Abstract
Psychostimulants like amphetamine and methylphenidate (MPD) are used to treat attention deficit hyperactivity disorder (ADHD), which is marked by developmentally inappropriate inattention, hyperactivity, and impulsivity. Neuropsychological analyses indicate that ADHD patients are impaired on tasks of behavioral inhibition, reward reversal, and working memory, which are functions of the prefrontal cortex (PFC) and are modulated by the mesocortical dopamine (DA) system. Non-specific electrical lesioning of PFC eliminated the expression of behavioral sensitization elicited by chronic MPD administration. Behavioral sensitization is the progressive augmentation of locomotor activity as a result of repetitive (chronic) exposure to the drug. It is believed that the sensitization to chronic drug treatment is caused due to an increase in DA in the mesocorticolimbic DA system, which includes the PFC. Therefore, this study investigated the role of PFC DA in mediating the behavioral sensitization to repeated administration of MPD in adult male Sprague Dawley rats. On experimental day (ED) 1, the behavior was recorded post a saline injection. On ED 2, the rats were divided into three groups – control, sham and bilateral 6-OHDA treated group, and the sham and 6-OHDA treated groups underwent respective surgeries. After five days of rest following surgery, the post-surgery baseline was recorded on ED 8 following a saline injection. All three groups received 2.5 mg/kg MPD for 6 days (from ED 9 to ED 14), followed by a three day washout period (ED 15 to ED 18). On ED 19, a rechallenge injection of 2.5 mg/kg MPD was given and locomotor activity was recorded. It was found that the 6-OHDA lesion group failed to exhibit behavioral sensitization to MPD. The involvement of the dopaminergic afferents of PFC in behavioral sensitization to MPD is discussed.
1. Introduction
Psychostimulants like amphetamine and methylphenidate (MPD) are used to treat attention deficit hyperactivity disorder (ADHD), which is marked by developmentally inappropriate inattention, hyperactivity, and impulsivity (Askenasy et. al., 2007; Elia et. al., 1999), and affects 5 - 15% of school going children in US (Anderson et. al., 1987; Barabesi et. al., 2002; Yang et. al., 2007). The pathophysiology of ADHD is often linked to dysfunction of the prefrontal cortex (PFC) (Arnsten et al., 1996; Sullivan and Brake, 2003; Yang et. al., 2007). Neuropsychological analyses indicate that impairments of ADHD patients are similar to those with prefrontal lesions, for example, problems with tasks of behavioral inhibition, reward reversal, and working memory (Bedard et al, 2003; Itami and Uno, 2002). The mesocortical dopamine (DA) system modulates these functions in the PFC (Sullivan and Brake, 2003) and ADHD patients are known to have dopaminergic aberrations in PFC (Arnsten et al., 1996; Yang et. al., 2007). It has also been suggested that the most important site for treating ADHD symptoms is to target the PFC with treatment (Sullivan and Brake, 2003). Treatment of ADHD with MPD has been linked to increased catecholaminergic transmission within the PFC compared to subcortical structures (Berridge et. al., 2006). MPD, when given systemically, results in the release of catecholamines and inhibition of their re-uptake, primarily DA (Patrick et. al., 1997; Roman et. al., 2001). As MPD does not stimulate the catecholamine receptors directly but rather facilitates the action of DA by binding to the DA transporter, it is considered as an indirect DA agonist (Ding et. al., 1994; Volkow et. al., 1999) that strengthens the DAergic transmission in the PFC (Grund et. al., 2006).
Electrical lesioning of PFC eliminated the behavioral sensitization elicited by chronic MPD administration (Lee et. al., 2008). Behavioral sensitization is also known as “reverse tolerance” i.e., progressively augmenting activity as a result of repetitive (chronic) exposure to the drug (Chao and Nestler, 2004; Gaytan et. al., 1997; Kalivas and Stewart, 1991; Lee et. al., 2008; Robinson, 1984; Robinson & Berridge, 1993; Yang et. al., 2003, 2007). Previous research suggests that the sensitization to chronic drug treatment is caused due to an increase in DA in the mesocorticolimbic DA system, which includes the PFC. (Beyer and Steketee, 1999; Kalivas and Stewart, 1991; Kalivas et. al., 1993).
Dopaminergic projections to PFC from the ventral tegmental area (VTA) [an area involved in the induction of behavioral sensitization (Pierce and Kalivas, 1997)] are recognized to have a role in motor attention (Krause et. al., 2003; Grillner and Mercuri, 2002). The PFC is also postulated to be able to regulate subcortical DA systems: administering dopamine receptor agonists into PFC causes decreases in DA transmission in Nucleus Accumbens (NAc) while injections of DA antagonists into PFC result in an increase of DA in NAc (Louilot et al., 1989). Similarly, elimination of DA from the PFC, through 6-OHDA injections in PFC, results in an increase of DA in NAc (Leccese and Lyness, 1987; Pycock et. al., 1980). The NAc is considered to be the site involved in the expression of behavioral sensitization following chronic psychostimulant administration (Dafny and Yang, 2006, Pierce and Kalivas, 1997; Wolf, 1998; Yang et. al., 2007). Therefore, dopaminergic afferents of PFC might be involved in the induction or expression of behavioral sensitization to MPD.
It was reported that dopaminergic afferents of PFC play a role in behavioral sensitization to amphetamine (Bjijou et. al. 2002), which shares chemical and pharmacological properties with MPD (Askenasy et. al., 2007; Kollins et. al., 2001). Bjijou et. al. (2002) reported that bilateral 6-hydroxydopamine (6-OHDA) injections into PFC, which selectively destroy dopaminergic afferents (Bjijou et al., 2002; Beyer and Steketee, 1999; Hanrott et. al., 2006), prevented the development of sensitization to repetitive amphetamine treatment. The objective of this study was to investigate the role of PFC DA in mediating behavioral sensitization to repeated administration of MPD in male adult Sprague Dawley (SD) rats. Previous MPD dose-response studies (Gaytan et. al., 1997; Yang et. al., 2007) have shown that 2.5mg/kg MPD elicits behavioral sensitization in SD rats. The experimental protocol consisted of pre and post surgery baselines, followed by six days of daily MPD injections, four days of washout period, and rechallenge injection of 2.5 mg/kg MPD. Response of the 6-OHDA lesion group on the rechallenge day was compared to that of the sham and control group to look at the effect of 6-OHDA lesion on behavioral sensitization to MPD.
2. Results
Effect of surgery on locomotor activity
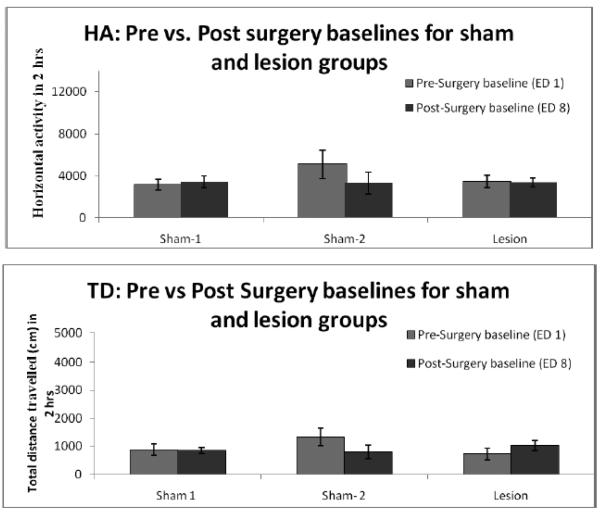
Baseline locomotor activity was recorded on ED 1 (pre-surgery baseline) and ED 8 (post-surgery baseline) for all the groups. Fig. 1 shows the baseline activities on ED 1of horizontal activity (HA), and total distance (TD) travelled for the two sham groups and the lesion group compared to ED 8. On ED 8, there was a slight decrease in activity in sham-2 group, but this decrease was found to be non-significant. Since the activities of the two sham groups were similar throughout the experimental days, henceforth their data is combined and presented as a single sham group. Bilateral 6-OHDA application in PFC did not result in any change in baseline HA and TD (Fig. 1). This data shows that the surgery and 6-OHDA administrations in the PFC did not have any significant effect on baseline activity.
Figure 1.
compares the pre and post surgery baseline for the lesion as well as both the sham groups. It was observed that the surgeries did not alter the baseline of any group. # - denotes significance (p < 0.05).
Acute effect of MPD administration (Comparing ED 9, the first day of MPD administration to ED 8, baseline after saline injection – Fig. 2)
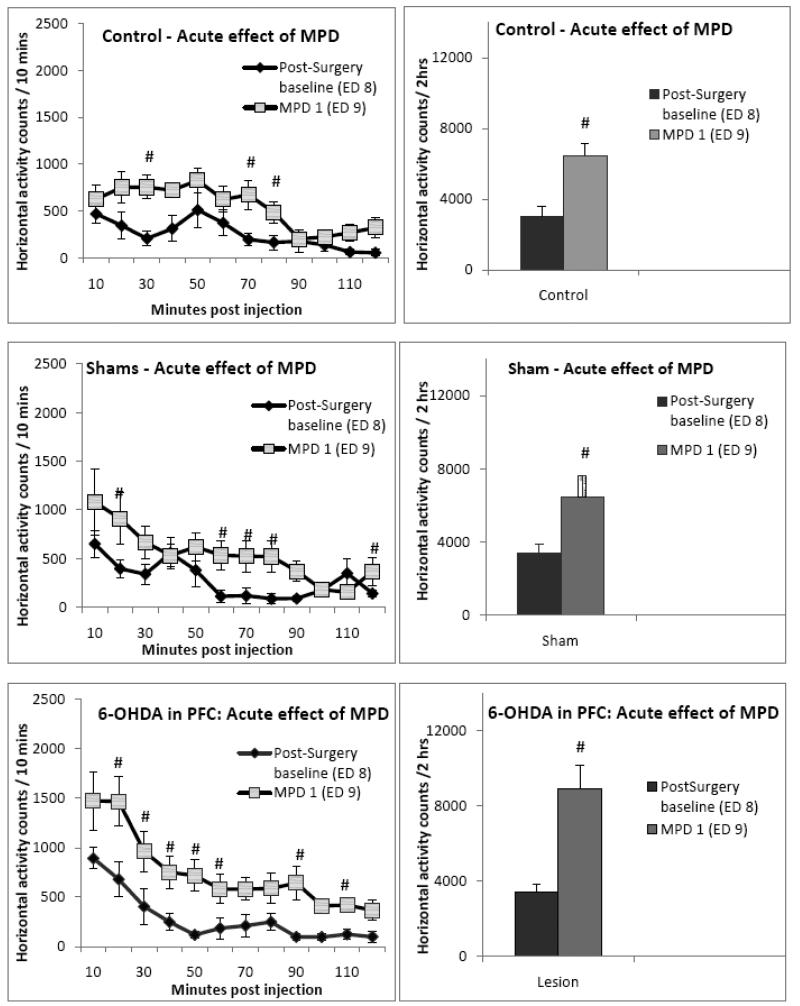
Figure 2.
contrasts the locomotor activity in all three groups recorded after the first injection of MPD (ED 9) with the post-surgery baseline (ED 8). The figures on the left indicate temporal graphs of the locomotor activity, while the histograms on the right indicate the sum of total horizontal activity in 2 hours post injection. MPD caused a significant increase in locomotor activity in all three groups. # - denotes significance (p < 0.05).
Figure 2 shows the sequential 10 min HA in the temporal graphs and the 2 hour histograms summarizing the activity under the temporal graph for control, sham and 6-OHDA in PFC groups and displays the acute effect of 2.5 mg/kg MPD. It was observed that acute injection of 2.5mg/kg MPD (i.p.) caused a significant (p < 0.05) increase in HA (Fig. 2) and in all locomotor indices analyzed of all the animal groups, and the group injected with 6-OHDA in PFC exhibited augmented increase in locomotion that is expressed in greater number of data points in the temporal graph.
Effect of repetitive MPD – The induction phase (comparing ED 14 to ED 9)
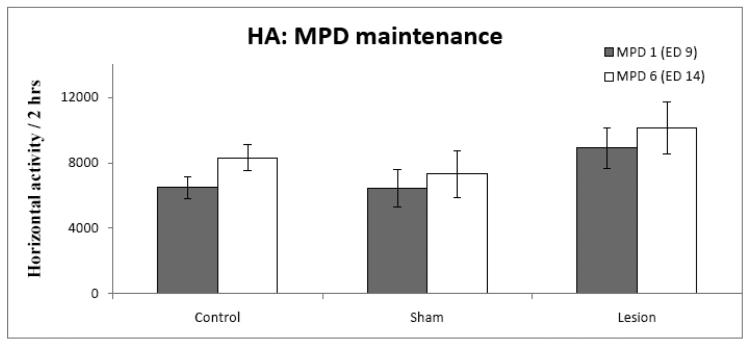
Figure 3 compares the locomotor activity after six daily injections of MPD (ED 14) to the initial MPD administration (ED 9). The locomotor activity on ED 14 exhibits some increase in activity compared to the locomotor activity observed after the administration of the same dose given on ED 9. However, this increase in activity was not found to be statistically significant for any group in any of the locomotor indices.
Figure 3.
indicates that the increase in locomotion observed after the first injection of MPD (ED 9) was maintained following the sixth consecutive injection of MPD (ED 14) in all three groups.
Locomotor activity during the washout phase (comparing ED's 15 and 18 to ED 8)
Figure 4 summarizes the HA sequential 10 minute activity in the temporal graphs and the two hour summary of the area under the temporal graphs in the histograms and compares the activity of the first and last washout days after six daily injections of 2.5 mg/kg MPD (ED 15, the first day of washout and ED 18, the last day of washout – Table 1) with that of pre MPD baseline injection (ED 8). It was found that the intact control group exhibited significantly elevated locomotor activity on both the first and last washout day at the time of previous recordings post MPD injections (06:45 – 08:45). The sham group also exhibited significantly increased locomotion on the first washout day (ED 15) but did not exhibit augmented activity on the last day of washout (ED 18). The 6-OHDA lesion group exhibited similar locomotor activity on all washout days as compared to the post surgery baseline (Fig. 4).
Figure 4.
analyzes the locomotor activity during the washout period as compared to the post-surgery baseline. In the temporal graphs on the left, ^ denotes activity on ED 15 was significantly different than activity at that time point on ED 8. * denotes activity on ED 18 was significantly different than activity at that time point on ED 8. The two hour histograms indicate that the control group exhibited significantly increased locomotion on both the first day of the washout, while the sham group exhibited significantly elevated activity on only the first and not the last day of washout. The lesion group exhibited baseline locomotor activity on both the first as well as last day of washout. # - denotes significance (p < 0.05).
Table 1.
Drug administration schedule:
| Group | ED 1 | ED 2 | EDs 3-7 | ED 8 | EDs 9 – 14 | EDs 15– 18 | ED 19 |
|---|---|---|---|---|---|---|---|
| Control | Saline | - | - | Saline | 2.5mg/kg | No treatment | 2.5mg/kg |
| MPD | MPD | ||||||
| Shams | Saline | Surgery | Recovery | Saline | 2.5mg/kg MPD |
No treatment | 2.5mg/kg MPD |
|
6-OHDA Lesion |
Saline | Surgery | Recovery | Saline | 2.5mg/kg MPD |
No treatment | 2.5mg/kg MPD |
Table 1 describes the experimental protocol for all three experimental groups. The animals received a saline injection on experimental days (ED) 1 and 8 to determine their pre and post-surgery baseline. The shams and lesion groups underwent surgery on ED2, and were given five days of rest. All three groups received daily intraperitoneal (i.p.) injections of 2.5mg/kg MPD on ED-9 to ED-14. Following the chronic MPD treatment, the animals were allowed a three day washout period (ED 15 – 18). The animals were then rechallenged with 2.5mg/Kg MPD on the last day.
Effect of rechallenge MPD injection – The Expression phase (comparing ED 19 to ED 9)
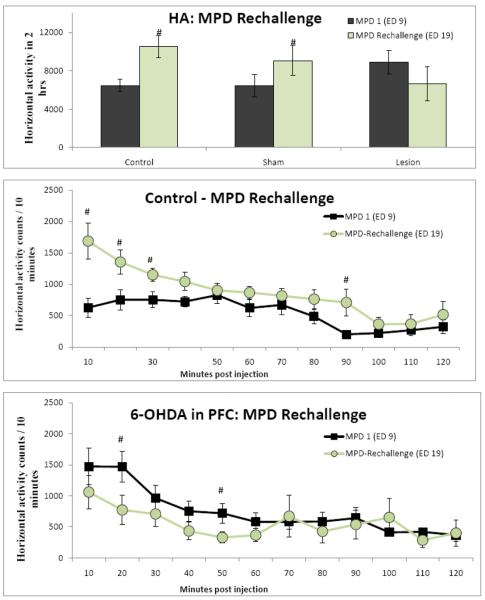
Figure 5 summarizes and compares the locomotor activity following the rechallenge injection of MPD (ED 19) with the first MPD injection (ED 9). Rechallenge treatment of MPD on ED 19 after six consecutive daily treatments of MPD and three days of washout shows that the locomotor activity of control and sham groups exhibited significant (p < 0.05) increases in locomotion on ED 19 as compared to the locomotor activity observed on ED 9, following the 1st MPD injection. The 6-OHDA lesion group failed to exhibit this increase in locomotion following MPD administration on ED 19 (Fig. 5). The increase in locomotor activity following MPD administration on ED 19 compared to ED 9 can be interpreted that behavioral sensitization was expressed. However, the PFC bilateral lesions with 6-OHDA group failed to exhibit this behavior in all the locomotor indices analyzed (Fig. 5).
Figure 5.
examines the locomotor activity following the MPD rechallenge injection (ED 19) as compared to the locomotor activity following the first injection of MPD (ED 9). It was noted that while control and sham group showed a significantly elevated activity following the rechallenge injection (sensitization), while the lesion group did not have a sensitized response. # - denotes significance (p < 0.05). In the temporal graph for the control group, it was observed that average 10 minute activity of the rechallenge injection was significantly higher than that of the acute injection for the first 30 minutes. In the 6-OHDA lesion in PFC group, this trend was reversed and the average 10 minute activity of the rechallenge injection was significantly lower than that of the acute injection at two time points.
3. Discussion
The role of PFC in behavioral sensitization to psychostimulants like amphetamine and cocaine has been investigated with selective neurotoxins like 6-OHDA and ibotenic acid, which elicit specific destruction of the PFC neurophyl (Cador et. al., 1999; Bjijou et al., 2002; Beyer and Steketee, 1999; Wanchoo et. al., 2009). Recently, it was reported that non-specific electric lesioning of the PFC prevents the expression of MPD induced behavioral sensitization (Lee et. al., 2008). The objective of this study was to use selective lesioning of the PFC DA neurophyl by bilateral 6-OHDA injection to the PFC. This study found that destroying DA afferents of PFC did not alter the acute effect of MPD while modulating the chronic effect of MPD by preventing the expression of behavioral sensitization to MPD, which suggests that DA afferents of PFC are involved in the neural circuitry underlying behavioral sensitization to MPD. Understanding this neural circuitry would be an essential step to understand the mechanism of MPD action. Additionally, it is essential to study the neural circuitry underlying behavioral sensitization since it is thought to be similar to that underlying compulsive drug addiction and drug craving (Dafny and Yang, 2006; Robinson, 1984; Robinson & Berridge, 1993; Wolf, 1998).
There are contradictory reports about the effects of 6-OHDA lesions in PFC on spontaneous locomotor activity (post surgery baseline) (Bjijou et. al., 2002; Joyce et. al., 1983; Clarke et. al., 1988; Carter and Pycock, 1980; Beyer and Steketee, 1999; Robinson and Stitt, 1981). Our data indicated that 6-OHDA lesion in PFC did not change the baseline locomotor activity, similar to other reports (Bjijou et. al., 2002; Joyce et. al., 1983; Clarke et. al., 1988; Carter and Pycock, 1980). However, decreased as well as increased post-surgery baseline locomotor activity has also been reported (Beyer and Steketee, 1999; Carter and Pycock, 1980; Robinson and Stitt, 1981). Carter and Pycock (1980) reported that different observations were noted depending upon whether or not NE uptake blocker was administered along with 6-OHDA. For example, only 6-OHDA injections to the PFC had no significant change on spontaneous activity, while 6-OHDA injections with NE uptake blocker resulted in significant enhancement of spontaneous motor activity (Carter and Pycock (1980)). However, Bjijou et. al. (2002) did not report changes in post-surgery baseline following 6-OHDA injection into PFC with NE uptake blocker. The reasons for these differences are not very clear but might be due to extent and location of the lesions produced by 6-OHDA and the time allowed between the 6-OHDA administrations and the subsequent recording of locomotor activity.
Greater increase in MPD induced hyper locomotor activity was observed in the 6-OHDA lesion group. Similar observations of significantly increased locomotion in 6-OHDA lesioned PFC group were reported following cocaine administration (Beyer and Steketee, 1999) i.e., the selective DA PFC neurophyl lesion by bilateral 6-OHDA injection resulted in higher behavioral responses elicited by cocaine administration at all doses tested and had a significantly different response than saline injection for a lower dose than the sham group. Therefore, it seems likely that MPD would also elicit a significant response in the lesion group as compared to sham and control groups at a lower dose. Contradictory findings are reported about the effect of 6-OHDA lesion in PFC on amphetamine-induced locomotion (Joyce et al., 1983; Clarke et al., 1988; Oades et al., 1986; Carter and Pyckok, 1980; Pycock et al., 1980; Bubser and Schmidt, 1990). As noted by Bjijou et. al. (2002), these discrepancies about the effect of 6-OHDA lesion in PFC on amphetamine-induced locomotion have been attributed to differences in stress levels of the animal (caused due to different level of illumination of the room), time elapsed since the lesion or by the level of DA depletion in the PFC after 6-OHDA lesion.
During the three days of washout when no injections were given, the intact MPD control group exhibited significantly higher locomotion at the time of previous MPD injections. Previous studies using similar experimental protocol of saline and MPD treatment have demonstrated that groups receiving injections of saline only do not exhibit an increase in locomotor activity during the washout period (Algahim et. al., 2009; Gaytan et. al., 2000a; Lee et. al., 2009), while the groups that received MPD exhibited increased locomotor activity, similar to the increase seen during washout days in this study. Therefore, the increase in locomotor activity in the control group during the washout days may be interpreted as anticipation to get the drug or withdrawal effects of the drug (Algahim et. al., 2009; Lee et. al., 2009). Alternatively, these results may be added to the body of literature that displays the presence of classical conditioning between psychostimulants and the environments in which they received the psychostimulants. For example, increased locomotor activity has been shown in drug-free rats in environments previously associated with psychostimulant administrations like amphetamine (Ahmed et.al., 1996; Pickens and Crowder, 1967; Tilson and Rech, 1973; Badiani et al. 1995) and cocaine (Adams et. al., 2000; Hinson and Poulos, 1981; Post et al., 1981; Pert, 1994). The sham group treated previously with MPD on the six preceding days exhibited higher locomotor activity only on the first washout day but not the last day of washout. It is possible that the stress of the surgery affected the long-term memory (Chen et. al., 2008) of the animal in some way such that the animal anticipated the drug on the first day of washout but not on the last day. The 6-OHDA lesion in PFC group exhibited similar activity to the baseline before drug treatment (ED 8) and the lack of anticipation might be attributed to the stress associated with surgery. Alternatively, as the role of DA receptors in PFC has been established for long-term memory (Nagai et. al. 2007), the lack of anticipation by the lesion group may be explained due to lack of DA in PFC.
Repeated administrations of MPD lead to behavioral sensitization in control and sham groups, similar to previous studies (Askenasy et. al., 2007; Gaytan et. al., 1997; Lee et. al., 2008, 2009; Yang et. al., 2006, 2007). However, expression of behavioral sensitization was not observed in number of stereotypy (NOS). This is in agreement with previous experiments that indicate that number of stereotypy movement at this MPD dose was not augmented, i.e., sensitized (Gaytan et. al., 1997). This observation suggests that HA and NOS are regulated by different neural circuits. The PFC 6-OHDA lesioned group failed to show behavioral sensitization in HA and TD. It is possible that this loss of behavioral sensitization was due to the loss of DA afferents of PFC. This observation with MPD is similar to that reported for amphetamine (Bjijou et. al., 2002) suggesting that chronic psychostimulant administration cause neuroadaptations at the levels of DA terminals in PFC that might account for behavioral sensitization. Since the DA afferents of PFC arise from VTA, it is possible that the neuroadaptations are at the level of the VTA (Bjijou et. al., 2002). Nevertheless, this experiment confirms that PFC dopaminergic afferents are critical to neural circuitry underlying behavioral sensitization. Thus, the role of PFC dopaminergic afferents in the expression of MPD sensitization is similar to that described for amphetamine elicited behavioral sensitization by Bjijou et. al. (2002).
Alternatively, these results may be explained by the impulsivity theory of Jentsch and Taylor (1999) in the light of the findings by van Gaalen et. al. (2006). According to Jentsch and Taylor (1999), drug seeking behavior can been theorized to be loss of inhibitory control over behavior by internal motivational states, also known as “impulses”, in addition to the increased salience of the rewarding properties of the drug. They hypothesized that repeated administrations of drugs of abuse cause cortical dopaminergic hypofunction which corresponds to faulty modulation of subcortical dopamine systems and the subsequent loss of cortical control on behavior elicited by reward-related cues (Jentsch and Taylor, 1999). While this hypothesis fit neural circuitry underlying cocaine sensitization (Beyer and Steketee, 1999), this hypothesis does not work for amphetamine (Bjijou et. al., 2002) or MPD (our results). These differences might just be due to pharmacological differences in mechanisms of actions of these drugs. However, contrary to Jentsch and Taylor's hypothesis about dopaminergic hypofunction, van Gaalen et. al. (2006) reported that behavioral disinhibition, which is often an effect of drugs of abuse and is considered a manifestation of impulsivity, results from increased dopaminergic transmission. But since the drugs modifying dopaminergic transmission were administered systemically in the van Gaalen et. al. (2006) study, the exact cerebral loci of DA action is not specified. Yet, the authors implicate that the PFC is generally involved in inhibitory control of behavior based on previous reports (Miller and Cohen, 2001; Dalley et. al., 2004). In the light of these results (van Gaalen et. al., 2006), the theory of Jentsch and Taylor (1999) can be interpreted as follows: augmented dopaminergic neurotransmission in the PFC, caused due to repeated administrations of drugs of abuse, results in behavioral disinhibition (impulsivity), which favors drug-seeking behavior. Thus, sensitization – the animal co-relate of drug seeking behavior - may be correlated to increased dopaminergic neurotransmission in the PFC, and then not surprisingly, the loss of DA in PFC corresponds to the loss of sensitization in the 6-OHDA treated group. However, this hypothesis has to be approached with caution since Stanis et. al. (2008) found no correlation between sensitization to amphetamine and impulsivity.
Previous experiments using identical experimental protocol of MPD administrations but with systemic drug injections of MK-801 (Gaytan et. al., 2000b, 2001, 2002; Sripada et. al., 1998; 2001) or valproate (Eckermann et. al., 2001; Yang et. al., 2000a and 2000b) or lithium chloride (Yang et. al., 2001b) instead of lesions were performed at different time points during the experimental protocol to elucidate the effect of MK-801, valproate and lithium chloride on MPD sensitization. Similarly, 6-OHDA can be administered in PFC at different time points – namely one day after the chronic MPD treatment or six days after the chronic MPD treatment to elucidate the role of PFC in neuronal circuitry underlying MPD sensitization to understand the circuitry of action of drug craving and dependence. Alternatively, D1/D5 antagonist or D2/D4 antagonist can be administered in lieu of 6-OHDA to elucidate the type of dopamine receptors involved in MPD sensitization.
It is possible that the effects of the 6-OHDA injection described above might also be due to some depletion of noradrenergic terminals of PFC or a combination of both (Hanrott et. al., 2006). Others have used nor-epinephrine (NE) reuptake inhibitor desipramine to prevent the destruction of noradrenergic afferents (Carter and Pycock, 1980; Banks and Gratton, 1995; Beyer and Steketee, 1999; Bjijou et. al., 2002). It is known that the extent of sparing NE innervations has considerable consequences to the behavioral effects of DA depletion of PFC: the modulatory influence of PFC on NAc DA function may depend on the complex interplay between the DA and NE neurons of PFC (Banks and Gratton, 1995; Pascucci et. al., 2007). Moreover, Carter and Pycock (1980) reported that 6-OHDA lesions in PFC had no significant change on spontaneous locomotor activity or stereotyped behavior induced following amphetamine administration, while 6-OHDA PFC lesions performed in the presence of NE uptake blocker significantly enhanced spontaneous motor activity and amphetamine- induced stereotypic behavior. Therefore, there remains the remote possibility that the results of the similar 6-OHDA lesions done in the presence of desipramine (NE reuptake inhibitor) might yield different results. However, other studies have demonstrated that NE lacks the ability to maintain self-administrative behavior in animals (White and Kalivas, 1998) and therefore may not have a prominent role in sensitization. Thus, the effect observed may be attributed to be due to destruction of DA neurophyl within the PFC. Some reports (Beyer and Steketee, 1999; Bjijou et. al., 2002) determine the level of DA and NE depletion post 6-OHDA administrations by performing High Pressure Liquid Chromatography/Electrochemical Detection (HPLC-EC).
In conclusion, selective DA PFC lesion by bilateral 6-OHDA injections into the PFC prevented the expression of behavioral sensitization following chronic MPD administration (Gaytan et. al., 1997, 2000a; Lee et. al., 2009; Yang et. al., 2007). This suggests that the PFC catecholamines have a role, atleast in part, in the neural circuitry underlying behavioral sensitization elicited by chronic MPD administration.
4. Materials and methods
4.1 Animals
Thirty two male Sprague-Dawley rats weighing around 160 g on the first day of their arrival (Harlan, Indianapolis, IN, USA) were used. They were housed, two per cage, and were given free access to food and water. They were allowed to acclimatize to the room for three to five days prior to the start of the experiment. The room was maintained at 21 ± 2 °C, and relative humidity of about 50 %. The animals were kept on 12:12 light/dark cycle; light period from 6:00 am to 6:00 pm. The animals weighed around 190 g on the first day of the experiment and around 280 g at the end of the experiment.
4.2 Procedure
After 20 to 30 minutes of acclimatization to the recording cage, the baseline locomotor activity was recorded on experimental day 1 (ED 1; Table 1) post saline injection (pre-surgery baseline). The next day (ED 2), animals were divided into control, shams, and lesion groups (n = 8 for control, n = 8 for sham1 (See ‘Surgeries’), n = 6 for sham 2(See ‘Surgeries’), and n = 10 for 6-OHDA lesion group), and the respective surgeries were performed on sham and lesion groups. The animals were given five days to recover (EDs 3 to 7) from the surgery (Table 1). On ED 8, after 20-30 minutes of acclimatization to the testing cage, the animals received the saline injection and their locomotor activity was recorded (postsurgery baseline). On EDs 9 – 14, the rats were given a single 2.5mg/kg MPD dose daily. The recordings were started immediately after each injection. No injections were given on EDs 15-18 (washout period) but locomotor activity was recorded at the usual time of recordings of previous injections days. The animals were rechallenged with 2.5mg/kg MPD dose on ED 19 followed by two hours of locomotor activity recording. This experimental protocol was adapted from previous experiments (Gaytan et. al., 1997, 2000a; Lee et. al., 2008, 2009; Yang et. al., 2006, 2007), All recordings were started immediately post injection.
4.2. a. Surgeries
On ED 2, the rats were weighed and anaesthetized with 40mg/kg pentobarbital and an incision was made on the scalp and muscles to expose the skull and two holes were drilled above the PFC at 3.2 mm anterior to the bregma, 0.6mm lateral on each side of the midline. The coordinates were derived from Paxinos and Watson Brain Atlas (1986). The cannula (27 gauge) containing 6-OHDA was inserted at 4.0mm deep and 5μl of 6-hydroxydopamine (6-OHDA) solution with 3% ascorbic acid was injected. The 6-OHDA solution was made by dissolving 3 mg 6-OHDA, 2 mg of ascorbic acid per ml of 0.9% isotonic saline. 3% ascorbic acid was added to prevent the rapid oxidation of 6-OHDA. The needle was kept in that position for additional six minutes to allow the drug at the tip of the needle to diffuse. The needle was then raised 1mm (so that the tip would be at a depth of 3.0 mm from the skull) and another 5μl of 6-OHDA solution with 3% ascorbic acid was injected, followed by an additional six minutes for the drug at the tip of the needle to diffuse. The same procedure was done on the other side of the PFC. After surgery, the skin was closed using wound closing staples. Two sham groups were used. For sham 1group, the exact same procedure was repeated. However, only 3% ascorbic acid and saline was injected. For the sham 2 group, similar surgical procedure was repeated without inserting the needle in the PFC. Two sham groups were used to check if injecting vehicle (saline with ascorbic acid) had any behavioral effects. The rats were then allowed to recover for five days. The recovery period was adapted from Bubser (1994) who demonstrated similar levels of DA depletion following 4 - 5, 10 - 12 and 32 - 36 days post surgery.
4.3 Drugs
Methylphenidate hydrochloride (MPD) was dissolved in 0.9% isotonic saline and injected intraperitoneally (i.p.). The animals were weighed every day, and were injected with 2.5 mg/kg of MPD doses on scheduled days before locomotor recording (Table 1). All injections were equalized to a volume of 0.8ml with 0.9% isotonic saline. The injections were given intraperitoneally between 6:30 and 7:00am, after 15-20 minutes of acclimatization to the testing chamber. The 2.5 mg/kg dose was chosen based on previous dose response experimental protocol that indicated that this dosage elicited behavioral sensitization (Gaytan et. al., 1997, 2000a; Lee et. al., 2008, 2009; Yang et. al., 2003, 2006, 2007). The time of administration was decided similarly based on previous experiments that indicated that behavioral sensitization is best elicited if the drug is administered at the beginning of the light cycle (Gaytan et. al., 2000a). The route of administration was determined to be i.p. since methylphenidate abuse in humans is noted more with intravenous administrations than oral usage (Parran and Jasinksi, 1991) and that the behavioral model to study addiction is sensitization (Dafny & Yang, 2006; Robinson & Berridge, 1993; Wolf, 1998). Moreover, behavioral sensitization was not observed in studies using oral administrations of MPD (Fregly and Black, 1964; Kuczenski and Segal, 2002) but sensitization was observed in studies that administered MPD parentarally by either i.p. or subcutaneous injections (reviewed by Askenasy et. al., 2007). Finally, previous experiments from our lab (Gaytan et. al., 1996, 1997; Lee et. al., 2008; Yang et. al., 2006) have used i.p. injections successfully to elicit sensitization. Therefore, the i.p. route of MPD administration was chosen in this study.
4.4 Apparatus
Computerized animal activity monitoring (CAAM; AccuScan Instruments, Inc., Columbus, OH) monitored the animal's locomotor activity during the recording sessions. The system consisted of open field cages (40.5cm × 40.5cm × 31.5cm) with two levels of 16 infrared beams and sensors that got interrupted when the animal moved. The motion sensors were 6 and 12.5 cm from the base of the cage. AccuScan Analyzer counted the number of beams interrupted every ten minutes and downloaded the data into OASIS program. The OASIS program further sorted the data into different locomotor indices. The locomotor activity was analyzed using the following indices: horizontal activity (HA), i.e., the total number of beam interruptions that occur in the horizontal sensor during the recording time and total distance (TD), i.e, the total distance travelled by the animal in centimeters during the recording time (Algahim et. al., 2009; Gaytan et. al., 1997, 2000; Lee et. al., 2008, 2009; Yang et. al., 2003, 2006, 2007).
4.5 Histology
The animals were over-dosed with sodium barbital after the last day of the experiment and perfused with 10% formaldehyde. The brains were removed and soaked in 10% formaldehyde for at least 48 hours. Brains were sliced at 40 μm using a Microm HM 505E cryostat (Microm GmbH, Germany) and allowed to dry for at least 24 hours, and then stained with Cresyl violet, similar to the procedure described by Riickert et. al., (1997). The histological sections were examined under high magnification microscope using the atlas of Paxinos and Watson (1986) to determine the accuracy and size of the bilateral lesions produced by 6-OHDA injections to PFC (Fig. 6).
Figure 6.
shows the site of the syringe tip location in the brain. The black dots indicate the location of the injection sites for the 6-OHDA administrations (sections 3.7, 3.2 and 2.7 mm anterior to bregma) in the PFC, whereas the grey shaded area indicates the spread of 6-OHDA in the PFC.
4.6 Data analysis
To investigate the effect of bilateral 6-OHDA administration to the PFC on the acute and the chronic effect of MPD on locomotor activity, horizontal activity (HA), the overall locomotor activity, total distance (TD) i.e., the total distance travelled in cm and number of stereotypic movements (NOS), i.e., the number of separate episodes of stereotypic activity were evaluated (Askenasy et. al., 2007). For each locomotor index analyzed, five comparisons were made: 1) the effect of surgery on locomotor baseline i.e., comparing ED 8 with ED 1, 2) the acute effect of MPD i.e., comparing ED 9 to ED 8, 3) the effect of the sixth repetitive daily injection of single MPD dose on locomotor activity compared to the initial MPD effect i.e., the induction phase – comparing ED 9 to ED 14, 4) the effect of six repetitive days of MPD injection on baseline activity i.e., washout activity - comparing EDs 15 and 18 with ED 8, and 5) comparing the activity elicited by the MPD rechallenge injection (ED 19) to ED 9 to observe whether sensitization is expressed.
The sum of the locomotor activity of the 2 hours was compared for different days. Significance of difference between experimental days was set at 0.05 and was judged using one way ANOVA and Post hoc analysis with LSD test (Gaytan et. al., 1996, 2000a; Lee et. al., 2008; Yang et. al., 2003, 2007).
Acknowledgement
This study was supported in part by the Pat Rutherford Chair in Psychiatry and NIH R01-DA027222.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Adams JU, Careri JM, Efferen TR, Rotrosen J. Conditioned locomotor stimulant effects of cocaine in rats do not result from interference with habituation. Psychopharmacology. 2000;151:13–18. doi: 10.1007/s002130000431. [DOI] [PubMed] [Google Scholar]
- 2.Ahmed SH, Oberling P, DiScala G, Sandner G. Amphetamine-induced conditioned activity does not result from a failure of rats to habituate to novelty. Psychopharmacology. 1996;123:325–332. doi: 10.1007/BF02246642. [DOI] [PubMed] [Google Scholar]
- 3.Algahim MF, Yang PB, Wilcox VT, Burau KD, Swann AC, Dafny N. Prolonged methylphenidate treatment alters the behavioral diurnal activity pattern of adult male Sprague-Dawley rats. Pharmacology, biochemistry, and behavior. 2009;92:93–99. doi: 10.1016/j.pbb.2008.10.021. [DOI] [PubMed] [Google Scholar]
- 4.Anderson JC, Williams S, McGee R, Silva PA. DSM-III disorders in preadolescent children: Prevalence in a large sample from the general population. Arch Gen Psychiatry. 1987;44:69–76. doi: 10.1001/archpsyc.1987.01800130081010. [DOI] [PubMed] [Google Scholar]
- 5.Arnsten AF, Steere JC, Hunt RD. The contribution of alpha 2-noradrenergic mechanisms of prefrontal cortical cognitive function. Potential significance for attention-deficit hyperactivity disorder. Arch Gen Psychiatry. 1996;53:448–455. doi: 10.1001/archpsyc.1996.01830050084013. [DOI] [PubMed] [Google Scholar]
- 6.Askenasy EP, Taber KH, Yang PB, Dafny N. Methylphenidate (Ritalin): behavioral studies in the rat. Int. J. Neurosci. 2007;117:757–794. doi: 10.1080/00207450600910176. [DOI] [PubMed] [Google Scholar]
- 7.Badiani A, Browman KE, Robinson TE. Influence of novel versus home environments on sensitization to the psychomotor stimulant effects of cocaine and amphetamine. Brain Res. 1995;674:291–298. doi: 10.1016/0006-8993(95)00028-o. [DOI] [PubMed] [Google Scholar]
- 8.Banks KE, Gratton A. Possible involvement of medial prefrontal cortex in amphetamine-induced sensitization of mesolimbic dopamine function. European Journal of Pharmacology. 1995;282:157–167. doi: 10.1016/0014-2999(95)00306-6. [DOI] [PubMed] [Google Scholar]
- 9.Barbaresi W, Katusic S, Colligan R, Oankratz V, Weber K, Mrazek D, et al. How common is attention deficit/hyperactivity disorder? Incidence in a population-based birth cohort in Rochester, Minn. Arch Pediatric Adolesc Medicine. 2002;156:217–224. doi: 10.1001/archpedi.156.3.217. [DOI] [PubMed] [Google Scholar]
- 10.Bedard AC, Ickowicz A, Logan GD, Hogg-Johnson S, Schachar R, Tannock R. Selective inhibition in children with attention-deficit hyperactivity disorder off and on stimulant medication. J Abnorm Child Psychol. 2003;31:315–327. doi: 10.1023/a:1023285614844. [DOI] [PubMed] [Google Scholar]
- 11.Berridge CW, Devilbiss DM, Andrzejewski ME, Arnsten AFT, Kelley AE, Schmeichel B, et al. Methylphenidate preferentially increases catecholamine neurotransmission within the prefrontal cortex at low doses that enhance cognitive function. Biol. Psychiatry. 2006;60:1111–1120. doi: 10.1016/j.biopsych.2006.04.022. [DOI] [PubMed] [Google Scholar]
- 12.Beyer CE, Steketee JD. Dopamine depletion in the medial prefrontal cortex induces sensitized-like behavioral and neurochemical responses to cocaine. Brain Res. 1999;883:133–141. doi: 10.1016/s0006-8993(99)01485-7. [DOI] [PubMed] [Google Scholar]
- 13.Bjijou Y, Deurwaerdere PD, Spampinato U, Stinus L, Cador M. d-amphetamine induced behavioral sensitization: effect of lesioning dopaminergic terminals in the medial prefrontal cortex, the amygdala and the entorhinal cortex. Neurosci. 2002;109:499–516. doi: 10.1016/s0306-4522(01)00508-5. [DOI] [PubMed] [Google Scholar]
- 14.Bubser M, Schmidt WJ. 6-Hydroxydopamine lesion of the rat prefrontal cortex increases locomotor activity, impairs acquisition of delayed alternation tasks, but does not affect un-interrupted tasks in the radial maze. Behav. Brain Res. 1990;37:157–168. doi: 10.1016/0166-4328(90)90091-r. [DOI] [PubMed] [Google Scholar]
- 15.Bubser M. 6-Hydroxydopamine Lesions of the Medial Prefrontal Cortex of Rats Do Not Affect Dopamine Metabolism in the Basal Ganglia at Short and Long Postsurgical Intervals. Neurochemical Res. 1994;19:421–425. doi: 10.1007/BF00967319. [DOI] [PubMed] [Google Scholar]
- 16.Cador M, Bjijou Y, Cailhol S, Stinus L. d-amphetamine-induced behavioral sensitization: implication of a glutamatergic medial prefrontal cortex-ventral tegmental area innervation. Neurosci. 1999;94:705–721. doi: 10.1016/s0306-4522(99)00361-9. [DOI] [PubMed] [Google Scholar]
- 17.Chao J, Nestler EJ. Molecular Neurobiology of drug addiction. Annual Rev. Med. 2004;55:113–132. doi: 10.1146/annurev.med.55.091902.103730. [DOI] [PubMed] [Google Scholar]
- 18.Chen Y, Dubé CM, Rice CJ, Baram TZ. Rapid loss of dendritic spines after stress involves derangement of spine dynamics by corticotropin-releasing hormone. J Neurosci. 2008;28:2903–2911. doi: 10.1523/JNEUROSCI.0225-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Clarke PBS, Jakubovic A, Fibiger HC. Anatomical analysis of the involvement of mesolimbocortcal dopamine in the locomotor stimulant actions of D-amphetamine and apomorphine. Psychopharmacol. 1988;96:511–520. doi: 10.1007/BF02180033. [DOI] [PubMed] [Google Scholar]
- 20.Carter CJ, Pycock CJ. Behavioral and biochemical effects of dopamine and noradrenaline depletion within the medial prefrontal cortex of the Rat. Brain Res. 1980;192:163–176. doi: 10.1016/0006-8993(80)91016-1. [DOI] [PubMed] [Google Scholar]
- 21.Dafny N, Yang PB. The role of age, genotype, sex, and route of acute and chronic administration of methylphenidate: a review of its locomotor effects. Brain Res. Bul. 2006;68:393–405. doi: 10.1016/j.brainresbull.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 22.Dalley JW, Cardinal RN, Robbins TW. Prefrontal executive and cognitive functions in rodents: neural and neurochemical substrates. Neurosci Biobehav Rev. 2004;28:771–784. doi: 10.1016/j.neubiorev.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 23.Ding YS, Fowler JS, Volkow ND, Gatley SJ, Logan J, Dewey SL, Alexoff D, Fazzini E, Wolf AP. Pharmacokinetics and in vivo specificity of [11C]dl-threomethylphenidate for the presynaptic dopaminergic neuron. Synapse. 1994;18:152–160. doi: 10.1002/syn.890180207. [DOI] [PubMed] [Google Scholar]
- 24.Eckermann K, Beasley A, Yang P, Gaytan O, Swann A, Dafny N. Methylphenidate sensitization is modulated by valproate. Life Sci. 2001;69:47–57. doi: 10.1016/s0024-3205(01)01095-5. [DOI] [PubMed] [Google Scholar]
- 25.Elia J, Ambrosini PJ, Rapoport JL. Treatment of attention-deficit-hyperactivity disorder. Drug Therapy. 1999;340:780–788. doi: 10.1056/NEJM199903113401007. [DOI] [PubMed] [Google Scholar]
- 26.Fregly MJ, Black BA. Effect of methylphenidate on spontaneous activity, food intake, and cold tolerance of propylthiouracil-treated rats. Canad. J.Physiol. Pharmacol. 1964;42:415–419. doi: 10.1139/y64-051. [DOI] [PubMed] [Google Scholar]
- 27.Gaytan O, al-Rahim S, Swann A, Dafny N. Sensitization to locomotor effects of methylphenidate in rat. LifeSci. 1997;61:101–107. doi: 10.1016/s0024-3205(97)00598-5. [DOI] [PubMed] [Google Scholar]
- 28.Gaytan O, Yang PB, Swann A, Dafny N. Diurnal differences in sensitization to methylphenidate. Brain Res. 2000a;864:24–39. doi: 10.1016/s0006-8993(00)02117-x. [DOI] [PubMed] [Google Scholar]
- 29.Gaytan O, Nason R, Alagugurusamy R, Swann A, Dafny N. MK-801 blocks the development of sensitization to the locomotor effects of methylphenidate. Brain Res. Bull. 2000b;51:485–492. doi: 10.1016/s0361-9230(99)00268-3. [DOI] [PubMed] [Google Scholar]
- 30.Gaytan O, Sripada S, Swann A, Dafny N. Blockade of sensitization to methylphenidate by MK-801: Partial dissociation from motor effects. Neuropharmacol. 2001;40:298–309. doi: 10.1016/s0028-3908(00)00122-2. [DOI] [PubMed] [Google Scholar]
- 31.Grillner P, Mercuri NB. Intrinsic membrane properties and synaptic inputs regulating the firing activity of the dopamine neurons. Behav Brain Res. 2002;130:149–69. doi: 10.1016/s0166-4328(01)00418-1. [DOI] [PubMed] [Google Scholar]
- 32.Grund T, Lehmann K, Bock N, Rothenberger A, Teuchert-Noodt G. Influence of methylphenidate on brain development – an update of recent animal experiments. Behav Brain Func. 2006;2:2. doi: 10.1186/1744-9081-2-2. doi:10.1186/1744-9081-2-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hanrott K, Gudmunsen L, O'Neill MJ, Wonnacott S. 6-Hydroxydopamine-induced apoptosis is mediated via extracellular auto-oxidation and caspase 3-dependent activation of protein Kinase Cδ*. Journal of Biological Chemistry. 2006;281:5373–5382. doi: 10.1074/jbc.M511560200. [DOI] [PubMed] [Google Scholar]
- 34.Hinson RE, Poulos CX. Sensitization to the behavioral effects of cocaine: modification by Pavlovian conditioning. Pharmacol Biochem Behav. 1981;15:559–562. doi: 10.1016/0091-3057(81)90208-2. [DOI] [PubMed] [Google Scholar]
- 35.Itami S, Uno H. Orbitofrontal cortex dysfunction in attention-deficit hyperactivity disorder revealed by reversal and extinction tasks. NeuroReport. 2002;13:2453–2457. doi: 10.1097/00001756-200212200-00016. [DOI] [PubMed] [Google Scholar]
- 36.Jentsch JD, Taylor JR. Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacol. 1999;146:373–390. doi: 10.1007/pl00005483. [DOI] [PubMed] [Google Scholar]
- 37.Joyce EM, Stinus L, Iversen SD. Effects of injections of 6-OHDA into either nucleus accumbens septi or frontal cortex on spontaneous and drug-induced activity. Neuropharmacol. 1983;22:1141–1145. doi: 10.1016/0028-3908(83)90051-5. [DOI] [PubMed] [Google Scholar]
- 38.Kalivas PW, Stewart J. Dopamine transmission in the initiation and expression of drug- and stress-induced sensitization of motor activity. Brain Res. Review. 1991;16:223–244. doi: 10.1016/0165-0173(91)90007-u. [DOI] [PubMed] [Google Scholar]
- 39.Kalivas PW, Sorg BA, Hooks MS. The pharmacology and neural circuitry of sensitization to psychostimulants. Behav. Pharmac. 1993;4:315–334. [PubMed] [Google Scholar]
- 40.Kollins SH, MacDonald EK, Rush CR. Assessing the abuse potential of methylphenidate in nonhuman and human subjects: a review. Pharmacol. Biochem. Behavior. 2001;68:611–627. doi: 10.1016/s0091-3057(01)00464-6. [DOI] [PubMed] [Google Scholar]
- 41.Krause KH, Dresel SH, Krause J, Fougere CL, Ackenheil M. The dopamine transporter and neuroimaging in attention deficit hyperactivity disorder. Neuroscience and Biobehavioral Reviews. 2003;27:605–613. doi: 10.1016/j.neubiorev.2003.08.012. [DOI] [PubMed] [Google Scholar]
- 42.Kuczenski R, Segal DS. Exposure of adolescent rats to oral methylphenidate: preferential effects on extracellular norepinephrine and absence of sensitization and cross-sensitization to methamphetamine. J. Neurosci. 2002;22:7264–7271. doi: 10.1523/JNEUROSCI.22-16-07264.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Leccesse AP, Lyness WH. Lesions of dopamine neurons in the medial prefrontal cortex: effects on self-administration of amphetamine and dopamine synthesis in the brain of the rat. Neuropharmacol. 1987;26:1303–1308. doi: 10.1016/0028-3908(87)90091-8. [DOI] [PubMed] [Google Scholar]
- 44.Lee MJ, Swann AC, Dafny N. Methylphenidate sensitization is prevented by prefrontal cortex lesion. Brain Res. Bull. 2008;76:131–140. doi: 10.1016/j.brainresbull.2007.12.004. [DOI] [PubMed] [Google Scholar]
- 45.Lee MJ, Yang PB, Wilcox VT, Burau KD, Swann AC, Dafny N. Does repetitive Ritalin injection produce long-term effects on SD female adolescent rats? Neuropharmacol. 2009;57:201–207. doi: 10.1016/j.neuropharm.2009.06.008. [DOI] [PubMed] [Google Scholar]
- 46.Louilot A, Le Moal M, Simon H. Opposite influences of dopaminergic pathways to the prefrontal cortex or the septum on the dopaminergic transmission in the nucleus accumbens. An in vivo voltammetric study. Neurosci. 1989;29:45–56. doi: 10.1016/0306-4522(89)90331-x. [DOI] [PubMed] [Google Scholar]
- 47.Miller EK, Cohen JD. An integrative theory of prefrontal cortex function. Annu Rev Neurosci. 2001;24:167–202. doi: 10.1146/annurev.neuro.24.1.167. [DOI] [PubMed] [Google Scholar]
- 48.Nagai T, Takuma K, Karnei H, Ito Y, Nakamichi N, Ibo D, et al. Dopamine D1 Receptors regulate protein synthesis-dependent long term recognition memory via extracellular signal-regulated kinase ½ in the prefrontal cortex. Learning & Memory. 2007;14:117–125. doi: 10.1101/lm.461407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Oades RD, Taghzouti K, Rivet J-M, Simon H, Le Moal M. Locomotor activity in relation to dopamine and noradrenaline in the nucleus accumbens, septal and frontal areas: a 6-hydroxydopamine study. Neuropsychobiol. 1986;16:37–42. doi: 10.1159/000118294. [DOI] [PubMed] [Google Scholar]
- 50.Parran TV, Jasinski DR. Intravenous methylphenidate abuse: prototype for prescription drug abuse. Arch Intern Med. 1991;151:781–783. [PubMed] [Google Scholar]
- 51.Pascucci T, Ventura R, Latagliata EC, Cabib S, Puglisi-Allegra S. The medial prefrontal cortex determines the accumbens dopamine response to stress throught the opposing influences of norepinephrine and dopamine. Cerebral Cortex. 2007;17:2796–2804. doi: 10.1093/cercor/bhm008. [DOI] [PubMed] [Google Scholar]
- 52.Patrick KS, Markowitz JS. Pharmacology of methylphenidate, amphetamine enantiomers, and penoline in attention deficit/ hyperactivity disorder. Hum. Psychopharmacol. 1997;12:527–546. [Google Scholar]
- 53.Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. 2nd ed. Academic Press; Orlando: 1986. [Google Scholar]
- 54.Pert A. Neurobiological mechanisms underlying the acquisition and expression of incentive motivation by cocaine-associated stimuli: relationship to craving. NIDA Res Monogr. 1994;145:118–146. [PubMed] [Google Scholar]
- 55.Pickens RW, Crowder WF. Effects of CS-US interval on conditioning of drug response, with assessment of speed of conditioning. Psychopharmacologia. 1967;11:88–94. doi: 10.1007/BF00401511. [DOI] [PubMed] [Google Scholar]
- 56.Pierce RC, Kalivas PW. A circuitry model of the expression of behavioral sensitization to amphetamine-like psychostimulants. Brain Res. Rev. 1997;25:192–216. doi: 10.1016/s0165-0173(97)00021-0. [DOI] [PubMed] [Google Scholar]
- 57.Post RM, Lockfeld A, Squillace KM, Contel NR. Drug environment interaction: context-dependency of cocaine induced behavioral sensitization. Life Sci. 1981;28:755–760. doi: 10.1016/0024-3205(81)90157-0. [DOI] [PubMed] [Google Scholar]
- 58.Pycock CJ, Carter CJ, Kerwin RW. Effect of 6-hydroxydopamine lesions of the medial prefrontal cortex on neurotransmitter systems in subcortical sites in the rat. J. Neurochem. 1980;34:91–99. doi: 10.1111/j.1471-4159.1980.tb04625.x. [DOI] [PubMed] [Google Scholar]
- 59.Riickert N, Bubser M, Schmidt WJ. 6-Hydroxydopamine lesion of locus coeruleus and the antiparkinsonian potential of NMDA-receptor antagonists in rats. J Neural Transm. 1997;104:363–377. doi: 10.1007/BF01277657. [DOI] [PubMed] [Google Scholar]
- 60.Robinson RG, Stitt TG. Intracortical 6-OHDA induces an asymmetrical behavioral response in the rat. Brain Res. 1981;213:387–395. doi: 10.1016/0006-8993(81)90243-2. [DOI] [PubMed] [Google Scholar]
- 61.Robinson TE. Behavioral sensitization: characterization of enduring changes in rotational behavior produced by intermittent injections of amphetamine in male and female rats. Psychopharmacol. 1984;84:466–475. doi: 10.1007/BF00431451. [DOI] [PubMed] [Google Scholar]
- 62.Robinson TE, Berridge KC. The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res. Rev. 1993;18:247–291. doi: 10.1016/0165-0173(93)90013-p. [DOI] [PubMed] [Google Scholar]
- 63.Roman T, Schmitz M, Polanczyk G, Eizirik M, Rohde LA, Hutz MH. Attention-deficit hyperactivity disorder: a study of association with both the dopamine transporter gene and the dopamine D4 receptor gene. Am. J. Med. Gen. 2001;105:471–478. doi: 10.1002/ajmg.1408. [DOI] [PubMed] [Google Scholar]
- 64.Sripada S, Gaytan O, Al-rahim S, Swann A, Dafny N. Dose-related effects of MK-801 on acute and chronic methylphenidate administration. Brain Res. 1998;814:78–85. doi: 10.1016/s0006-8993(98)01035-x. [DOI] [PubMed] [Google Scholar]
- 65.Sripada S, Gaytan O, Swann A, Dafny N. The role of MK-801 in sensitization to stimulants. Brain Res. Rev. 2001;35:97–114. doi: 10.1016/s0165-0173(00)00046-1. [DOI] [PubMed] [Google Scholar]
- 66.Stanis JJ, Avila HM, White MD, Gulley JM. Dissociation between long-lasting behavioral sensitization to amphetamine and impulsive choice in rats performing a delay-discounting task. Psychopharmacol. 2008;199:539–548. doi: 10.1007/s00213-008-1182-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sullivan RM, Brake WG. What the rodent prefrontal cortex can teach us about attention-deficit/hyperactivity disorder: the critical role of early developmental events on prefrontal function. Behav Brain Res. 2003;146:43–55. doi: 10.1016/j.bbr.2003.09.015. [DOI] [PubMed] [Google Scholar]
- 68.Tilson HA, Rech RH. Conditioned drug effects and absence of tolerance to d-amphetamine induced motor activity. Pharmacol Biochem Behav. 1973;1:149–153. [Google Scholar]
- 69.van Gaalen MM, Brueggeman RJ, Bronius PFC, Schoffelmeer ANM, Vanderschuren LJMJ. Behavioral disinhibition requires dopamine receptor activation. Psychopharmacol. 2006;187:73–85. doi: 10.1007/s00213-006-0396-1. [DOI] [PubMed] [Google Scholar]
- 70.Volkow ND, Wang GJ, Fowler JS, Fischman M, Foltin R, Abumrad NN, et al. Methylphenidate and cocaine have a similar in vivo potency to block dopamine transporters in the human brain. Life Sci. 1999;65:L7–L12. doi: 10.1016/s0024-3205(99)00225-8. [DOI] [PubMed] [Google Scholar]
- 71.Wanchoo SJ, Swann AC, Dafny N. Descending glutamatergic pathways of PFC are involved in acute and chronic action of methylphenidate. Brain Res. 2009 doi: 10.1016/j.brainres.2009.08.095. in print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.White FJ, Kalivas PW. Neuroadaptations involved in amphetamine and cocaine addiction. Drug and Alcohol Dependence. 1998;51:141–153. doi: 10.1016/s0376-8716(98)00072-6. [DOI] [PubMed] [Google Scholar]
- 73.Wolf ME. The role of excitatory amino acids in behavioral sensitization to psychomotor stimulants. Prog. Neurobiol. 1998;54:679–720. doi: 10.1016/s0301-0082(97)00090-7. [DOI] [PubMed] [Google Scholar]
- 74.Yang P, Beasley A, Eckermann K, Swann A, Dafny N. Valproate modulates the expression of methylphenidate (Ritalin) sensitization. Brain Res. 2000a;874:216–220. doi: 10.1016/s0006-8993(00)02500-2. [DOI] [PubMed] [Google Scholar]
- 75.Yang P, Beasley A, Eckermann K, Swann A, Dafny N. Valproate prevents the induction of sensitization to methylphenidate (Ritalin) in rats. Brain Res. 2000b;887:276–284. doi: 10.1016/s0006-8993(00)02996-6. [DOI] [PubMed] [Google Scholar]
- 76.Yang P, Singhal N, Modi G, Swann A, Dafny N. Effect of lithium on induction and expression of methylphenidate sensitization. Eur. J. of Pharmacol. 2001;426:65–72. doi: 10.1016/s0014-2999(01)01213-4. [DOI] [PubMed] [Google Scholar]
- 77.Yang P, Swann A, Dafny N. Valproate prevents the induction and the expression of MK-801 sensitization. Brain Res. 2002;954:151–159. doi: 10.1016/s0006-8993(02)03141-4. [DOI] [PubMed] [Google Scholar]
- 78.Yang PB, Amini B, Swann AC, Dafny N. Strain differences in the behavioral responses of male rats to chronically administered methylphenidate. Brain Res. 2003;971:139–152. doi: 10.1016/s0006-8993(02)04240-3. [DOI] [PubMed] [Google Scholar]
- 79.Yang PB, Swann AC, Dafny N. Dose-response characteristics of methylphenidate on locomotor behavior and on sensory evoked potentials recorded from the VTA, NAc, and PFC in freely behaving rats. Behavioral and Brain Functions. 2006;2:3. doi: 10.1186/1744-9081-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Yang PB, Swann AC, Dafny N. Chronic administration of methylphenidate produces neurophysiological and behavioral sensitization. Brain Res. 2007;1145:66–80. doi: 10.1016/j.brainres.2007.01.108. [DOI] [PMC free article] [PubMed] [Google Scholar]