Abstract
Background
Transannular patch (TAP) repair of tetralogy of Fallot often results in significant right ventricular outflow tract (RVOT) dilation and distortion. We hypothesized that insertion of Melody valves into the proximal right and left branch pulmonary arteries (PAs) would reduce pulmonary regurgitation fraction (PRF) in an ovine model of pulmonary insufficiency and dilated RVOT.
Methods and Results
Ten sheep underwent baseline cardiac catheterization, surgical pulmonary valvectomy, and TAP placement. A subset (n=5) had Melody valves (2 devices per animal) inserted into the proximal right and left PAs during the surgical procedure. Melody valves were placed distal to the right-upper-lobe (RUL) artery branch, leaving the RUL “unprotected.” Preoperative MRIs (n=5) were used to determine baseline RV ejection fraction (RVEF) and left ventricular (LV) EF. All surviving animals (n=9) underwent MRI and catheterization 6 weeks postsurgery.
Mean PRF was lower in the Melody valve group (15±6% versus 37±3%; P=0.014). The unprotected RUL was responsible for 64% of the PRF measured in the Melody valve group. In the non-Melody group, the RVEF was lower than baseline (P=0.003) and than in the Melody group (P=0.05). The LVEF was also lower in the non-Melody group versus baseline (P=0.004) and versus Melody (P=0.01).
Conclusions
Bilateral branch PA Melody valve implantation significantly reduced PRF and altered RV and LV function favorably in a model of TAP for tetralogy of Fallot. This novel intervention may offer potential benefit in treating patients with anatomically heterogeneous disease of the RVOT.
Keywords: MRI, heart valve prosthesis implantation, pulmonary heart disease, tetralogy of Fallot, pulmonary valve regurgitation, pulmonary valve stenosis
Surgical repair of tetralogy of Fallot (ToF), the most common cyanotic congenital heart disease, almost always results in pulmonary valve insufficiency and often in proximal branch pulmonary artery (PA) stenosis.1-6 These lesions are the major cause of late morbidity and mortality after successful repair of ToF, leading to right ventricular dilation; exercise intolerance; ventricular dysfunction; arrhythmia; increased risk for sudden death; and, eventually, reoperation in adolescents and young adults.1-3,6,7
Percutaneous transcatheter pulmonary valve replacement has been introduced recently,8-10 and early and intermediate clinical experience has been reported with modest success.11-13 These prior studies demonstrated the feasibility of this general approach, and this important breakthrough has the potential to delay and reduce the need for multiple operative procedures in children with ToF. However, this approach is currently limited almost exclusively to patients in whom a conduit connecting the right ventricle (RV) to the main PA (MPA) was placed at the time of their initial surgery.10,12,14 Unfortunately, they represent only a fraction of those patients after repair of ToF who are followed clinically.14 The vast majority of others have had a transannular patch (TAP) repair, a procedure that effectively destroys the pulmonary valve, resulting in free pulmonary insufficiency.1,2,6,15
In the absence of an RV-to-PA conduit, severe RV outflow tract (RVOT) failure with marked anatomic distortion and dilation of that region can develop. This distortion/dilation makes it technically impossible to implant the currently available transcatheter valves in the orthotopic (usual) pulmonary position through a transcatheter approach,10,16,17 and therefore, repeat surgery (at relatively higher risk) is required to restore pulmonary valve competence.
Several experimental approaches to transcatheter valve replacement in dilated RVOTs have been described,16-19 but most have required surgical reduction in the size of the RVOT before valve implantation, and none has been widely used in humans. Most recently, the first-in-human implantation of a novel, custom-designed percutaneous pulmonary valve was reported.20,21 This exciting, innovative approach to valve replacement offers a glimpse into what may be the future of device therapy. However, given the anatomic variability of the RVOT and the concomitant occurrence of branch PA disease frequently encountered in this patient population, alternative approaches to valve replacement should continue to be explored.
In the current study, we tested the hypothesis that implantation of a transcatheter valve into both the right and left proximal branch PAs is feasible and will reduce regurgitant (backward) flow into the RV. The ultimate goal is to broaden the indications for percutaneous valve replacement to include patients with large, distorted outflow tracts by exploring unorthodox locations within the PA tree for valve implantation. Implantation of a durable valve into the branch PAs could potentially alleviate pulmonary regurgitation, preserve RV function, and reduce late postoperative morbidity without the risks inherent in open heart surgery.
Methods
Following approval by The University of Pennsylvania’s Institute of Animal Care and Use Committee, female Dorsett hybrid sheep were subjected to study. Ten sheep underwent baseline cardiac catheterization, surgical pulmonary valvectomy, and TAP placement. A subset (n=5) also underwent baseline cardiac MRI before surgery and had Melody valves (Medtronic, Inc; Minneapolis, MN) (2 devices per animal) inserted into the proximal right PA (RPA) and left PA (LPA) during the surgical procedure. Baseline MRIs (n=5) were used to determine control RV and left ventricular (LV) volumes and ejection fractions (EFs). All animals underwent MRI and catheterization 6 weeks after surgery.
Surgical Pulmonary Valvectomy TAP Placement
A left-side thoracotomy was performed, and the pericardium was incised to expose the RVOT and MPA. An oval Gore-Tex patch was prepared, measuring 45 mm long by 25 mm wide by 0.6 mm thick. A Satinsky clamp was placed across the RVOT to subtotally occlude it, incorporating the pulmonary annulus. The RVOT was then incised along its length with the pulmonary annulus at the midpoint of this incision, and 1 complete pulmonary valve leaflet was excised. The patch then was sutured to the edges of the incised RVOT with a 6-0 running prolene suture.
Melody Valve Implantation
At the time of the surgery, 5 animals underwent bilateral branch PA Melody valve implantation in addition to pulmonary valvectomy and TAP placement. These animals comprised the Melody valve group.
Baseline angiography was performed to delineate anatomy and determine branch PA diameter. In the ovine PA tree, the proximal RPA and LPA are shorter than in humans, with the segmental upper-lobe branches originating just beyond the MPA bifurcation. Thus, deployment of the Melody device, which effectively is a covered stent, excludes the upper-lobe arteries if implanted into the proximal part of the branch PAs. We observed that the left upper lobe (LUL) of the lung is slightly smaller than the right upper lobe (RUL), and that the LUL branch of the LPA generally appeared to be smaller than the corresponding RUL branch of the RPA. To minimize the percentage of lung excluded by Melody valve implantation, we deployed the device in the proximal LPA, completely jailing and thus excluding the LUL artery. Conversely, we deployed the Melody valve just distal to the origin of the RUL RPA, leaving the RUL branch patent but “unprotected” by the Melody valve.
In each animal, the Melody valves were crimped onto angioplasty balloons measuring 16 to 18 mm in diameter by 4 cm in length. Balloon diameter was chosen according to baseline angiographic measurements. In general, the balloon was oversized by 3 to 5 mm. To minimize the profile of the Melody valve, crimping was accomplished in a stepwise manner by sequentially elongating the device on progressively smaller Hegar dilators starting at 14 mm in diameter down to 7 mm. This technique allowed for a uniform and symmetrical reduction in device profile. The balloon-Melody complex was then front loaded into a modified 20-F sheath, which was introduced directly into the MPA over a guidewire through an incision made at the level of the pulmonary valve annulus. Hemostasis was maintained using a purse string suture. If necessary for hemostatic control, in lieu of a single purse string, the introducer sheaths were inserted through a short segment of vascular tube graft sewn to the MPA. In both approaches, the incision used for sheath insertion was extended proximally across the pulmonary valve annulus for valvectomy and TAP placement (see previous surgical description). Once in position, the devices were deployed through standard balloon inflation technique. Postdeployment angiography was performed to assess valve position and function bilaterally.
Cardiac MRI Methods
All MRIs were performed under general anesthesia using a 3-T Siemens Avanto (Siemens Corp; New York, NY) scanner and analyzed by a single observer. A high-fidelity pressure transducer (SPC-350; Millar Instruments Inc; Houston, TX) was inserted percutaneously through the femoral artery into the LV, allowing for cardiac gated image acquisition.
Phase-Contrast MRI Methodology
Phase-contrast magnetic resonance acquisition was applied at the MPA, branch PAs, and the aorta for flow quantification. The typical imaging parameters were an echo time of 3.5 ms; temporal resolution, 40 ms; flip angle, 25°; and voxel size, 2.0×1.5×5.0 mm (slice thickness) with a resulting signal-to-noise ratio of approximately 1.0. Signal averaging was used to improve signal-to-noise ratio and minimize respiratory artifact. Data analysis involved contouring regions of interest throughout the cardiac cycle. The regurgitant fraction (RF) through a region of interest was defined as follows: RF=(reverse flow/forward flow)×100. Fractional branch PA pulmonary blood flow (PBF) distribution was calculated as follows: fractional branch PA PBF=(net branch PA flow/net total PBF)×100. Indexed values were obtained by calculating body surface area based on an accepted conversion formula for sheep: body surface area (m2)=0.097×weight (kg)0.656.22
Ventricular Volume Acquisition and Analysis
High temporal and spatial resolution gradient echo cine short-axis imaging of the ventricles were acquired from base to apex. The typical imaging parameters were an echo time of 2.7 ms; temporal resolution, 31.7 ms; bandwidth, 401 mHz; and voxel size, 2.1×2.1×5.0 mm (slice thickness) with a resulting signal-to-noise ratio of approximately 1.0. The ventricular systolic function analysis involved contouring the blood pool at end diastole and end systole at each level data set, thereby quantifying the end-diastolic volume (EDV) and end-systolic volume (ESV). The stroke volume is defined as the difference between the EDV and the ESV, and the EF is defined as (stroke volume/EDV)×100.
Six weeks after the initial surgery, all surviving animals underwent repeat MRI (n=9) as described previously. For the Melody valve group, the proximal RUL and LUL flows were acquired distal to the valve. Flow data also were acquired for the RUL branch of the RPA.
Statistical Analysis
Summary statistics are reported as mean±SD. Because nearly all catheter- and MRI-derived continuous variables are normally distributed (Shapiro-Wilk test for normality), parametric analyses were used. Differences in MRI measures between the baseline group and the Melody group were compared using paired t test (because these studies were on the same patients). Differences in MRI measures between the baseline group and the non-Melody group as well as differences between the Melody group and the non-Melody group were compared using unpaired t test. Because there were no differences between the Melody and non-Melody groups in the baseline hemodynamic variables obtained at cardiac catheterization, differences between these 2 groups at terminal catheterization were compared directly using unpaired t test. Statistical significance was established at P<0.05. All statistical analyses were performed using STATA version 10 (STATA Corp, College Station, TX) software.
Results
Ten Dorsett hybrid sheep were enrolled in the study, with 9 surviving the entire protocol. One animal in the Melody valve group died of hemorrhagic shock from a femoral arterial bleed in the recovery room immediately after surgery. The 2 groups of animals (Melody valve group, n=4; non-Melody valve group, n=5) were assessed by MRI and cardiac catheterization 6 weeks after the initial surgery. The preoperative cardiac catheterization (n=10) and MRI (n=5) were used as baseline control data for comparison.
Cardiac MRI Results
The cardiac MRI data are summarized in Figures 1 to 6. Six weeks after surgery, the mean MPA pulmonary RF (PRF%) measured at the level of the pulmonary valve annulus was significantly lower in the Melody valve group than in the non-Melody valve group (15±6% versus 37±3%; P<0.001). Figures 1 and 2 graphically show the PRF% and the relative contribution to the regurgitant volume made by each segment of the PA tree. In the Melody valve group, the unprotected RUL was responsible for the majority (64%) of the regurgitant volume measured at the MPA. The mean RUL RF was 81±20% (Figure 3A and 3B), contributing a mean volume per heartbeat of 4.8±2.1 mL to the total regurgitant volume per heartbeat of 7.5±4.0 mL in the MPA. The PA segments distal to the implanted Melody valves contributed very little to the regurgitant volume (mean RPA regurgitant volume, 0.6±0.3 mL; mean LPA regurgitant volume, 0.7±0.2 mL) (Figure 2), leaving 1.4 mL per heartbeat of regurgitant volume unaccounted for.
Figure 1.
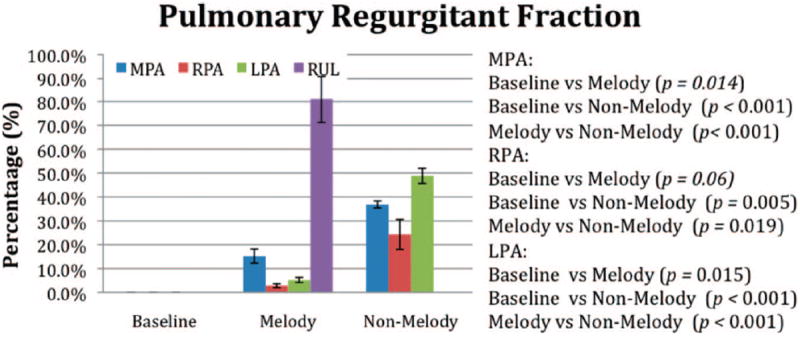
The RF [RF=(reverse flow/forward flow)×100] measured at various locations in the ovine PA tree 6 weeks after the initial surgery. Baseline indicates preoperative (control) animals.
Figure 6.
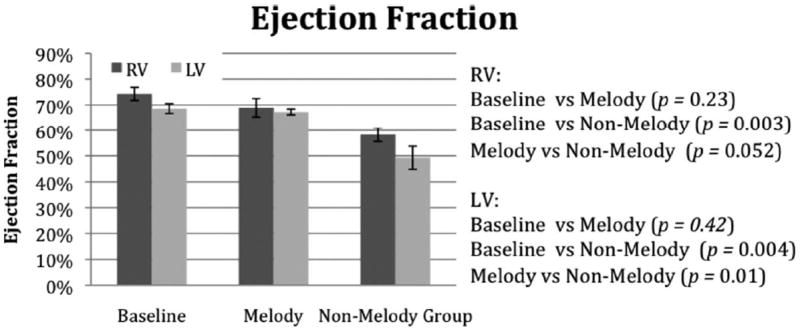
The RVEF and LVEF measurements made by cardiac MRI assessment at baseline and then again 6 weeks postoperatively.
Figure 2.
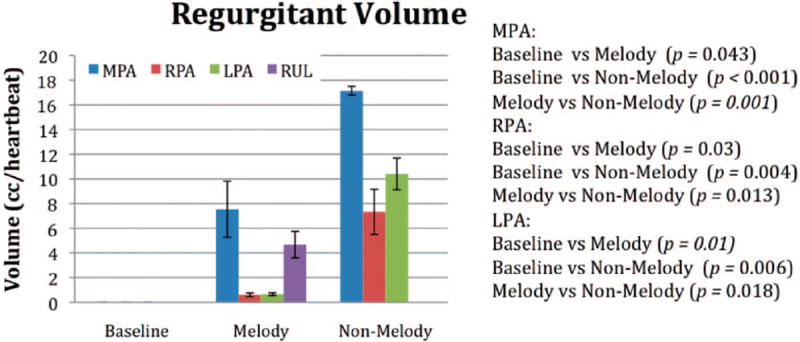
The volume of reverse flow per heartbeat from each PA segment. In the Melody valve group, the majority of reverse flow measured in the MPA originated from the RUL.
Figure 3.
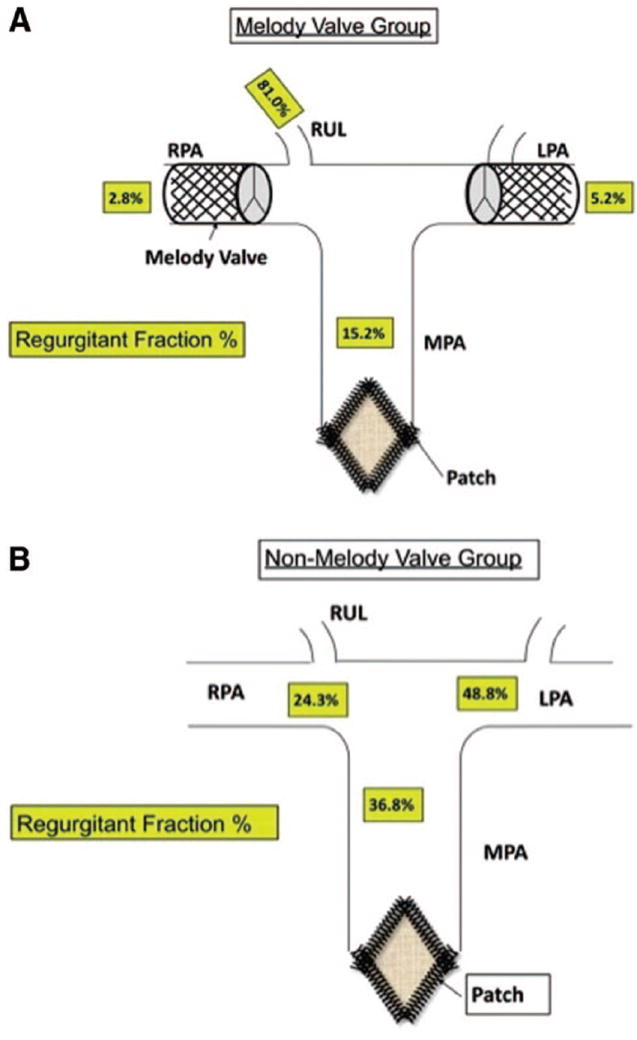
A, A schematic representation of the Melody valve group. Each animal underwent bilateral branch PA Melody valve implantation and surgical pulmonary valve resection with TAP placement. The mean PRF% from each arterial segment measured 6 weeks postoperatively is shown. The unprotected RUL artery had an PRF% of 81%. B, A schematic representation of the non-Melody valve group animals and mean PRF% from each arterial segment. Patch indicates TAP; regurgitant fraction%, mean PRF measured at each location in the PA tree assessed by MRI.
There were differences noted in ventricular size and function between the groups. The ventricular volumes for the RV and LV are shown in Figures 4 and 5, respectively. Compared with indexed baseline values, the RV volume measurements were increased in both the Melody and the non-Melody valve groups, with the non-Melody valve group having the largest RV EDV and ESV versus baseline (P=0.001). The Melody valve group had lower diastolic volumes compared to the non-Melody valve group (65.1±12 mL/m2 versus 81.6±9.8 mL/m2); however, the difference did not reach statistical significance (P=0.087). The RV ESV was significantly lower in the Melody valve group than in the non-Melody valve group (19.9±4 mL/m2 versus 34.5±8 mL/m2; P=0.019). In the LV, there were no significant differences in the diastolic volumes; however, the ESV was significantly higher in the non-Melody valve group (versus baseline, P=0.019; versus Melody valve, P=0.023). The corresponding ventricular EFs (RVEF and LVEF) were lower in the non-Melody valve group compared with both Melody valve and baseline values (Figure 6). The non-Melody valve group mean RVEF was 58±6% (versus baseline, P=0.003; versus Melody valve, P=0.05). The Melody valve group mean LVEF was significantly higher than that of the non-Melody group (67±2% versus 49±10%; P=0.01).
Figure 4.
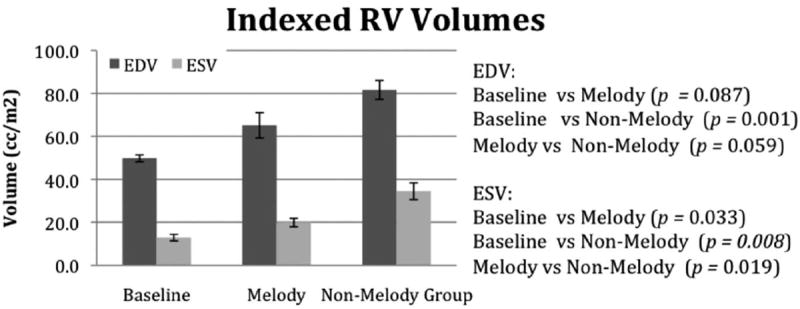
The RV diastolic and systolic volumes measured in the unoperated animals (baseline) and then again 6 weeks postoperatively in the Melody and non-Melody valve groups.
Figure 5.
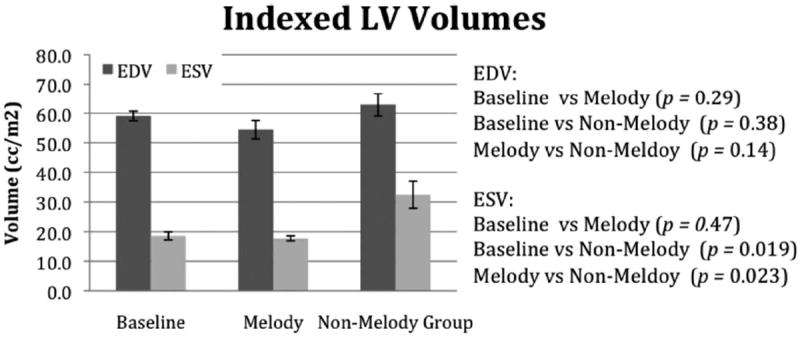
The LV diastolic and systolic volumes measured in the unoperated animals (baseline) and then again 6 weeks postoperatively in the Melody and non-Melody valve groups.
Cardiac Catheterization Results
Animals in each group underwent cardiac catheterization for hemodynamic assessment preoperatively (n=10) and again 6 weeks postoperatively at the time of the terminal MRI (Melody valve group, n=4; non-Melody valve group, n=5). The hemodynamics are summarized in Table. There were no significant differences in the baseline hemodynamics between the Melody valve and non-Melody valve groups and no difference within the Melody valve group at the terminal catheterization compared with baseline. However, there were significant differences noted between the groups at the terminal catheterization (Table).
Table.
Terminal Catheterization Data
| Variable | Non-Melody Valve Group (n=5) | Melody Valve Group (n=4) | P |
|---|---|---|---|
| Aortic systolic pressure, mm Hg | 138.2±16.8 | 133±17.6 | 0.67 |
| Aortic diastolic pressure, mm Hg | 106±12.8 | 92.5±7.7 | 0.06 |
| Aortic mean pressure, mm Hg | 119±14 | 108±9.1 | 0.21 |
| Heart rate, bpm | 105±9.1 | 91±12.4 | 0.08 |
| MPA systolic pressure, mm Hg* | 32.8±3.6 | 29±6.3 | 0.28 |
| MPA diastolic pressure, mm Hg* | 18.4±1.7 | 6.3±3 | 0.0001 |
| MPA mean pressure, mm Hg* | 24.6±0.9 | 16.7±6.1 | 0.02 |
| LPA systolic pressure, mm Hg | 32.8±3.6 | 26.5±7.1 | 0.13 |
| LPA diastolic pressure, mm Hg | 18.4±1.7 | 14.5±3.1 | 0.046 |
| LPA mean pressure, mm Hg | 24.6±0.9 | 18.8±5.4 | 0.048 |
| RLL systolic pressure, mm Hg | 32.8±3.6 | 26.5±8.1 | 0.16 |
| RLL diastolic pressure, mm Hg | 18.4±1.7 | 15.3±4.3 | 0.18 |
| RLL mean pressure, mm Hg | 24.6±0.9 | 20±5.5 | 0.102 |
| PCWP mean, mm Hg | 19.4±1.9 | 12±4.5 | 0.01 |
| RV systolic pressure, mm Hg | 38.2±2.2 | 30.5±6.1 | 0.03 |
| RV diastolic pressure, mm Hg | 15.6±4.8 | 5.8±2.5 | 0.007 |
| RA pressure mean, mm Hg | 18.2±2.5 | 9.8±4.1 | 0.006 |
| Cardiac output, L/min | 3.8±1 | 3.3±0.5 | 0.42 |
| PVR, Wood units | 1.4±0.3 | 2.1±0.4 | 0.04 |
The hemodynamic data (mean±SD) from the terminal catheterization procedure, which was performed 6 weeks after the initial surgery. There were no significant differences in the baseline hemodynamics at the preoperative catheterization between the 2 groups. PCWP indicates pulmonary capillary wedge pressure; RLL, right lower lobe; RA, right atrium.
The RUL arterial pressures were the same as the MPA in the non-Melody group.
The right- and left-sided filling pressures in the non-Melody valve group were significantly elevated compared to those of the Melody valve group, as was the central venous pressure. There was no significant difference noted in cardiac output.
In the Melody valve group, there was no significant difference noted in systolic pressure between the MPA and the distal PAs (P=0.14), meaning that there was no gradient across the implanted Melody valves. However, diastolic pressure distal to the valves was significantly higher compared with the MPA (P<0.01). The pulmonary vascular resistance (PVR) was higher in the Melody group, but the absolute values were within normal limits (for humans) for both. Angiographically, the Melody valves appeared competent and unobstructed (Figures 7 and 8).
Figure 7.
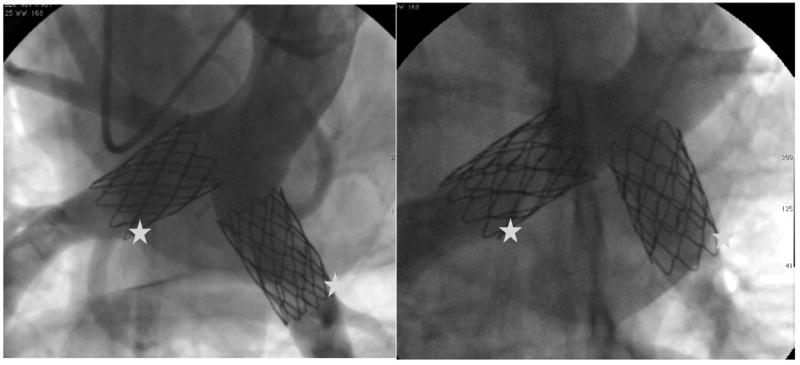
MPA angiogram taken 6 weeks after bilateral Melody valve implantation in 2 separate animals shows 1 device positioned within each branch PA. The stars mark the Melody valves.
Figure 8.
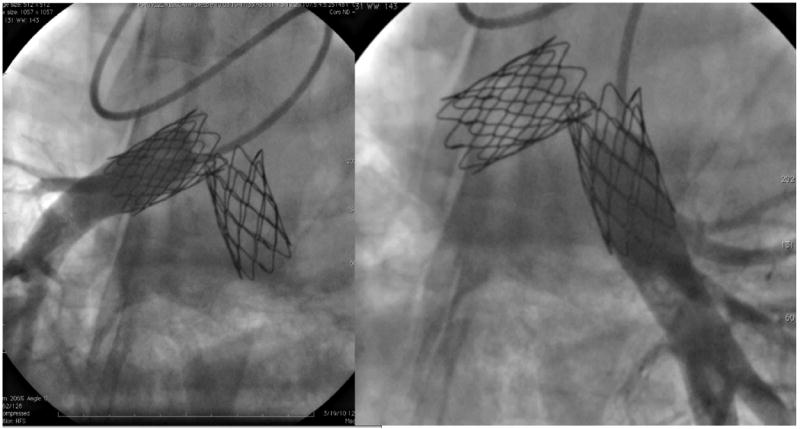
Bilateral branch pulmonary arteriograms taken 6 weeks after Melody valve implantation into the right and left branch PAs, respectively. The contrast injection is distal to the valves, revealing no regurgitation.
Discussion
Despite remarkable advances in transcatheter pulmonary valve replacement, <20% of patients with congenital heart disease who develop postoperative RVOT dysfunction (pulmonary insufficiency and obstruction) are currently eligible for treatment with existing approaches and technologies.14,23 Patients with chronic pulmonary insufficiency after surgical TAP repair of ToF are particularly underserved by existing techniques. In these patients, chronic regurgitation leads to massive dilation, dynamic distortion, and significant morphological variability of the RVOT,6,14,24 creating a truly inhospitable landing zone for any device, and is therefore untreatable with currently available transcatheter valves10,13,19,23 deployed in the usual position. Given this anatomic complexity, a single approach (or single device) for transcatheter pulmonary valve replacement is unlikely to work for all—or even most—patients, and the number of devices necessary to cover the full range of RVOT morphologies is difficult to know. Using advanced imaging and finite element analysis along with polymer models, Capelli et al21 described the development of a novel device and its applicability to 5 morphological RVOT subtypes (derived based on real patient data). They expanded this work beyond the theoretical realm by constructing a patient-specific device that was subsequently implanted with good results.20 This approach offers an exciting glimpse into what the future of percutaneous valve therapy may hold. However, despite this promising work, we believe that RVOT morphological variability is likely to remain a significant clinical challenge, especially because RVOT abnormalities often are complicated by branch PA pathology4,14,25 that frequently requires stent placement for the alleviation of proximal stenosis. To maximize the number of available therapeutic options, alternative strategies for treatment of pulmonary insufficiency and obstruction, including unorthodox sites for valve implantation, should be pursued.
The purpose of the present study was to reproduce the RVOT dysfunction and asymmetry encountered clinically and to test the effect that implantation of 2 Melody valves (1 into each branch PA) would have on pulmonary regurgitation into the RV. We accomplished 2 main goals. First, we demonstrated that RVOT dilation and distortion consistent with TAP repair of ToF could be reliably mimicked with our animal model. Second, we showed that Melody valve implantation into the right and left branch PAs resulted in a significant reduction in pulmonary regurgitation, with preserved biventricular function demonstrated by MRI and catheterization.
When compared with the non-Melody valve group, animals in the Melody valve group had significantly less PRF% (P=0.001) and, as a result, had less RV dilation. It seems obvious that the insertion of Melody valves into the branch PAs—unorthodox though it may be—was responsible for this finding. In other words, the Melody valves reduced PRF% by virtue of being “valves.” However, there are other potential explanations for the difference, but none that are easily assessed. For example, it is possible that the RV compliance and impedance to forward flow through the pulmonary vasculature (estimated by PVR),26,27 were higher in the non-Melody valve group. RV compliance was not quantified in this study, and PVR calculation in the non-Melody valve group was confounded by severe pulmonary insufficiency and, therefore, likely underestimated. Thus, we cannot definitively comment on their effect.
In addition to the reduced PRF%, we also found that the systolic function of both the RV and the LV was better in the Melody valve group as assessed by MRI (Figure 6). Furthermore, catheter measurements demonstrated that the animals in the Melody group had lower filling pressures on both the right and the left sides of the heart. The non-Melody valve animals had higher end diastolic pressure (wedge pressure) with no significant difference in EDV, suggesting that the LV was considerably less compliant (ie, stiffer) than in the Melody group. The higher PRF% in these animals also had an adverse impact on LV systolic function. Although the exact mechanism is unclear, 1 theory pertaining to the RV-LV interaction in patients with ToF is that LV contractile performance is diminished by leftward displacement of the ventricular septum. Septal displacement reduces LV preload and thus the recruitable stroke work, resulting in diminished contractility.28 In the present study, the LVEF was significantly lower in the non-Melody valve group, even though the LV diastolic volume did not differ significantly. Therefore, a reduced preload recruitable stroke work based on a diminished LV diastolic volume does not completely explain reduced LVEF. Perhaps, RV dilation contributed to LV systolic dysynchrony, as has been described in the echocardiographic literature.29 We are unable to comment of the issue of mechanical dysynchrony because it was beyond the scope of our study.
There were significant limitations encountered in this study, mostly due to the anatomic constraints of the ovine PA tree. Specifically, the segmental arteries supplying the RUL and LUL originate proximally from their respective source vessels. Because of these constraints, implantation of the Melody valves resulted in exclusion of the LUL artery in all animals. We decided to leave the RUL unguarded by the Melody valves to a variable degree in each animal for fear that if we jailed both upper lobes completely with the covered Melody device, the animals would have inadequate effective PBF and, thus, might not survive. The end result of this strategy was that the PA branch to the RUL was unprotected and, thus, was subject to increased regurgitation relative to the PA tree distal to the Melody valves (mean RUL RF, 81%) (Figure 3A). The amount of regurgitation at the MPA correlated significantly with the reverse flow from the RUL. If one controls for this RUL reverse flow volume by subtracting it from the total reverse flow measured at the level of the pulmonary valve, the mean RF% decreased dramatically to 4%—a trivial amount.
In an ideal setting, all segments of the PA arterial tree would be distal to the valve, and the covered frame of the implanted device would exclude no arterial segments. This strategy was not possible with the current technology in our ovine model, but it is one that we are exploring. Human postoperative anatomy is more variable, but comparatively, it may be more forgiving in terms of the size, shape, and length of the proximal branch PA landing zone. It is conceivable, that branch PA implantation of the current generation of Melody valves is achievable in humans now, without detrimental exclusion of segmental PA branches.
A question that remains to be answered is the long-term fate of the MPA segment distal to the native annulus and proximal to the branch PA Melody valves. Our study was limited to a follow-up period of 6 weeks, so we were unable to make any meaningful conclusions in this regard. We found that even preoperatively, the MPA segment was dynamic in nature, with its size changing dramatically in systole and diastole. It would seem to act as a capacitance vessel that dilates and “charges” in systole and then shrinks as it “discharges” in diastole. Some of the discharged volume goes forward into the PAs, and in the absence of a functioning native pulmonary valve, some goes backward into the RV as pulmonary regurgitation. If the branch PA valves are functioning appropriately and all lung segments are distal to the valves themselves, this volume of blood should be the only source of reverse flow. We did not directly measure this “MPA capacitance volume”; however, by subtracting out reverse flow from the RUL from the reverse flow at the native pulmonary annulus, we found that it is a trivial amount. That said, it is possible that ongoing insufficiency from the MPA, even in small amounts, will lead to progressive dilation of that segment with increasing amounts of regurgitation and eventually to enlargement of the RV. Longer-term follow-up is needed to fully understand the implications of this phenomenon.
This study was an acute experiment to test the feasibility of implantation and short-term effects of branch PA Melody valves. As such, we are unable to say anything about the potential for RV remodeling in patients with chronic postoperative pulmonary insufficiency. In addition, we cannot comment on the durability of the Melody devices when deployed into this position.
Conclusions
In this study, we accurately modeled postoperative RVOT dysfunction and distortion following TAP repair and demonstrated the feasibility and potential benefits of Melody valve implantation into the proximal RPA and LPA. This strategy may be an alternative approach to surgical valve replacement in patients with pulmonary insufficiency after TAP repair of ToF in whom anatomic complexity precludes orthotopic percutaneous valve replacement.
There is ongoing, worldwide interest in developing devices and strategies to treat these complex issues percutaneously. Further study using anatomically appropriate preclinical models to design and evaluate new approaches to this vexing issue is warranted.
CLINICAL PERSPECTIVE.
Pulmonary insufficiency and consequent right ventricular dilation and dysfunction are common and important problems after transannular patch repair of tetralogy of Fallot. Transcatheter pulmonary valve therapy has provided a means of treating postoperative right ventricular outflow tract dysfunction (pulmonary insufficiency and obstruction) with less morbidity than standard surgical pulmonary valve replacement. However, most patients with chronic pulmonary insufficiency after transannular patch repair of tetralogy of Fallot are ineligible for treatment with the current approaches and technologies because of severe right ventricular outflow tract dilation and distortion, which develops over time, often with concomitant branch pulmonary artery pathology. This anatomic complexity makes it technically impossible to implant currently available and approved valves in the orthotopic (usual) pulmonary position through a transcatheter approach, and therefore, surgery is required to restore pulmonary valve competence. The purpose of this study was to explore alternative locations within the pulmonary arterial tree for valve implantation. We demonstrated that Melody valve insertion in the branch pulmonary arteries was feasible and effective at reducing pulmonary insufficiency and had a favorable effect on both right and left ventricular function in the short term. We currently are investigating the effectiveness of this approach in a chronic model. The ultimate goal of this project is to expand the choices that are available to clinicians charged with treating these complex patients.
Acknowledgments
Sources of Funding
An American Heart Association Beginning Grant In Aid (to Dr Gillespie) funded this research. Drs R. Gorman and J. Gorman are supported by Individual Established Investigator Awards from the American Heart Association.
Footnotes
Disclosures
Medtronic Inc (Minneapolis, MN) donated the Melody valves for this study.
Melody is a trademark of Medtronic, Inc (Minneapolis, MN).
References
- 1.Therrien J, Provost Y, Merchant N, Williams W, Colman J, Webb G. Optimal timing for pulmonary valve replacement in adults after tetralogy of Fallot repair. Am J Cardiol. 2005;95:779–782. doi: 10.1016/j.amjcard.2004.11.037. [DOI] [PubMed] [Google Scholar]
- 2.Horneffer PJ, Zahka KG, Rowe SA, Manolio TA, Gott VL, Reitz BA, Gardner TJ. Long-term results of total repair of tetralogy of Fallot in childhood. Ann Thorac Surg. 1990;50:179–183. doi: 10.1016/0003-4975(90)90728-o. discussion 183–175. [DOI] [PubMed] [Google Scholar]
- 3.Gatzoulis MA, Balaji S, Webber SA, Siu SC, Hokanson JS, Poile C, Rosenthal M, Nakazawa M, Moller JH, Gillette PC, Webb GD, Redington AN. Risk factors for arrhythmia and sudden cardiac death late after repair of tetralogy of Fallot: a multicentre study. Lancet. 2000;356:975–981. doi: 10.1016/S0140-6736(00)02714-8. [DOI] [PubMed] [Google Scholar]
- 4.Rothman A, Perry SB, Keane JF, Lock JE. Early results and follow-up of balloon angioplasty for branch pulmonary artery stenoses. J Am Coll Cardiol. 1990;15:1109–1117. doi: 10.1016/0735-1097(90)90250-s. [DOI] [PubMed] [Google Scholar]
- 5.Kavey RE, Thomas FD, Byrum CJ, Blackman MS, Sondheimer HM, Bove EL. Ventricular arrhythmias and biventricular dysfunction after repair of tetralogy of Fallot. J Am Coll Cardiol. 1984;4:126–131. doi: 10.1016/s0735-1097(84)80329-0. [DOI] [PubMed] [Google Scholar]
- 6.Bove EL, Byrum CJ, Thomas FD, Kavey RE, Sondheimer HM, Blackman MS, Parker FB., Jr The influence of pulmonary insufficiency on ventricular function following repair of tetralogy of Fallot. Evaluation using radionuclide ventriculography. J Thorac Cardiovasc Surg. 1983;85:691–696. [PubMed] [Google Scholar]
- 7.Gatzoulis MA, Till JA, Somerville J, Redington AN. Mechanoelectrical interaction in tetralogy of Fallot. QRS prolongation relates to right ventricular size and predicts malignant ventricular arrhythmias and sudden death. Circulation. 1995;92:231–237. doi: 10.1161/01.cir.92.2.231. [DOI] [PubMed] [Google Scholar]
- 8.Bonhoeffer P, Boudjemline Y, Saliba Z, Hausse AO, Aggoun Y, Bonnet D, Sidi D, Kachaner J. Transcatheter implantation of a bovine valve in pulmonary position: a lamb study. Circulation. 2000;102:813–816. doi: 10.1161/01.cir.102.7.813. [DOI] [PubMed] [Google Scholar]
- 9.Bonhoeffer P, Boudjemline Y, Qureshi SA, Le Bidois J, Iserin L, Acar P, Merckx J, Kachaner J, Sidi D. Percutaneous insertion of the pulmonary valve. J Am Coll Cardiol. 2002;39:1664–1669. doi: 10.1016/s0735-1097(02)01822-3. [DOI] [PubMed] [Google Scholar]
- 10.Khambadkone S, Nordmeyer J, Bonhoeffer P. Percutaneous implantation of the pulmonary and aortic valves: indications and limitations. J Cardiovasc Med (Hagerstown) 2007;8:57–61. doi: 10.2459/01.JCM.0000247437.05194.8e. [DOI] [PubMed] [Google Scholar]
- 11.Zahn EM, Hellenbrand WE, Lock JE, McElhinney DB. Implantation of the melody transcatheter pulmonary valve in patients with a dysfunctional right ventricular outflow tract conduit early results from the U.S. clinical trial. J Am Coll Cardiol. 2009;54:1722–1729. doi: 10.1016/j.jacc.2009.06.034. [DOI] [PubMed] [Google Scholar]
- 12.McElhinney DB, Hellenbrand WE, Zahn EM, Jones TK, Cheatham JP, Lock JE, Vincent JA. Short- and medium-term outcomes after transcatheter pulmonary valve placement in the expanded multicenter US Melody valve trial. Circulation. 122:507–516. doi: 10.1161/CIRCULATIONAHA.109.921692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Khambadkone S, Coats L, Taylor A, Boudjemline Y, Derrick G, Tsang V, Cooper J, Muthurangu V, Hegde SR, Razavi RS, Pellerin D, Deanfield J, Bonhoeffer P. Percutaneous pulmonary valve implantation in humans: results in 59 consecutive patients. Circulation. 2005;112:1189–1197. doi: 10.1161/CIRCULATIONAHA.104.523266. [DOI] [PubMed] [Google Scholar]
- 14.Schievano S, Coats L, Migliavacca F, Norman W, Frigiola A, Deanfield J, Bonhoeffer P, Taylor AM. Variations in right ventricular outflow tract morphology following repair of congenital heart disease: implications for percutaneous pulmonary valve implantation. J Cardiovasc Magn Reson. 2007;9:687–695. doi: 10.1080/10976640601187596. [DOI] [PubMed] [Google Scholar]
- 15.Bove EL, Kavey RE, Byrum CJ, Sondheimer HM, Blackman MS, Thomas FD. Improved right ventricular function following late pulmonary valve replacement for residual pulmonary insufficiency or stenosis. J Thorac Cardiovasc Surg. 1985;90:50–55. [PubMed] [Google Scholar]
- 16.Boudjemline Y, Agnoletti G, Bonnet D, Sidi D, Bonhoeffer P. Percutaneous pulmonary valve replacement in a large right ventricular outflow tract: an experimental study. J Am Coll Cardiol. 2004;43:1082–1087. doi: 10.1016/j.jacc.2003.10.037. [DOI] [PubMed] [Google Scholar]
- 17.Boudjemline Y, Schievano S, Bonnet C, Coats L, Agnoletti G, Khambadkone S, Bonnet D, Deanfield J, Sidi D, Bonhoeffer P. Off-pump replacement of the pulmonary valve in large right ventricular outflow tracts: a hybrid approach. J Thorac Cardiovasc Surg. 2005;129:831–837. doi: 10.1016/j.jtcvs.2004.10.027. [DOI] [PubMed] [Google Scholar]
- 18.Dittrich S, Gloeckler M, Arnold R, Sarai K, Siepe M, Beyersdorf F, Schlensak C. Hybrid pulmonary valve implantation: injection of a self-expanding tissue valve through the main pulmonary artery. Ann Thorac Surg. 2008;85:632–634. doi: 10.1016/j.athoracsur.2007.08.010. [DOI] [PubMed] [Google Scholar]
- 19.Momenah TS, El Oakley R, Al Najashi K, Khoshhal S, Al Qethamy H, Bonhoeffer P. Extended application of percutaneous pulmonary valve implantation. J Am Coll Cardiol. 2009;53:1859–1863. doi: 10.1016/j.jacc.2008.08.061. [DOI] [PubMed] [Google Scholar]
- 20.Schievano S, Taylor AM, Capelli C, Coats L, Walker F, Lurz P, Nordmeyer J, Wright S, Khambadkone S, Tsang V, Carminati M, Bonhoeffer P. First-in-man implantation of a novel percutaneous valve: a new approach to medical device development. EuroIntervention. 5:745–750. doi: 10.4244/eijv5i6a122. [DOI] [PubMed] [Google Scholar]
- 21.Capelli C, Taylor AM, Migliavacca F, Bonhoeffer P, Schievano S. Patient-specific reconstructed anatomies and computer simulations are fundamental for selecting medical device treatment: application to a new percutaneous pulmonary valve. Philos Transact A Math Phys Eng Sci. 368:3027–3038. doi: 10.1098/rsta.2010.0088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Berman A. Effects of body surface area estimates on predicted energy requirements and heat stress. J Dairy Sci. 2003;86:3605–3610. doi: 10.3168/jds.S0022-0302(03)73966-6. [DOI] [PubMed] [Google Scholar]
- 23.Garay F, Webb J, Hijazi ZM. Percutaneous replacement of pulmonary valve using the Edwards-Cribier percutaneous heart valve: first report in a human patient. Catheter Cardiovasc Interv. 2006;67:659–662. doi: 10.1002/ccd.20753. [DOI] [PubMed] [Google Scholar]
- 24.Schievano S, Migliavacca F, Coats L, Khambadkone S, Carminati M, Wilson N, Deanfield JE, Bonhoeffer P, Taylor AM. Percutaneous pulmonary valve implantation based on rapid prototyping of right ventricular outflow tract and pulmonary trunk from MR data. Radiology. 2007;242:490–497. doi: 10.1148/radiol.2422051994. [DOI] [PubMed] [Google Scholar]
- 25.Shaffer KM, Mullins CE, Grifka RG, O’Laughlin MP, McMahon W, Ing FF, Nihill MR. Intravascular stents in congenital heart disease: short- and long-term results from a large single-center experience. J Am Coll Cardiol. 1998;31:661–667. doi: 10.1016/s0735-1097(97)00535-4. [DOI] [PubMed] [Google Scholar]
- 26.Chaturvedi RR, Redington AN. Pulmonary regurgitation in congenital heart disease. Heart. 2007;93:880–889. doi: 10.1136/hrt.2005.075234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Redington AN, Oldershaw PJ, Shinebourne EA, Rigby ML. A new technique for the assessment of pulmonary regurgitation and its application to the assessment of right ventricular function before and after repair of tetralogy of Fallot. Br Heart J. 1988;60:57–65. doi: 10.1136/hrt.60.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brookes C, Ravn H, White P, Moeldrup U, Oldershaw P, Redington A. Acute right ventricular dilatation in response to ischemia significantly impairs left ventricular systolic performance. Circulation. 1999;100:761–767. doi: 10.1161/01.cir.100.7.761. [DOI] [PubMed] [Google Scholar]
- 29.Kapetanakis S, Kearney MT, Siva A, Gall N, Cooklin M, Monaghan MJ. Real-time three-dimensional echocardiography: a novel technique to quantify global left ventricular mechanical dyssynchrony. Circulation. 2005;112:992–1000. doi: 10.1161/CIRCULATIONAHA.104.474445. [DOI] [PubMed] [Google Scholar]


