Significance
Cardiovascular thrombotic events are a leading cause of mortality and morbidity worldwide. Hypercoagulability increases the risk of thrombosis. This study shows that elevated sodium concentration stimulates endothelial production of a key initiator of the clotting cascade, von Willebrand factor, leading to increased coagulation and thrombogenesis. In everyday life, increases in blood sodium often occur as the result of insufficient drinking, excessive water loss, or high salt intake. Therefore, our results indicate that water and salt intake are modifiable factors affecting coagulability and risk of thrombosis. In clinical practice, sustained elevation of plasma sodium is a part of widely used hypertonic saline therapy. Our results suggest that monitoring of coagulation during the therapy might be beneficial to prevent thrombotic complications.
Keywords: blood clotting, HUVEC, osmotic stress
Abstract
Hypercoagulability increases risk of thrombi that cause cardiovascular events. Here we identify plasma sodium concentration as a factor that modulates blood coagulability by affecting the production of von Willebrand factor (vWF), a key initiator of the clotting cascade. We find that elevation of salt over a range from the lower end of what is normal in blood to the level of severe hypernatremia reversibly increases vWF mRNA in endothelial cells in culture and the rate of vWF secretion from them. The high NaCl increases expression of tonicity-regulated transcription factor NFAT5 and its binding to promoter of vWF gene, suggesting involvement of hypertonic signaling in vWF up-regulation. To elevate NaCl in vivo, we modeled mild dehydration, subjecting mice to water restriction (WR) by feeding them with gel food containing 30% water. Such WR elevates blood sodium from 145.1 ± 0.5 to 150.2 ± 1.3 mmol/L and activates hypertonic signaling, evidenced from increased expression of NFAT5 in tissues. WR increases vWF mRNA in liver and lung and raises vWF protein in blood. Immunostaining of liver revealed increased production of vWF protein by endothelium and increased number of microthrombi inside capillaries. WR also increases blood level of D-dimer, indicative of ongoing coagulation and thrombolysis. Multivariate regression analysis of clinical data from the Atherosclerosis Risk in Communities Study demonstrated that serum sodium significantly contributes to prediction of plasma vWF and risk of stroke. The results indicate that elevation of extracellular sodium within the physiological range raises vWF sufficiently to increase coagulability and risk of thrombosis.
Cardiovascular thrombotic events are a leading cause of mortality and morbidity worldwide (1–3). A critical step in thrombosis is inappropriate initiation of the coagulation cascade. The trigger is not always apparent. In some cases the triggering mechanism is vascular injury, similar to the initiation of hemostasis. For example, arterial thrombosis can be triggered by rupture of atherosclerotic plaques that leads to endothelial damage. However, venous thrombosis can also occur in the presence of an intact endothelium, making the triggering mechanism less apparent (1, 4, 5).
von Willebrand factor (vWF) and platelets are the key components of blood that initiate clots. vWF is a large multimeric glycoprotein. It is produced by endothelial cells, stored in the form of ultralarge (UL) vWF multimers in Weibel–Palade bodies, then secreted by exocytosis of the Weibel–Palade bodies (6, 7). Upon secretion, UL vWF multimers are cleaved by ADAMTS13 protease into smaller and less hemostatically active multimers that circulate in blood. Clot formation initiates when platelets bind to specific binding sites on vWF through the platelet membrane glycoprotein Ib. While vWF is circulating in blood in the globular latent form, the binding sites are not exposed. A special elongated confirmation is necessary for binding. This conformation occurs, for example, after secretion of vWF from endothelial cells before cleavage by ADAMTS13 protease, or under high shear stress in stenosed arteries. Numerous coagulation factors are then activated, resulting in growth of blood clots (4, 5, 8–10). A number of population-based studies have suggested that elevated levels of vWF are a risk factor for thrombogenesis, especially in patients with previous cardiovascular diseases and older people (9).
Hypernatremia has been defined as a rise in serum sodium to above 145 mmol/L (11). The most common cause of hypernatremia is reduced body water. Dehydration and hypernatremia are often accompanied by thrombosis. Thus, complications owing to thrombosis, such as disseminated intravascular coagulation, intracranial thrombosis, and peripheral thrombosis are commonly encountered during the hypernatremia associated with dehydration (12–14), the hyperosmolality associated with diabetes (15), and heatstroke (16). Increased sodium concentration is a common feature of the thrombosis in these studies. This led to recognition that hypernatremia might contribute to the thrombosis observed in such cases, but the mechanism had not been identified (17).
In the present study we demonstrate that high NaCl increases production and secretion of vWF from endothelial cells in cell culture and in tissues of mice in vivo. In mice, the modest increase of blood sodium caused by mild water restriction raises endothelial vWF secretion sufficiently to increase coagulability of blood and induce thrombosis. Further, analysis of data from the Atherosclerosis Risk in Communities (ARIC) Study demonstrates that serum sodium is positively associated with the level of vWF in blood and the risk of stroke in humans. Our results identify a mechanism for hypernatremia-induced thrombosis and suggest that hydration and salt intake are modifiable factors that affect coagulability and thrombosis through high salt-dependent secretion of vWF from endothelial cells. Fig. S1 summarizes the findings and the proposed implications of our study.
Results
High Salt-Induced vWF Secretion: Human Umbilical Vein Endothelial Cells.
vWF is critical for the development of venous thrombosis when blood flow is restricted by stenosis in mice (18). The thrombosis depends on the level of vWF. Thus, vWF knock-out mice do not develop the thrombi, and mice that have low vWF have reduced thrombosis. We hypothesized that the thrombosis that occurs during hypernatremia might also be caused by increased vWF secretion, albeit from intact endothelial cells, in response to the elevated NaCl in blood.
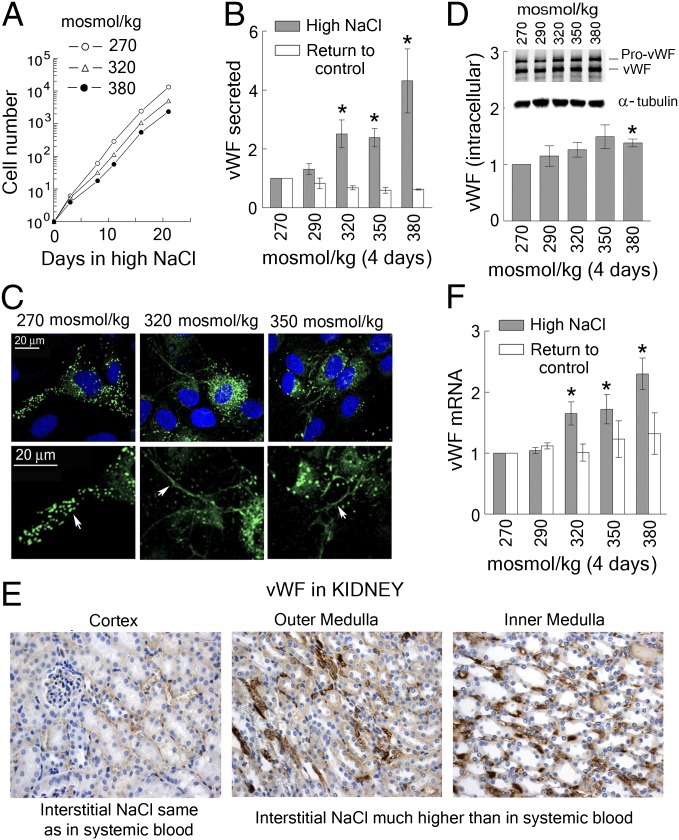
To test whether high NaCl affects production of vWF, we exposed human umbilical vein endothelial cells (HUVECs) in culture to the range of NaCl that occurs in humans during hypernatremia. We added up to 55 mmol/L of NaCl, elevating the medium osmolality from 270 mosmol/kg (lower end of the normal physiological range) to up to 380 mosmol/kg (severe hypernatremia). HUVECs adapt well up to at least 380 mosmol/kg, maintaining a normal appearance (Fig. S2A), and logarithmic growth (Fig. 1A), for weeks. High NaCl reversibly increases secretion of vWF (Fig. 1B), and the rate of secretion remains elevated for up to 5 wk, provided that NaCl remains elevated (Fig. S2B). Immunofluorescent staining demonstrates that HUVECs secrete long strings of vWF multimers while exposed to high NaCl, but, when not exposed to high NaCl, retain most of their vWF compactly stored in Weibel–Palade bodies (Fig. 1C). When NaCl is elevated, the level of intracellular vWF does not change much (Fig. 1D), indicating that the newly synthesized vWF is secreted nearly as fast as it is made. The rate of secretion returns to the basal level soon after NaCl is lowered, demonstrating that the salt-dependent increase is reversible (Fig. 1B). High NaCl also reversibly elevates vWF mRNA, consistent with increased transcription (Fig. 1F).
Fig. 1.
High NaCl increases production and secretion of vWF from vascular endothelial cells. (A–D and F) HUVECs were exposed to media in which NaCl was elevated for 4 d to the total osmolality indicated in the figure panels. (A) HUVECs adapt to the range of elevated NaCl that occurs in hypernatremia, maintaining logarithmic growth (see also Fig. S2A for images of the cells). (B) High NaCl increases secretion of vWF, but when the elevated NaCl is lowered to the control level (270 mosmol/kg) for 2 d vWF secretion returns to its control level (mean ± SEM, *P < 0.05, t test, n = 5, linearly dependent on NaCl concentration, P = 0.0009). (C) At 270 mosmol/kg vWF multimers are compactly stored in Weibel–Palade bodies (arrow), but when NaCl is increased for 4 d the cells release long fibers of uncondensed vWF multimers (arrow). Green, vWF; blue, nuclei stained with DAPI. The lower panel is a higher magnification of the upper panel. (D) High NaCl has little effect on the level of intracellular vWF protein. (Upper) Representative Western blot image. (Lower) Quantification, relative to 270 mosmol/kg, normalized to tubulin (mean ± SEM, *P < 0.05, t test, n = 3). (F) High NaCl increases vWF mRNA in HUVECs. vWF mRNA level returns to the basal when NaCl is lowered for 2 d (mean ± SEM, *P < 0.05, t test, n = 5, linearly dependent on NaCl concentration, P < 0.0001). (E) vWF protein is higher in the kidney medulla, where interstitial NaCl is very high, than in kidney cortex, where the interstitial concentration of NaCl is similar to that in systemic blood. Immunohistochemical staining for vWF (brown). See also Fig. S2C for colocalization with CD31staining (endothelial cell marker).
Having found a salt-dependent increase of vWF secretion from HUVECs, we questioned whether high salt produces the same effect on endothelial cells in vivo. The kidney was a convenient organ to start with because of the wide range of NaCl normally present in the different parts of the kidney. The level of NaCl in blood perfusing the cortex of the kidney is the same as that in systemic blood, whereas blood perfusing the renal medulla normally contains very high NaCl. In fact, vWF is higher in inner and outer renal medullas of mice than it is in the renal cortex (Fig. 1E and Fig. S2C). This finding supports the conclusion that a high salt-dependent increase of vWF production by endothelial cells occurs in vivo, as well as in cell culture.
Role of the Osmotically Regulated Transcription Factor NFAT5 in High Salt-Induced vWF Secretion.
NaCl is a functionally impermeant solute, so elevation of extracellular NaCl is hypertonic. High NaCl causes osmotic efflux of water from cells, leading to decreased cell volume and increased intracellular ionic strength. Adaptive cellular responses are activated to compensate for the dehydration and its consequences. NFAT5 is the master transcription factor that is activated by hypertonicity. Hypertonicity increases expression of NFAT5 mRNA and protein, increases NFAT5 transcriptional and transactivating activities, and mediates transcription of many NFAT5 target genes that are directly or indirectly involved in adaptation to high NaCl (19).
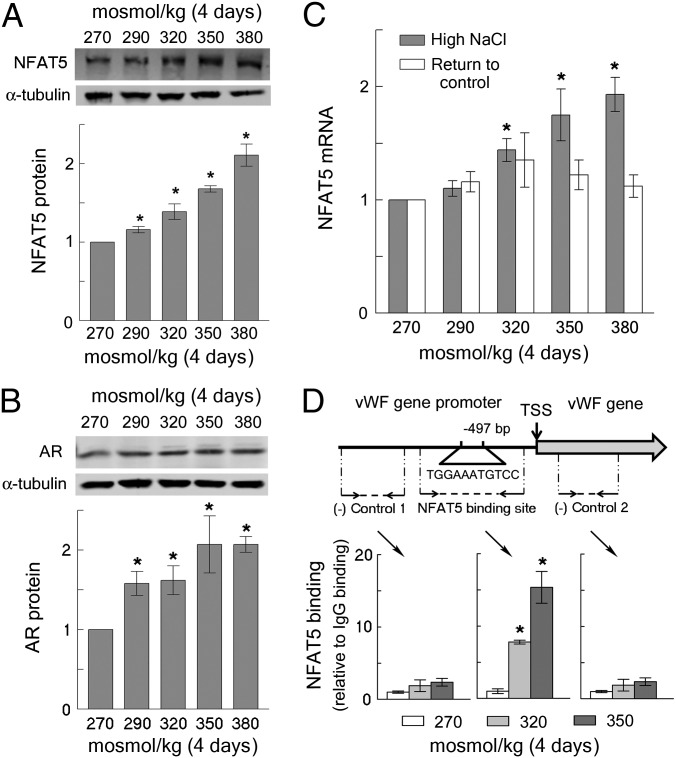
Therefore, we tested whether vWF is an NFAT5 target gene in HUVECs. High NaCl elevates vWF mRNA, which is consistent with increased transcription (Fig. 1F). The vWF promoter contains an osmotic response element (20) (ORE, NFAT5 binding site) close to the transcription start site (Fig. 2D). In HUVECs, high NaCl increases NFAT5 mRNA (Fig. 2C) and NFAT5 protein (Fig. 2A) in correlation with the salt-dependent increases of vWF mRNA and protein secretion (Fig. 1 B and F). In addition, the NFAT5 target, aldose reductase, also increases (Fig. 2B), consistent with up-regulation of NFAT5 transcriptional activity. Finally, ChIP analysis confirms binding of NFAT5 to the ORE in the vWF gene promoter in proportion to the level of NaCl (Fig. 2D). These results indicate that high NaCl-induced increase of NFAT5 activity contributes to the increased vWF production in endothelial cells.
Fig. 2.
(A–C) High NaCl-induced secretion of vWF from HUVECs is accompanied by increased expression of the osmoregulated transcription factor NFAT5. HUVECs were exposed to high NaCl, as in Fig 1. (A) High NaCl increases NFAT5 protein. (Upper) Representative Western blot. (Lower) Quantification, relative to 270 mosmol/kg, normalized to tubulin (mean ± SEM, *P < 0.05, t test, n = 4, linearly dependent on NaCl concentration, P < 0.0001). (B) High NaCl increases expression of aldose reductase (AR), which is a transcriptional target of NFAT5. Western blot, analyzed as in A (mean ± SEM, *P < 0.05, t test, n = 3, linearly dependent on NaCl concentration, P = 0.006). (C) High NaCl increases NFAT5 mRNA. Quantification is relative to 270 mosmol/kg (mean ± SEM, *P < 0.05, t test, n = 5, linearly dependent on NaCl concentration, P < 0.0001). The NFAT5 mRNA returns to the basal level when NaCl is lowered for 2 d. (D) High NaCl increases binding of NFAT5 to the DNA element that is an NFAT5 binding site in the promoter of the vWF gene. (Upper) Diagram showing location of the NFAT5 binding site upstream of transcription start site (TSS) of the vWF gene, and the positions of primers that were used to analyze NFAT5 binding by ChIP. (Lower) ChIP results, relative to 270 mosmol/kg (mean ± SEM, *P < 0.05, t test, n = 3).
Dehydration and Hypertonic Signaling in Mice.
We next tested whether hypernatremia results in increased production of vWF in endothelial cells in vivo. We chose dehydration as a model to increase NaCl in vivo. Dehydration is defined as net loss of body water resulting from decreased water intake or increased water loss. Dehydration leads to elevated osmolality of plasma and other extracellular fluids (21).
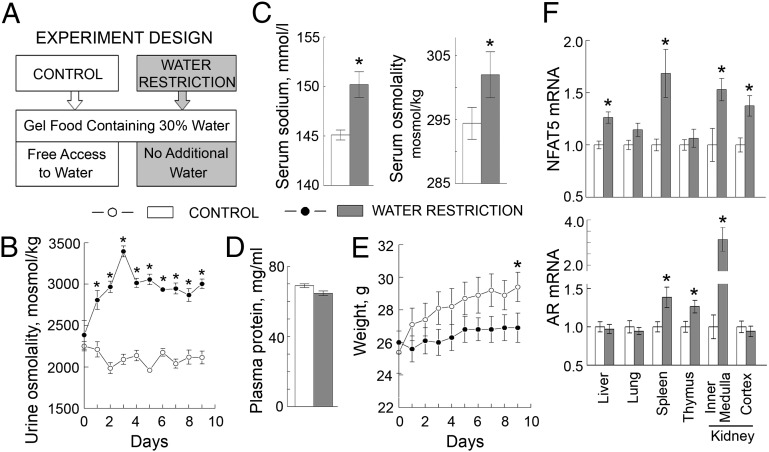
We controlled the amount of water that the mice consumed by feeding them with gel food containing 30% water as their only source of water intake or, as a control, feeding the same food, but with free access to drinking water (Fig. 3A). To assess the degree of dehydration, we measured urine osmolality, body weight, serum sodium, serum osmolality, and plasma protein concentration. Water restriction increased urine osmolality, indicating activation of the renal concentrating mechanism to conserve water (Fig. 3B). The water-restricted mice did not lose weight, but their growth was retarded, so they weighed about 2% less than control mice by the end of 9 d of water restriction (Fig. 3E). Plasma protein concentration was unchanged (Fig. 3D). The dehydration of our water-restricted mice is of a magnitude recognized to be quite mild (22). Still, serum sodium and serum osmolality increased (Fig. 3C). Thus, our model produced a small increase of plasma sodium within the physiological range. Despite the fact that the increase in serum sodium and osmolality (Fig. 3C and Table S1) were very modest, we detected activation of a hypertonic response in several tissues, as evidenced by increased expression of NFAT5 and its transcriptional target, aldose reductase (Fig. 3F).
Fig. 3.
Water restriction elevates serum sodium and activates hypertonic signaling in mouse tissues. To elevate NaCl in vivo, mice were subjected to water restriction for 9 d. (A–E) The water restriction produced mild subclinical dehydration (mean ± SEM, n = 5, *P < 0.05, t test). (A) Experiment design. To limit the amount of water, mice were fed with gel food containing 30% water and were not given any additional water. Control group were fed the same gel food but had free access to water. (B) Water restriction increased urine osmolality. (C) Water restriction increased serum sodium (by 5 mmol/L) and serum osmolality. See also Table S1 for other serum parameters. (D) Water restriction did not change plasma protein concentration. (E) Water restriction retarded growth but did not acutely reduce weight. (F) Water restriction increased mRNA of NFAT5 and of its transcriptional target, aldose reductase (AR) in several mouse tissues.
Dehydration, vWF Secretion, and Thrombosis in Mice.
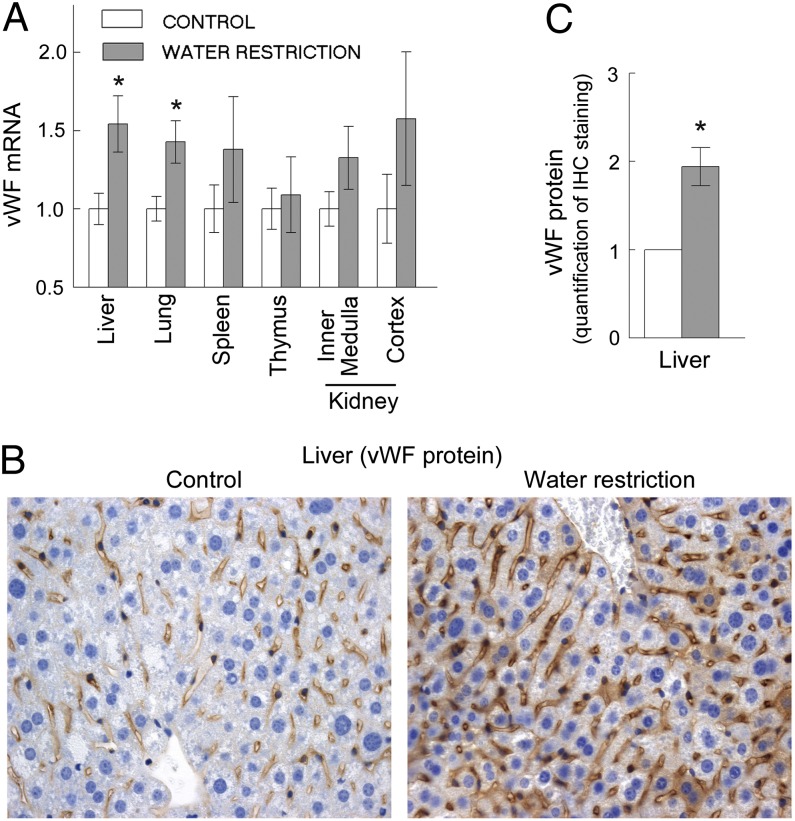
In HUVECs, high NaCl increases vWF production (Figs. 1 and 2). To see whether mild dehydration, caused by water restriction, affects production of vWF by endothelial cells in mouse tissues, we analyzed vWF mRNA and protein in tissues of water-restricted mice. Mild water restriction increases vWF mRNA significantly in the liver and lungs and also increases it in some other tissues, but not to a statistically significant degree (Fig. 4A). We measured vWF protein in endothelial cells by immunohistochemistry of the liver tissue sections. The mild water restriction significantly increases vWF in endothelial cells of the liver (Fig. 4 B and C and Fig. S3). vWF apparently rises in endothelial cells, as evidenced by correlation of staining for vWF with staining for the endothelial cell marker CD31 (Fig. S4).
Fig. 4.
Water restriction increases vWF in endothelial cells of mice (mean ± SEM, n = 5, *P < 0.05, t test). Water was restricted for 9 d. (A) vWF mRNA increased in liver and lung. (B and C) Water restriction increased vWF protein in endothelial cells in the liver. (B) Representative images from immunohistochemical staining for vWF protein (brown) in the liver tissue sections. See Fig. S4 for pattern of blood capillaries in the liver (same sections stained for endothelial cells marker CD31). (C) Quantification of vWF in tissue sections. See Materials and Methods and Fig. S3 for details about image analysis.
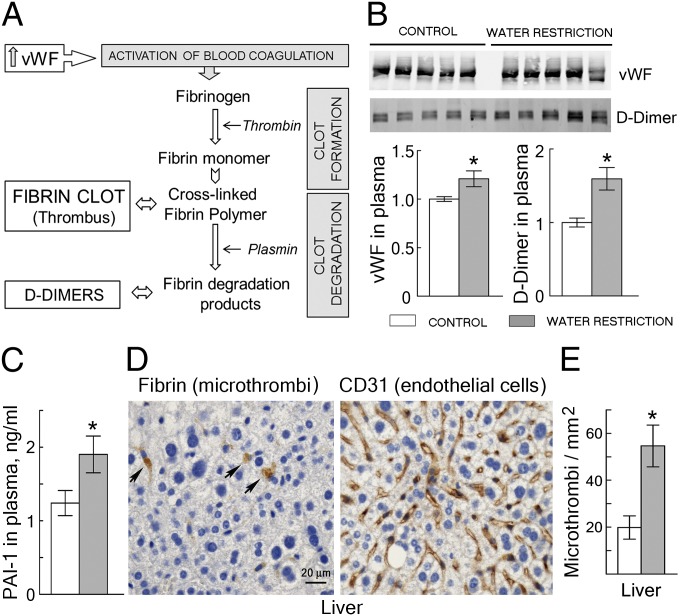
We hypothesized that dehydration-induced increase of secretion of vWF from endothelial cells in vivo might elevate vWF in blood and promote thrombosis. To assess the effect of water restriction on coagulation, we analyzed some of the factors depicted on Fig. 5A. Interaction of vWF with platelets activates thrombin and converts soluble plasma fibrinogen into insoluble, cross-linked fibrin polymers that stabilize blood clots (thrombi) (5, 10). Concurrently, clots are degraded by activation of other factors, principally the protease plasmin. Degradation of the fibrin polymers by plasmin leads to the appearance of fibrin degradation products (FDPs) in blood. D-dimer is the FDP whose level is used as a clinical indicator of ongoing coagulation. D-dimer increases in thrombotic conditions such as disseminated intravascular coagulation, deep venous thrombosis, and pulmonary embolism (5, 10, 23). We analyzed the levels of vWF and D-dimer in mouse blood and the number of microthrombi in the liver. Water restriction increases vWF and D-dimer in plasma (Fig. 5B), as well as the number of microthrombi in capillaries of the liver (Fig. 5 D and E). In addition, plasminogen activator inhibitor 1 (PAI-1) also increases (Fig. 5C), which limits the activation of plasmin and favors thrombosis. Thus, mild water restriction increases endothelial cell secretion of vWF enough to elevate the level of vWF in blood and to promote thrombosis in mice.
Fig. 5.
Water restriction increases vWF in the blood of mice and activates thrombogenesis (mean ± SEM, n = 5, *P < 0.05, t test). Water was restricted for 9 d. (A) Overview of blood clotting. Increase of vWF causes platelet activation and aggregation, leading to coagulation and formation of fibrin clots (thrombi). Concurrent activation of fibrinolysis begins degrading clots, which increases D-dimer in blood. (Note that measurement of D-dimer is also a clinical test for thrombogenesis.) (B) Water restriction increases vWF and D-dimer in plasma of mice. (Upper) Western blots. (Lower) Quantification, normalized to control. (C) Water restriction increases PAI-1 in plasma of mice. Note that PAI-1 inhibits fibrinolysis, which delays degradation of fibrin clots. (D) Representative images of immunohistochemical staining (brown) of liver tissue sections for fibrin and for the endothelial cell marker CD31. The staining for fibrin identifies microthrombi (arrows) inside liver capillaries. (E) Quantification of microthrombi in tissue sections from liver. Water restriction increases the number of microthrombi per square millimeter.
Positive Association of Serum Sodium Concentration with Blood Level of vWF and Stroke Risk in Humans.
To access the relevance of our findings to humans, we analyzed whether serum sodium and vWF have a positive association in humans, using data from the ARIC Study. ARIC is a study of cardiovascular disease in a cohort of 15,792 45- to 64-y-old persons sampled from four US communities in 1987–1989 (24). We used the results of the baseline clinical examination of the participants during their first visit.
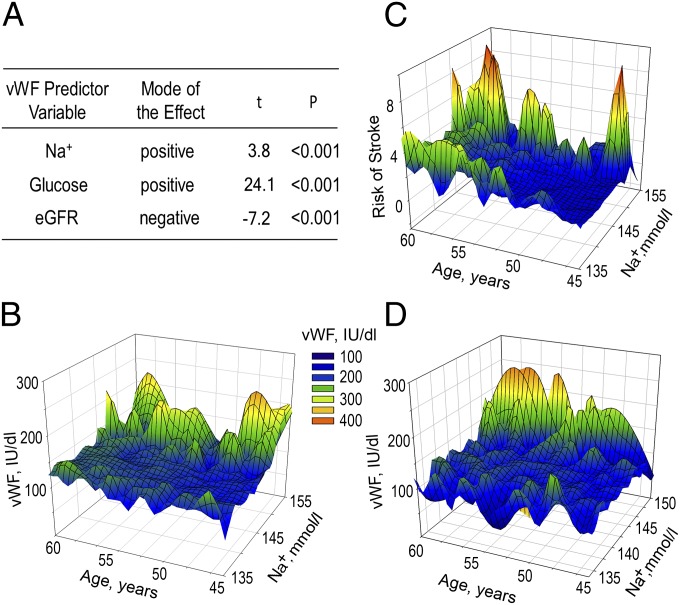
To evaluate effect of Na on vWF, we conducted multiple regression analysis with the following predictor variables: serum sodium, glucose, and estimated glomerular filtration rate (eGFR) (Fig. 6A). See SI Materials and Methods, Fig. S5, and Tables S2–S7 for the reasons those variables were selected, information about the basic descriptive statistics for the variables, zero-order correlations between the variables, and regression coefficients. The overall model is statistically significant [F(3, 14,675) = 210, P < 0.001] with all variables significantly contributing to predicting plasma level of vWF (P < 0.001) (Fig. 6A and Tables S4 and S5). The positive regression coefficient for serum Na+ indicates that the increase in Na+ is accompanied by increased vWF, consistent with overall findings of our study. Additionally, a 3D plot of blood vWF level vs. serum sodium concentration and age demonstrates that higher levels of vWF occur in participants with higher concentrations of serum sodium (Fig. 6B). The same is true for 10-y stroke risk (Fig. 6C). The stroke risk at first visit was retrospectively calculated for every participant of the ARIC Study based on the study outcomes and included in ARIC datasets (25). Multivariable regression analysis of 10-y stroke risk with sodium, glucose, and eGFR as predictor variables demonstrates that plasma sodium significantly contributes to prediction of stroke (Table S6). Diabetes and chronic kidney disease (CKD) lead to increased levels of VWF (26, 27). Consistently, there is significant zero-order correlation of plasma glucose and eGFR with level of vWF (Table S3). To assess whether a positive association of plasma sodium with vWF and risk of stroke is still present in participants without diabetes and CKD, we performed the analysis on a cohort of participants without diabetes and without CKD (eGFR >60 mL⋅min−1⋅1.73 m−2) who had normal weight [body mass index (BMI) = 18.5–25 kg/m2]. In this “healthy” cohort (n = 3,345), higher levels of vWF and risk of stroke occur also in participants who have higher concentrations of plasma sodium (Fig. 6D and Table S7). In summary, the analyses indicate that serum sodium is positively associated with the level of vWF and the risk of stroke in humans.
Fig. 6.
Plasma sodium is positively associated with blood level of vWF and 10-y risk of stroke in the ARIC Study. (A) Multiple linear regression analysis was used to assess effect of serum sodium on level of vWF. The results demonstrate that serum Na+ as well as glucose and eGFR significantly contribute to predicting the level of vWF. See SI Materials and Methods, Fig. S5, and Tables S2–S5 for details of the analysis. (B–D) Three-dimensional mesh plots, visualizing level of vWF and risk of stroke, as functions of serum sodium concentration and age. (B and D) Level of vWF is higher in participants with higher levels of serum sodium, both in B [all ARIC participants (n = 14,679)] and in D [cohort of 3,345 participants without diabetes who had eGFR >60 mL⋅min−1⋅1.73 m−2 and normal weight (BMI = 18.5–25 kg/m2)]. (C) Ten-year risk of stroke is increased in participants with higher level of serum sodium. All ARIC participants were included in the analysis. See also Tables S6 and S7.
Discussion
A model that describes the link between sodium, vWF, and thrombosis is shown on Fig. S1. In this study we have shown that elevation of extracellular NaCl stimulates production of vWF by vascular endothelial cells both in cell culture (HUVECs) and in mice in vivo. In HUVECs, secretion of vWF and activation of the osmoregulated transcription factor NFAT5 increase in parallel when NaCl is added to elevate osmolality of the medium over the range from 270 mosmol/kg, which is the lower end of the normal physiological range, to 380 mosmol/kg, which is the high end of severe hypernatremia. In mice, elevation of serum sodium by 5 mmol/L as the result of water restriction increases expression of NFAT5 and stimulates endothelial vWF production in several tissues. The mild dehydration increases vWF secretion enough to elevate vWF significantly in systemic blood and to cause thrombosis, as evidenced by the appearance of microthrombi and activation of fibrinolysis. Further, blood sodium and vWF are positively associated in humans.
Our findings identify the mechanism probably involved in the thrombosis seen in cases of hypernatremic dehydration (12–14), hyperosmolality associated with diabetes (15), and heatstroke (16). In clinical practice, infusion of hypertonic saline to elevate plasma sodium up to 155 mmol/L, and even higher, is routinely used in head injury, ischemic stroke, and intracerebral hemorrhage and is the subject of heated debate (28, 29). The results of our study indicate that elevation of plasma sodium during such therapy could increase blood coagulability, leading to thrombotic complications.
There are also broader implications of our study, as follows. Inadequate water intake increases plasma sodium concentration (21, 30), as does the high consumption of salt that is prevalent worldwide (31). Thus, 400 mL of soup containing 6 g of salt elevates plasma sodium for many hours by about 2 mmol/L (32), and increasing salt intake from 600 mg to 10 g per day for several days elevates plasma sodium by about 3 mmol/L (33). How much salt and water should be consumed, and how the consumption affects health, are issues that are the focus of intensive research and discussion (30, 34, 35). Perhaps monitoring the level of vWF or other coagulation markers could enhance investigation of these issues.
Subclinical dehydration may impair health because of increased vWF. Old age predisposes one to dehydration because of decreased thirst, impaired urinary concentrating ability, and increased insensible water loss (36, 37). Diabetes mellitus predisposes one to dehydration because of the osmotic diuresis caused by high urinary glucose. Further investigation is required to determine whether chronic subclinical dehydration in the elderly and in diabetics is the reason for elevated vWF and increased risk of thrombosis.
Do dehydration and hypernatremia cause hypercoagulability in humans? There are previous indications that they do. Namely, increased circulating vWF and risk of thrombosis were found to be associated with diabetes (26, 38), which predisposes one to dehydration; small clinical trials demonstrated association of platelet aggregation (39), plasma fibrinogen, D-dimer, and vWF (40) with high salt intake, and the 200-fold increase in deep vein thrombosis in shelters in Japan after the 2011 earthquake was attributed to dehydration (41).
Is health in humans likely to be adversely affected by hypercoagulability and increased vWF induced by subclinical dehydration? That seems likely, considering that hypercoagulability is linked to cardiovascular disease and atherosclerosis (42), and increased vWF is a risk factor for thromboembolism, myocardial infarction, and stroke (7, 26, 43).
Do other factors besides elevated NaCl contribute to the increased blood vWF and coagulability induced by water restriction? In addition to elevating blood sodium, water restriction increases antidiuretic hormone, and antidiuretic hormone increases vWF in blood (44). Infusion of the antidiuretic hormone analog desmopressin (DDAVP) is used clinically to treat bleeding disorders by elevating vWF (45). However, the increased production of vWF caused by high NaCl in HUVECs occurs independent of any increase of antidiuretic hormone (Fig. 1). In addition, the therapeutic use of DDAVP involves much greater antidiuretic hormone activity than the maximal level reached during dehydration (17, 45, 46). Furthermore, DDAVP produces only a transient increase of vWF in blood because it causes release of preexisting intracellular stores of vWF, whereas we observe a prolonged transcriptionally regulated increase in mice. Therefore, although the mild water restriction that we used undoubtedly elevated antidiuretic hormone, that increase does not explain the prolonged increase of vWF that we observe. Increased blood viscosity and hemoconcentration as a result of dehydration might also contribute to increased coagulability and thrombogenesis. However, our water restriction protocol does not cause acute weight loss (Fig. 3E) or increased plasma protein concentration (Fig. 3D), which speaks against these possibilities.
In summary, we find that high salt-induced secretion of vWF from endothelial cells causes hypercoagulability, which could induce thrombosis in conditions associated with hypernatremia, including dehydration, old age, diabetes, high dietary salt intake, and hyperosmotic therapy. We propose that hydration and salt intake are modifiable factors that affect coagulability and thrombogenesis.
Materials and Methods
HUVEC cells and growth medium were obtained from ATCC. Osmolality of this medium (control medium) was 270 mosmol/kg. High NaCl medium was prepared by adding NaCl to the total osmolality of 290–380 mosmol/kg. vWF was measured in HUVEC supernatants using the vWF Human ELISA Kit (Abcam). HUVECs protein extraction, Western blot, immunofluorescent staining, and ChIP were performed as described (47). Target sequences in ChIPed DNA were quantified by real-time PCR with SYBR-Green PCR Kit (Qiagen). RNA was extracted from HUVECs with RNeasy Mini Kit (Qiagen) and from mouse tissues using AllPrep DNA/RNA Mini Kit (QIAGEN). mRNA was quantified by quantitative RT-PCR using TaqMan gene expression assays (Life Technologies). All mouse studies were done under approval of the National Heart, Lung, and Blood Institute animal study protocols. Mice were water-restricted by feeding them with gel food containing 30% water. Blood was collected from the tails. Total plasma protein was measured using BCA Protein Assay (Pierce). vWF and D-dimer in plasma were measured by Western blot. PAI-1 in plasma was measured using Luminex immunoassay technique (48). PAI-1 agarose beads and assay reagents were obtained from EMD Millipore. Paraffin embedding of mouse tissues and immunohistochemical staining were performed as described (49). To quantify vWF protein expression on the diaminobenzidine-stained sections, we used the yellow-CMYK channel method (50). The Python script that performs yellow-CMYK channel extraction and quantification is available upon request. Further details of the study methods are provided in SI Materials and Methods.
Supplementary Material
Acknowledgments
We thank Daniil A. Kitchaev for writing Python script for CMYK yellow channel extraction from immunohistochemistry images, Daniela Malide and Christian Combs at the National Heart, Lung, and Blood Institute (NHLBI) Light Microscopy Core facility for help with microscopy, Leigh Samsel at NHLBI Flow Cytometry Core Facility for help with Luminex analysis, and Eric S. Leifer at the NHLBI Office of Biostatistics Research for expert advice on the clinical data analysis. This manuscript was prepared using Atherosclerosis Risk in Communities research materials obtained from the NHLBI Biologic Specimen and Data Repository Information Coordinating Center. The study was supported by the Intramural Program of NHLBI.
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1404809111/-/DCSupplemental.
References
- 1.Goldhaber SZ, Bounameaux H. Pulmonary embolism and deep vein thrombosis. Lancet. 2012;379(9828):1835–1846. doi: 10.1016/S0140-6736(11)61904-1. [DOI] [PubMed] [Google Scholar]
- 2.Roger VL, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee Heart disease and stroke statistics—2011 update: A report from the American Heart Association. Circulation. 2011;123(4):e18–e209. doi: 10.1161/CIR.0b013e3182009701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mackman N. Triggers, targets and treatments for thrombosis. Nature. 2008;451(7181):914–918. doi: 10.1038/nature06797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Engelmann B, Massberg S. Thrombosis as an intravascular effector of innate immunity. Nat Rev Immunol. 2013;13(1):34–45. doi: 10.1038/nri3345. [DOI] [PubMed] [Google Scholar]
- 5.Reininger AJ. Function of von Willebrand factor in haemostasis and thrombosis. Haemophilia. 2008;14(Suppl 5):11–26. doi: 10.1111/j.1365-2516.2008.01848.x. [DOI] [PubMed] [Google Scholar]
- 6.Michaux G, Cutler DF. How to roll an endothelial cigar: The biogenesis of Weibel-Palade bodies. Traffic. 2004;5(2):69–78. doi: 10.1111/j.1600-0854.2004.00157.x. [DOI] [PubMed] [Google Scholar]
- 7.Sadler JE. New concepts in von Willebrand disease. Annu Rev Med. 2005;56:173–191. doi: 10.1146/annurev.med.56.082103.104713. [DOI] [PubMed] [Google Scholar]
- 8.Chen JM, López JA. Interactions of platelets with subendothelium and endothelium. Microcirculation. 2005;12(3):235–246. doi: 10.1080/10739680590925484. [DOI] [PubMed] [Google Scholar]
- 9.Denis CV, Lenting PJ. von Willebrand factor: At the crossroads of bleeding and thrombosis. Int J Hematol. 2012;95(4):353–361. doi: 10.1007/s12185-012-1041-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Versteeg HH, Heemskerk JWM, Levi M, Reitsma PH. New fundamentals in hemostasis. Physiol Rev. 2013;93(1):327–358. doi: 10.1152/physrev.00016.2011. [DOI] [PubMed] [Google Scholar]
- 11.Adrogué HJ, Madias NE. Hypernatremia. N Engl J Med. 2000;342(20):1493–1499. doi: 10.1056/NEJM200005183422006. [DOI] [PubMed] [Google Scholar]
- 12.van Amerongen RH, Moretta AC, Gaeta TJ. Severe hypernatremic dehydration and death in a breast-fed infant. Pediatr Emerg Care. 2001;17(3):175–180. doi: 10.1097/00006565-200106000-00006. [DOI] [PubMed] [Google Scholar]
- 13.Hbibi M, et al. Severe hypernatremic dehydration associated with cerebral venous and aortic thrombosis in the neonatal period. BMJ Case Rep. 2012 doi: 10.1136/bcr.07.2011.4426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shroff R, Hignett R, Pierce C, Marks S, van’t Hoff W. Life-threatening hypernatraemic dehydration in breastfed babies. Arch Dis Child. 2006;91(12):1025–1026. doi: 10.1136/adc.2006.095497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Keenan CR, Murin S, White RH. High risk for venous thromboembolism in diabetics with hyperosmolar state: Comparison with other acute medical illnesses. J Thromb Haemost. 2007;5(6):1185–1190. doi: 10.1111/j.1538-7836.2007.02553.x. [DOI] [PubMed] [Google Scholar]
- 16.Bouchama A, et al. Tissue factor/factor VIIa pathway mediates coagulation activation in induced-heat stroke in the baboon. Crit Care Med. 2012;40(4):1229–1236. doi: 10.1097/CCM.0b013e3182387bef. [DOI] [PubMed] [Google Scholar]
- 17.Grant PJ, Tate GM, Hughes JR, Davies JA, Prentice CR. Does hypernatraemia promote thrombosis? Thromb Res. 1985;40(3):393–399. doi: 10.1016/0049-3848(85)90274-9. [DOI] [PubMed] [Google Scholar]
- 18.Brill A, et al. von Willebrand factor-mediated platelet adhesion is critical for deep vein thrombosis in mouse models. Blood. 2011;117(4):1400–1407. doi: 10.1182/blood-2010-05-287623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Burg MB, Ferraris JD, Dmitrieva NI. Cellular response to hyperosmotic stresses. Physiol Rev. 2007;87(4):1441–1474. doi: 10.1152/physrev.00056.2006. [DOI] [PubMed] [Google Scholar]
- 20.Ferraris JD, Williams CK, Ohtaka A, García-Pérez A. Functional consensus for mammalian osmotic response elements. Am J Physiol. 1999;276(3 Pt 1):C667–C673. doi: 10.1152/ajpcell.1999.276.3.C667. [DOI] [PubMed] [Google Scholar]
- 21.Senay LC, Christensen ML. Changes in blood plasma during progressive dehydration. J Appl Physiol. 1965;20(6):1136. [Google Scholar]
- 22.Cheuvront SN, Kenefick RW, Charkoudian N, Sawka MN. Physiologic basis for understanding quantitative dehydration assessment. Am J Clin Nutr. 2013;97(3):455–462. doi: 10.3945/ajcn.112.044172. [DOI] [PubMed] [Google Scholar]
- 23.Adam SS, Key NS, Greenberg CS. D-dimer antigen: Current concepts and future prospects. Blood. 2009;113(13):2878–2887. doi: 10.1182/blood-2008-06-165845. [DOI] [PubMed] [Google Scholar]
- 24.Williams OD. The Atherosclerosis Risk in Communities (ARIC) Study: Design and objectives. The ARIC investigators. Am J Epidemiol. 1989;129(4):687–702. [PubMed] [Google Scholar]
- 25.Chambless LE, Heiss G, Shahar E, Earp MJ, Toole J. Prediction of ischemic stroke risk in the Atherosclerosis Risk in Communities Study. Am J Epidemiol. 2004;160(3):259–269. doi: 10.1093/aje/kwh189. [DOI] [PubMed] [Google Scholar]
- 26.Blann AD. Plasma von Willebrand factor, thrombosis, and the endothelium: The first 30 years. Thromb Haemost. 2006;95(1):49–55. [PubMed] [Google Scholar]
- 27.Shen L, et al. Von Willebrand factor, ADAMTS13 activity, TNF-α and their relationships in patients with chronic kidney disease. Exp Ther Med. 2012;3(3):530–534. doi: 10.3892/etm.2011.432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Diringer MN. New trends in hyperosmolar therapy? Curr Opin Crit Care. 2013;19(2):77–82. doi: 10.1097/MCC.0b013e32835eba30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Grände PO, Romner B. Osmotherapy in brain edema: A questionable therapy. J Neurosurg Anesthesiol. 2012;24(4):407–412. doi: 10.1097/01.ana.0000419730.29492.8b. [DOI] [PubMed] [Google Scholar]
- 30.Armstrong LE. Challenges of linking chronic dehydration and fluid consumption to health outcomes. Nutr Rev. 2012;70(Suppl 2):S121–S127. doi: 10.1111/j.1753-4887.2012.00539.x. [DOI] [PubMed] [Google Scholar]
- 31.Brown IJ, Tzoulaki I, Candeias V, Elliott P. Salt intakes around the world: implications for public health. Int J Epidemiol. 2009;38(3):791–813. doi: 10.1093/ije/dyp139. [DOI] [PubMed] [Google Scholar]
- 32.Suckling RJ, He FJ, Markandu ND, MacGregor GA. Dietary salt influences postprandial plasma sodium concentration and systolic blood pressure. Kidney Int. 2012;81(4):407–411. doi: 10.1038/ki.2011.369. [DOI] [PubMed] [Google Scholar]
- 33.He FJ, Markandu ND, Sagnella GA, de Wardener HE, MacGregor GA. Plasma sodium: Ignored and underestimated. Hypertension. 2005;45(1):98–102. doi: 10.1161/01.HYP.0000149431.79450.a2. [DOI] [PubMed] [Google Scholar]
- 34.He FJ, Macgregor GA. Salt intake, plasma sodium, and worldwide salt reduction. Ann Med. 2012;44(Suppl 1):S127–S137. doi: 10.3109/07853890.2012.660495. [DOI] [PubMed] [Google Scholar]
- 35.Valtin H. “Drink at least eight glasses of water a day.” Really? Is there scientific evidence for “8 x 8”? Am J Physiol Regul Integr Comp Physiol. 2002;283(5):R993–R1004. doi: 10.1152/ajpregu.00365.2002. [DOI] [PubMed] [Google Scholar]
- 36.Cowen LE, Hodak SP, Verbalis JG. Age-associated abnormalities of water homeostasis. Endocrinol Metab Clin North Am. 2013;42(2):349–370. doi: 10.1016/j.ecl.2013.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dmitrieva NI, Burg MB. Increased insensible water loss contributes to aging related dehydration. PLoS ONE. 2011;6(5):e20691. doi: 10.1371/journal.pone.0020691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Carr ME. Diabetes mellitus: a hypercoagulable state. J Diabetes Complications. 2001;15(1):44–54. doi: 10.1016/s1056-8727(00)00132-x. [DOI] [PubMed] [Google Scholar]
- 39.Nara Y, et al. Dietary effect on platelet aggregation in men with and without a family history of essential hypertension. Hypertension. 1984;6(3):339–343. doi: 10.1161/01.hyp.6.3.339. [DOI] [PubMed] [Google Scholar]
- 40.Liu F, et al. Potassium supplement ameliorates salt-induced haemostatic abnormalities in normotensive subjects. Acta Cardiol. 2011;66(5):635–639. doi: 10.1080/ac.66.5.2131090. [DOI] [PubMed] [Google Scholar]
- 41.Ueda S, Hanzawa K, Shibata M, Suzuki S. High prevalence of deep vein thrombosis in tsunami-flooded shelters established after the great East-Japan earthquake. Tohoku J Exp Med. 2012;227(3):199–202. doi: 10.1620/tjem.227.199. [DOI] [PubMed] [Google Scholar]
- 42.Loeffen R, Spronk HMH, ten Cate H. The impact of blood coagulability on atherosclerosis and cardiovascular disease. J Thromb Haemost. 2012;10(7):1207–1216. doi: 10.1111/j.1538-7836.2012.04782.x. [DOI] [PubMed] [Google Scholar]
- 43.Wiman B, et al. Plasma levels of tissue plasminogen activator/plasminogen activator inhibitor-1 complex and von Willebrand factor are significant risk markers for recurrent myocardial infarction in the Stockholm Heart Epidemiology Program (SHEEP) study. Arterioscler Thromb Vasc Biol. 2000;20(8):2019–2023. doi: 10.1161/01.atv.20.8.2019. [DOI] [PubMed] [Google Scholar]
- 44.Nussey SS, Bevan DH, Ang VTY, Jenkins JS. Effects of arginine vasopressin (AVP) infusions on circulating concentrations of platelet AVP, factor VIII: C and von Willebrand factor. Thromb Haemost. 1986;55(1):34–36. [PubMed] [Google Scholar]
- 45.Mannucci PM. Desmopressin (DDAVP) in the treatment of bleeding disorders: the first 20 years. Blood. 1997;90(7):2515–2521. [PubMed] [Google Scholar]
- 46.Grant PJ, Davies JA, Tate GM, Boothby M, Prentice CR. Effects of physiological concentrations of vasopressin on haemostatic function in man. Clin Sci (Lond) 1985;69(4):471–476. doi: 10.1042/cs0690471. [DOI] [PubMed] [Google Scholar]
- 47.Dmitrieva NI, Cui K, Kitchaev DA, Zhao K, Burg MB. DNA double-strand breaks induced by high NaCl occur predominantly in gene deserts. Proc Natl Acad Sci USA. 2011;108(51):20796–20801. doi: 10.1073/pnas.1114677108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Khan SS, Smith MS, Reda D, Suffredini AF, McCoy JP., Jr Multiplex bead array assays for detection of soluble cytokines: Comparisons of sensitivity and quantitative values among kits from multiple manufacturers. Cytometry B Clin Cytom. 2004;61(1):35–39. doi: 10.1002/cyto.b.20021. [DOI] [PubMed] [Google Scholar]
- 49.Dmitrieva NI, Burg MB. High NaCl promotes cellular senescence. Cell Cycle. 2007;6(24):3108–3113. doi: 10.4161/cc.6.24.5084. [DOI] [PubMed] [Google Scholar]
- 50.Pham N-A, et al. Quantitative image analysis of immunohistochemical stains using a CMYK color model. Diagn Pathol. 2007;2:8. doi: 10.1186/1746-1596-2-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.