had been feeding on B. rapa. Indolic glucosinolates were present and induced in all feeding-damaged host plant species, but only 1-methoxy-indol-3-ylmethyl glucosinolate levels increased significantly and only in adults feeding on B. rapa and A. thaliana.
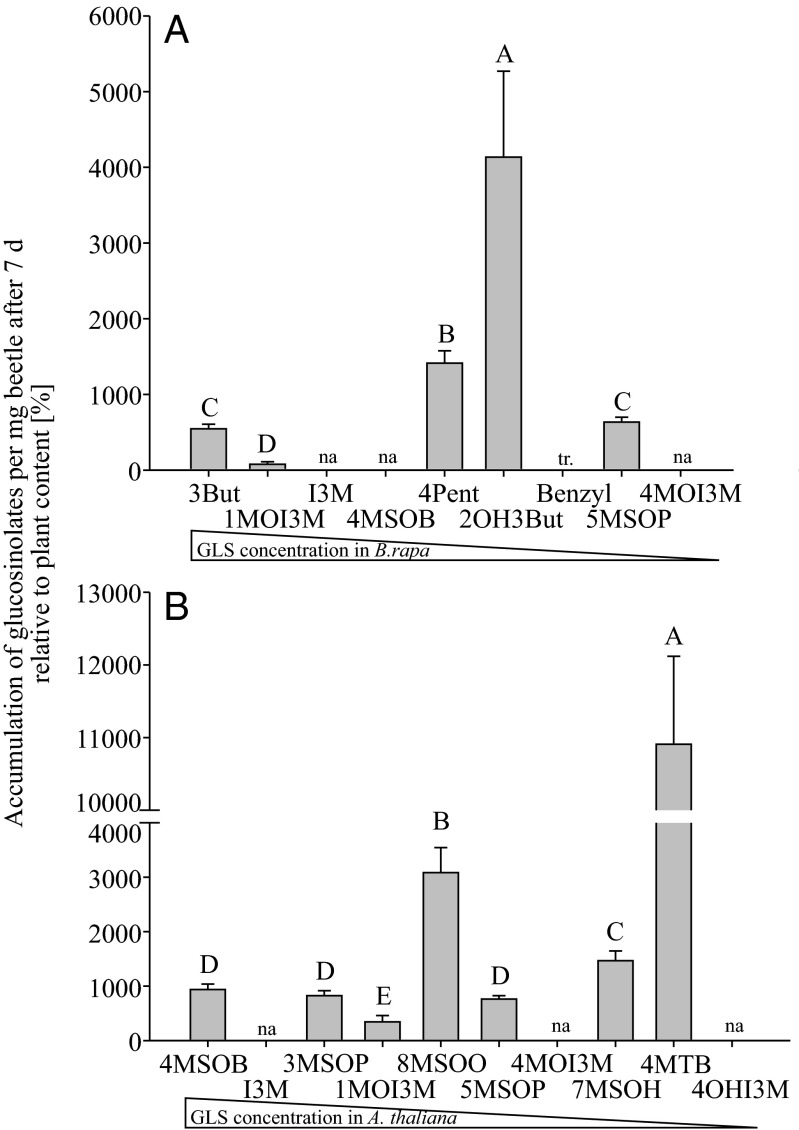
These differences among glucosinolate classes resulted in widely divergent accumulation patterns relative to plant content after 7 d (Fig. 2). For example, 2-hydroxy-3-butenyl glucosinolate was detected at a more than 40-fold higher concentration in beetles than in B. rapa, but 1-methoxy-indol-3-ylmethyl glucosinolate concentrations in beetles were even lower than the plant concentration (Fig. 2A). The latter compound was also sequestered only 3.5-fold in beetles feeding on A. thaliana. However, the 4-methylthiobutyl (4MTB) glucosinolate concentration in the same insects was about 100-fold higher than in the plant (Fig. 2B).
Fig. 2.
Glucosinolates are selectively sequestered in P. striolata. Depicted are glucosinolate sequestration patterns in P. striolata relative to the plant glucosinolate content (100%) after feeding on either B. rapa (A) or A. thaliana (B) for 7 d (n = 11–12, +SEM). The order of glucosinolates corresponds to the concentration in the plant from the most abundant glucosinolate to the least abundant glucosinolate. Several compounds from both host plants were not accumulated (na); benzyl glucosinolate was detected in traces (tr) in beetles feeding on B. rapa. Bars with different letters are significantly different [general least squares (GLS), n = 11–12; P < 0.05]. 3But, 3-butenyl; I3M, indol-3-ylmethyl; 1MO13M, 1-methoxyindol-3-ylmethyl; 7MSOH, 7-methylsulfinylheptyl; 8MSOO, 8-methylsulfinyloctyl; 3MSOP, 3-methylsulfinylpropyl; 5MSOP, 5-methylsulfinylpentyl; 2OH3But, 2-hydroxy-3-butenyl; 4OHI3M, 4-hydroxyindol-3-ylmethyl; 4Pent, 4-pentenyl.
After 7 d, the highest total glucosinolate concentration was detected in adults that had been feeding on B. juncea (31.2 ± 1.9 nmol × mg−1 per beetle, ±SEM, n = 12). An additional group of beetles was fed for several weeks with B. juncea, and their glucosinolate content was afterward determined to be 47.9 ± 1.8 nmol × mg−1 per beetle (n = 5), of which 95% was accounted for by alkenyl glucosinolates. Additionally, traces of indolic glucosinolates and benzyl glucosinolate were detected in beetles after feeding on B. juncea. The total glucosinolate content corresponded to 1.75% of body weight.
Identification of a P. striolata Myrosinase.
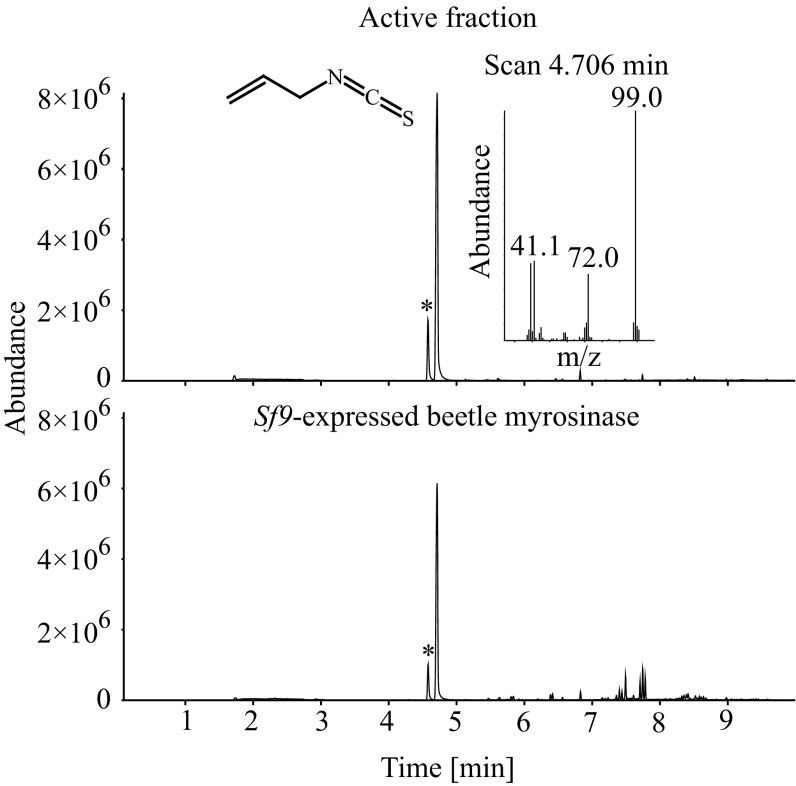
Endogenous flea beetle β-thioglucosidase activity was detected by myrosinase activity assays of crude beetle protein extract, and ITCs were identified as major products (SI Results and Fig. S3). Myrosinase activity was partially purified using three chromatographic steps. Following size exclusion chromatography, the active fraction was subjected to Con A chromatography in which glycosylated proteins bind to the immobilized lectin. Myrosinase activity was recovered in the bound fraction, indicating that the enzyme is glycosylated. A third purification step using anion exchange chromatography yielded an active fraction, which, after separation on a 1D SDS/PAGE gel, was shown to contain several proteins. Sixteen subfractions covering the apparent molecular mass range from 25 to 100 kDa (Fig. S4) were prepared for protein liquid chromatography mass spectrometry (LC-MS) analyses. Separated proteins were characterized using complementary mass spectrometric approaches: data-independent acquisition (DIA; LC-MSE) and data-dependent acquisition (DDA; LC-MS/MS). The data obtained were used to search a protein database derived from the P. striolata transcriptome. In total, 45 proteins were identified by at least one of the MS methods (Dataset S2). According to the nomenclature for carbohydrate-active enzymes (31), six of these proteins were predicted to belong to the GH1 and to have β-glucosidase activity. Because all plant myrosinases, as well as the only previously characterized insect myrosinase from the cabbage aphid, B. brassicae, are GH1 enzymes with high similarity to β-O-glucosidases, we focused on these six candidates. These GH1 proteins were positively identified using LC-MSE and LC-MS/MS in proteome samples 4, 5, and 7 (apparent molecular mass between 55 and 70 kDa), except for GH1-6, which was only identified with LC-MSE in sample 4 (Dataset S2). The full-length sequences of all six GH1 candidates were cloned into the pIB/V5 His TOPO TA vector (Invitrogen) and expressed in Sf9 cell cultures. The recombinant enzymes were detected as secreted proteins in the cell culture medium using Western blotting (Fig. S5). Activity assays revealed that only one of the heterologously expressed GH1 enzymes was able to hydrolyze allyl glucosinolate, resulting in the release of the product allyl ITC as observed in crude and purified protein extracts from P. striolata (Fig. 3).
Fig. 3.
GC-MS analyses (total ion chromatograms) of myrosinase activity assays using partially purified myrosinase from the P. striolata protein extract (Upper) and heterologously expressed P. striolata myrosinase (Lower) and allyl glucosinolate as a substrate. The volatile hydrolysis product allyl ITC collected by solid-phase microextraction was detected at 4.7 min. The preceding peak designated with an asterisk corresponds to allyl thiocyanate, which is a thermal artifact of allyl ITC (SI Materials and Methods). Allyl ITC was not detected in control assays conducted with pure buffer (without protein) or without substrate.
Characterization of a P. striolata Myrosinase.
The ORF of the myrosinase gene encodes 495 amino acids, including a predicted N-terminal signal peptide of 20 amino acids. Furthermore, three predicted N-glycosylation sites and the recovery of myrosinase activity in the bound fraction after Con A chromatography suggest that the difference between the calculated molecular mass (54.9 kDa) and the apparent molecular mass of the corresponding protein band in the active fraction (Fig. S4, sample 5, estimated apparent molecular mass of 64.4 kDa) and of the recombinant protein detected using Western blotting is due to glycosylation.
The P. striolata myrosinase shares 30% sequence identity with the plant myrosinase from white mustard, S. alba, and 40% identity with the aphid myrosinase from B. brassicae. An amino acid alignment of the three myrosinases shows that both insect proteins possess the two glutamate residues involved in catalysis, one of which acts as a nucleophile and the other in acid/base catalysis hydrolysis of the glycosidic bond (32), whereas in the S. alba myrosinase, only the glutamate acting as a nucleophile is present, with the other glutamate residue replaced by a glutamine (Fig. S6). The S. alba enzyme is thought to mediate hydrolysis by the good leaving group character of the glucosinolate aglycone and the use of the cofactor ascorbate as a base (33). The glucose recognition site is identical among the three enzymes; however, only two of the seven residues forming the aglycone binding site in plant myrosinases are identical in the P. striolata myrosinase (Fig. S6).
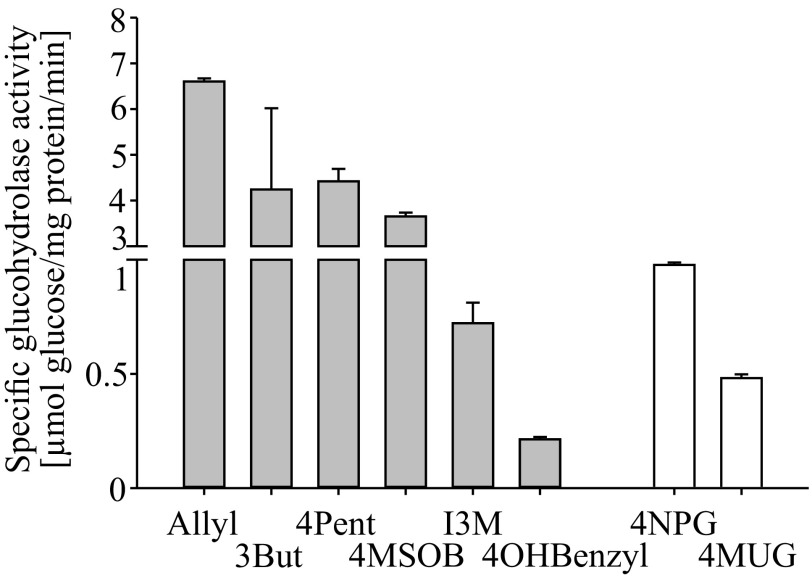
We used recombinant enzyme that was partially purified via the vector-encoded C-terminal His-tag for preliminary characterization of the activity. The pH optimum of the enzyme was determined to be 6.5, whereas at pH 5.5 and pH 7.6, activity was reduced by more than 75%. From all glucosinolates tested in enzyme assays, the highest myrosinase activity was detected with allyl glucosinolate. Activity with 3-butenyl glucosinolate, 4-pentenyl glucosinolate, and 4-methylsulfinylbutyl (4MSOB) glucosinolate was 50–70% of that with allyl glucosinolate (Fig. 4), but indol-3-ylmethyl glucosinolate and 4-hydroxybenzyl glucosinolate were hydrolyzed at only 11% and 3%, respectively, of the rate with allyl glucosinolate. To determine if the flea beetle myrosinase exhibited β-O-glucosidase as well as β-thioglucosidase activity, the general β-O-glucosidase substrates 4-nitrophenyl- and 4-methylumbelliferyl-β-d-glucopyranoside were tested. These substrates were hydrolyzed at only 15% and 7%, respectively, of the activity toward allyl glucosinolate.
Fig. 4.
Substrate specificity of the P. striolata myrosinase. Activity of the partially purified Sf9-expressed beetle myrosinase is shown with the glucosinolates (β-thioglucosides) allyl glucosinolate, 3But, 4Pent, 4MSOB, I3M, and 4-hydroxybenzyl glucosinolate (4OHBenzyl), and with the β-O-glucosides 4-nitrophenylglucopyranoside (4NPG) and 4-methylumbelliferyl-β-d-glucopyranoside (4MUG). All substrates were tested at a substrate concentration of 1.25 mM (n = 3, +SD).
GH1 β-Glucosidases Constitute a Large Multigene Family in P. striolata.
Based on sequence similarity, we identified 28 putative GH1 β-glucosidase sequences, including the myrosinase in our P. striolata cDNA library, and recovered 25 full-length and three partial sequences using rapid amplification of cDNA ends PCR (Dataset S3 and Table S3; GenBank accession nos. KF377828–KF377855). An amino acid alignment of the predicted full-length protein sequences revealed a high level of sequence divergence in the GH1 β-glucosidase family of P. striolata, with sequence identities between 25% and 75%. Interestingly, several of these predicted enzymes may not be active β-glucosidases because one or both catalytic Glu residues are substituted with residues that have different biochemical properties (Fig. S7).
The relative expression levels of GH1 β-glucosidases in male and female P. striolata, as revealed by digital gene expression analysis, were similar for most genes, including the beetle myrosinase (Table S3). However, a few genes showed sex-specific expression patterns [i.e., four genes were female-specific (GH1-3, GH1-10, GH1-11, and GH1-26), but only one gene was male-specific (GH1-21)].
Evolution of the P. striolata Myrosinase.
A maximum likelihood-inferred phylogeny of all predicted P. striolata GH1 β-glucosidases, the aphid myrosinase, as well as putative GH1 β-glucosidases from other Coleoptera and Hemiptera, showed species-specific diversification of the enzyme family (Fig. S8). Only one predicted sequence from Tribolium castaneum clustered together with two P. striolata sequences, suggesting that they are true orthologs. The beetle myrosinase, together with five other putative P. striolata GH1 β-glucosidases, formed a distinct clade, which is strongly supported by high bootstrap and posterior probability values (99/1). Within this clade, the myrosinase clusters together with GH1-10 (58.9% amino acid identity), again with strong node support (89/1). We expressed GH1-10, as well as GH1-1, GH1-3, and GH1-8 in Sf9 cells (Fig. S5), but none of these heterologously expressed proteins showed β-thioglucosidase activity in our experiments. However, the aphid myrosinase clusters separately with three predicted GH1 enzymes designated “myrosinase 1-like” from the pea aphid, Acyrthosiphon pisum, which indicates that both insect myrosinases evolved independently of each other.
Discussion
Several genera in the Chrysomelidae have evolved the ability to use crucifers as host plants (22) despite the presence of the glucosinolate-myrosinase defense system (6). The flea beetle genus Phyllotreta is particularly interesting in this regard because virtually all species (about 180 in total) are specialized on plants in the order Brassicales, and it includes several devastating pest species of crucifer crops, such as P. striolata and P. cruciferae (34). Although the ITC metabolites of glucosinolates are effective plant defense compounds against Phyllotreta (35), P. striolata may be able to release volatile ITCs without being negatively affected (30). However, the origin of flea beetle ITCs and their mechanism of formation have not been previously understood.
Here, we show that P. striolata beetles not only selectively accumulate substantial amounts of aliphatic glucosinolates from their host plants to up to 1.75% of their body weights but also possess their own myrosinase for the formation of bioactive hydrolysis products, which exhibits specificity for aliphatic glucosinolates. This sophisticated beetle glucosinolate-myrosinase system represents the first example in crucifer-feeding Coleoptera of an adaptive strategy for coping with dietary glucosinolates.
For chewing insects, such as Phyllotreta flea beetles, the plant myrosinase represents a major barrier to the sequestration of intact glucosinolates, because the spatial separation of glucosinolates and the myrosinase in the plant tissue is typically destroyed during feeding, leading to glucosinolate hydrolysis (7). The observed accumulation of intact glucosinolates in P. striolata thus requires physiological adaptations to either directly or indirectly circumvent plant myrosinase activity. All other known glucosinolate-sequestering herbivores, except for sawfly larvae of the genus Athalia, are sucking insects that may be able to feed without destroying glucosinolate-myrosinase compartmentation (7). In Athalia rosae, glucosinolates are further metabolized to desulfoglucosinolates, glucosinolate-sulfates, and desulfoglucosinolate-sulfates before excretion (36), but sulfatase activity comparable to that described in Plutella xylostella (12) could not be detected in A. rosae protein extracts (37). Sulfatase activity assays performed with P. striolata protein extracts also revealed no formation of desulfoglucosinolates (SI Materials and Methods). To date, the mechanisms that enable P. striolata and Athalia spp. to sequester intact glucosinolates remain unclear.
Sequestration of host plant glucosinolates by P. striolata is highly selective and not simply dependent on the relative glucosinolate concentration in the plant (Fig. 2). Aliphatic glucosinolates are clearly preferred over indolic and aromatic glucosinolates. However, the accumulation patterns of aliphatic glucosinolates depended on the host plant and were specific as well (Fig. 2).
The much higher amount of 4MTB glucosinolate in comparison to 4MSOB glucosinolate in beetles that had been feeding on A. thaliana is surprising (Fig. 2B). The ∼1-mg–sized beetles could accumulate the observed amount of 4MSOB by consuming 1.3 mg of foliage per beetle per day but would have to consume 15.5 mg of leaves to account for direct sequestration of 4MTB. There are two possible explanations for this result: P. striolata may be able to biosynthesize glucosinolates de novo, or, alternatively, beetles may enzymatically modify some available glucosinolates, whereas others may only be acquired directly from a host plant source. The latter explanation is more likely because the de novo biosynthesis of glucosinolates is complex and requires a number of different enzymes. For example, because 4MSOB is so abundant in the plant, the amount present in only 0.24 mg of consumed foliage per beetle per day would account for the observations if converted to 4MTB by a methionine sulfoxide reductase-like activity.
Glucosinolates are selectively sequestered by other crucifer-feeding insects as well (7, 16, 20). For example, larvae of Athalia spp. sawflies show species-specific sequestration patterns in the hemolymph that were suggested to reflect adaptations to different host plants (16, 38). In the crucifer aphid, B. brassicae, glucosinolate accumulation in the hemolymph differs quantitatively between wingless and winged morphs feeding on the same diet, which indicates regulated transport between the gut and hemolymph (17). We also assume that transporters are involved in glucosinolate uptake from the gut in P. striolata. Considering the structural diversity of glucosinolates, the selective accumulation observed could result from the substrate specificity of one or more transporters. Given transporters with a hierarchical preference for various glucosinolates, differences in plant glucosinolate profiles could lead to divergence in glucosinolate accumulation rates for the same compound, which may be responsible for the observed accumulation of 4MSOB glucosinolate from A. thaliana but not from B. rapa (Fig. 2). Furthermore, other aspects of the host plant background, independent of the glucosinolate profile, could influence sequestration. It was recently shown that the sequestration of different ratios of iridoid glycosides by the curculionid weevil Cionus hortulanus was determined by the host plant genotype apart from its iridoid chemistry (39). Further experiments will be needed to disentangle the impact of glucosinolate structure and host plant background on glucosinolate sequestration in P. striolata.
The previously observed pattern of ITC emission by feeding P. striolata adults did not correspond to the glucosinolate chemistry of their current food plant, suggesting that beetles are able to store glucosinolates from previous feeding bouts. Indeed, we detected substantial amounts of aliphatic glucosinolates in adults that had only had access to indolic glucosinolates for 4 wk, which indicates that these aliphatic glucosinolates had been stored for at least this period (Table S1). Similarly, feeding studies with the harlequin bug, Murgantia histrionica, revealed traces of aliphatic glucosinolates after bugs had been maintained on a glucosinolate-free plant for several weeks (13).
The activation of sequestered glucosinolates in P. striolata requires β-thioglucosidase activity, which was readily detectable in crude beetle protein extracts. By combining proteomic and transcriptomic approaches, a gene encoding myrosinase activity in P. striolata was identified. The substrate specificity of the heterologously expressed enzyme was found to be in remarkable agreement with the glucosinolate sequestration pattern of the adults (Fig. 4). Aliphatic glucosinolates were the preferred substrates of the flea beetle myrosinase, with aromatic and indolic glucosinolates and two different standard β-O-glucoside substrates being hydrolyzed at much lower efficiencies. The previously identified aphid myrosinase from B. brassicae (40–42) displays much broader substrate specificity (41, 43).
The P. striolata myrosinase evolved independently of the B. brassicae myrosinase (Fig. S8). Both share only 40% amino acid identity and have much higher similarity to other GH1 β-glucosidases from their own insect orders. However, none of the closely related GH1 β-glucosidases from P. striolata that were expressed showed myrosinase activity, suggesting that only a single myrosinase gene is present in the beetle. Phylogenetic analyses further suggest that the flea beetle myrosinase and GH1-10 are products of gene duplication, but it is not clear whether GH1-10 has lost myrosinase activity or whether the β-thioglucosidase activity represents neofunctionalization after the duplication event.
The major hydrolysis products of the P. striolata myrosinase are ITCs (Fig. S3); however, we cannot exclude that other breakdown products, such as nitriles, are also formed depending on cellular conditions or by an insect specifier protein. Further research is required to understand glucosinolate degradation in this beetle.
The convergent evolution of an enzymatically activated defense in a specialized herbivore with one component being sequestered from the host plant and the other supplied by the herbivore has also been reported in another activated plant defense system (i.e., cyanogenesis). Burnet moths, Zygaena spp., sequester cyanogenic glucosides from their fabaceous host plants (44) and are additionally able to synthesize the same compounds de novo (45). In Zygaena trifolii larvae, a specific β-glucosidase (linamarase) involved in cyanogenic glycoside cleavage and cyanide release is colocalized with its substrate in the hemolymph but remains almost inactive at the prevailing pH (46). Interestingly, cyanogenic glucosides and derived metabolites were shown to play multiple roles throughout the Zygaena life cycle in protection, development, and communication (44).
In summary, we have shown that P. striolata flea beetles selectively accumulate aliphatic glucosinolates from their host plants and apparently avoid their hydrolysis by the plant myrosinase. The beetles have evolved their own myrosinase, which is correspondingly most active on aliphatic glucosinolates. The mechanism by which the beetle controls the hydrolysis of its stored glucosinolates to avoid autointoxication remains to be determined but is likely to be actively controlled. In contrast, the “mustard-oil bomb” in aphids is activated when an individual is attacked by a predator, which benefits the survival of the clonal aphid colony by repelling the predator (17, 19, 47). Although we cannot rule out the participation of sequestered glucosinolates in a similar defense in Phyllotreta, an additional signaling role is also suggested by previous studies showing that ITCs enhance the response to the male-produced aggregation pheromone (26–29). Elucidation of the spatial organization of the glucosinolate-myrosinase system, as well as the roles of glucosinolate hydrolysis products in communication and defense, will be required to understand the ecological significance of the remarkable myrosinase activity in Phyllotreta beetles that has evolved convergently with myrosinase activity in its host plant.
Materials and Methods
Feeding studies were performed with field-collected P. striolata adults on the host plants S. alba, B. rapa, B. juncea, and A. thaliana Col-0. Beetles were allowed to feed for 1, 3, and 7 d on the plant, and the corresponding glucosinolate profiles in beetles and host plants were analyzed with HPLC-UV. To analyze the accumulation of host plant glucosinolates (calculated as the percentage relative to the induced plant glucosinolate content) in beetles, regressions were used. Myrosinase activity was detected in crude beetle extracts and was partially purified via three chromatographic steps using an Äkta FPLC system (GE Healthcare). The final active fraction was separated on a 1D SDS/PAGE gel, and protein bands between 25 and 100 kDa were further analyzed using LC-MS/MS and LC-MSE. Peptide data were searched against a P. striolata EST database. Candidates were cloned into the pIB/V5 His TOPO TA vector (Invitrogen) and transiently expressed in the Sf9 cell line. Expression was confirmed by Western blotting, and enzyme activity was analyzed in assays using allyl glucosinolate as a substrate. The hydrolysis product allyl ITC was detected by solid-phase microextraction coupled with GC-MS. The identified beetle myrosinase was stably expressed in Sf9 cells and purified via the vector-encoded His-tag for characterization of substrate specificity. Details about organisms used in this study, analytical procedures, data analysis, and all other methods used are provided in SI Materials and Methods.
Supplementary Material
Acknowledgments
We thank Daniel Giddings-Vassão, Peter Rahfeld, Roy Kirsch, Katharina Schramm, and Nicole Joußen for discussion and technical support; Yvonne Hupfer for preparing the proteomic samples for MS analysis; Steffi Gebauer-Jung for support of database searches; Kerstin Weniger for support of GC-MS analyses; Domenica Schnabelrauch for DNA sequencing services; Lin Mei-ying and Hu Ruey-chuan from AVRDC–The World Vegetable Center in Taiwan for providing P. striolata for experiments; and the greenhouse team of the Max Planck Institute for Chemical Ecology for taking care of plants. Financial support was provided by the Max-Planck-Gesellschaft.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
Data deposition: The sequences reported in this paper have been deposited in the GenBank database (accession nos. KF377828–KF377855).
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1321781111/-/DCSupplemental.
References
- 1.Mithöfer A, Boland W. Plant defense against herbivores: Chemical aspects. Annu Rev Plant Biol. 2012;63:431–450. doi: 10.1146/annurev-arplant-042110-103854. [DOI] [PubMed] [Google Scholar]
- 2.Opitz SEW, Müller C. Plant chemistry and insect sequestration. Chemoecology. 2009;19(3):117–154. [Google Scholar]
- 3.Nishida R. Sequestration of defensive substances from plants by Lepidoptera. Annu Rev Entomol. 2002;47:57–92. doi: 10.1146/annurev.ento.47.091201.145121. [DOI] [PubMed] [Google Scholar]
- 4.Hopkins RJ, van Dam NM, van Loon JJA. Role of glucosinolates in insect-plant relationships and multitrophic interactions. Annu Rev Entomol. 2009;54:57–83. doi: 10.1146/annurev.ento.54.110807.090623. [DOI] [PubMed] [Google Scholar]
- 5.Textor S, Gershenzon J. Herbivore induction of the glucosinolate-myrosinase defense system: Major trends, biochemical bases and ecological significance. Phytochem Rev. 2009;8(1):149–170. [Google Scholar]
- 6.Halkier BA, Gershenzon J. Biology and biochemistry of glucosinolates. Annu Rev Plant Biol. 2006;57:303–333. doi: 10.1146/annurev.arplant.57.032905.105228. [DOI] [PubMed] [Google Scholar]
- 7.Winde I, Wittstock U. Insect herbivore counteradaptations to the plant glucosinolate-myrosinase system. Phytochemistry. 2011;72(13):1566–1575. doi: 10.1016/j.phytochem.2011.01.016. [DOI] [PubMed] [Google Scholar]
- 8.Agerbirk N, Olsen CE. Glucosinolate structures in evolution. Phytochemistry. 2012;77:16–45. doi: 10.1016/j.phytochem.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 9.Burow M, Wittstock U. Regulation and function of specifier proteins in plants. Phytochem Rev. 2009;8(1):87–99. [Google Scholar]
- 10.Schramm K, Vassão DG, Reichelt M, Gershenzon J, Wittstock U. Metabolism of glucosinolate-derived isothiocyanates to glutathione conjugates in generalist lepidopteran herbivores. Insect Biochem Mol Biol. 2012;42(3):174–182. doi: 10.1016/j.ibmb.2011.12.002. [DOI] [PubMed] [Google Scholar]
- 11.Wittstock U, et al. Successful herbivore attack due to metabolic diversion of a plant chemical defense. Proc Natl Acad Sci USA. 2004;101(14):4859–4864. doi: 10.1073/pnas.0308007101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ratzka A, Vogel H, Kliebenstein DJ, Mitchell-Olds T, Kroymann J. Disarming the mustard oil bomb. Proc Natl Acad Sci USA. 2002;99(17):11223–11228. doi: 10.1073/pnas.172112899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aliabadi A, Renwick JAA, Whitman DW. Sequestration of glucosinolates by harlequin bug Murgantia histrionica. J Chem Ecol. 2002;28(9):1749–1762. doi: 10.1023/a:1020505016637. [DOI] [PubMed] [Google Scholar]
- 14.Müller C, et al. Sequestration of host plant glucosinolates in the defensive hemolymph of the sawfly Athalia rosae. J Chem Ecol. 2001;27(12):2505–2516. doi: 10.1023/a:1013631616141. [DOI] [PubMed] [Google Scholar]
- 15.Weber G, Oswald S, Zöllner U. Suitability of rape cultivars with a different glucosinolate content for Brevicoryne brassicae (L) and Myzus persicae (Sulzer) (Hemiptera, Aphididae) Z Pflanzenk Pflanzen. 1986;93(2):113–124. [Google Scholar]
- 16.Opitz SEW, Jensen SR, Müller C. Sequestration of glucosinolates and iridoid glucosides in sawfly species of the genus Athalia and their role in defense against ants. J Chem Ecol. 2010;36(2):148–157. doi: 10.1007/s10886-010-9740-3. [DOI] [PubMed] [Google Scholar]
- 17.Kazana E, et al. The cabbage aphid: A walking mustard oil bomb. Proc Biol Sci. 2007;274(1623):2271–2277. doi: 10.1098/rspb.2007.0237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dawson GW, Griffiths DC, Pickett JA, Wadhams LJ, Woodcock CM. Plant-derived synergists of alarm pheromone from turnip aphid, Lipaphis (Hyadaphis) erysimi (Homoptera, Aphididae) J Chem Ecol. 1987;13(7):1663–1671. doi: 10.1007/BF00980207. [DOI] [PubMed] [Google Scholar]
- 19.Bridges M, et al. Spatial organization of the glucosinolate-myrosinase system in brassica specialist aphids is similar to that of the host plant. Proc Biol Sci. 2002;269(1487):187–191. doi: 10.1098/rspb.2001.1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Francis F, Lognay G, Wathelet JP, Haubruge E. Effects of allelochemicals from first (Brassicaceae) and second (Myzus persicae and Brevicoryne brassicae) trophic levels on Adalia bipunctata. J Chem Ecol. 2001;27(2):243–256. doi: 10.1023/a:1005672220342. [DOI] [PubMed] [Google Scholar]
- 21.Feeny P, Paauwe KL, Demong NJ. Flea beetles and mustard oils—Host plant specificity of Phyllotreta cruciferae and P. striolata adults (Coleoptera: Chrysomelidae) Ann Entomol Soc Am. 1970;63(3):832–841. [Google Scholar]
- 22.Nielsen JK. Crucifer-feeding Chrysomelidae: Mechanisms of host plant finding and acceptance. In: Jolivet PH, Petitpierre E, Hsiao TH, editors. Biology of Chrysomelidae. Dordrecht, The Netherlands: Kluwer; 1988. pp. 25–40. [Google Scholar]
- 23.Vig K. Biology of Phyllotreta (Alticinae), with emphasis on Hungarian and middle European species. In: Jolivet P, Santiago-Blay JA, Schmitt M, editors. New Developments in the Biology of Chrysomelidae. Amsterdam: SPB Academic; 2004. pp. 565–578. [Google Scholar]
- 24.Vig K. Host plant selection by Phyllotreta vittula (Redtenbacher, 1849) In: Biondi M, Daccordi M, Furth DG, editors. Proceedings of the Fourth International Symposium on Chrysomelidae, XX International Congress of Entomology. Torino, Italy: Museo Regionale di Scienze Naturali; 1998. pp. 233–251. [Google Scholar]
- 25.Siemens DH, Mitchell-Olds T. Evolution of pest-induced defenses in Brassica plants: Tests of theory. Ecology. 1998;79(2):632–646. [Google Scholar]
- 26.Beran F, et al. Male Phyllotreta striolata (F.) produce an aggregation pheromone: Identification of male-specific compounds and interaction with host plant volatiles. J Chem Ecol. 2011;37(1):85–97. doi: 10.1007/s10886-010-9899-7. [DOI] [PubMed] [Google Scholar]
- 27.Tóth M, Csonka E, Bartelt RJ, Cossé AA, Zilkowski BW. Similarities in pheromonal communication of flea beetles Phyllotreta cruciferae Goeze and Ph. vittula Redtenbacher (Coleoptera, Chrysomelidae) J Appl Entomol. 2012;136(9):688–697. [Google Scholar]
- 28.Tóth M, et al. Pheromonal activity of compounds identified from male Phyllotreta cruciferae: Field tests of racemic mixtures, pure enantiomers, and combinations with allyl isothiocyanate. J Chem Ecol. 2005;31(11):2705–2720. doi: 10.1007/s10886-005-7621-y. [DOI] [PubMed] [Google Scholar]
- 29.Soroka JJ, Bartelt RJ, Zilkowski BW, Cossé AA. Responses of flea beetle Phyllotreta cruciferae to synthetic aggregation pheromone components and host plant volatiles in field trials. J Chem Ecol. 2005;31(8):1829–1843. doi: 10.1007/s10886-005-5929-2. [DOI] [PubMed] [Google Scholar]
- 30.Beran F. Phyllotreta striolata. Berliner ökophysiologische und phytomedizinische Schriften. Vol 19. Uelvesbüll, Germany: Der Andere; 2011. Host preference and aggregation behavior of the striped flea beetle. p 143. German. [Google Scholar]
- 31.Cantarel BL, et al. The Carbohydrate-Active EnZymes database (CAZy): An expert resource for Glycogenomics. Nucleic Acids Res. 2009;37(Database issue):D233–D238. doi: 10.1093/nar/gkn663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Burmeister WP, et al. The crystal structures of Sinapis alba myrosinase and a covalent glycosyl-enzyme intermediate provide insights into the substrate recognition and active-site machinery of an S-glycosidase. Structure. 1997;5(5):663–675. doi: 10.1016/s0969-2126(97)00221-9. [DOI] [PubMed] [Google Scholar]
- 33.Burmeister WP, Cottaz S, Rollin P, Vasella A, Henrissat B. High resolution X-ray crystallography shows that ascorbate is a cofactor for myrosinase and substitutes for the function of the catalytic base. J Biol Chem. 2000;275(50):39385–39393. doi: 10.1074/jbc.M006796200. [DOI] [PubMed] [Google Scholar]
- 34.Lamb RJ. Entomology of oilseed Brassica crops. Annu Rev Entomol. 1989;34:211–229. [Google Scholar]
- 35.Mitchell-Olds T, Siemens D, Pedersen D. Physiology and costs of resistance to herbivory and disease in Brassica. Entomol Exp Appl. 1996;80(1):231–237. [Google Scholar]
- 36.Opitz SEW, Mix A, Winde IB, Müller C. Desulfation followed by sulfation: Metabolism of benzylglucosinolate in Athalia rosae (Hymenoptera: Tenthredinidae) ChemBioChem. 2011;12(8):1252–1257. doi: 10.1002/cbic.201100053. [DOI] [PubMed] [Google Scholar]
- 37.Müller C, Wittstock U. Uptake and turn-over of glucosinolates sequestered in the sawfly Athalia rosae. Insect Biochem Mol Biol. 2005;35(10):1189–1198. doi: 10.1016/j.ibmb.2005.06.001. [DOI] [PubMed] [Google Scholar]
- 38.Abdalsamee MK, Müller C. Effects of indole glucosinolates on performance and sequestration by the sawfly Athalia rosae and consequences of feeding on the plant defense system. J Chem Ecol. 2012;38(11):1366–1375. doi: 10.1007/s10886-012-0197-4. [DOI] [PubMed] [Google Scholar]
- 39.Baden CU, Franke S, Dobler S. Host dependent iridoid glycoside sequestration patterns in Cionus hortulanus. J Chem Ecol. 2013;39(8):1112–1114. doi: 10.1007/s10886-013-0323-y. [DOI] [PubMed] [Google Scholar]
- 40.Jones AME, Bridges M, Bones AM, Cole R, Rossiter JT. Purification and characterisation of a non-plant myrosinase from the cabbage aphid Brevicoryne brassicae (L.) Insect Biochem Mol Biol. 2001;31(1):1–5. doi: 10.1016/s0965-1748(00)00157-0. [DOI] [PubMed] [Google Scholar]
- 41.Pontoppidan B, Ekbom B, Eriksson S, Meijer J. Purification and characterization of myrosinase from the cabbage aphid (Brevicoryne brassicae), a brassica herbivore. Eur J Biochem. 2001;268(4):1041–1048. doi: 10.1046/j.1432-1327.2001.01971.x. [DOI] [PubMed] [Google Scholar]
- 42.Jones AME, Winge P, Bones AM, Cole R, Rossiter JT. Characterization and evolution of a myrosinase from the cabbage aphid Brevicoryne brassicae. Insect Biochem Mol Biol. 2002;32(3):275–284. doi: 10.1016/s0965-1748(01)00088-1. [DOI] [PubMed] [Google Scholar]
- 43.Francis F, Lognay G, Wathelet JP, Haubruge E. Characterisation of aphid myrosinase and degradation studies of glucosinolates. Arch Insect Biochem Physiol. 2002;50(4):173–182. doi: 10.1002/arch.10042. [DOI] [PubMed] [Google Scholar]
- 44.Zagrobelny M, Møller BL. Cyanogenic glucosides in the biological warfare between plants and insects: The Burnet moth-Birdsfoot trefoil model system. Phytochemistry. 2011;72(13):1585–1592. doi: 10.1016/j.phytochem.2011.02.023. [DOI] [PubMed] [Google Scholar]
- 45.Jensen NB, et al. Convergent evolution in biosynthesis of cyanogenic defence compounds in plants and insects. Nat Commun. 2011;2:273. doi: 10.1038/ncomms1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Franzl S, Ackermann I, Nahrstedt A. Purification and characterization of a β-glucosidase (linamarase) from the hemolymph of Zygaena trifolii Esper, 1783 (Insecta, Lepidoptera) Experientia. 1989;45(8):712–718. [Google Scholar]
- 47.Chaplin-Kramer R, et al. Chemically mediated tritrophic interactions: Opposing effects of glucosinolates on a specialist herbivore and its predators. J Appl Ecol. 2011;48(4):880–887. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.