Abstract
Herein we considered the role of oxidative stress on deficiencies of functional physical performance that could affect a future pre-frailty condition. Using principal component analyses (PCA), we created new variables to better describe the functionality regarding the physical performance of the upper and lower body limbs. Gait speed and the Short Physical Performance Battery (SPPB) score were classified by PCA to describe functional performance of the lower body limbs. Variables describing the general physical status, including weekly consumption of kilocalories and the musculoskeletal index, were classified together with grip strength of the dominant hand as indicators of functional performance of the upper body limbs. An intimate association between the functional physical performance of the upper body limbs and the total antioxidant capacity was observed in older subjects. Low levels of total antioxidant capacity were found in women 76 years or younger with deficiencies in the physical performance of both upper and lower body limbs. Similarly, we observed a close association between the functional physical performance of the lower body limbs and the levels of hemoglobin. In particular, low levels of hemoglobin were mostly found in men older than 76 years of age, showing impaired functional physical performance. In addition, the physical performance of the lower body limbs was shown to be more important than that of the upper body limbs in the statistical association with pre-frailty in the elderly. Therefore, specific low levels of hemoglobin and deficient oxidative defense in the elderly could significantly affect the functional physical performance and future outcomes of pre-frailty.
Keywords: Oxidative stress, Antioxidant defense, Hemoglobin, Physical performance, Pre-frailty, The FRADEA study
Introduction
Frailty is normally defined as a biologic syndrome of decreased reserve and resistance to stressors, resulting from a cumulative decline across multiple physiologic systems, leading to loss of homeostatic capability and vulnerability to adverse outcomes (Campbell and Buchner 1997). Fried et al. (2001) described the phenotype of frailty for the first time. Frailty is defined by the presence of three or more Fried’s criteria, specifically unintentional weight loss, weakness, exhaustion, slow walking speed, or low level of physical activity. These measures have been demonstrated to be good criteria and have been shown to be valid in different cohorts of elderly people (Bandeen-Roche et al. 2006). The prevalence of frailty can range from 4 to 23.1 % according to different studies, depending on the setting, sociodemographic conditions, and the criteria used (Fried et al. 2001, 2004; Santos-Eggimann et al. 2009; Alcala et al. 2010; Abizanda et al. 2011a, b; Garcia-Garcia et al. 2011). In particular, frailty increases exponentially with age, reaching approximately 23.1 % at 90 years of age (Fried et al. 2001; Fernandez-Bolanos et al. 2008; Jurschik Gimenez et al. 2011). The frailty syndrome is considered to be an important predictor of adverse outcomes, such as death (up to 45 % a year), institutionalization, falls, worsening mobility, increased dependence on basic and instrumental activities of daily living, and hospitalization in elderly people (Fried et al. 2004; Bandeen-Roche et al. 2006). Therefore, frailty is considered to be one of the main preceding adverse conditions before the development of disability and/or functional dependence in the elderly (Bandeen-Roche et al. 2006; Topinkova 2008).
Recently, the operative criteria for frailty proposed by Fried et al. (2001) were applicable to an elderly Spanish population by way of studies on frailty and dependence in Albacete (the FRADEA study), which is considered to be representative of an urban population in Spain (Abizanda et al. 2011b, 2013). In this study, the prevalence of frailty in the cohort was 16.9 %, increasing to 26.4 % in people showing moderate to severe disability (Abizanda et al. 2011a, b, 2013). The frailty phenotype in the FRADEA cohort was independently associated with age, female gender, and disability in basic activities of daily living, high comorbidity, depression, and cognitive status (Abizanda et al. 2011b). Frail subjects in the FRADEA study presented a higher risk of exhibiting adverse outcomes, including incident disability in basic and instrumental activities of daily living, mobility impairment, a higher risk of hospitalization, and most importantly, a higher risk of mortality (Abizanda et al. 2011b, 2013). Notably, a high prevalence (ranging from 48.5 to 48.8 %) of a previous state of frailty, known as the pre-frailty condition, was found in this elderly Spanish population (Abizanda et al. 2011a, b). This pre-frail phenotype was defined as the presence of one or two of Fried’s criteria (Fried et al. 2001). The pre-frail elderly have an increased risk of becoming frail within 3 years (Fried et al. 2004).
The clinical phenotype of frailty can be associated with common age-related pathologic situations, which suggest a possible pathogenesis in the frailty syndrome involving hormonal deregulation, immune-aging, and pro-coagulative and pro-inflammatory status (Topinkova 2008). It should be noted that an age-related chronic inflammation state has also been described as a good predictor of adverse outcomes, such as 1-year hospitalization and mortality in the dependent elderly (de Gonzalo-Calvo et al. 2012a). Furthermore, we have recently described different biological oxidative stress-related parameters as potential blood clinical markers to define functional dependence in the institutionalized elderly (de Gonzalo-Calvo et al. 2012b, c). In these studies, the functional decline in the dependent elderly was associated with increased levels of carbonylated proteins (de Gonzalo-Calvo et al. 2012c) as well as some specific pro-inflammatory biomarkers, such as interleukin-6 (IL-6) and the soluble tumor necrosis factor receptor I (s-TNF-RI) (de Gonzalo-Calvo et al. 2012b). Serum levels of IL-6 and circulating levels of C-reactive protein have also been associated with frailty (Balducci 2007). Therefore, in the present work, we tested the hypothesis that deregulation of common clinical parameters and oxidative stress-related markers is associated with specific deficiencies of the functional physical performance of the lower or upper body limbs in the elderly population. In particular, we studied levels of carbonylated protein, lipid peroxidation, antioxidant capacity, C-reactive protein, hemoglobin levels, and total blood protein among elderly living in Albacete (Spain) and specifically included in the FRADEA urban cohort (study of frailty and dependence in Albacete). The ultimate objective of the present study was to identify a possible associative relationship of the pre-frail phenotype, which has a high prevalence in the FRADEA cohort, with the specific decline of the functional physical performance of the lower and/or upper body limbs.
Methods
Design and ethical aspects
The population (N = 200) was randomly selected from the concurrent study of the FRADEA cohort (frailty and dependence in Albacete). The inclusion criteria were age ≥ 70 years and living in Albacete city for at least 3 months a year. We have carried out a secondary data analysis of this representative age and gender-stratified cohort, whose methodology and design have been fully described in previous studies (Abizanda et al. 2011a, b, 2012, 2013). The only exclusion criteria considered was refusal to participate in the study or to sign the informed consent form. Geriatricians carried out the initial evaluations, which included an exhaustive medical and pharmacological history review and physical examination of each patient. Each participant or participant’s guardian received information about the purposes and objectives of the study and signed an informed consent form. The performance tests to assess functional limitations were conducted on the same day as the interview by geriatricians in the geriatric day hospital unit or by nurses when this assessment was performed at the participant’s home. The information was provided by the participants themselves or by the main caregiver if the participant was unable to do so. Four trained nurses completed the questionnaires and administered the Barthel and Lawton indexes, Charlson Index, Mini-Mental State Examination (MMSE), and Yesavage’s Geriatric Depression Scale (GDS). The reliability and validity of all these questionnaires (Barthel and Lawton indexes, Charlson Index, MMSE, and GSD) have been previously demonstrated in Spanish elderly population (Baztán et al. 1993; Bermejo et al. 1999; Martí et al. 2000; Miralles Basseda and Esperanza Sanjuán 2007; Torres Moreno et al. 2009). The study complied with the Declaration of Helsinki. It was also approved by the Institutional Review Board of the Albacete Health Area and the Clinical Research Committee of the University Hospital of Albacete.
Instrumental tests related to physical fitness status and functional performance
The methodology for application of the assessment tools used and the bibliographical references can be found in the original and previous studies of FRADEA (Abizanda et al. 2011a, b, 2012, 2013). The assessment of balance and gait were studied by the speed of walking 4 m at normal pace in meters per second (m/s) (Podsiadlo and Richardson 1991; Abizanda et al. 2011a). Gait speed is both a predictor of adverse outcomes and an indicator of physical frailty in the elderly (Gill 2010; Peel et al. 2013), and it can be analyzed alone and/or as part of the short physical performance battery (SPPB). This way, we assayed independently both values of gait speed and the SPPB score as both are considered valid for assessments of the functional performance and mobility in the elderly (Guralnik et al. 1994; Landi et al. 2007a, b; Wells et al. 2012). The SPPB score evaluates balance, gait, strength, and endurance by testing one’s ability to stand with feet together in three positions (i.e., side by side, semi-tandem, and tandem), time required to walk 8 ft, and time required to rise from a chair and return to the seated position five times (Guralnik et al. 1994; Landi et al. 2007b; Abizanda et al. 2011a, b, 2012). Subjects unable to perform or complete the five chair stands were not considered for analysis. The physical fitness status was studied considering the following variables: muscle strength by measurement of the grip strength of the dominant hand using a JAMAR® digital handheld dynamometer and the number of kilocalories expended weekly, which was obtained from information given by the patient using the Calcumed® instrument, as previously described (Abizanda et al. 2011a). The Calcumed® estimates the number of hours of sleep, very low-intensity physical activity (e.g., sitting, standing, driving, sewing, playing instruments, ironing), low-intensity physical activity (e.g., walking 4 to 5 km/h, housework, cooking, hand-laundry, shopping, golf, table tennis, sailing), moderate physical activity (e.g., walking 5.5 to 6.5 km/h, hard shopping, bicycling, dancing, tennis, skiing, mopping the floor, moderate weight load), and hard physical activity (e.g., hard weight load or work, basketball, swimming, climbing, football) and transforms these activities in energy expenditure (kcal) with an algorithmic method (Abizanda et al. 2011a). The physical fitness status also considers the muscular mass in kilograms (kg), which was calculated using the Janssen equation, as previously described (Abizanda et al. 2011a), and the musculoskeletal index (MSI), which was calculated by dividing the muscular mass by height in square meters (kg/m2) (Abizanda et al. 2011a).
The Barthel and Lawton indexes were used to study the functional abilities of the participants and to identify disabilities in performing basic or instrumental activities of daily living (Mahoney and Barthel 1965; Lawton and Brody 1969). The Barthel index is a ten-item scale of the following items: feeding, grooming, bathing, toilet use, dressing, walking, transfers, climbing stairs, fecal incontinence, and urinary incontinence. The highest score is 100 (independence) and the lowest is 0 (total dependence). Groups of different functional dependence were described based on previous criteria from expert geriatricians: an independent group (100–95), a moderately dependent group (94–60), and a severely dependent group (59–0) (de Gonzalo-Calvo et al. 2012c). The Lawton scale regarding instrumental activities of daily living assesses a person’s ability to perform eight tasks: food preparation, shopping, doing laundry, housekeeping, telephone use, responsibility for medications, mode of transportation, and handling finances (Lawton and Brody 1969; Graf 2008). Persons are scored according to their highest level of functionality in these categories. A summary score ranges from 0–4 (low function, dependent) and 5–7 (low function, moderately dependent) to 8 (high function, independent).
Frailty was studied using Fried’s frailty criteria (Fried et al. 2001), with some slight modifications (Abizanda et al. 2011a). Specifically, we assessed the following items: unintentional weight loss equal to or greater than 4.6 kg or equal to or greater than 5 % of body weight in the last year; weakness as measured by grip strength in the lowest 20 %, adjusted for gender and body mass index, according to Fried’s original data; poor energy and endurance, as indicated by self-reported exhaustion determined by two questions from the Center for Epidemiologic Studies-Depression (CES-D) scale according to Fried’s criteria; slowness, as determined by measuring the time taken to walk 4 m within the lowest 20th percentile, adjusted for gender and height according to Fried’s original data; low physical activity levels, determined by calculating the number of kilocalories expended weekly, within the lowest quintile for each gender with Fried’s original cutoff points. Subjects were considered as frail if three or more criteria were present and pre-frail if one or two were present (Abizanda et al. 2011a, b, 2012, 2013).
Study co-variables
Socio-demographic data including age and gender were collected. The anthropometric data considered included weight (kg), height (cm), and waist circumference (cm). The body mass index (kg/m2) including the individual’s weight divided by the square of their height was also calculated. The percentage of total fat mass was calculated by bioelectrical impedance analysis (BIA) using the Tanita® instrument (BC-418 MA® Instruments, Tanita Corporation, Japan), and the resting metabolic rate (kcal/day) was measured using the FitMate® instrument (COSMED, Italy) of indirect calorimetry (Abizanda et al. 2011a). Comorbidity was evaluated using Charlson Index, with a rating of 3 or higher considered to be high comorbidity (Charlson et al. 1987). Cognitive status was assayed using the MMSE, with a score of 24 or lower considered to indicate cognitive decline (Folstein et al. 1975). Risk of depression was considered to be present with a score of 5 or more on Yesavage’s GDS (Yesavage et al. 1983). Information regarding chronic diseases of the participants including diabetes, dyslipidemia, high arterial hypertension, and cardiac insufficiency, as well as the number of medications they were taking, was obtained from their medical records.
Biochemical analysis
Blood samples were obtained by venipuncture after an overnight fast and a 15-min rest in the morning. All venous blood samples were taken before 10:00 AM to preclude circadian variation. Blood samples were drawn into vacutainer tubes containing EDTA for plasma (BD, NJ, USA). After processing, the plasma was divided into aliquots and stored at −80 °C until further analysis, as previously described (de Gonzalo-Calvo et al. 2012a, b, c). Plasma protein concentrations were measured using Bradford method (Bradford 1976). Carbonylated proteins were determined by the method developed by Levine et al. (1990) with some modifications, as previously described (Coto-Montes and Hardeland 1999). Data are presented as nanomole of protein carbonyls per milligram of protein. Lipid peroxidation was assessed by measurement of the levels of reactive aldehydes, malondialdehyde (MDA), and 4-hydroxy-2(E)-nonenal (4-HNE) using the LPO Assay Kit from Calbiochem, as previously described (de Gonzalo-Calvo et al. 2012b). Results are expressed as nanomole (MDA + 4-HNE) per gram of protein. Plasma total antioxidant activity (TAA) was determined using the ABTS/H202/HRP method modified for plasma (de Gonzalo-Calvo et al. 2010, 2012b). Results are expressed in equivalents of milligrams of Trolox per gram of protein. Intra-assay coefficients of variation were 5.6 % for carbonylated proteins, 3.8 % for lipid peroxidation, and 4.3 % for TAA. Inter-assay coefficients of variations were 13.3 % for carbonylated proteins, 10.2 % for lipid peroxidation, and 10.5 % for TAA. Regarding clinical variables, hemoglobin levels (g/dL) were measured by an automated hematology analyzer ADVIA 2120 (Siemens), while the total blood proteins (g/dL) and C-reactive protein levels (mg/L) were measured using a modular system D and P, respectively (Roche).
Statistical analysis
The statistical software package SPSS 15.0 for Windows (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses. The principal component analysis (PCA) was applied to reduce our instrumental tests of functional performance, including parameters related to general physical fitness, balance, and gait, into a smaller set of principal components that account for most of the variance of the studied variables [gait speed at 4 m, SPPB score, grip strength of the dominant hand, weekly consumption of kilocalories, and musculoskeletal index]. The number of components retained were based on eigenvalues (the amount of the total variance that is explained by each component) of 1 or greater. A varimax rotation was used to obtain a set of independent and best interpretable components. The Varimax rotation method minimizes the number of variables that have high saturations in each component. It simplifies the interpretation of the components by optimizing the solution. The components were interpreted based on the loadings that relate the parameter to the components. Loadings greater than 0.5 were used to identify the variables comprising a component, and this cutoff point also provided good separation of the components. Principal component scores were calculated for each participant and standardized to yield a sample mean equal to 0. Independent cluster analyses were then applied for each PCA component, with the objective to classify each participant in two groups (poor versus good functional performance) according to the extreme values reached by the variables specifically included in each component of the PCA analysis.
Descriptive statistics were used to characterize the study population and to describe study parameters at baseline, including oxidative and clinical biomarkers. Data are presented as frequencies (percentages) for categorical variables and means (error standard of the mean) for continuous variables. Categorical variables were compared among groups with a different functional physical performance using the chi-squared test. Continuous variables were compared among groups using one-way analysis of variance (ANOVA) for normal samples. The non-parametric H of the Kruskall–Wallis test was used for samples without a normal distribution. The normality of the data was analyzed by the Kolmogorov–Smirnov test. Differences were always considered as statistically significant when p ≤ 0.05.
Bivariate logistic regression models were applied to study associations between each of the oxidative stress-related parameters and clinical variables with functional physical performance. These studies were carried out independently for the functional performance of the lower and upper body limbs. The models were adjusted for the covariate age, gender, anthropometric data (waist circumference, total fat mass, and body mass index, including also independent values of weight and height), the Barthel and Lawton indexes, risk of depression, and drug use (models 1 and 3) to take into account any potential confusion of these variables. To establish whether frailty could influence the observed associations of our biomarkers with functional physical performance, sequential logistic regression models were also adjusted for frailty (models 2 and 4). The results are presented as odds ratios (OR) and 95 % confidence interval (CI).
Finally, to establish associative models of pre-frailty and the functional physical performance of the lower or upper body limbs, univariate linear regression analysis was applied after excluding frail subjects. Covariates considered in this logistic regression analysis included age, gender, anthropometric data (waist circumference, total fat mass, and body mass index, including also independent values of weight and height), and the Barthel and Lawton indexes (model 1) to take into account any potential confusion of these variables. To establish whether risk of depression (mode 2) and drug use (model 3) could influence the associative analysis, sequential linear regression models were also adjusted for these variables. The results are presented as standardized betas (β) and 95 % CI.
Results
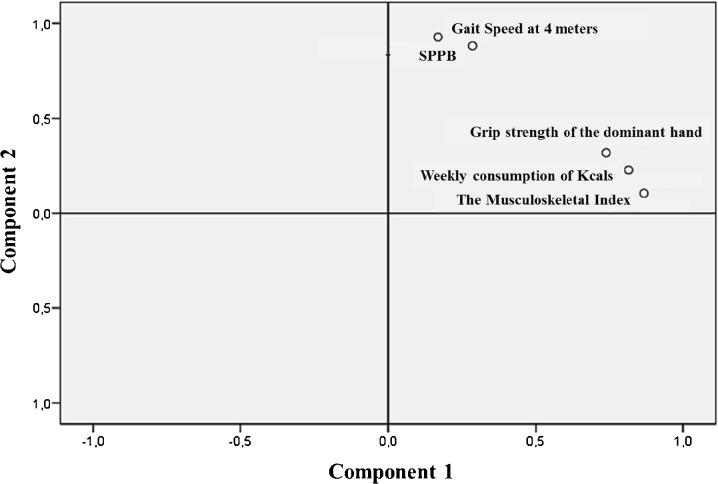
In order to reduce the number of instrumental tests related to functional performance, including balance and gait as well as physical fitness status, we applied the PCA statistical data reduction tool to create new variables that could provide, in a simple way, a complete description of the functional physical performance of the upper and lower body limbs (Fig. 1). The PCA resulted in two main eigenvalues greater than 1. These two components explained 77.6 % of the total variance data (57. 5 % component 1 and 20.2 % component 2). As shown in Table 1, three variables (grip strength of the dominant hand, the weekly consumption of kilocalories, and the musculoskeletal index) loaded highest on the first component (component 1). The other two variables (gait speed at 4 m and SPPB score) loaded highest on the second component (component 2). The Kaiser–Meyer–Olkin measure of sampling adequacy was 0.732, showing a good and appropriate use of the factorial analysis with our sample data. Likewise, the observed significance level of Bartlett’s test of sphericity was 0.00, which was considered to be a significant and relevant correlation between the variables and the factor model. We also considered including other clinical variables in the PCA (e.g., the resting metabolic rate); however, this rendered the factorial model unsuitable, and it explained less total variance (data not shown).
Fig. 1.
Graphic representation of the rotated component loadings of the PCA. Three variables (grip strength of the dominant hand, the weekly consumption of kilocalories, and the musculoskeletal index) loaded highest on the first PCA component (component 1), while two variables (gait speed at 4 m and SPPB) loaded highest in the second PCA component (component 2). SPPB short physical performance battery
Table 1.
Descriptive analysis of different instrumental variables related to functional performance and rotated component loading of the PCA
| Variables | Rotated component loadings | |
|---|---|---|
| Component 1 | Component 2 | |
| Gait speed | 0.286 | 0.882 |
| SPPB | 0.169 | 0.928 |
| Musculoskeletal Index | 0.868 | 0.106 |
| Grip strength of the dominant hand | 0.740 | 0.319 |
| Weekly consumption of kilocalories | 0.816 | 0.228 |
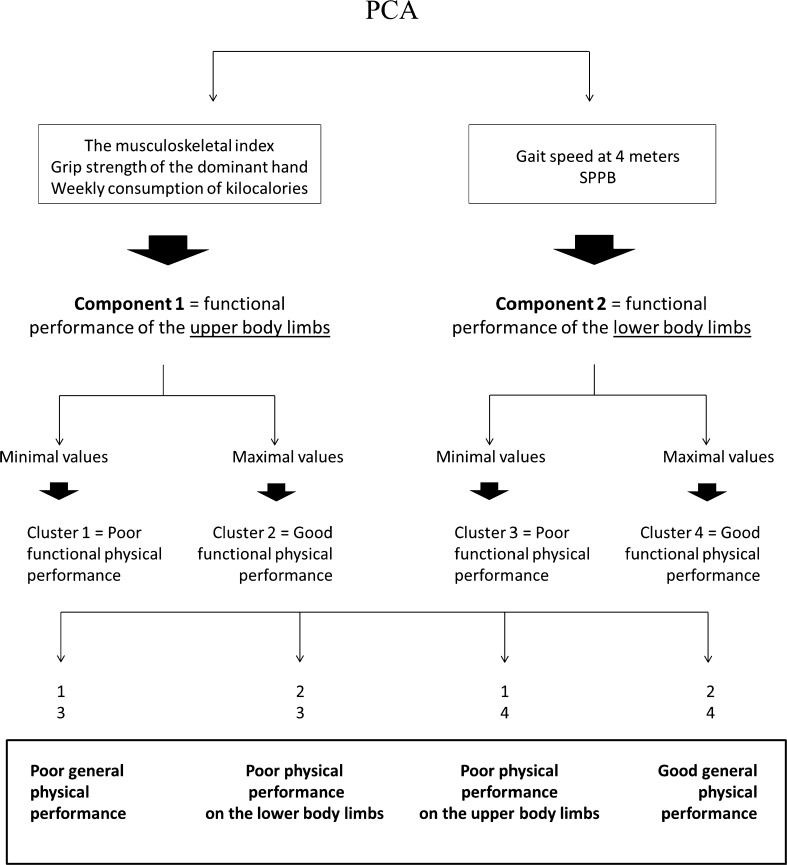
As shown in Fig. 2, cluster analysis was carried out to group our study population into 2 different functional groups depending on the physical performance of their upper and lower body limbs. For this purpose, subjects were classified into two groups (poor or good physical performance) according to the extreme values reached by the specific variables included in each component of the PCA. For instance, minimal values of grip strength of the dominant hand, the weekly consumption of kilocalories, and the musculoskeletal index were considered to be the centers of cluster 1, which was defined as “poor functional physical performance of the upper body limbs”. Maximal values of these variables were considered to be the centers of cluster 2, which was defined as “good functional physical performance of the upper body limbs”. Similarly, minimal values of gait speed at 4 m and SPPB in the study population were considered to be the centers of cluster 3, which was defined as “poor functional physical performance of the lower body limbs”. Maximal values of gait speed and SPPB were considered to be the centers of cluster 4, which was defined as “good functional physical performance of the lower body limbs”. Finally, after combining both clusters of each component derived from the PCA, we were able to classify our study population according to a new variable with four categories, describing different levels of functional physical performance: poor general physical performance, poor physical performance of the lower body limbs, poor physical performance of the upper body limbs, and good general physical performance (Fig. 2).
Fig. 2.
Summary showing the different statistical analyses applied to create a new variable to better describe functional physical performance in the elderly. After applying the PCA, we obtained two components to describe the functional physical performance of the upper (component 1) or lower (component 2) body limbs, as detailed in Fig. 1. The subjects were then classified in two groups (poor or good physical performance) using cluster analysis according to the extreme values reached by the specific variables included in each component of the PCA. After combining both clusters of each component derived from the PCA, we classified our study population according to a new variable with four categories, describing different levels of functional physical performance: poor general physical performance, poor physical performance of lower body limbs, poor physical performance of upper body limbs, and good general physical performance
As shown in Table 2, the study shows that 53 participants had good general physical performance, 62 participants had poor general physical performance, 67 participants had deficiencies in the physical performance of their upper body limbs, and only 18 participants had deficiencies in the physical performance of their lower body limbs. The mean age of the participants in the four groups was significantly different, with the older subjects (above 76 years) showing different impairments on physical performance. Men mostly showed good general physical performance (48.8 %), while women were mostly included (46.6 %) in the group with poor general physical performance. In both sexes, the upper body limbs were mainly affected by deficiencies in physical performance (males 28.6 % and females 37.1 %). As we expected, the anthropometric characteristics of the participants in the groups with different functional physical performance were also significantly different. Regarding clinical characteristics, we should note that disability in basic or instrumental activities of daily living, as described by Barthel and Lawton indexes, respectively, showed that our population is made up of mostly independent adults (49–77.5 % of the total population) or those with moderate dependence (20.5–41 % of the total population). Likewise, our participants mostly have a pre-frail phenotype (57.5 % of total population). Consequently, only 2 % of the total population had severe dependence, as indicated by Barthel and Lawton indexes, and only 11 % of the total population consisted of frail adults. Interestingly, we observed that among older adults showing a depressive mood, a high percentage of subjects had significant deficiencies in functional physical performance; in particular, 48.1 % had poor general physical performance and 32.7 and 7.7 % had deficiencies in functional physical performance on the upper and lower body limbs, respectively. In addition, the number of medications taken per subject was higher in the groups showing some deficiencies on physical performance as compared with the group with good performance (Table 2).
Table 2.
Baseline characteristics of the FRADEA population according to different levels of functional physical performance
| Poor general physical performance | Poor physical performance on the lower body limbs | Poor physical performance on the upper body limbs | Good general physical performance | P-value | |
|---|---|---|---|---|---|
| N = 62 | N = 18 | N = 67 | N = 53 | ||
| Age (years) | 78.1 (0.6) | 79.4 (0.9) | 76.8 (0.5) | 74.2 (0.4) | 0 |
| Gender | 0 | ||||
| Men | 8 (9.5) | 11 (13.1) | 24 (28.6) | 41 (48.8) | |
| Women | 54 (46.6) | 7 (6.0) | 43 (37.1) | 12 (10.3) | |
| Weight (kg) | 68.4 (1.5) | 81.1 (3.2) | 66.6 (1.1) | 77.8 (1.5) | 0 |
| Height (cm) | 151.2 (0.8) | 160.3 (1.6) | 155.2 (0.9) | 161.3 (1.1) | 0 |
| Waist circumference (cm) | 100.5 (1.3) | 108.1 (1.9) | 97.9 (1.6) | 105.1 (1.6) | 0 |
| BMI (kg/m2) | 29.9 (0.6) | 31.5 (1.0) | 27.7 (0.5) | 29.9 (0.6) | 0 |
| Total fat mass (%) | 35.2 (0.9) | 33.8 (1.3) | 32.3 (0.9) | 30.9 (1.1) | 0.018 |
| Resting metabolic rate (kcal/day) | 1,071.3 (37.4) | 1,010.4 (65.2) | 980.3 (40.8) | 1,108.4 (44.0) | 0.133 |
| Barthel Index (score) | 0.002 | ||||
| Independent (100–95) | 37 (23.9) | 15 (9.7) | 58 (37.4) | 45 (29) | |
| Moderately dependent (94–60) | 21 (51.2) | 3 (7.3) | 9 (22.0) | 8 (19.5) | |
| Severely dependent (59–0) | 4 (100) | – | – | – | |
| Lawton Index (score) | 0 | ||||
| Independent (8) | 34 (34.7) | 9 (9.2) | 42 (42.9) | 13 (13.3) | |
| Moderately dependent (5–7) | 14 (17.1) | 7 (8.5) | 23 (28) | 38 (46.3) | |
| Severely dependent (0–4) | 14 (70) | 2 (10) | 2 (10) | 2 (10) | |
| Frailty | 0 | ||||
| Non-frail | 3 (4.8) | 4 (6.3) | 25 (39.7) | 31 (49.2) | |
| Pre-frail | 39 (33.9) | 14 (12.2) | 41 (35.7) | 21 (18.3) | |
| Frail | 20 (90.9) | – | 1 (4.5) | 1 (4.5) | |
| Depressive mood (GSD) | 25 (48.1) | 4 (7.7) | 17 (32.7) | 6 (11.5) | 0.005 |
| Cognitive decline (MMSE) | 8 (34.8) | 2 (8.7) | 10 (43.5) | 3 (13) | 0.445 |
| High comorbidity (CCI) | 9 (39.1) | 1 (4.3) | 8 (34.8) | 5 (21.7) | 0.702 |
| Diabetes | 20 (39.2) | 2 (3.9) | 19 (37.3) | 10 (19.6) | 0.174 |
| Dyslipidemia | 29 (35.4) | 5 (6.1) | 33 (40.2) | 15 (18.3) | 0.056 |
| HTA | 49 (32.9) | 10 (6.7) | 51 (34.9) | 38 (25.5) | 0.201 |
| Cardiac insufficiency | 4 (80) | – | 1 (20) | – | 0.108 |
| Number of drugs used | 6.6 (0.4) | 5.6 (0.9) | 5.2 (0.3) | 3.3 (0.4) | 0 |
Data presented as means (standard error of the mean) for continuous variables or as frequencies (percentages) for categorical variables. P-values by one-way analysis of variance (ANOVA) or Kruskall–Wallis H Test for continuous variables according to normality as assayed by Kolmogorov–Smirnov test. P-values by chi-square test for categorical variables
BMI body mass index, CCI The Charlson Comorbidity Index, GSD geriatric depression scale of Yesavage, HTA high arterial hypertension, MMSE Mini-Mental State Examination of Folstein
Different biological markers related to oxidative stress (antioxidant capacity, carbonylated proteins, and lipid peroxidation) as well as common clinical markers (total blood proteins, hemoglobin, and C-reactive protein) were also studied in our population according to specific deficiencies of functional physical performance (Tables 3, 4, and 5). To get a clear understanding about the differences related to functional physical performance, these parameters were independently studied according to gender and age group (≤ 76 years, > 76 years). As shown in Table 3, women 76 years or younger showed significant differences in their total antioxidant capacity among groups with different functional physical performance. Given that the levels of total blood proteins were significantly different among the groups, we also calculated the percentages of the total antioxidant capacity with respect to the levels of total blood proteins. Notably, these percentages of total antioxidant capacity remained statistically significant among groups with different physical performance. However, women aged more than 76 years only showed differences in the levels of hemoglobin and C-reactive protein among groups with different functional physical performance (Table 4). Men 76 years or younger did not show significant differences in these biological and clinical markers among groups with different functional physical performance (data not shown). In contrast, men aged more than 76 years showed significant differences exclusively in the levels of hemoglobin among the groups with different physical performance (Table 5).
Table 3.
Oxidative stress-related variables and clinical blood parameters in women ≤76 years of age according to different levels of functional physical performance
| Poor general physical performance | Poor physical performance on the lower body limbs | Poor physical performance on the upper body limbs | Good general physical performance | P-value | |
|---|---|---|---|---|---|
| N = 24 | N = 2 | N = 22 | N = 9 | ||
| Total antioxidant capacity | 2.2 (0.2) | 3.4 (1.4) | 3.6 (0.5) | 2.5 (0.3) | 0.023 |
| (mg Trolox/g protein) | |||||
| % total antioxidant capacity (with respect to total blood protein levels) | 30.1 (2.5) | 49.6 (22.3) | 49.7 (6.6) | 36.8 (4.5) | 0.014 |
| Carbonylated proteins | 2.5 (0.2) | 2.7 (0.5) | 2.6 (0.1) | 2.1 (0.2) | 0.379 |
| (nmol carbonyls/mg protein) | |||||
| Lipid peroxidation | 20.1 (1.8) | 13.7 (1.9) | 22.6 (3.2) | 19.1 (3.9) | 0.686 |
| (nmol MDA + 4-HNE/g protein) | |||||
| Total blood proteins (g/dL) | 7.4 (0.1) | 6.9 (0.3) | 7.14 (0.1) | 6.9 (0.1) | 0.009 |
| C-reactive protein (mg/L) | 4.1 (0.7) | 4.0 (0.6) | 4.7 (2.5) | 3.9 (0.9) | 0.546 |
| Hemoglobin (g/dL) | 13.6 (0.2) | 13.1 (0.6) | 13.9 (0.2) | 13.7 (0.3) | 0.521 |
All data are presented as means (standard error of the mean). P-values by one-way analysis of variance (ANOVA) or the Kruskall–Wallis H test according to normality as assayed by Kolmogorov–Smirnov Test. Differences were always considered to be statistically significant when p ≤ 0.05
MDA malondialdehyde, 4-HNE 4-hydroxy-2(E)-nonenal
Table 4.
Oxidative stress-related variables and clinical blood parameters in women > 76 years of age according to different levels of functional physical performance
| Poor general physical performance | Poor physical performance on the lower body limbs | Poor physical performance on the upper body limbs | Good general physical performance | P-value | |
|---|---|---|---|---|---|
| N = 30 | N = 5 | N = 21 | N = 3 | ||
| Total antioxidant capacity | 2.4 (0.2) | 1.9 (0.3) | 2.2 (0.2) | 1.9 (0.3) | 0.788 |
| (mg Trolox/g protein) | |||||
| % total antioxidant capacity (with respect to total blood protein levels) | 33.9 (3.2) | 27.7 (4.4) | 31.1 (2.6) | 27.5 (3.1) | 0.82 |
| Carbonylated proteins (nmol carbonyls/mg protein) | 2.3 (0.1) | 2.5 (0.7) | 2.1 (0.1) | 2.0 (0.3) | 0.742 |
| Lipid peroxidation (nmol MDA + 4-HNE/g protein) | 19.2 (1.1) | 20.2 (4.6) | 21.8 (2.0) | 20.4 (1.6) | 0.698 |
| Total blood proteins (g/dL) | 7.2 (0.1) | 7.2 (0.2) | 7.3 (0.1) | 6.9 (0.2) | 0.608 |
| C-reactive protein (mg/L) | 5.9 (1.5) | 3.6 (3.1) | 2.7 (0.7) | 3.6 (1.0) | 0.029 |
| Hemoglobin (g/dL) | 12.9 (0.2) | 13.2 (0.3) | 13.9 (0.2) | 12.9 (0.5) | 0.037 |
All data are presented as means (standard error of the mean). P-values by one-way analysis of variance (ANOVA) or Kruskall–Wallis H test according to normality as assayed by Kolmogorov–Smirnov test. Differences were always considered to be statistically significant when p ≤ 0.05
MDA malondialdehyde, 4-HNE 4-hydroxy-2(E)-nonenal
Table 5.
Oxidative stress-related variables and clinical blood parameters in men > 76 years of age according to different levels of functional physical performance
| Poor general physical performance | Poor physical performance on the lower body limbs | Poor physical performance on the upper body limbs | Good general physical performance | P-value | |
|---|---|---|---|---|---|
| N = 6 | N = 9 | N =14 | N = 6 | ||
| Total antioxidant capacity | 2.7 (0.6) | 1.9 (0.2) | 2.1 (0.3) | 2.0 (0.3) | 0.738 |
| (mg Trolox/g protein) | |||||
| % Total antioxidant capacity (respect to total blood protein levels) | 37.9 (8.8) | 27.7 (2.9) | 30.4 (3.7) | 27.0 (4.5) | 0.749 |
| Carbonylated proteins (nmol carbonyls/mg protein) | 1.8 (0.2) | 2.5 (0.2) | 2.4 (0.2) | 2.1 (0.2) | 0.086 |
| Lipid peroxidation (nmol MDA + 4-HNE/g protein) | 21.9 (3.9) | 27.3 (3.2) | 21.2 (2.5) | 14.5 (3.4) | 0.102 |
| Total blood proteins (g/dL) | 7.1 (0.2) | 6.9 (0.2) | 6.9 (0.1) | 7.2 (0.1) | 0.588 |
| C-reactive protein (mg/L) | 2.9 (0.7) | 3.1 (0.9) | 5.3 (2.0) | 3.9 (1.7) | 0.743 |
| Hemoglobin (g/dL) | 13.3 (0.3) | 14.5 (0.2) | 13.7 (0.3) | 15.8 (0.4) | 0 |
All data are presented as means (standard error of the mean). P-values by one-way analysis of variance (ANOVA) or Kruskall–Wallis H test according to normality as assayed by Kolmogorov–Smirnov test. Differences were always considered to be statistically significant when p ≤ 0.05
MDA malondialdehyde, 4-HNE 4-hydroxy-2(E)-nonenal
Bivariate logistic regression models were used to examine the relationship between oxidative stress-related biomarkers and deficiencies of the functional physical performance of either the upper and lower body limbs, as summarized in Table 6. Considering patient age, gender, anthropometric characteristics, Barthel and Lawton indexes, drug use, and depressive mood, the total antioxidant capacity was the only parameter significantly associated with functional physical performance of the upper body limbs (Table 6, model 1). This relationship remained statistically significant after adjusting the model also for frailty (Table 6, model 2). In regards to the functional physical performance of the lower body limbs, the only parameter significantly associated was the levels of hemoglobin (Table 6, models 3 and 4). Notably, when participants with severe dependence, as indicated by Barthel and Lawton indexes, were excluded from the analysis, these associations remained statistically significant (data not shown).
Table 6.
Associations between levels of studied biomarkers with the functional physical performance of the upper and lower body limbs
| The upper body limbs | The lower body limbs | |||
|---|---|---|---|---|
| Variables | Model 1 | Model 2 | Model 3 | Model 4 |
| OR (95 % CI) P-value | OR (95 % CI) P-value | OR (95 % CI) P-value | OR (95 % CI) P-value | |
| Total antioxidant activity | 0.56 (0.34–0.93) 0.025 | 0.58 (0.34–0.97) 0.039 | 1.18 (0.87–1.62) 0.288 | 1.28 (0.89–1.83) 0.187 |
| % total antioxidant activity (respect to total blood protein levels) | 0.96 (0.93–0.99) 0.040 | 0.97 (0.93–1.00) 0.052 | 1.01 (0.99–1.03) 0.344 | 1.02 (0.99–1.04) 0.241 |
| Carbonylated proteins | 0.96 (0.55–1.67) 0.883 | 0.95 (0.54–1.68) 0.862 | 0.77 (0.48–1.23) 0.277 | 0.72 (0.43–1.21) 0.216 |
| Lipid peroxidation | 1.00 (0.96–1.05) 0.843 | 1.00 (0.96–1.05) 0.868 | 0.99 (0.96–1.03) 0.661 | 0.99 (0.95–1.03) 0.495 |
| Total blood proteins | 0.37 (0.13– 1.06) 0.064 | 0.47 (0.15–1.43) 0.183 | 0.86 (0.36–2.04) 0.733 | 1.12 (0.42–3.01) 0.816 |
| C-reactive protein | 0.98 (0.93–1.04) 0.514 | 0.98 (0.03–1.03) 0.441 | 1.03 (0.97–1.09) 0.380 | 1.02 (0.96–1.09) 0.504 |
| Hemoglobin | 1.46 (0.97–2.19) 0.073 | 1.28 (0.83–1.98) 0.272 | 1.67 (1.15–2.42) 0.007 | 1.62 (1.09–2.4) 0.018 |
All data are presented as odds ratio (95 % confidence interval). Models 1 and 3: adjusted for gender, age, anthropometric data (weight, height, waist circumference, body mass index, total fat mass), Barthel and Lawton indexes, risk of depression, and drug use. Models 2 and 4: also adjusted for frailty. Differences were always considered to be statistically significant when p ≤ 0.05
OR odds ratio, CI confidence interval
Table 7 displays the univariate linear regression model for studying the associative relationship between the physical performance of the upper or lower body limbs and prevalence of the pre-frail phenotype. For this purpose, frail subjects were excluded from this logistic analysis. In the univariate models that were adjusted for age, gender, anthropometric data, and Barthel and Lawton indexes, we found a statistically significant association to explain 23.3 % of the variance of pre-frailty from the values of functional physical performance of the lower and upper body limbs (Table 7, model 1). Notably, physical performance of the lower body limbs had a greater weight [β = 0.27 (95 % CI, 0.12–0.42), p ≤ 0.001] in this logistic model (Table 7, model 1). After adjustment for depressive mood, this association remained statistically significant (Table 7, model 2). However, after adding drug use in the univariate linear regression models (Table 7, model 3), we observed that only the association between pre-frailty and physical performance of the lower body limbs remained statistically significant [β = 0.25 (95 % CI, 0.09–0.40), p ≤ 0.01].
Table 7.
Associations between the functional performance of the lower and upper body limbs and the pre-frail phenotype
| Pre-frailty | |||
|---|---|---|---|
| Model 1 | Model 2 | Model 3 | |
| β (95 % CI) P-value | β (95 % CI) P-value | β (95 % CI) P-value | |
| Lower body limbs | 0.27 (0.12–0.42) 0.001 | 0.27 (0.12–0.42) 0.001 | 0.25 (0.09–0.40) 0.002 |
| Upper body limbs | 0.19 (0.02–0.35) 0.028 | 0.18 (0.02–0.34) 0.032 | 0.16 (−0.02–0.32) 0.075 |
All data are presented as standardized beta (95 % confidence interval). Model 1: adjusted for gender, age, anthropometric data (weight, height, waist circumference, body mass index, total fat mass), Barthel and Lawton indexes. Model 2: also adjusted for risk of depression. Model 3: also adjusted for risk of depression and drug use. Differences were always considered to be statistically significant when p ≤ 0.05
β standardized beta, CI confidence interval
Discussion
The purpose of our present study was to determine the degree of association of common clinical variables (hemoglobin levels, C-reactive protein, and total blood protein) and oxidative stress-related markers (protein damage, lipid peroxidation, antioxidant capacity) with the decline on the functional physical performance of the lower and upper body limbs in elderly of the FRADEA cohort. We also studied which deficiencies of functional physical performance, on the lower or upper body limbs, could be better associated with the pre-frail phenotype in the elderly. For these purposes, we have created a new variable by applying PCA on different instrumental tests of functional performance in order to describe properly the different states of functional physical performance. In particular, grip strength of the dominant hand, kilocalories consumed per week, and the musculoskeletal index were selected as the most appropriate variables for describing functional performance of the upper body limbs (component 1 of the PCA), while gait speed and SPPB were grouped in the other component to describe functional performance of the lower body limbs (component 2 of the PCA). The relation between gait speed and the SPPB score with functional performance of the lower limbs has been well described (Guralnik et al. 1994; Sayers et al. 2004; Kuo et al. 2007; Abizanda et al. 2011a, b; Shin et al. 2013a). Likewise, grip strength of the dominant hand describes functional performance of the upper limbs (Hairi et al. 2010; Abizanda et al. 2011a, b). However, there are no existing data showing correlation between weekly consumption of kilocalories and the musculoskeletal index with functional performance of the upper extremities. Nonetheless, both of these variables were significantly classified together with grip strength of the dominant hand in component 1 of our PCA. In addition, we grouped our study population by cluster analysis into different functional groups depending on the physical performance of their upper and lower body limbs, as detailed in Fig. 2.
Our study population is mainly independent or moderately dependent and mostly comprises the pre-frail phenotype. However, the greatest number of subjects was found in the group with poor physical performance of the upper body limbs and in the group with poor general physical performance as they presented functional impairments in both the upper and lower body limbs. Age, gender, and specific body composition, including a high level of fat mass together with a low percentage of lean mass, morbidity, and a higher body mass index have recently been associated with deficiencies in physical performance among older adults (Cabrero-Garcia et al. 2012; Hardy et al. 2013; Shin et al. 2013a). Accordingly, we observed significant differences on functional physical performance in our population in relation to gender, age, and anthropometric characteristics. For instance, subjects with deficiencies in the physical performance of their lower body limbs had a higher waist circumference, total fat mass, weight, height and, a higher body mass index compared with subjects with deficiencies of mainly their upper body limbs. Subjects with poor general physical performance were mostly women with the highest percentage of total fat mass. Surprisingly, the chronic diseases studied (diabetes, dyslipidemia, hypertension, and cardiac insufficiency) did not show significant statistical differences among groups with different functional physical performance. In the recent literature, poorer functional physical performance has been related to malnutrition, weak muscle strength, sarcopenia, cognitive impairment, and different chronic diseases including heart failure, peripheral neuropathy, chronic lung and kidney diseases, and rheumatoid arthritis (Koufaki and Kouidi 2010; Vogiatzis et al. 2012; Alosco et al. 2013; Ylitalo et al. 2013; Hardy et al. 2013; Kiesswetter et al. 2013; Landi et al. 2013; Shin et al. 2013b). In our previous studies, chronic conditions such as cognitive decline, osteoarthritis, and hypertension were significantly presented in older institutionalized subjects with moderate to severe dependence (de Gonzalo-Calvo et al. 2012b, c). However, our present population, representative of an urban Spanish population, did not show such a great proportion of chronic pathologies among older subjects. Notably, among participants with pathology, we found a higher number of subjects with general impairments of physical performance or specific deficiencies of the upper body limbs. In addition, risk of depression significantly also affected a better or worse functional physical performance. Depressive mood is intimately associated with poor functional performance (Cabrero-Garcia et al. 2012; Wells et al. 2012). Likewise, depressive mood has recently been associated with a high prevalence of frailty in the FRADEA study (Abizanda et al. 2011b, 2012) as well as in other elderly Spanish cohorts, such as the Toledo Study for Healthy Aging (Garcia-Garcia et al. 2011). Finally, the use of drugs was also implicated on differences of functional physical performance; in particular, subjects with poor general physical performance and those with specific functional deficiencies of their upper or lower body limbs presented higher drug use. Previous studies have also observed that the inappropriate use of drugs is associated with impaired physical performance among frail elderly subjects (Landi et al. 2007b).
It is well known that oxidative stress and inflammation are closely associated with human diseases during aging (Caballero et al. 2013; de Gonzalo-Calvo et al. 2010, 2011). Similarly, oxidative stress can favor mobility limitations and severe disability among older subjects (Cesari et al. 2005; Howard et al. 2007; Semba et al. 2007). Oxidative protein damage has been associated with moderate to severe disability as well as functional dependence in the institutionalized elderly (Dayhoff-Brannigan et al. 2008; de Gonzalo-Calvo et al. 2012c). Inflammatory markers that also play a key role as biological markers of disability or functional dependence among the elderly include the tumor necrosis factor alpha (TNFα), interleukin-6 (IL-6), and C-reactive protein (de Gonzalo-Calvo et al. 2012b, c; Kuo et al. 2012). In addition, chronic inflammation has recently been described as a predictor of age-related adverse outcomes, including hospitalization and mortality in the dependent elderly population (de Gonzalo-Calvo et al. 2012a). However, we did not find a significant association between oxidative damage in proteins or lipids and specific deficiencies of functional physical performance in our population. Although C-reactive protein, as a clinical marker related to inflammation and disability (Ridker et al. 2000; Pepys and Hirschfield 2003; Kuo et al. 2012), showed significant differences in older women (> 76 years) with different functional physical performance, bivariate regression analysis did not find a close association between this biomarker and functional physical performance. Other studies have observed that oxidative stress, in particular oxidative damage in protein and lipids, is associated with a severe walking disability and poor grip strength, indicative of functional deficiencies in performance on the lower and upper body limbs, respectively (Semba et al. 2007; Howard et al. 2007). The lack of association between these biomarkers and physical performance in the present study can be explained by the fact that our population showed a lower level of severe dependence (2 %) and frailty (11 %). Interestingly, our data showed statistically significant differences among the groups with different functional physical performance in regards to the total antioxidant capacity of women who are 76 years or younger. However, differences in the total antioxidant capacity were not detected in older women (> 76 years) or in men independently of age. A direct association between total antioxidant capacity and functional dependence in the elderly has not been observed previously (de Gonzalo-Calvo et al. 2012c). Notably, in our population, which is characterized by being mainly independent or moderately dependent, we found a close association between the total antioxidant capacity and functional physical performance of the upper body limbs exclusively. Despite the low values of the ORs, this association remained statistically significant even after adjusting our bivariate regression models for different confounding variables (age, gender, anthropometric parameters, Barthel and Lawton indexes, depressive mood, drug use, and frailty). When participants with severe dependence were excluded from the analysis, this association also remained statistically significant. Previous studies have already observed an association between low levels of antioxidants and adverse clinical outcomes in the elderly, including the presence of frailty (Ble et al. 2006) and increased risk of death (Li et al. 2011; Ray et al. 2006; Shardell et al. 2011). In accordance with these previous studies, our data showed the lowest total antioxidant capacity in subjects with impaired functional physical performance of both their lower and upper body limbs. Upper extremity function is considered to be an independent predictor of adverse events in the hospitalized elderly (Abizanda et al. 2007). Thus, our present data suggest that deficiencies of physical performance of the upper body limbs could more significantly affect the mobility capacity of older subjects not characterized by a severe dependence, which is related to a decreased antioxidant defense. In previous studies, we have demonstrated an association between oxidative damage and inflammation mainly in situations of severe functional dependence (de Gonzalo-Calvo et al. 2012b, c). Taking into account both our present results from older mainly independent or moderately dependent subjects and our previous studies in institutionalized and dependent elderly, we consider that the balance between the systemic antioxidant defense and oxidative damages plays a key role on the physical functional performance in the elderly. It is even possible to follow a time sequence in which we first lose antioxidant capability, which leads to the consequent future oxidative damage.
Moreover, we also assayed levels of hemoglobin, a common marker that is generally assayed in clinical analyses. Decreased levels of hemoglobin were mainly observed in older men (> 76 years) with worse physical performance of either their upper and/or lower body limbs. Low levels of hemoglobin in older subjects could be indicative of anemia, which is considered a risk factor in functional and cognitive decline, frailty, hospitalization, and risk of mortality in the elderly (Patel and Guralnik 2009; Riva et al. 2009). In the elderly, we frequently find low levels of hemoglobin and oxygen supply in relation to hypoxia situations that can lead to increased oxidative tissue damage, which can affect physical performance (Lundby et al. 2009; de Gonzalo-Calvo et al. 2011). Despite large CIs, our bivariate logistic regression models showed a clear and significant association between blood hemoglobin levels and functional physical performance of the lower body limbs exclusively. This association remained statistically significant even after adjustment of our regression models for different confounding variables (age, gender, anthropometric parameters, Barthel and Lawton indexes, depressive mood, drug use, and frailty). Previous studies have recently found weak but significant associations between hemoglobin levels and functional dependence as well as risk of hospitalization in the institutionalized elderly population (de Gonzalo-Calvo et al. 2012a, b). Other clinical markers related to mobility impairments of the lower extremities have been described, such as low levels of bilirubin (Kao et al. 2012), which is considered a risk factor of functional dependence. Elevated levels of homocysteine have also been associated with disability on the lower body limbs (Kuo et al. 2007). The red blood cell distribution width (RDW) showed a strong association with functional dependence and adverse outcomes, including hospitalization and mortality in the institutionalized elderly (de Gonzalo-Calvo et al. 2012a, b). In accordance with these data, the present study shows that age and gender-dependent alteration of hemoglobin levels in the elderly should be related to deficiencies of the functional physical performance of the lower body limbs.
Notably, our elderly Spanish FRADEA population is characterized by a high prevalence (48.5 to 48.8 %) of a previous state of frailty, better known as a pre-frailty condition (Abizanda et al. 2011a, b). Due to this, we decided to exclude frail subjects to study possible associations between the functional physical performance of the lower and upper body limbs and the pre-frail phenotype. Our univariate linear logistic regression models showed significant associations of pre-frailty and physical performance of both the upper and lower body limbs. The model explained 23.3 % of the variance of the pre-frailty phenotype. These associations remained statistically significant even after adjustment of our linear regression models for different confounding variables (age, gender, anthropometric parameters, Barthel and Lawton indexes, and depressive mood). However, after an additional adjustment for drug use, the association of the physical performance of the upper body limbs with pre-frailty was almost statistically significant. Notably, we observed a better and more significant association between pre-frailty and functional physical performance of the lower body limbs in our linear regression models. This specific association of physical performance of the lower body limbs and pre-frailty always remained statistically significant even after adjustment also for drug use. In accordance with our data, it has recently been described that muscle strength of the lower extremities is a better predictor of physical disability and functional limitation than that of the upper extremities (Hairi et al. 2010). Likewise, adopting physical training to increase muscle strength in mainly the lower limbs has been demonstrated to reduce the risk of adverse outcomes (e.g., falls), which frequently occur in the elderly (Marini et al. 2008). Therefore, given these premises and our present data, deficiencies of functional physical performance of the lower body limbs could be more significantly associated with a future condition of pre-frailty in the elderly.
Several limitations of this study must be considered. First, a population sample of more than 200 participants would have been desirable. With respect to the low number of participants with severe dependence and frailty, it should be noted that our cohort was randomly selected from older people living in Albacete (Spain), which means maintaining the original percentages of functional dependence and frailty in the city. Thus, our cohort should be considered representative of this urban Spanish population. Additionally, better knowledge of the type of diet and previous exercise and physical training would also be desirable. Furthermore, as this was an observational study, we were not able to determinate the causal directions of the observed associations between the total antioxidant capacity and hemoglobin levels with specific functional physical performance of the upper and lower body limbs, respectively. We should also remind that we performed a cross-sectional study, such that longitudinal studies should be performed to better document the validity and selectivity of our statistical associations. Finally, we cannot exclude the effects of other diseases or medications that were not recorded in the present study as well as subsequent adverse outcomes. Despite these limitations, the present study has demonstrated that variables normally related to a general physical status, such as the weekly consumption of kilocalories and the musculoskeletal index, could be incorporated into clinical practice in relation to functional performance of the upper body limbs. Likewise, taken together, our results suggest an intimate association between functional physical performance of the upper body limbs and the total antioxidant capacity of elderly subjects. A close association between the functional physical performance of the lower body limbs and levels of hemoglobin was also specifically observed. As our data suggest, the physical performance of the lower body limbs has more importance than that of the upper body limbs with regard to future mobility limitations, which could favor a pre-frail phenotype in the elderly. Since the pre-frail elderly could be particularly vulnerable to oxidative stress, we consider that their identification is paramount. The pre-frailty in the elderly could lead not only to a higher risk for developing a future condition of frailty but also to developing a disease as well as disability and/or functional dependence.
Acknowledgments
The authors thank Dr. Mamen Oliván for her excellent statistical advice. Beatriz Caballero, Ana Coto-Montes, Pedro Abizanda, and Juan José Solano are participants of the National Net RETICEF for Aging studies [Red Temática de Investigación Cooperativa en Envejecimiento y Fragilidad (RETICEF)]. Likewise, Ana Coto is a member of INEUROPA.
References
- Abizanda P, Navarro JL, Romero L, Leon M, Sanchez-Jurado PM, Dominguez L. Upper extremity function, an independent predictor of adverse events in hospitalized elderly. Gerontology. 2007;53(5):267–273. doi: 10.1159/000102541. [DOI] [PubMed] [Google Scholar]
- Abizanda P, Lopez-Torres J, Romero L, et al. Fragilidad y dependencia en Albacete (estudio FRADEA): razonamiento, diseno y metodología. Rev Esp Geriatr Gerontol. 2011;46:81–88. doi: 10.1016/j.regg.2010.10.004. [DOI] [PubMed] [Google Scholar]
- Abizanda P, Sanchez-Jurado PM, Romero L, Paterna G, Martinez-Sanchez E, Atienzar-Nunez P. Prevalence of frailty in a Spanish elderly population: the frailty and dependence in Albacete study. J Am Geriatr Soc. 2011;59:1356–1359. doi: 10.1111/j.1532-5415.2011.03463.x. [DOI] [PubMed] [Google Scholar]
- Abizanda P, Romero L, Sanchez-Jurado PM, Atienzar-Nunez P, Esquinas-Requena JL, Garcia-Noguera I. Association between functional assessment instruments and frailty in older adults: the FRADEA study. J Frailty Aging. 2012;1:1–7. doi: 10.14283/jfa.2012.25. [DOI] [PubMed] [Google Scholar]
- Abizanda P, Romero L, Sanchez-Jurado PM, Martinez-Reig M, Gomez-Arnedo L, Alfonso SA. Frailty and mortality, disability and mobility loss in a Spanish cohort of older adults: the FRADEA study. Maturitas. 2013;74:54–60. doi: 10.1016/j.maturitas.2012.09.018. [DOI] [PubMed] [Google Scholar]
- Alcala MV, Puime AO, Santos MT, Barral AG, Montalvo JI, Zunzunegui MV. Prevalence of frailty in an elderly Spanish urban population. Relationship with comorbidity and disability. Aten Primaria. 2010;42(10):520–527. doi: 10.1016/j.aprim.2009.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alosco ML, Brickman AM, Spitznagel MB, Griffith EY, Narkhede A, Raz N, Cohen R, Sweet LH, Colbert LH, Josephson R, Hughes J, Rosneck J, Gunstad J. Poorer physical fitness is associated with reduced structural brain integrity in heart failure. J Neurol Sci. 2013;328:51–57. doi: 10.1016/j.jns.2013.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balducci L. Aging, frailty and chemotherapy. Cancer Control. 2007;14:7–12. doi: 10.1177/107327480701400102. [DOI] [PubMed] [Google Scholar]
- Bandeen-Roche K, Xue QL, Ferrucci L, Walston J, Guralnik JM, Chaves P, Zeger SL, Fried LP. Phenotype of frailty: characterization in the women's health and aging studies. J Gerontol A Biol Sci Med Sci. 2006;61:262–266. doi: 10.1093/gerona/61.3.262. [DOI] [PubMed] [Google Scholar]
- Baztán JJ, Pérez del Molino J, Alarcón T, San Cristóbal E, Izquierdo G, Manzarbeitia J. Índice de Barthel: instrumento válido para la valoración funcional de pacientes con enfermedad cerebrovascular. Rev Esp Geriatr Gerontol. 1993;28:32–40. [Google Scholar]
- Bermejo F, Morales JM, Valerga C, Del Ser T, Artolazábal J, Gabriel R. Comparación entre dos versiones españolas abreviadas de evaluación del estado mental en el diagnóstico de demencia. Datos de un estudio en ancianos residentes en la comunidad. Med Clin (Barc) 1999;112:330–334. [PubMed] [Google Scholar]
- Ble A, Cherubini A, Volpato S, Bartali B, Walston JD, Windham BG, Bandinelli S, Lauretani F, Guralnik JM, Ferrucci L. Lower plasma vitamin E levels are associated with the frailty syndrome: the InCHIANTI study. J Gerontol A Biol Sci Med Sci. 2006;61:278–283. doi: 10.1093/gerona/61.3.278. [DOI] [PubMed] [Google Scholar]
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Caballero B, Veenman L, Gavish M. Role of mitochondrial translocator protein (18 kDa) on mitochondrial-related cell death processes. Recent Pat Endocr Metab Immune Drug Discov. 2013;7:86–101. doi: 10.2174/1872214811307020002. [DOI] [PubMed] [Google Scholar]
- Cabrero-Garcia J, Munoz-Mendoza CL, Cabanero-Martinez MJ, Gonzalez-Llopis L, Ramos-Pichardo JD, Reig-Ferrer A. Short physical performance battery reference values for patients 70 years-old and over in primary health care. Aten Primaria. 2012;44:540–548. doi: 10.1016/j.aprim.2012.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell AJ, Buchner DM. Unstable disability and the fluctuations of frailty. Age Ageing. 1997;26:315–318. doi: 10.1093/ageing/26.4.315. [DOI] [PubMed] [Google Scholar]
- Cesari M, Kritchevsky SB, Nicklas BJ, Penninx BW, Holvoet P, Koh-Banerjee P, Cummings SR, Harris TB, Newman AB, Pahor M. Lipoprotein peroxidation and mobility limitation: results from the Health, Aging, and Body Composition Study. Arch Intern Med. 2005;165:2148–2154. doi: 10.1001/archinte.165.18.2148. [DOI] [PubMed] [Google Scholar]
- Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- Coto-Montes A, Hardeland R. Antioxidative effects of melatonin in Drosophila melanogaster: antagonization of damage induced by the inhibition of catalase. J Pineal Res. 1999;27:154–158. doi: 10.1111/j.1600-079X.1999.tb00610.x. [DOI] [PubMed] [Google Scholar]
- Dayhoff-Brannigan M, Ferrucci L, Sun K, Fried LP, Walston J, Varadhan R, Guralnik JM, Semba RD. Oxidative protein damage is associated with elevated serum interleukin-6 levels among older moderately to severely disabled women living in the community. J Gerontol A Biol Sci Med Sci. 2008;63:179–183. doi: 10.1093/gerona/63.2.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Gonzalo-Calvo D, Neitzert K, Fernandez M, Vega-Naredo I, Caballero B, Garcia-Macia M, Suarez FM, Rodriguez-Colunga MJ, Solano JJ, Coto-Montes A. Differential inflammatory responses in aging and disease: TNF-alpha and IL-6 as possible biomarkers. Free Radic Biol Med. 2010;49:733–737. doi: 10.1016/j.freeradbiomed.2010.05.019. [DOI] [PubMed] [Google Scholar]
- de Gonzalo-Calvo D, Neitzert K, Fernandez M, Vega-Naredo I, Caballero B, Garcia-Macia M, Suarez FM, Rodriguez-Colunga MJ, Solano JJ, Coto-Montes A. Defective adaption of erythrocytes during acute hypoxia injury in an elderly population. J Gerontol A Biol Sci Med Sci. 2011;66:376–384. doi: 10.1093/gerona/glq204. [DOI] [PubMed] [Google Scholar]
- de Gonzalo-Calvo D, de Luxan-Delgado B, Martinez-Camblor P, Rodriguez-Gonzalez S, Garcia-Macia M, Suarez FM, Solano JJ, Rodriguez-Colunga MJ, Coto-Montes A. Chronic inflammation as predictor of 1-year hospitalization and mortality in elderly population. Eur J Clin Invest. 2012;42:1037–1046. doi: 10.1111/j.1365-2362.2012.02689.x. [DOI] [PubMed] [Google Scholar]
- de Gonzalo-Calvo D, de Luxan-Delgado B, Rodriguez-Gonzalez S, Garcia-Macia M, Suarez FM, Solano JJ, Rodriguez-Colunga MJ, Coto-Montes A. Interleukin 6, soluble tumor necrosis factor receptor I and red blood cell distribution width as biological markers of functional dependence in an elderly population: a translational approach. Cytokine. 2012;58:193–198. doi: 10.1016/j.cyto.2012.01.005. [DOI] [PubMed] [Google Scholar]
- de Gonzalo-Calvo D, de Luxan-Delgado B, Rodriguez-Gonzalez S, Garcia-Macia M, Suarez FM, Solano JJ, Rodriguez-Colunga MJ, Coto-Montes A. Oxidative protein damage is associated with severe functional dependence among the elderly population: a principal component analysis approach. J Gerontol A Biol Sci Med Sci. 2012;67:663–670. doi: 10.1093/gerona/glr215. [DOI] [PubMed] [Google Scholar]
- Fernandez-Bolanos M, Otero A, Zunzunegui MV, Beland F, Alarcon T, de Hoyos C, Castell MV. Sex differences in the prevalence of frailty in a population aged 75 and older in Spain. J Am Geriatr Soc. 2008;56:2370–2371. doi: 10.1111/j.1532-5415.2008.02032.x. [DOI] [PubMed] [Google Scholar]
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:M146–M156. doi: 10.1093/gerona/56.3.M146. [DOI] [PubMed] [Google Scholar]
- Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G. Untangling the concepts of disability, frailty, and comorbidity: implications for improved targeting and care. J Gerontol A Biol Sci Med Sci. 2004;59:255–263. doi: 10.1093/gerona/59.3.M255. [DOI] [PubMed] [Google Scholar]
- Garcia-Garcia FJ, Gutierrez Avila G, Alfaro-Acha A, et al. The prevalence of frailty syndrome in an older population from Spain. The Toledo Study for Healthy Aging. J Nutr Health Aging. 2011;15:852–856. doi: 10.1007/s12603-011-0075-8. [DOI] [PubMed] [Google Scholar]
- Gill TM. Assessment of function and disability in longitudinal studies. J Am Geriatr Soc. 2010;58(Suppl 2):S308–S312. doi: 10.1111/j.1532-5415.2010.02914.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf C. The Lawton instrumental activities of daily living scale. Am J Nurs. 2008;108:52–62. doi: 10.1097/01.NAJ.0000314810.46029.74. [DOI] [PubMed] [Google Scholar]
- Guralnik JM, Simonsick EM, Ferrucci L, Glynn RJ, Berkman LF, Blazer DG, Scherr PA, Wallace RB. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85–M94. doi: 10.1093/geronj/49.2.M85. [DOI] [PubMed] [Google Scholar]
- Hairi NN, Cumming RG, Naganathan V, Handelsman DJ, Le Couteur DG, Creasey H, Waite LM, Seibel MJ, Sambrook PN. Loss of muscle strength, mass (sarcopenia), and quality (specific force) and its relationship with functional limitation and physical disability: the Concord Health and Ageing in Men Project. J Am Geriatr Soc. 2010;58:2055–2062. doi: 10.1111/j.1532-5415.2010.03145.x. [DOI] [PubMed] [Google Scholar]
- Hardy R, Cooper R, Aihie Sayer A, Ben-Shlomo Y, Cooper C, Deary IJ, Demakakos P, Gallacher J, Martin RM, McNeill G, Starr JM, Steptoe A, Syddall H, Kuh D. Body mass index, muscle strength and physical performance in older adults from eight cohort studies: the HALCyon programme. PLoS One. 2013;8:e56483. doi: 10.1371/journal.pone.0056483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard C, Ferrucci L, Sun K, Fried LP, Walston J, Varadhan R, Guralnik JM, Semba RD. Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol. 2007;103:17–20. doi: 10.1152/japplphysiol.00133.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jurschik Gimenez P, Escobar Bravo MA, Nuin Orrio C, Botigue Satorra T. Frailty criteria in the elderly: a pilot study. Aten Primaria. 2011;43:190–196. doi: 10.1016/j.aprim.2010.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kao TW, Chou CH, Wang CC, Chou CC, Hu J, Chen WL. Associations between serum total bilirubin levels and functional dependence in the elderly. Intern Med J. 2012;42:1199–1207. doi: 10.1111/j.1445-5994.2011.02620.x. [DOI] [PubMed] [Google Scholar]
- Kiesswetter E, Pohlhausen S, Uhlig K, Diekmann R, Lesser S, Heseker H, Stehle P, Sieber CC, Volkert D. Malnutrition is related to functional impairment in older adults receiving home care. J Nutr Health Aging. 2013;17:345–350. doi: 10.1007/s12603-012-0409-1. [DOI] [PubMed] [Google Scholar]
- Koufaki P, Kouidi E. Current best evidence recommendations on measurement and interpretation of physical function in patients with chronic kidney disease. Sports Med. 2010;40:1055–1074. doi: 10.2165/11536880-000000000-00000. [DOI] [PubMed] [Google Scholar]
- Kuo HK, Liao KC, Leveille SG, Bean JF, Yen CJ, Chen JH, Yu YH, Tai TY. Relationship of homocysteine levels to quadriceps strength, gait speed, and late-life disability in older adults. J Gerontol A Biol Sci Med Sci. 2007;62:434–439. doi: 10.1093/gerona/62.4.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo HK, Al Snih S, Kuo YF, Raji MA. Chronic inflammation, albuminuria, and functional disability in older adults with cardiovascular disease: the National Health and Nutrition Examination Survey, 1999-2008. Atherosclerosis. 2012;222:502–508. doi: 10.1016/j.atherosclerosis.2012.03.004. [DOI] [PubMed] [Google Scholar]
- Landi F, Russo A, Cesari M, Pahor M, Bernabei R, Onder G. HDL-cholesterol and physical performance: results from the ageing and longevity study in the sirente geographic area (ilSIRENTE Study) Age Ageing. 2007;36:514–520. doi: 10.1093/ageing/afm105. [DOI] [PubMed] [Google Scholar]
- Landi F, Russo A, Liperoti R, Barillaro C, Danese P, Pahor M, Bernabei R, Onder G. Impact of inappropriate drug use on physical performance among a frail elderly population living in the community. Eur J Clin Pharmacol. 2007;63:791–799. doi: 10.1007/s00228-007-0321-5. [DOI] [PubMed] [Google Scholar]
- Landi F, Cruz-Jentoft AJ, Liperoti R, Russo A, Giovannini S, Tosato M, Capoluongo E, Bernabei R, Onder G. Sarcopenia and mortality risk in frail older persons aged 80 years and older: results from ilSIRENTE study. Age Ageing. 2013;42:203–209. doi: 10.1093/ageing/afs194. [DOI] [PubMed] [Google Scholar]
- Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9:179–186. doi: 10.1093/geront/9.3_Part_1.179. [DOI] [PubMed] [Google Scholar]
- Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG, Ahn BW, Shaltiel S, Stadtman ER. Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol. 1990;186:464–478. doi: 10.1016/0076-6879(90)86141-H. [DOI] [PubMed] [Google Scholar]
- Li C, Ford ES, Zhao G, Balluz LS, Giles WH, Liu S. Serum alpha-carotene concentrations and risk of death among US Adults: the Third National Health and Nutrition Examination Survey Follow-up Study. Arch Intern Med. 2011;171:507–515. doi: 10.1001/archinternmed.2010.440. [DOI] [PubMed] [Google Scholar]
- Lundby C, Calbet JA, Robach P. The response of human skeletal muscle tissue to hypoxia. Cell Mol Life Sci. 2009;66:3615–3623. doi: 10.1007/s00018-009-0146-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahoney FI, Barthel DW. Functional evaluation: the Barthel Index. Md State Med J. 1965;14:61–65. [PubMed] [Google Scholar]
- Marini M, Sarchielli E, Brogi L, Lazzeri R, Salerno R, Sgambati E, Monaci M. Role of adapted physical activity to prevent the adverse effects of the sarcopenia. A pilot study. Ital J Anat Embryol. 2008;113:217–225. [PubMed] [Google Scholar]
- Martí D, Miralles R, Llorach I, García-Palleiro P, Esperanza A, Guillén J, et al. Trastornos depresivos en una unidad de convalecencia: experiencia y validación de una versión española de 15 preguntas de la escala de depresión geriátrica de Yesavage. Rev Esp Geriatr Gerontol. 2000;35:7–14. [Google Scholar]
- Miralles Basseda R, Esperanza Sanjuán A (2007) Instrumentos y escalas de valoración. In: Sociedad Española de Geriatría y Gerontología (ed) Tratado de Geriatría para Residentes. Madrid, pp 771–89.
- Patel KV, Guralnik JM. Prognostic implications of anemia in older adults. Haematologica. 2009;94:1–2. doi: 10.3324/haematol.2008.001289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peel NM, Kuys SS, Klein K. Gait speed as a measure in geriatric assessment in clinical settings: a systematic review. J Gerontol A Biol Sci Med Sci. 2013;68:39–46. doi: 10.1093/gerona/gls174. [DOI] [PubMed] [Google Scholar]
- Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest. 2003;111:1805–1812. doi: 10.1172/JCI200318921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39:142–148. doi: 10.1111/j.1532-5415.1991.tb01616.x. [DOI] [PubMed] [Google Scholar]
- Ray AL, Semba RD, Walston J, Ferrucci L, Cappola AR, Ricks MO, Xue QL, Fried LP. Low serum selenium and total carotenoids predict mortality among older women living in the community: the women's health and aging studies. J Nutr. 2006;136:172–176. doi: 10.1093/jn/136.1.172. [DOI] [PubMed] [Google Scholar]
- Ridker PM, Hennekens CH, Buring JE, Rifai N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med. 2000;342:836–843. doi: 10.1056/NEJM200003233421202. [DOI] [PubMed] [Google Scholar]
- Riva E, Tettamanti M, Mosconi P, Apolone G, Gandini F, Nobili A, Tallone MV, Detoma P, Giacomin A, Clerico M, Tempia P, Guala A, Fasolo G, Lucca U. Association of mild anemia with hospitalization and mortality in the elderly: the health and anemia population-based study. Haematologica. 2009;94:22–28. doi: 10.3324/haematol.13449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos-Eggimann B, Cuenoud P, Spagnoli J, Junod J. Prevalence of frailty in middle-aged and older community-dwelling Europeans living in 10 countries. J Gerontol A Biol Sci Med Sci. 2009;64:675–681. doi: 10.1093/gerona/glp012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayers SP, Jette AM, Haley SM, Heeren TC, Guralnik JM, Fielding RA. Validation of the late-life function and disability instrument. J Am Geriatr Soc. 2004;52:1554–1559. doi: 10.1111/j.1532-5415.2004.52422.x. [DOI] [PubMed] [Google Scholar]
- Semba RD, Ferrucci L, Sun K, Walston J, Varadhan R, Guralnik JM, Fried LP. Oxidative stress and severe walking disability among older women. Am J Med. 2007;120:1084–1089. doi: 10.1016/j.amjmed.2007.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shardell MD, Alley DE, Hicks GE, El-Kamary SS, Miller RR, Semba RD, Ferrucci L. Low-serum carotenoid concentrations and carotenoid interactions predict mortality in US adults: the Third National Health and Nutrition Examination Survey. Nutr Res. 2011;31:178–189. doi: 10.1016/j.nutres.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin H, Liu PY, Panton LB, Ilich JZ. Physical performance in relation to body composition and bone mineral density in healthy, overweight, and obese postmenopausal women. J Geriatr Phys Ther. 2013 doi: 10.1519/JPT.0b013e31828af203. [DOI] [PubMed] [Google Scholar]
- Shin SY, Julian L, Katz P. The relationship between cognitive function and physical function in rheumatoid arthritis. J Rheumatol. 2013;40:236–243. doi: 10.3899/jrheum.120871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topinkova E. Aging, disability and frailty. Ann Nutr Metab. 2008;52(Suppl 1):6–11. doi: 10.1159/000115340. [DOI] [PubMed] [Google Scholar]
- Torres Moreno B, Núñez González E, Pérez Hernández Dde G, Simón Turriate JP, Alastuey Giménez C, Díaz Melián J, et al. Barthel and Charlson indexes for the prognosis of mortality and institutionalization in hospitalized geriatric patients. Rev Esp Geriatr Gerontol. 2009;44:209–212. doi: 10.1016/j.regg.2009.02.002. [DOI] [PubMed] [Google Scholar]
- Vogiatzis I, Zakynthinos G, Andrianopoulos V. Mechanisms of physical activity limitation in chronic lung diseases. Pulm Med. 2012 doi: 10.1155/2012/634761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wells M, Avers D, Brooks G. Resilience, physical performance measures, and self-perceived physical and mental health in older Catholic nuns. J Geriatr Phys Ther. 2012;35:126–131. doi: 10.1519/JPT.0b013e318237103f. [DOI] [PubMed] [Google Scholar]
- Yesavage JABT, Rose TL, Lum O. Development and validation of a geriatric depression scale: a preliminary report. J Psychiatr Res. 1983;17:37–49. doi: 10.1016/0022-3956(82)90033-4. [DOI] [PubMed] [Google Scholar]
- Ylitalo KR, Herman WH, Harlow SD. Performance-based physical functioning and peripheral neuropathy in a population-based cohort of women at midlife. Am J Epidemiol. 2013;177:810–817. doi: 10.1093/aje/kws327. [DOI] [PMC free article] [PubMed] [Google Scholar]