Abstract
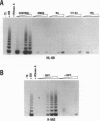
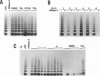
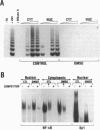
Telomerase, a ribonucleic acid-protein complex, adds hexameric repeats of 5'-TTAGGG-3' to the ends of mammalian chromosomal DNA (telomeres) to compensate for the progressive loss that occurs with successive rounds of DNA replication. Although somatic cells do not express telomerase, germ cells and immortalized cells, including neoplastic cells, express this activity. To determine whether the phenotypic differentiation of immortalized cells is linked to the regulation of telomerase activity, terminal differentiation was induced in leukemic cell lines by diverse agents. A pronounced downregulation of telomerase activity was produced as a consequence of the differentiated status. The differentiation-inducing agents did not directly inhibit telomerase activity, suggesting that the inhibition of telomerase activity is in response to induction of differentiation. The loss of telomerase activity was not due to the production of an inhibitor, since extracts from differentiated cells did not cause inhibition of telomerase activity. By using additional cell lineages including epithelial and embryonal stem cells, down-regulation of telomerase activity was found to be a general response to the induction of differentiation. These findings provide the first direct link between telomerase activity and terminal differentiation and may provide a model to study regulation of telomerase activity.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Counter C. M., Gupta J., Harley C. B., Leber B., Bacchetti S. Telomerase activity in normal leukocytes and in hematologic malignancies. Blood. 1995 May 1;85(9):2315–2320. [PubMed] [Google Scholar]
- Haber D. A. Telomeres, cancer, and immortality. N Engl J Med. 1995 Apr 6;332(14):955–956. doi: 10.1056/NEJM199504063321412. [DOI] [PubMed] [Google Scholar]
- Harley C. B. Telomere loss: mitotic clock or genetic time bomb? Mutat Res. 1991 Mar-Nov;256(2-6):271–282. doi: 10.1016/0921-8734(91)90018-7. [DOI] [PubMed] [Google Scholar]
- Higgins K. A., Perez J. R., Coleman T. A., Dorshkind K., McComas W. A., Sarmiento U. M., Rosen C. A., Narayanan R. Antisense inhibition of the p65 subunit of NF-kappa B blocks tumorigenicity and causes tumor regression. Proc Natl Acad Sci U S A. 1993 Nov 1;90(21):9901–9905. doi: 10.1073/pnas.90.21.9901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim N. W., Piatyszek M. A., Prowse K. R., Harley C. B., West M. D., Ho P. L., Coviello G. M., Wright W. E., Weinrich S. L., Shay J. W. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994 Dec 23;266(5193):2011–2015. doi: 10.1126/science.7605428. [DOI] [PubMed] [Google Scholar]
- Mantell L. L., Greider C. W. Telomerase activity in germline and embryonic cells of Xenopus. EMBO J. 1994 Jul 1;13(13):3211–3217. doi: 10.1002/j.1460-2075.1994.tb06620.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin G. B. Is telomerase a universal cancer target? J Natl Cancer Inst. 1995 Jun 21;87(12):859–861. doi: 10.1093/jnci/87.12.859. [DOI] [PubMed] [Google Scholar]
- Murnane J. P., Sabatier L., Marder B. A., Morgan W. F. Telomere dynamics in an immortal human cell line. EMBO J. 1994 Oct 17;13(20):4953–4962. doi: 10.1002/j.1460-2075.1994.tb06822.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narayanan R., Higgins K. A., Perez J. R., Coleman T. A., Rosen C. A. Evidence for differential functions of the p50 and p65 subunits of NF-kappa B with a cell adhesion model. Mol Cell Biol. 1993 Jun;13(6):3802–3810. doi: 10.1128/mcb.13.6.3802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pugh B. F., Tjian R. Mechanism of transcriptional activation by Sp1: evidence for coactivators. Cell. 1990 Jun 29;61(7):1187–1197. doi: 10.1016/0092-8674(90)90683-6. [DOI] [PubMed] [Google Scholar]
- Sokoloski J. A., Sartorelli A. C., Rosen C. A., Narayanan R. Antisense oligonucleotides to the p65 subunit of NF-kappa B block CD11b expression and alter adhesion properties of differentiated HL-60 granulocytes. Blood. 1993 Jul 15;82(2):625–632. [PubMed] [Google Scholar]
- Strickland S., Smith K. K., Marotti K. R. Hormonal induction of differentiation in teratocarcinoma stem cells: generation of parietal endoderm by retinoic acid and dibutyryl cAMP. Cell. 1980 Sep;21(2):347–355. doi: 10.1016/0092-8674(80)90471-7. [DOI] [PubMed] [Google Scholar]
- Tabilio A., Pelicci P. G., Vinci G., Mannoni P., Civin C. I., Vainchenker W., Testa U., Lipinski M., Rochant H., Breton-Gorius J. Myeloid and megakaryocytic properties of K-562 cell lines. Cancer Res. 1983 Oct;43(10):4569–4574. [PubMed] [Google Scholar]
- Tahara H., Nakanishi T., Kitamoto M., Nakashio R., Shay J. W., Tahara E., Kajiyama G., Ide T. Telomerase activity in human liver tissues: comparison between chronic liver disease and hepatocellular carcinomas. Cancer Res. 1995 Jul 1;55(13):2734–2736. [PubMed] [Google Scholar]