Abstract
Glioblastoma multiforme (GBM) tumors are the most common malignant primary brain tumors in adults. Although many GBM tumors are believed to be caused by self-renewing, glioblastoma-derived stem-like cells (GSCs), the mechanisms that regulate self-renewal and other oncogenic properties of GSCs are only now being unraveled. Here we showed that GSCs derived from GBM patient specimens express varying levels of the transcriptional repressor REST, suggesting heterogeneity across different GSC lines. Loss- and gain-of-function experiments indicated that REST maintains self-renewal of GSCs. High REST-expressing GSCs (HR-GSCs) produced tumors histopathologically distinct from those generated by low REST-expressing GSCs (LR-GSCs) in orthotopic mouse brain tumor models. Knockdown of REST in HR-GSCs resulted in increased survival in GSC-transplanted mice and produced tumors with higher apoptotic and lower invasive properties. Conversely, forced expression of exogenous REST in LR-GSCs produced decreased survival in mice and produced tumors with lower apoptotic and higher invasive properties, similar to HR-GSCs. Thus, based on our results, we propose that a novel function of REST is to maintain self-renewal and other oncogenic properties of GSCs and that REST can play a major role in mediating tumorigenicity in GBM.
Keywords: REST, glioblastoma stem cells, neural stem cells, glioblastoma multiforme, invasion, self-renewal
Introduction
Glioblastoma multiforme (GBM) tumors are the most common malignant primary brain tumors in adults 1–3 However, despite decades of basic science and clinical research, the outcome for GBM patients remains dismal, with average median survival of only 12–16 months4–15. The lack of progress in GBM research can be attributed to the highly heterogeneous, invasive, necrotic, and chemotherapy- and radiation-resistant nature of GBM tumor cells16. Thus, even after aggressive multimodal therapy, the invading tumor cells often survive and cause tumor relapse. The generation of invaluable animal models have provided new insights into GBM biology and alternate therapeutic approaches17–19. In addition, the recent advent of the “cancer stem cell” hypothesis has brought a new perspective to our understanding of GBM biology and therapy16, 20–23. According to this hypothesis, tumors contain stem-like cells, which have the capacity for long-term self-renewal. This hypothesis further proposes that stem-like cells are the main cancer-initiating cells due to their long-term self-renewal capacity. However, the mechanisms that lead to self-renewal and tumorigenicity in glioblastoma-derived stem-like cells (GSCs) are still unclear.
The transcriptional repressor REST was originally discovered as a repressor of many neuronal differentiation genes in non-neural cells and results derived from Rest mutant mice showed that it was indeed a repressor of neurogenesis24–26. However, more recent genome-wide analysis revealed that REST can bind to the chromatin of more than a thousand genes and can potentially affect many biological processes of different tissue types27. Our previous studies indicated that REST maintains self-renewal of embryonic stem cells28 and that this role of REST was dependent on the specific cell line as well as the cellular environment (unpublished data). More recently, REST was found to maintain stemness of normal adult neural stem cells 29 . Our previous studies indicated that REST is involved in medulloblastoma tumorigenesis, in which it functions by blocking differentiation of neuronal stem/progenitor cells30–32. Thus, we reasoned that REST might have a critical role in maintaining self-renewal and other oncogenic properties of GSCs. Our studies indicate that REST indeed regulates such functions in GSCs. Our studies also suggest that targeting REST can potentially represent a novel therapeutic strategy in GBM.
Materials and Methods
Cells and cultures
GSCs used in this study were obtained by acute cell dissociation of human glioblastoma multiforme (GBM) surgical specimens from patients treated at The University of Texas MD Anderson Cancer Center, Houston, Texas, as described previously33–38. The specific HR-GSC1, HR-GSC2 and/or LR-GSC lines have been described previously33–38. These cell lines were maintained under proliferation conditions in Dulbecco’s modified Eagle’s medium (DMEM)/F12 supplemented with B27 1:50 (Invitrogen), 20 ng/ml EGF (Sigma), and 20 ng/ml bFGF as neural stem cell–permissive medium, according to procedures described elsewhere39, 40. These cell lines form neurospheres and demonstrate the ability to express multiple lineage markers and reliably form tumors that recapitulate many of the pathological features of the original tumor after orthotopic xenografting36, 37, 41.
Immunofluorescence and TUNEL Assay
Cells were fixed with 4% paraformaldehyde (PFA) in PBS for 15 minutes. After washing with PBS, cells were blocked with 4% goat serum and 0.3% Triton-X for 1 hour at room temperature. Primary antibodies were added overnight at 4°C, rabbit anti-REST (Sigma, 1:100), mouse anti-REST (1:100), Sox-2 (Stemgent, 1:50), rabbit anti-YKL40 (Quidel, 1:500). After washing with PBS, cells were incubated with Alexa fluor secondary antibodies (Invitrogen) for 1 hour at room temperature. Cells were mounted with mounting media containing 4,6 diamino-2-phenylindole (DAPI) (Vector Laboratories) and photographed using a Nikon TE2000 controlled with Metamorph software (Molecular Devices).
Paraffin sections were hydrated through alcohol grades after clearing with CitriSolv (Fisher Scientific). Antigen retrieval was performed with citrate buffer at 90°C for 20 minutes. Sections were blocked with 4% goat serum followed by human specific primary antibody (Nestin 1:200 (Millipore)) overnight at 4°C. After washing twice with PBS sections were incubated with secondary antibodies followed by mounting with DAPI. In vivo TUNEL Assay was conducted as per manufacturer’s recommendations and positive cells were counted from 10 different fields (Roche cat # 11684817910).
Neurosphere and self-renewal assay
For GSCs, neurosphere assay was done as previously described33–38. In brief, cells were dissociated into single cell suspensions, 10 cells per well for self renewal assay for multiple generations or 100 cells per well self renewal for single generation were seeded in 96-well plates, and the number of neurospheres was counted after 15 days. For serial neurosphere assay, cells were collected after the first passage and dissociated into single cells and plated at the same density as the first passage.
In vitro invasion assay
Control and Rest altered lines were counted and 5×105 cells were seeded into the upper compartment of either the invasion or control chambers, as chemo-attractant 10%FBS containing media was added to the lower chamber. Cells were incubated for 24 hrs. The nonadherent cells were removed by washing with PBS and the noninvading cells were removed by cotton swabs. The control and invasion membranes were then fixed with 100% Methanol and stained with crystal violet. Cells were counted under the microscope and a ratio of invading cells on the matrigel matrix to invasive cells on the control membranes of the control versus the Rest altered lines was compared.
Cell transfections and transductions
Loss-of-function experiments
Stable knockdown of REST in GSCs was done using Mission-lentiviruses expressing short hairpin RNA (shRNA) (Sigma), either non-targeting shRNA (shNT) as control or targeting REST (shRest). Cells were incubated the day prior to transduction with 8 µg/ml hexadimethrine bromide. Cells were then incubated with the virus particles at one multiplicity of infection (1MOI) in normal proliferation media for 48 hours. Cells were puromycin (1.5ug/ml) selected. Five individual clones for each line were selected, cultured and REST knockdown efficiency was determined by western blot analysis. The clone with maximum knockdown for each line (shRest construct #1 for HR-GSC1: 42% knockdown; shRest construct #3 for HR-GSC2: 70% knockdown) was selected for further experimentation.
Gain-of-function experiments
GSCs were transfected with plasmids expressing a control vector, or Rest using the the Amaxa nucleofector kit. In brief, 4×106 cells were suspended in 90 µl of nucleofector solution with REST or control plasmids and then transfected using an Amaxa transfection device. Cells were puromycin (1.5ug/ml) selected and REST over expression was confirmed by western blot analysis.
RNA preparation and qRT-PCR
Total RNA prepared using Trizol (Invitrogen) according to the manufacturer’s instructions. Approximately 500 ng total RNA was used as a template for cDNA synthesis using the Verso cDNA kit (Thermo-Scientific). Real-time PCR was done using SYBR green master-mix (Applied Biosystems) as per the manufacturer’s instructions. All real-time PCR experiments were done on an ABI7900HT sequence detection system (Applied Biosystems).
Genome-wide expression analysis of GSCs with loss- and gain-of-function manipulations
Total RNA was used and Affymetrix HG-133 Plus 2.0 whole genome microarrays were performed (LC sciences, Houston) for two HR-GSCs and one LR-GSC plus the corresponding control lines. Genes with differential fold expression in HR- versus LR-GSCs were determined using the Agilent GeneSpring GX 11.5 software. Robust Multiarray Average (RMA) was used for normalization and genes greater than 1.5 fold differentially expressed were included to determine gene function and disease ontologies using Ingenuity Pathway Analysis (IPA, www.ingenutiy.com).
Western blot procedures
Whole cell extracts were prepared from cell lines and 30 µg protein was loaded on gradient SDS-PAGE gel (Pierce). Then proteins were electrophoretically transferred to nitrocellulose membranes. The membranes were probed with the following antibodies: rabbit anti-REST (1:1000, Upstate), mouse anti-Sox2 (1:350 R&D), and rabbit anti-Nestin (Millipore, 1:10,000), mouse anti Bmi-1 (Millipore, 1:500), and goat anti-YKL40 (Santacruz, 1:200). Mouse anti-actin (1:10,000, Sigma) was used as the internal control. The membranes were incubated with the primary antibodies overnight before they were incubated with corresponding fluorescent-labeled secondary antibodies (1:15000, Licor) for 1 hour at room temperature. The protein-antibody complexes were visualized by scanning the membrane using the Odyssey detection system (Li-Cor Biosciences).
Proliferation assays
Proliferation assays for HR-GSC1 and HR-GSC2 cell lines were done using colorimetric BrdU cell proliferation assay kit (Roche 11647229-01).The assay was performed according to manufacturer’s instructions. In short GSC 1 or GSC 2 cells were seeded at 3000 cells per well in a 96 well plate. Allowed them to grow for overnight and incubated with BrdU labeling agent for 24 hours. Cells were fixed for 30min and incubated with Anti-BrdU for 2 hours and the substrate was added and on color development, colorimetric measurements were performed at 492nm.
Intracranial tumorigenicity assay
Intracranial transplantation of GSCs into nude male mice was done as described previously41. A total of 56 animals (7 animals/treatment condition) were used. To standardize GSC transplantation into nude mice brain, a guide-screw system was implanted 2.5mm lateral and 1mm anterior to the bregma accepting a 26 gauge Hamilton syringe. GSCs cultured were collected and dissociated into single cells. 50,000 cells (or 5,000 cells for Supplementary Fig. 2) in 5ul of media were simultaneously injected using an automated syringe pump (Harvard Apparatus). Animals were carefully monitored for survival.
Statistical Analysis
Values are reported as mean+/− standard error of means. Microsoft Excel 2010 and SPSS (IBM) software was used to conduct t-test or ANOVA for each of the experiments. Microarray data was analyzed with Agilent GeneSpring GX11.5 and the Genepattern suite (Broad Institute).
Results
Patient derived glioblastoma stem cells show varying levels of the REST protein
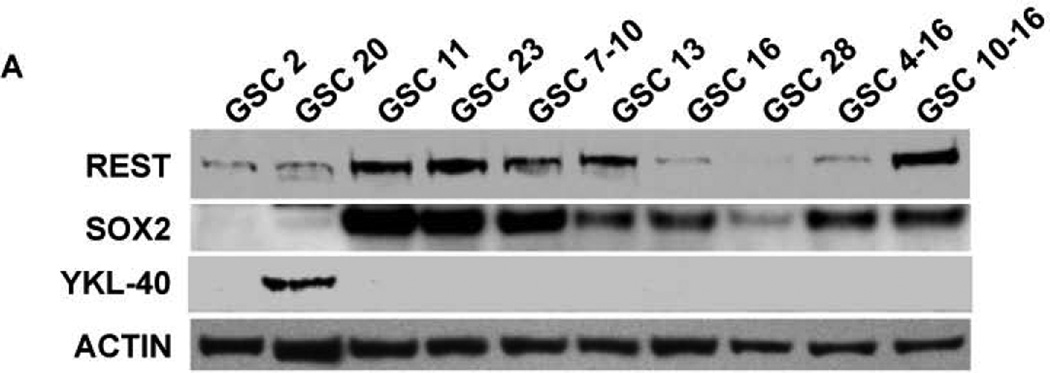
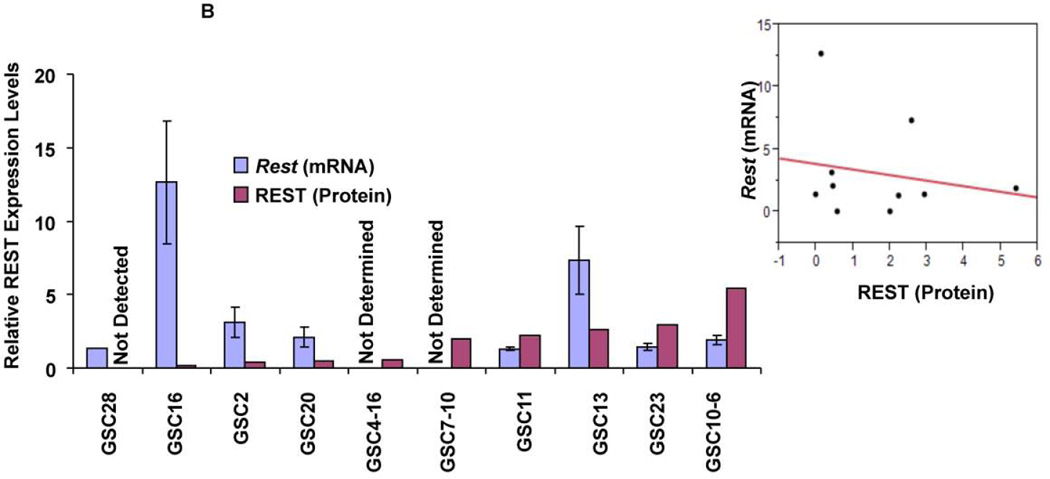
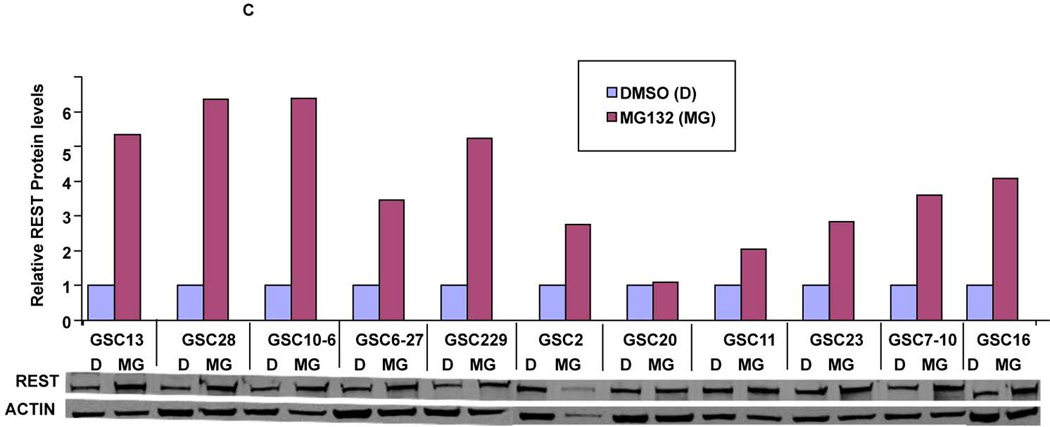
To understand the role of REST in GBM, we quantified REST protein expression in ten patient-derived GSC lines. These lines showed varying levels of REST protein with several fold differences between the lines (Fig. 1A). High REST expression (HR-GSCs) coincided with high levels of Sox2, a known neural stem cell self-renewal regulator42, 43, whereas low-REST expression (LR-GSCs) with low levels of Sox2, suggesting that REST plays a role in self-renewal of GSCs. Interestingly, only one of the GSC lines expressed YKL40, a mesenchymal marker9. To further understand the regulation of REST expression in GSCs, we measured the Rest transcript levels using real time RT-PCR. Comparison of the relative levels of REST protein and Rest transcript levels (Fig. 1B, left panel) and a scatter plot (Fig. 1B, right panel) are shown. The data indicated that the transcript levels did not correlate with the REST protein levels (correlation coefficient =0.04, Fig. 1B). To investigate the lack of correlation between protein and RNA levels, we treated GSCs with the proteasomal inhibitor (MG132) for 24 hours. Our results indicated significant REST protein accumulation in most of the 11 different GSC lines after MG132 treatment compared to the control DMSO treatment (Figure 1C). These results strongly suggested that REST expression is controlled mainly at the protein degradation level in GSCs, similar to what was observed in other cell types26, 44, 45.
Figure 1. (A) Patient glioblastoma-derived stem cells (GSCs) show varying REST levels.
Western blotting assays of GSCs obtained from 10 human GBM patient specimens showed varying levels of REST, Sox2 and YKL-40 proteins. Actin was used as an internal control. (B) Comparison of REST expression at the protein and transcript levels indicated that REST protein levels do not correspond to Rest transcript levels (left panel). A scatter plot is also shown (Right panel, Rsq=0.04). (C) Western blot based quantification of REST protein levels in GSCs treated with either the proteasomal inhibitor (MG132) or solvent (DMSO) indicated that REST protein is sensitive to proteasomal degradation in most of the GSC lines.
REST protein levels correlate with expression of stemness markers and neurosphere formation capacity of GSCs
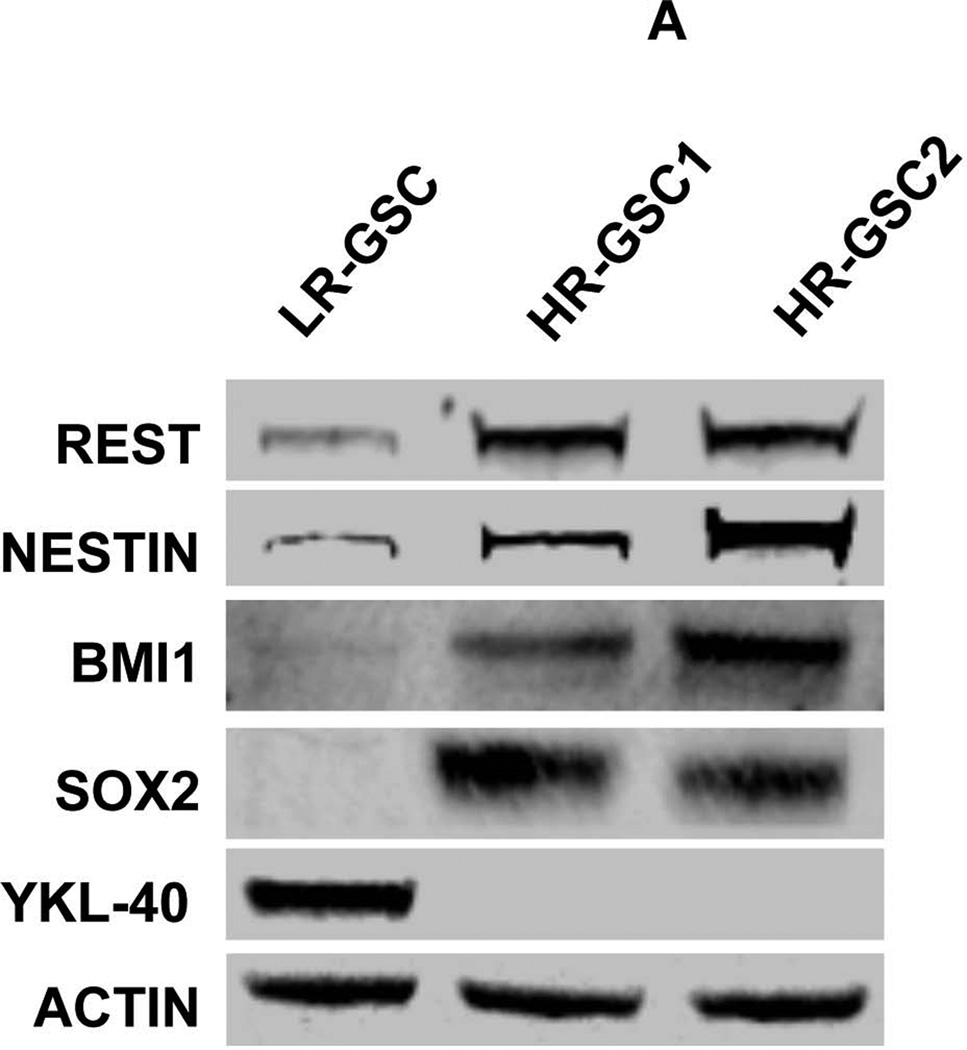
We selected two GSC lines with high levels of REST, HR-GSC1 and HR-GSC2 (GSC11 and GSC23, respectively), and one line with low levels of REST, LR-GSCs (GSC20), for further characterization by Western blotting (Fig. 2A) and double-immunofluorescence (Fig. 2B) analysis. As shown, both HR-GSC lines, compared to LR-GSCs, showed higher expression of the neural stemness markers Nestin, Sox2, and Bmi1, and absence YKL-40. These findings further confirmed the association of REST with stemness markers of GSCs.
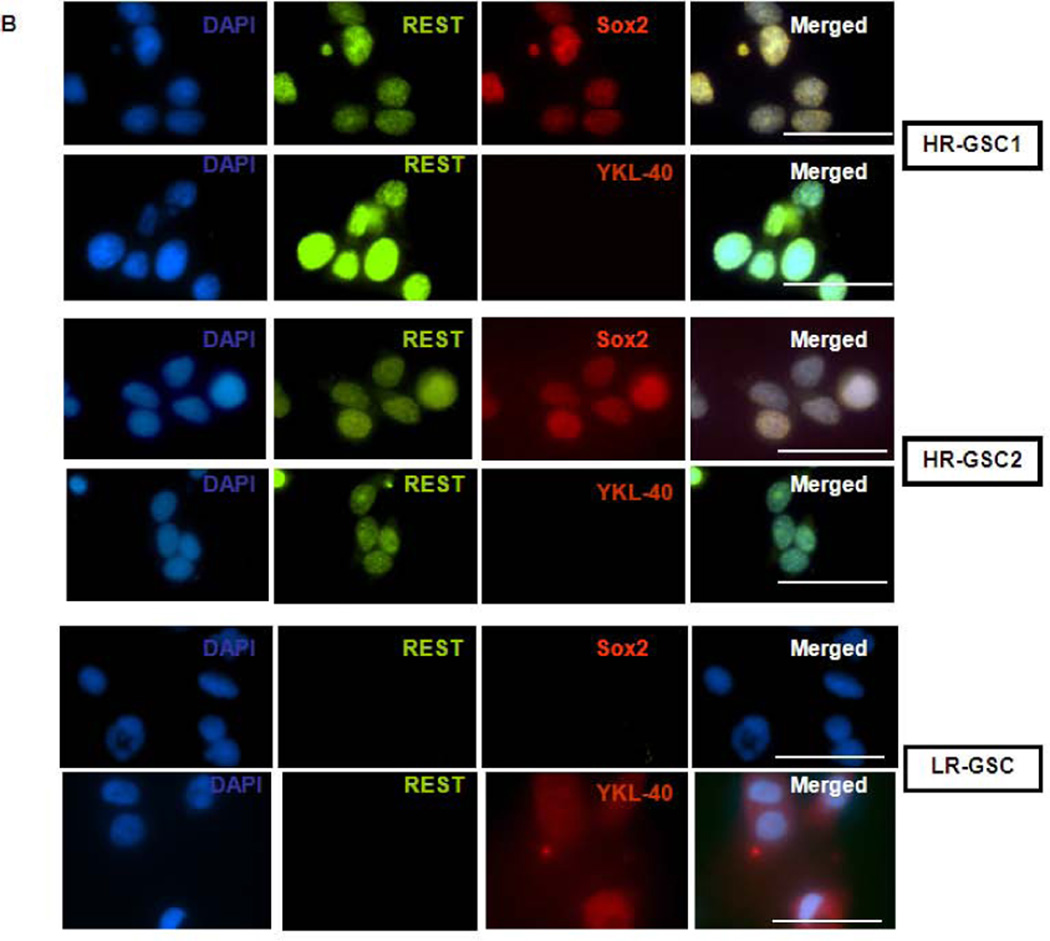
Figure 2. High REST protein expression correlates with stemness markers in GSCs.
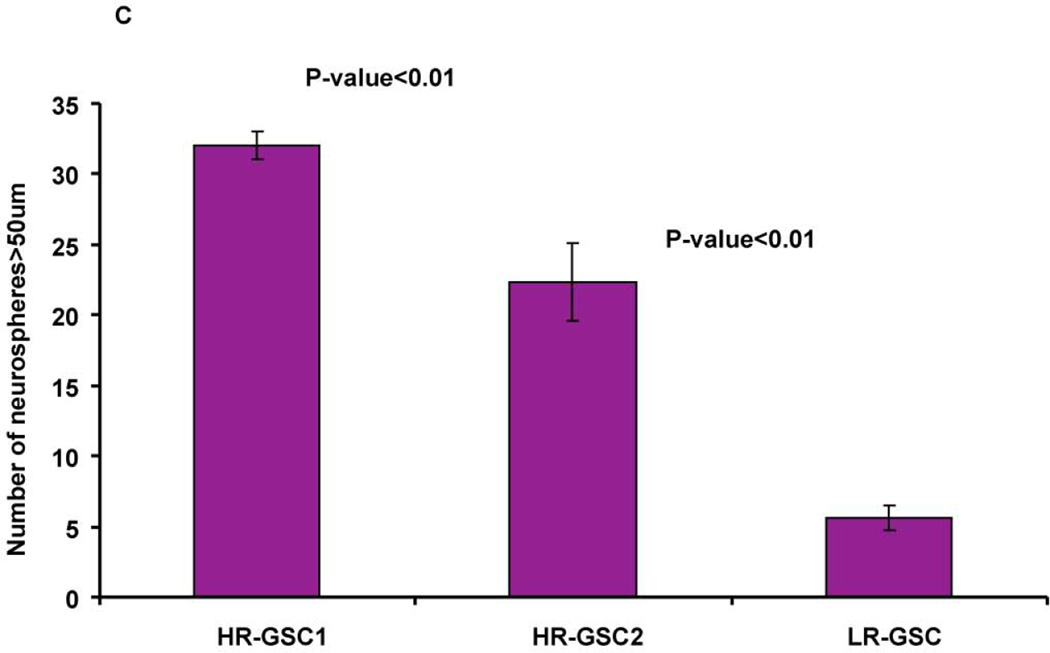
(A) Expression of REST, Nestin, Bmi1, Sox2, and YKL40 in two HR-GSCs (HR-GSC1 and HR-GSC2) and one LR-GSC were determined by Western blotting assays. Actin was used as an internal control. (B) Immunofluorescence analysis of the two HR-GSCs and one LR-GSC shows a distinct pattern of expression of Sox2, and YKL-40. The bar in the figures represents 50 micron. (C) HR-GSCs (HR-GSC1 and HR-GSC2) show higher neurosphere-formation ability compared to LR-GSCs.
To determine if high levels of REST in GSCs corresponded to increased functional stem-like properties, we measured their self-renewal efficiency using neurosphere assays. Our results showed greater neurosphere formation ability of both HR-GSC1 and HR-GSC-2 compared to LR-GSCs (Fig. 2C). Thus, our data indicated that high REST expression is associated with high stemness of GSCs both at marker and functional levels.
REST maintains self-renewal of GSCs
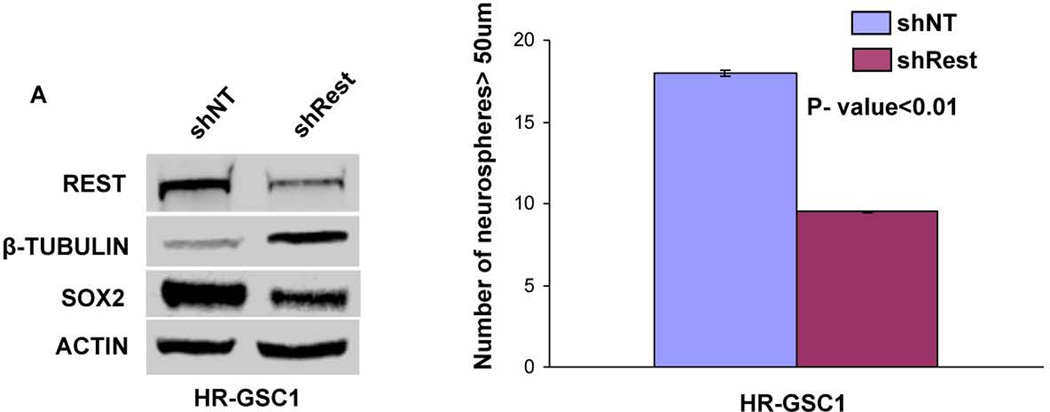
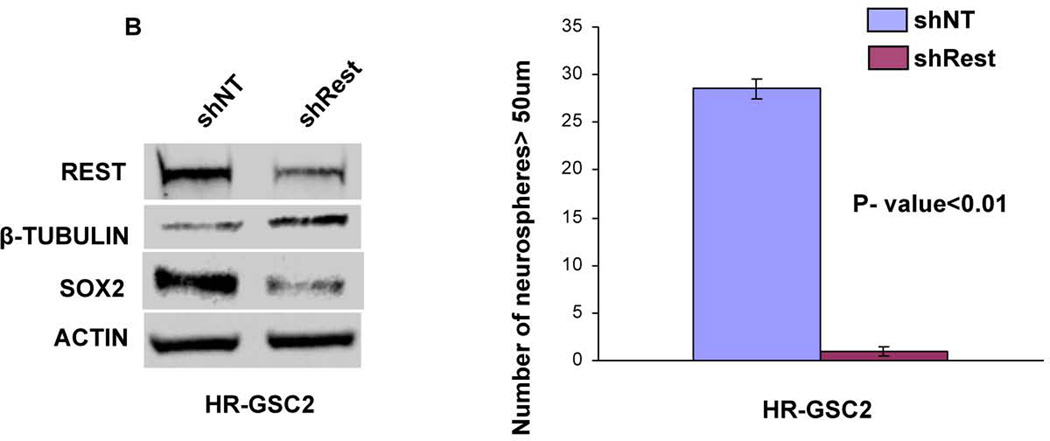
To determine the functional role of REST in GSCs, we generated stable lines expressing either shNT control or shRest in HR-GSC1 and HR-GSC2. As shown in Figures 3A and 3B, Western blot analysis confirmed REST protein knockdown in the shRest clones in both HR-GSC lines. The knockdown of REST corresponded to increased expression of neuronal β-tubulin, a direct target gene of Rest46, 47, indicating that shRest caused a functional REST knockdown. The loss of REST also produced concomitant loss of the stemness regulator Sox2. To determine whether loss of REST resulted in functional loss of stem like properties of GSCs, we performed self-renewal assays in both HR-GSCs lines. Our results showed that loss of REST resulted in decreased self-renewal in both the high REST lines over multiple passages (Supplementary Figs. 1A for HR-GSC1 and Fig. 1B for HR-GSC2), indicating that REST maintains self-renewal in both HR-GSC lines.
Figure 3. REST controls self-renewal of GSCs.
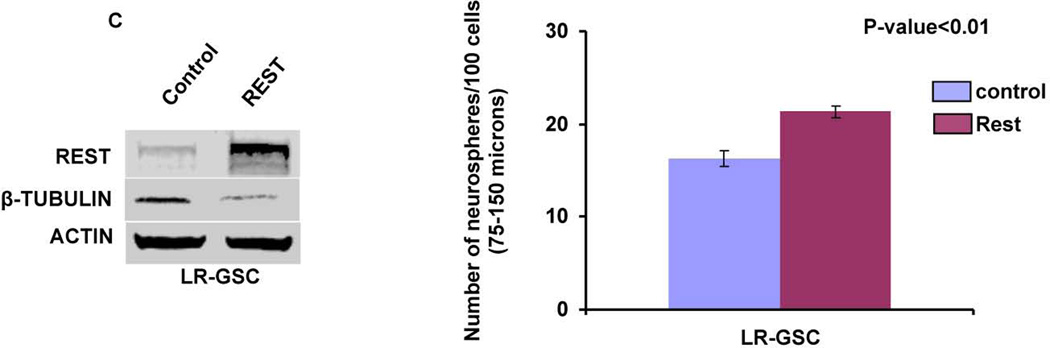
(A, B) Rest loss-of-function experiments. Knockdown of REST with shRest (shNT as a control) in HR-GSC1 (A) and HR-GSC2 (B) caused lowered expression of REST, higher expression of the direct REST target neuronal beta-tubulin and lower expression of neural stem cell self-renewal regulator Sox2 as determined by Western blotting assay (left panels), and lowered self-renewal ability (right panels) (n=48). (C) REST gain-of-function experiments. Expression of exogenous REST in LR-GSCs (expression of exogenous GFP served as a control) caused decreased β-tubulin expression (left panel) and increased self-renewal ability (right panel) (n=48).
To further establish the role of REST in GSCs, we performed complementary experiments in which we transfected LR-GSCs with expression vectors encoding either non targeting control or Rest, confirmed the increased REST protein expression by Western blot and determined the cells’ self-renewal capacity (Fig. 3C). As shown, higher REST expression produced lower expression of its target gene neuronal beta-tubulin and caused a significant increase in self-renewal. As expected from the loss-of-function experiments, additional REST caused an increase in the expression of Sox2 levels (Supplementary Fig. 2). Thus, taken together, these results showed that REST maintains self-renewal of GSCs.
HR-GSCs and LR-GSCs produce distinct brain tumors in mice
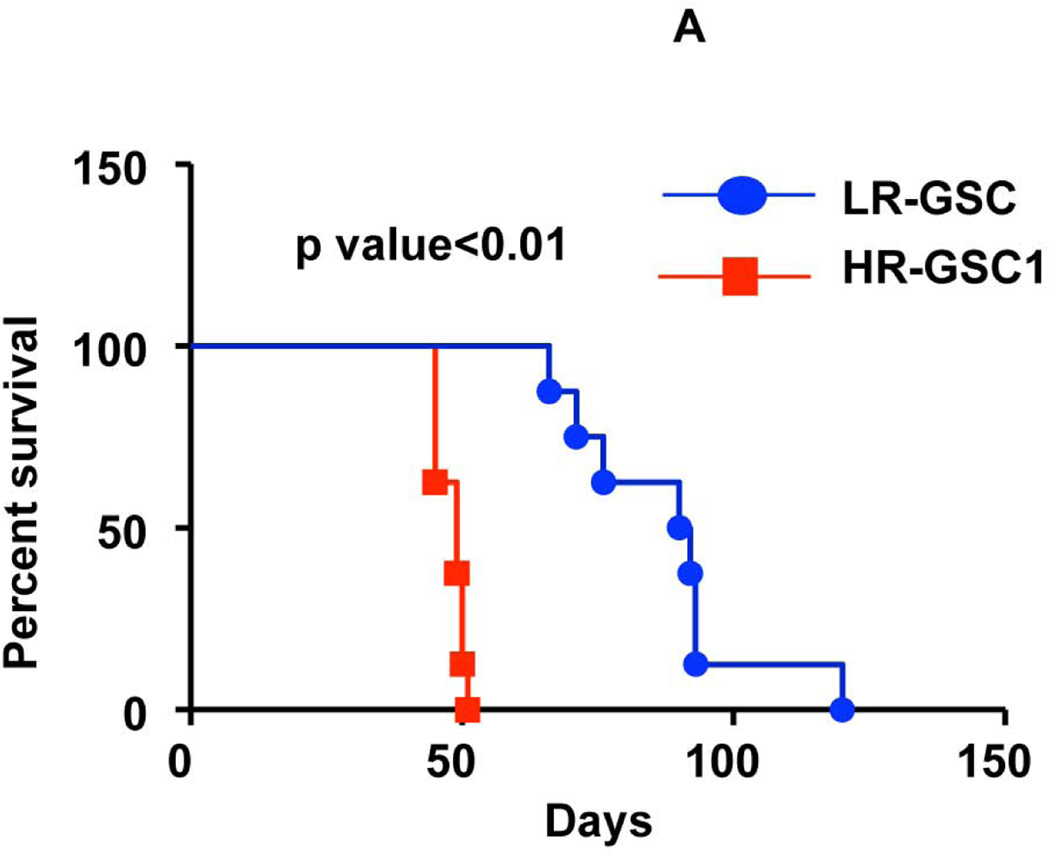
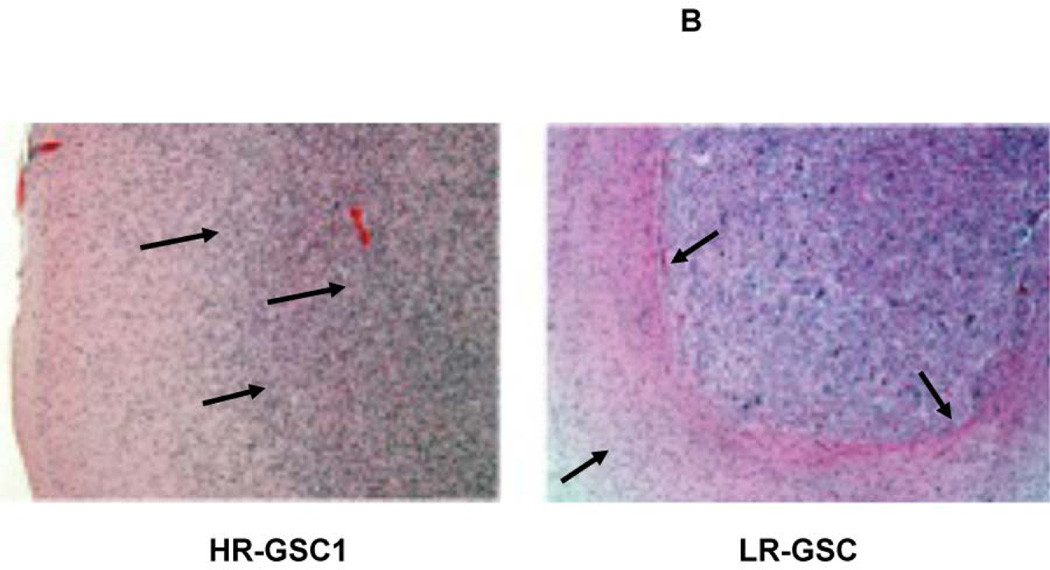
To determine the tumor-producing properties of the HR- and LR-GSCs, we transplanted HR-GSC1 and LR-GSC cells (5×104 cells) into the brains of nude mice and performed mouse survival assays (Fig. 4A) and histopathological examination of the resultant tumors (Fig. 4B). HR-GSC1 cells produced shorter survival in mice compared with LR-GSCs. This corresponded to their respective self-renewal capacity (Fig. 2C). While both HR- and the LR-GSC lines produced tumors, HR-GSC1 tumors morphologically showed a marked difference in growth pattern when compared with the LR-GSC tumors. The HR-GSC1 tumors were more diffusely infiltrative than the LR-GSC tumors, which grew as relatively circumscribed expansive masses.
Figure 4. REST regulates survival in mice bearing GSC-mediated brain tumors.
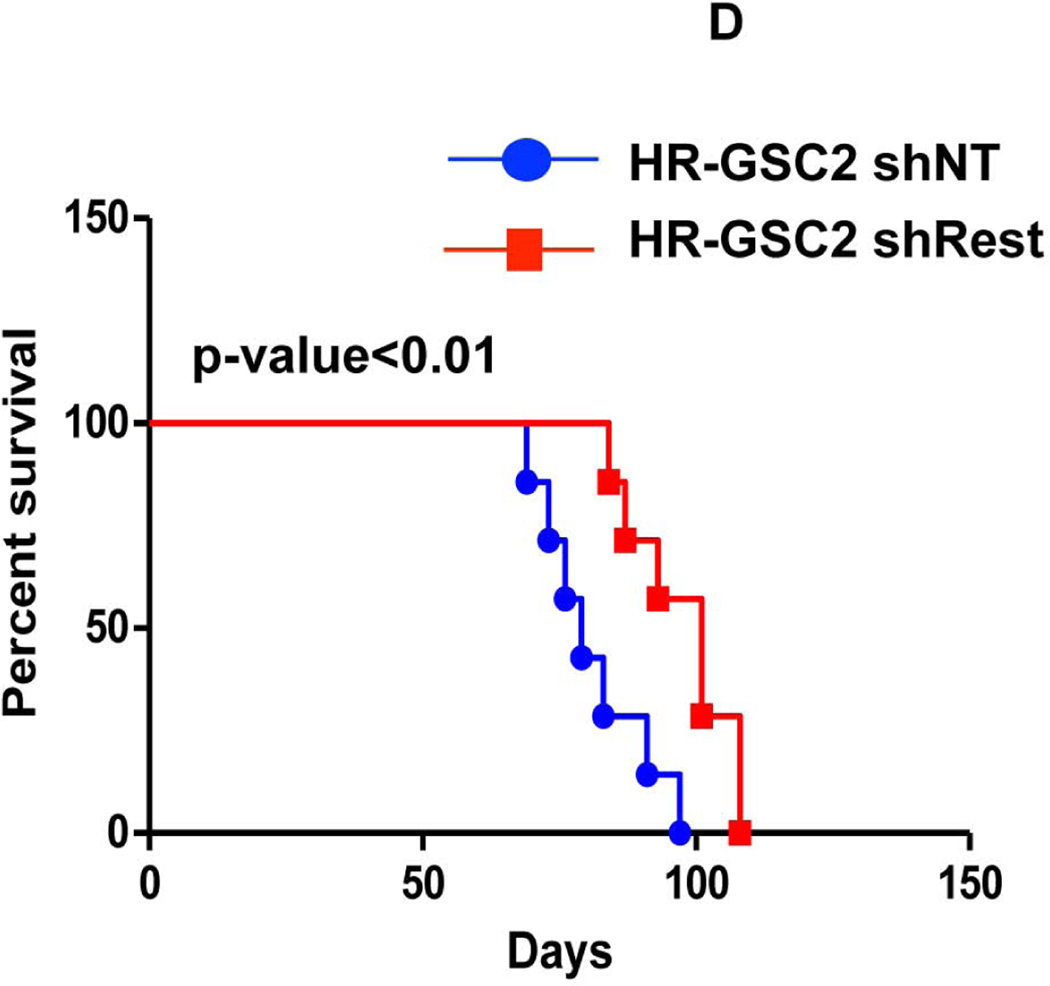
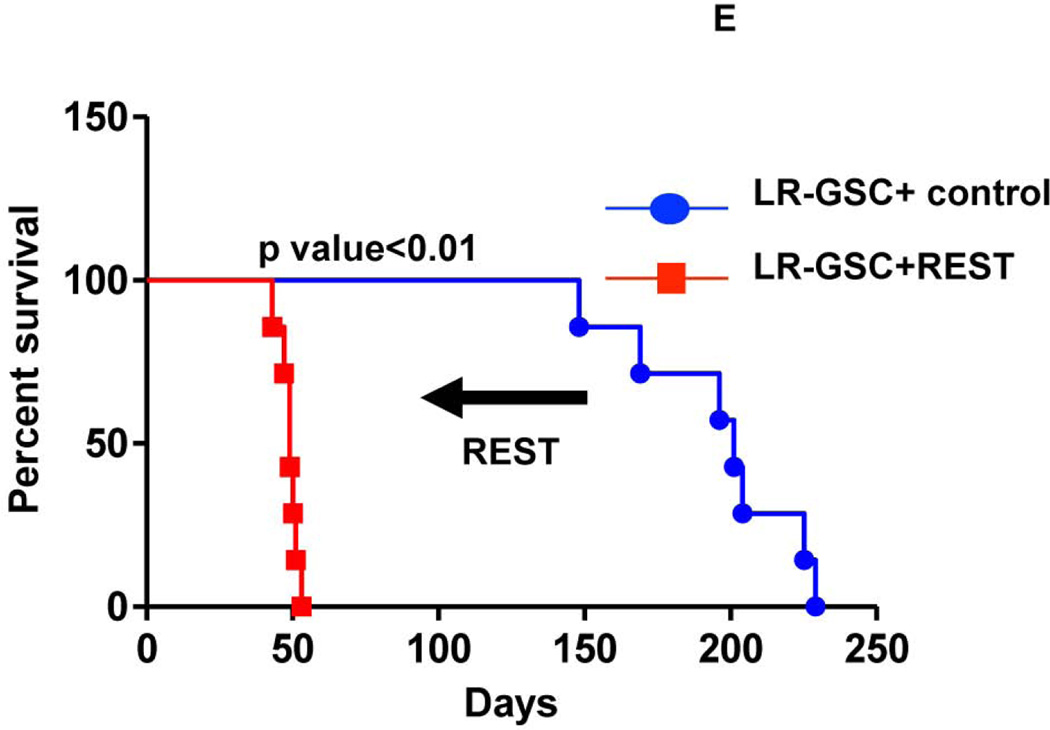
(A, B) HR-GSC1 and LR-GSC cells were transplanted into the brains of nude mice, the mice were assayed for survival using Kaplan-Meier method (A) and histopathology of the resulting tumors were examined by H&E staining (B) (n=7 mice per treatment group). HR-GSC1 tumors produced shorter survival in mice than LR-GSC tumors and HR-GSC1 tumors were more infiltrative than LR-GSC tumors, which showed circumscribed masses. Arrows in the same direction indicate more infiltrative cells while those in the opposite direction represent more restrictive structure. (C, D) Rest loss-of-function tumors. Stable lines of HR-GSC1 (C) and HR-GSC2 (D) cells expressing either shREST or shNT were transplanted into the brains of nude mice and mouse survival was compared. Knockdown of REST produced increased survival in tumor-bearing mice generated by both lines (n=7 mice per treatment group). (D) Rest gain-of-function tumors. LR-GSCs expressing either exogenous REST or GFP were transplanted into the brains of nude mice, the mice were assayed for survival. Additional REST converted LR-GSCs into more infiltrative tumors and decreased survival in tumor-bearing mice (n=7 mice per treatment group).
REST regulates survival in orthotropic mouse model
We performed loss-of-function experiments, in which we transplanted 5×104 cells of the stable lines of HR-GSC1 (Fig. 4C) and HR-GSC2 (Fig. 4D) expressing either shRest or shNTinto the brains of nude mice and assayed for their survival. Under these conditions, shRest treatment, as compared with shNT treatment, increased the survival of tumor-bearing mice. Although the effect in mouse survival was statistically significant, it was not robust and perhaps points to the notion that survival depended on multiple factors. To further confirm that these experiments were performed under conditions, in which the survival correlated with the number of cells injected, we compared the survival of mice harboring HR-GSC1 cells expressing either shRest or shNT and either 5×104 (Fig. 4C) or 5×103 (Supplementary Fig. 3) per mouse. All mice bearing shNT control cells died within 64 days for 5×104 cells compared to within 112 days for 5×103 cells, indicating that survival corresponded inversely to the number of tumor cells transplanted. Thus, these results suggest that by regulating self-renewal in GSCs, REST expression also correlated with survival of GSC tumor-bearing mice.
To further establish the control of survival by REST, we performed gain-of-function experiments, in which we transplanted LR-GSCs expressing REST that we previously found to express higher levels of the REST protein, lower levels of its target gene, neuronal β-tubulin, and concomitant higher self-renewal ability into the brains of nude mice. We then assayed for survival (Fig. 4E). As shown, the addition of exogenous REST, compared with that of non-targeting control, to LR-GSCs caused decreased survival in tumor-bearing mice. Thus, REST controls survival in mice bearing GSC-mediated brain tumors
REST suppresses apoptosis in GSC-mediated tumors
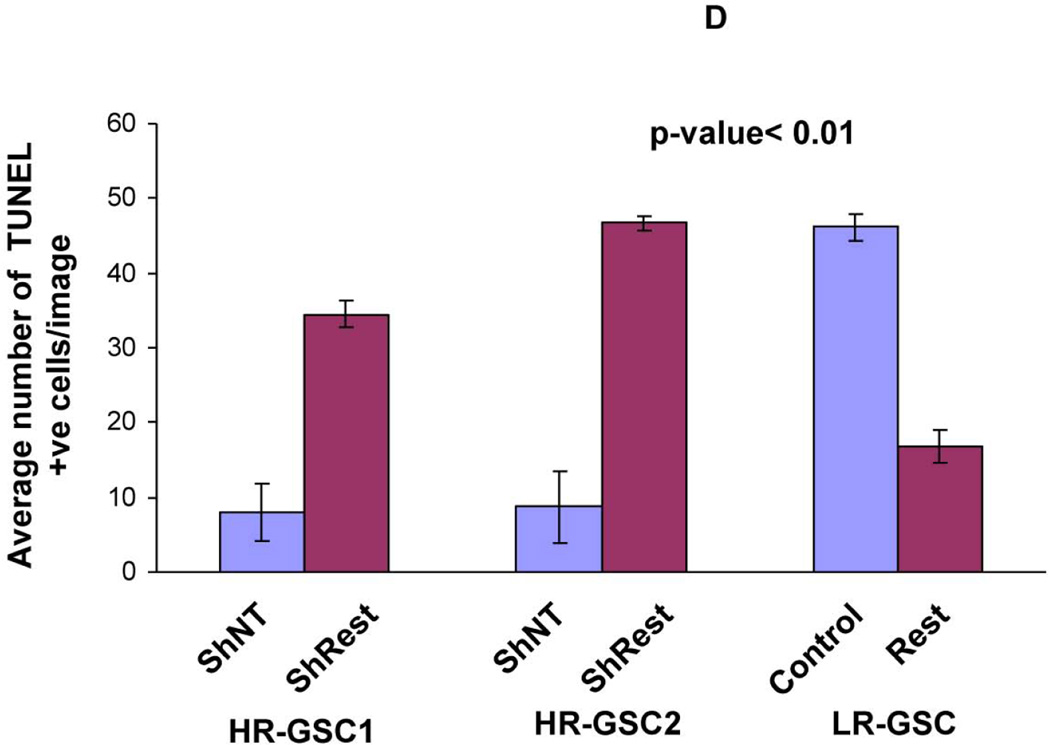
To determine if the tumorigenic effects of REST in mouse brain tumors were partly regulated by promoting GSC survival, we analyzed our orthotopic mouse tumor sections for apoptosis using TUNEL assays. The data showed increased cell death upon REST knockdown in both HR-GSC lines (Fig. 5A and Fig. 5B) and, conversely, decreased cell death upon expression of exogenous REST in the LR-GSC line (Fig. 5C). Quantification of the TUNEL assay is shown in Fig. 5D. The in vivo TUNEL staining was concordant with the BrdU-incorporation in cultured cells and showed a decrease in cell proliferation for the two HR-GSC lines upon REST knockdown (Supplementary Fig. 4). These results indicated that REST suppresses apoptosis in GSC tumors.
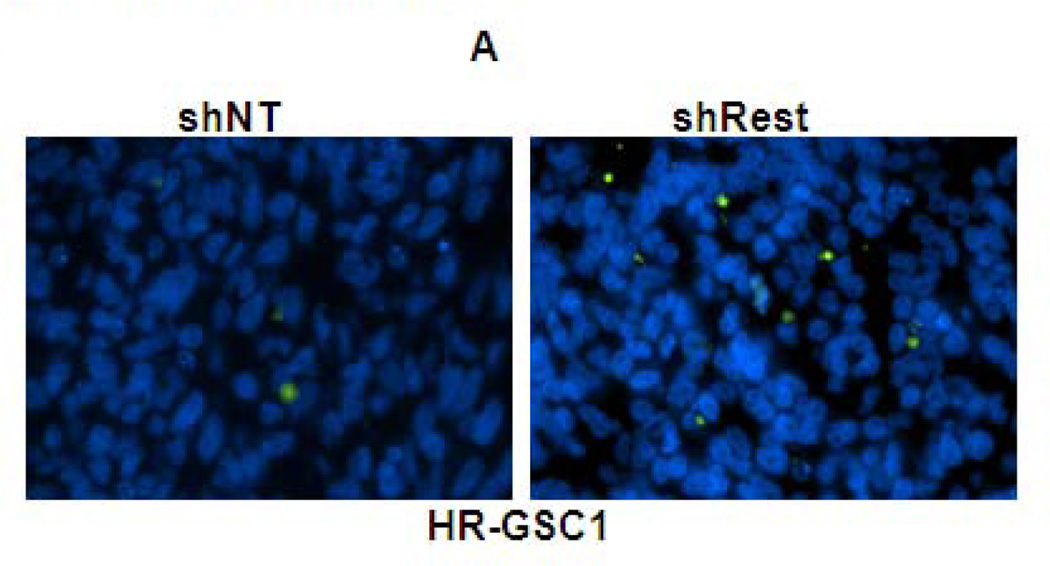
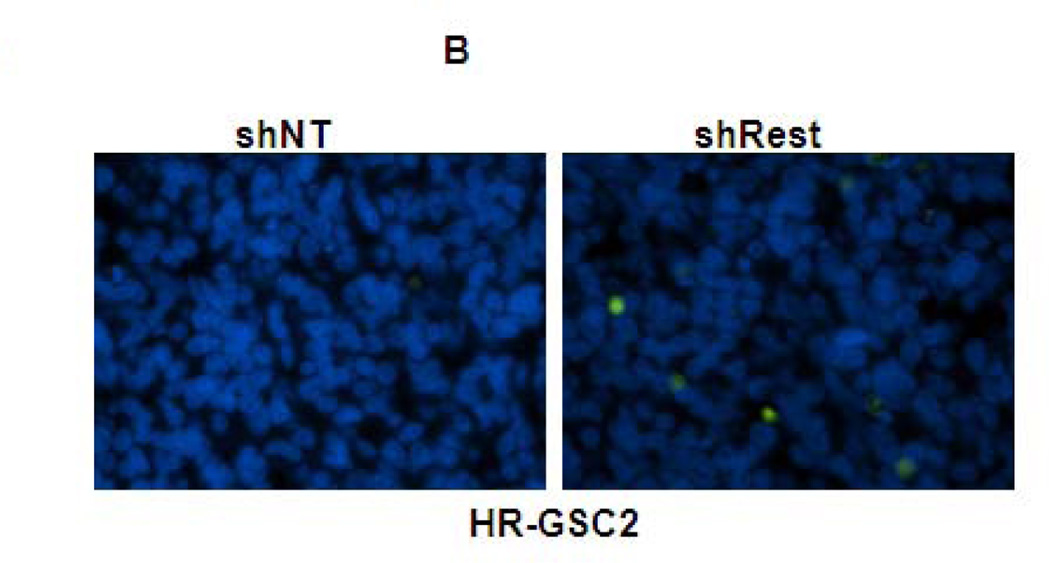
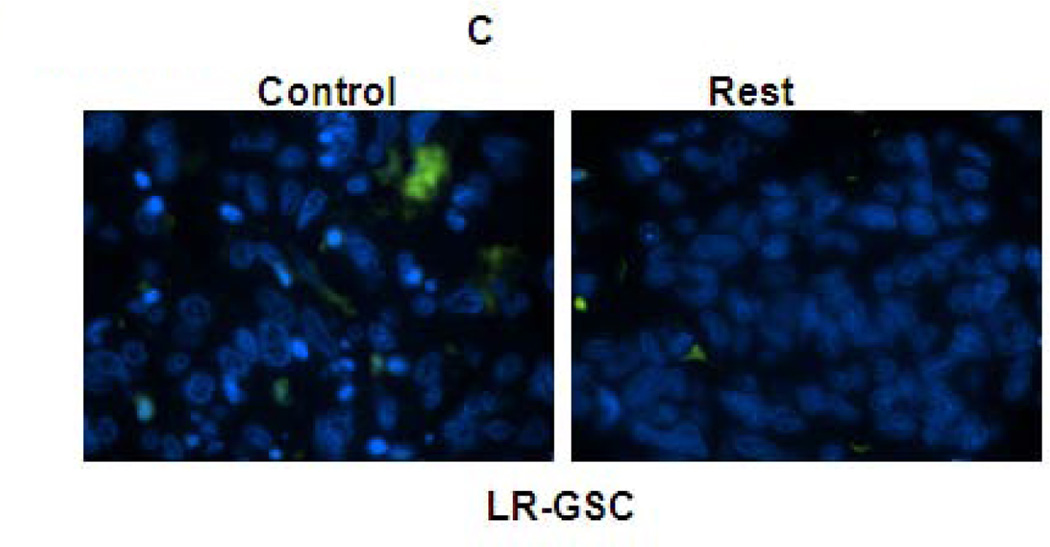
Figure 5. REST suppresses apoptosis in GSC-mediated brain tumors.
(A–C) In vivo TUNEL assay indicating increased apoptosis in mouse tumors upon knockdown of REST in HR-GSC1 (A) and HR-GSC2 (B). LR-GSCs showed reduced apoptosis upon expression of exogenous REST compared to exogenous GFP control (C). (D) quantification of in vivo TUNEL assays shown in A–C.
Genome-wide mRNA expression analysis reveals further potential oncogenic properties of REST
Because REST regulates numerous genes, it is likely that REST controls tumorigenicity of GSCs at multiple regulatory points. To determine what pathways could be affected by REST alteration in GSCs, in addition to self-renewal and cell viability, we compared genome-wide mRNA expression profiling of the six GSC lines that we characterized above: HR-GSC1+shNT, HR-GSC1+shRest, HR-GSC2+shNT, HR-GSC2+shRest, LR-GSC+GFP, and LR-GSC+Rest. We identified 301 genes that were more than 1.5 fold differentially expressed across the HR- versus the LR-GSC lines. We then identified core biological pathways and cellular functions associated with REST expression using the Ingenuity Pathway Analysis (Table 1). The pathways involving cellular movement, cellular assembly and organization, cellular development, cell-to-cell signaling and interaction, and cellular growth and proliferation were most significantly associated with REST.
Table 1. Potential pathways regulated by REST in GSCs.
Ingenuity Pathway Analysis of genome-wide expression profiles of GSCs with loss- and gain-of-function manipulations.
| Molecular and Cellular Functions | |||
|---|---|---|---|
| Name | p-value | # Molecules | |
| Cellular Movement | 1.99E-08 - 8.23E-03 | 63 | |
| Cellular Assembly and Organization | 2.75E-08 - 7.77E-03 | 49 | |
| Cellular Development | 4.61E-06 - 8.58E-03 | 80 | |
| Cell-To-Cell Signaling and Interaction | 6.96E-06 - 8.58E-03 | 68 | |
| Cellular Growth and Proliferation | 1.23E-05 - 8.62E-03 | 77 | |
| Physiological System Development and Function | |||
| Name | p-value | # Molecules | |
| Tissue Development | 2.75E-08 - 8.56E-03 | 75 | |
| Organismal Development | 5.97E-07 - 8.77E-03 | 64 | |
| Cardiovascular System Development and Function | 1.25E-06 - 8.02E-03 | 40 | |
| Connective Tissue Development and Function | 1.79E-06 - 7.77E-03 | 35 | |
| Skeletal and Muscular System Development and Function | 1.79E-06 - 8.77E-03 | 41 | |
| ID | Associated Network Functions | Score | |
| 1 | Drug Metabolism, Endocrine System Development and Function, Lipid Metabolism | 50 | |
| 2 | Cell-To-Cell Signaling and Interaction, Cellular Function and Maintenance, Drug Metabolism | 32 | |
| 3 | Cellular Movement, Cell Morphology, Immune Cell Trafficking | 30 | |
| 4 | Lipid Metabolism, Molecular Transport, Small Molecule Biochemistry | 27 | |
| 5 | Cell Death, Cellular Development, Cellular Growth and Proliferation | 26 | |
REST regulates invasion of GSC-mediated orthotopic tumors
Since the genome-wide results suggested REST to play a major role in cellular movement, we further investigated whether REST regulated invasion of GSCs using in vitro invasion assays. Our data showed a statistically significant decrease of invasion in the two HR-GSC lines upon REST knock down with HR-GSC1 showing comparatively higher degree of infiltration than HR-GSC2 (Supplementary Fig. 5).
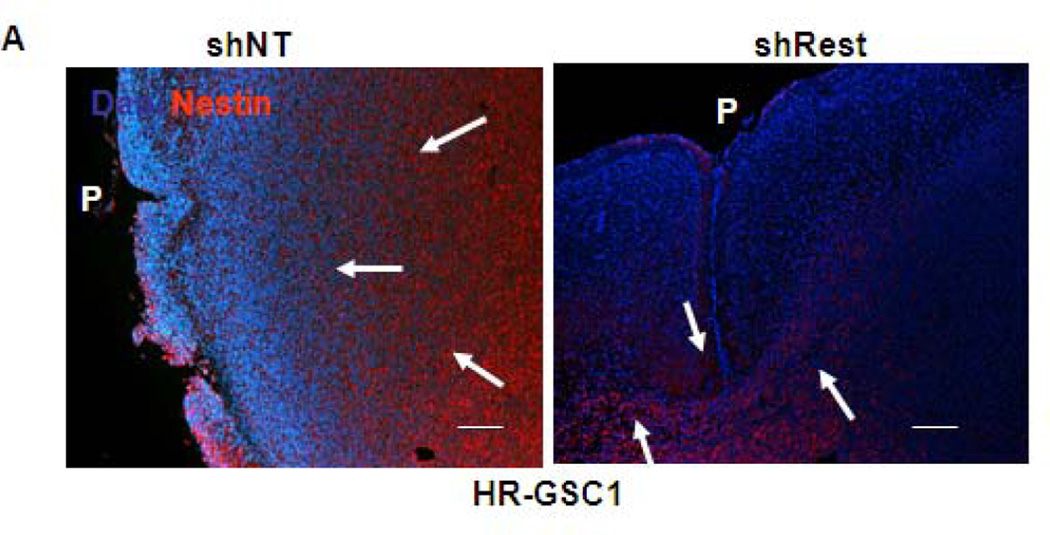
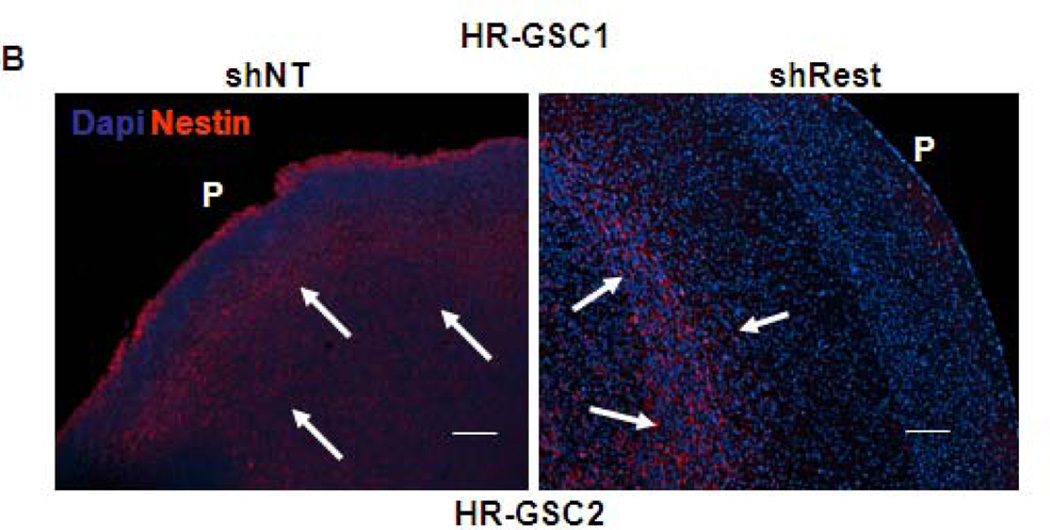
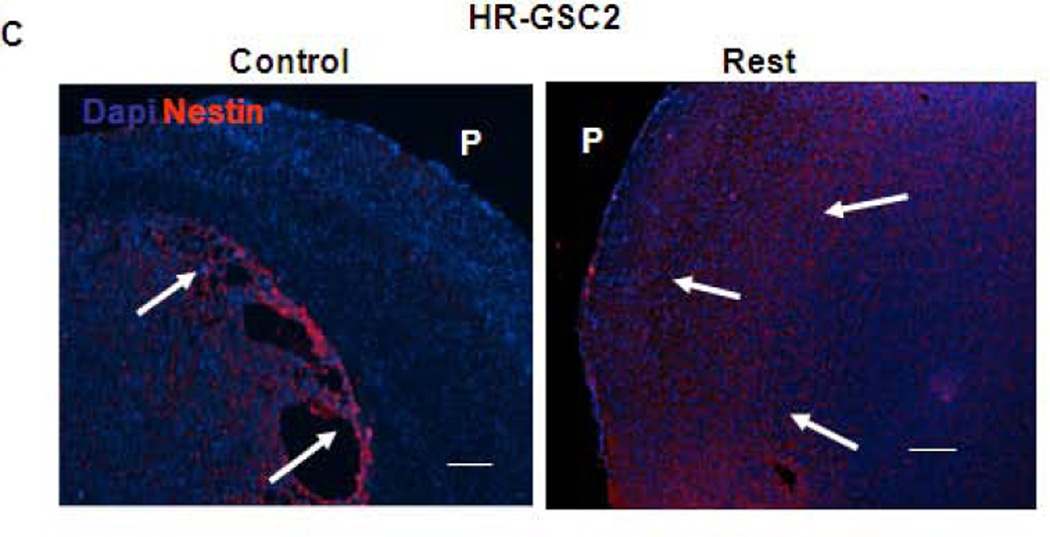
To further establish the role of REST in GSC-mediated invasion, we re-examined the tumor sections generated during orthotopic mouse experiments described in Fig 4 by staining the tumor sections with a human nestin antibody to distinguish the human tumor-derived cells from the surrounding mouse cells (Fig. 6A – 6C)48. This approach helps in visualizing the injected human cells in the midst of mouse brain cells present in the mouse xenograft tumors but cannot determine the cellular status of the injected cells, such as their stemness or differentiation properties. Our results indicated that the shNT-treated HR-GSC1 and HR-GSC2 lines showed very high degree of migration with cell invasion to the pial surface from the core of the tumor. In contrast, the corresponding shRest-treated cells showed a decreased migratory phenotype with a circumscribed tumor growth, where cells rarely reached the pial surface. In the complementary experiment, exogenous REST expression in LR-GSCs resulted in a diffuse tumor growth and significant increase of cellular invasion compared to the control cells, in which GFP was expressed. Remarkably, the expression of additional REST in LR-GSCs converted the tumors such that they were similar to HR-GSCs. Taken together, these results suggest a major in vitro and in vivo role of REST in regulating tumor phenotype and invasion in GSCs.
Figure 6. REST regulates invasion in GSC-mediated brain tumors.
(A–C) Both HR-GSC1 (A)- and HR-GSC2 (B)-mediated tumors showed decreased invasion upon shRest-treatment compared to control shNT-treatment. LR-GSC-mediated tumors showed increased invasion upon expression of exogenous REST compared to GFP control (C). Arrows in the same direction indicate more infiltrative cells while those in the opposite direction represent more restrictive structure.P = pial layer. Bar = 50 micron.
Discussion
Here we show that REST promotes oncogenic properties of GSCs with respect to self-renewal, cellular viability, and invasion and affects survival in mice bearing GSC-xenograft tumors. Our work does not reflect the GBM tumor, which will presumably contain many other types of cells than just the GSCs. Previously, we found that REST, which is also over-expressed in a major subtype of human medulloblastoma tumors that are mostly in the neuronal pathway, caused tumorigenesis by blocking the differentiation of cerebellar stem/progenitor cells30, 31. Thus, these results suggest that the conservation of stemness through REST contributes to both neuronal and glial tumors and that deregulation of normal REST expression is a major factor in producing these tumors. Our results also indicate that REST expression in GSCs is mainly regulated at the protein degradation level suggesting that patient outcome should be determined based on REST expression at the protein, and not transcript, levels. Thus, the public datasets, which are based on the transcript levels, cannot be used to determine the prognostic values of the HR- versus LR-GBM patient population.
Our results indicate that expression of exogenous REST in LR-GSCs produce more invasive tumors in the mouse brain. The mechanism by which REST regulates invasive properties is unclear. Neural stem cells carrying exogenous genes were found to specifically target mouse intracranial glioma tumors either when transplanted at a site distant from the tumor or injected into the tail veins, providing a promising means of delivery of therapeutic molecules to the tumor49. The targeting activity of the NSCs is presumably regulated by cell-cell and cell-microenvironment interactions. Whether the high-level expression of REST in HR-GSCs would affect the tumor-targeting process of NSCs is unknown. We are currently exploring ways to answer these questions.
Studies in ES cells have revealed that self-renewal in these cells is regulated by an interconnected regulatory circuit consisting of many factors, including Oct4, Sox2, and Nanog, rather than by a single molecule50. Similarly, REST was found to be a part of the interconnected network regulating ES cell self-renewal and pluripotency50–52. This suggests that self-renewal in GSCs, as well as in normal neural stem cells, might be controlled by a circuit, in which there is constant cross-talk between the regulators of self-renewal and the microenvironment (niche), producing a dynamic equilibrium; when individual regulators of the circuit are influenced by the niche, the equilibrium is re-set. To understand GBM tumorigenesis and to design a more appropriate targeted therapy for GBM than what is currently available, it will be important to identify and characterize the other players in this self-renewal circuit.
Supplementary Material
Supplementary Figure 1. REST regulates self-renewal of GSCs over multiple passages. Rest knockdown in both HR-GSC1 (A) and HR-GSC2 (B) with shRest, as compared to control shNT, caused decreased self-renewal over multiple passages (P1-P3).
Supplementary Figure 2. REST gain-of-function experiments. Expression of exogenous REST in LR-GSCs (expression of exogenous GFP served as a control) caused increased expression of Sox2. Actin was used as an internal control.
Supplementary Figure 3. REST regulates survival in mice bearing GSC-mediated brain tumors. Survival of mice bearing tumors generated by 5×103 cells of the stable lines of HR-GSC1 expressing either shRest or shNT (compare Fig. 4C for tumors with 5×104 cells) (n=7 mice per treatment group).
Supplementary Figure 4. REST regulates cell-proliferation of GSCs in culture. BrdU-incorporation assays indicated that knockdown of REST by shRest in both HR-GSC1 and HR-GSC2 lowered cell-proliferation ability when compared to shNT control.
Supplementary Figure 5. REST regulates invasion of GSC in vitro. Invasion chamber assays indicated decreased invasion property of both the two HR-GSC lines upon REST knock down using shRest treatment (shNT-treatment served as a control).
Acknowledgements
This work was partly supported by grants from the NCI (CA97124 and CA81255 to S.M.). P.S. is the recipient of Dodie P. Hawn Fellowship in Genetics.
Footnotes
Authors’ contribution(s):
MMK, PS, SKS, POZ, AM: Conception and design, Collection and/or assembly of data, Data analysis and interpretation, Manuscript writing, Final approval of manuscript
SL: Conception and design, Data analysis and interpretation, Manuscript writing
JG, DS: Provision of study material or patients, Collection and/or assembly of data, Data analysis and interpretation
HOE: Financial support, Administrative support
HC: Provision of study material or patients
GNF, FFL: Conception and design, Provision of study material or patients, Collection and/or assembly of data, Data analysis and interpretation
SM: Conception and design, Financial support, Administrative support, Data analysis and interpretation, Manuscript writing, Final approval of manuscript
The authors declare no competing financial interests.
References
- 1.Furnari FB, Fenton T, Bachoo RM, et al. Malignant astrocytic glioma: genetics, biology, and paths to treatment. Genes Dev. 2007;21:2683–2710. doi: 10.1101/gad.1596707. [DOI] [PubMed] [Google Scholar]
- 2.Gladson CL, Prayson RA, Liu WM. The pathobiology of glioma tumors. Annu Rev Pathol. 2010;5:33–50. doi: 10.1146/annurev-pathol-121808-102109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Louis DN, Ohgaki H, Wiestler OD, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114:97–109. doi: 10.1007/s00401-007-0243-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stupp R, Hegi ME, Mason WP, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. The Lancet Oncology. 2009;10:459–466. doi: 10.1016/S1470-2045(09)70025-7. [DOI] [PubMed] [Google Scholar]
- 5.Gilbertson RJ, Gutmann DH. Tumorigenesis in the brain: location, location, location. Cancer Res. 2007;67:5579–5582. doi: 10.1158/0008-5472.CAN-07-0760. [DOI] [PubMed] [Google Scholar]
- 6.Sathornsumetee S, Rich JN, Reardon DA. Diagnosis and treatment of high-grade astrocytoma. Neurol Clin. 2007;25:1111–1139. x. doi: 10.1016/j.ncl.2007.07.004. [DOI] [PubMed] [Google Scholar]
- 7.Hodgson JG, Yeh RF, Ray A, et al. Comparative analyses of gene copy number and mRNA expression in glioblastoma multiforme tumors and xenografts. Neuro Oncol. 2009;11:477–487. doi: 10.1215/15228517-2008-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huse JT, Holland EC. Targeting brain cancer: advances in the molecular pathology of malignant glioma and medulloblastoma. Nat Rev Cancer. 2010;10:319–331. doi: 10.1038/nrc2818. [DOI] [PubMed] [Google Scholar]
- 9.Phillips HS, Kharbanda S, Chen R, et al. Molecular subclasses of high-grade glioma predict prognosis, delineate a pattern of disease progression, and resemble stages in neurogenesis. Cancer Cell. 2006;9:157–173. doi: 10.1016/j.ccr.2006.02.019. [DOI] [PubMed] [Google Scholar]
- 10.TCGA. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature. 2008;455:1061–1068. doi: 10.1038/nature07385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Verhaak RG, Hoadley KA, Purdom E, et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell. 2010;17:98–110. doi: 10.1016/j.ccr.2009.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gerber DE, Laterra J. Emerging monoclonal antibody therapies for malignant gliomas. Expert Opin Investig Drugs. 2007;16:477–494. doi: 10.1517/13543784.16.4.477. [DOI] [PubMed] [Google Scholar]
- 13.Mineharu Y, King GD, Muhammad AK, et al. Engineering the brain tumor microenvironment enhances the efficacy of dendritic cell vaccination: implications for clinical trial design. Clin Cancer Res. 2011;17:4705–4718. doi: 10.1158/1078-0432.CCR-11-0915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Serwer LP, Noble CO, Michaud K, et al. Investigation of intravenous delivery of nanoliposomal topotecan for activity against orthotopic glioblastoma xenografts. Neuro Oncol. 2011 doi: 10.1093/neuonc/nor139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sawaya R. Fifty years of neurosurgery argue in favor of glioma resection. Clin Neurosurg. 2001;48:10–19. [PubMed] [Google Scholar]
- 16.Bao S, Wu Q, McLendon RE, et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature. 2006;444:756–760. doi: 10.1038/nature05236. [DOI] [PubMed] [Google Scholar]
- 17.Bryant MJ, Chuah TL, Luff J, et al. A novel rat model for glioblastoma multiforme using a bioluminescent F98 cell line. J Clin Neurosci. 2008;15:545–551. doi: 10.1016/j.jocn.2007.04.022. [DOI] [PubMed] [Google Scholar]
- 18.Llaguno SA, Chen J, Kwon CH, et al. Neural and cancer stem cells in tumor suppressor mouse models of malignant astrocytoma. Cold Spring Harb Symp Quant Biol. 2008;73:421–426. doi: 10.1101/sqb.2008.73.005. [DOI] [PubMed] [Google Scholar]
- 19.Weiss WA, Israel M, Cobbs C, et al. Neuropathology of genetically engineered mice: consensus report and recommendations from an international forum. Oncogene. 2002;21:7453–7463. doi: 10.1038/sj.onc.1205936. [DOI] [PubMed] [Google Scholar]
- 20.Jandial R, U H, Levy ML, et al. Brain tumor stem cells and the tumor microenvironmen. Neurosurg Focus. 2008;24:E27. doi: 10.3171/FOC/2008/24/3-4/E26. [DOI] [PubMed] [Google Scholar]
- 21.Singh SK, Clarke ID, Terasaki M, et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63:5821–5828. [PubMed] [Google Scholar]
- 22.Stiles CD, Rowitch DH. Glioma stem cells: a midterm exam. Neuron. 2008;58:832–846. doi: 10.1016/j.neuron.2008.05.031. [DOI] [PubMed] [Google Scholar]
- 23.James CD, Cavenee WK. Stem Cells for Treating Glioblastoma: How Close to Reality? Neuro-Oncology. 2009;11:101. doi: 10.1215/15228517-2009-016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kagalwala MN, Singh SK, Majumder S. Stemness Is Only a State of the Cell. Cold Spring Harbor Symposia on Quantitative Biology. 2008;73:227–234. doi: 10.1101/sqb.2008.73.042. [DOI] [PubMed] [Google Scholar]
- 25.Gopalakrishnan V. REST and the RESTless: in stem cells and beyond. Future Neurol. 2009;4:317–329. doi: 10.2217/fnl.09.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ballas N, Grunseich C, Lu DD, et al. REST and Its Corepressors Mediate Plasticity of Neuronal Gene Chromatin throughout Neurogenesis. Cell. 2005;121:645–657. doi: 10.1016/j.cell.2005.03.013. [DOI] [PubMed] [Google Scholar]
- 27.Bruce AW, López-Contreras AsJ, Flicek P, et al. Functional diversity for REST (NRSF) is defined by in vivo binding affinity hierarchies at the DNA sequence level. Genome Research. 2009;19:994–1005. doi: 10.1101/gr.089086.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Singh SK, Kagalwala MN, Parker-Thornburg J, et al. REST maintains self-renewal and pluripotency of embryonic stem cells. Nature. 2008;453:223–227. doi: 10.1038/nature06863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gao Z, Ure K, Ding P, et al. The Master Negative Regulator REST/NRSF Controls Adult Neurogenesis by Restraining the Neurogenic Program in Quiescent Stem Cells. The Journal of Neuroscience. 2011;31:9772–9786. doi: 10.1523/JNEUROSCI.1604-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fuller GN, Su X, Price RE, et al. Many human medulloblastoma tumors overexpress repressor element-1 silencing transcription (REST)/neuron-restrictive silencer factor, which can be functionally countered by REST-VP16. Molecular Cancer Therapeutics. 2005;4:343–349. doi: 10.1158/1535-7163.MCT-04-0228. [DOI] [PubMed] [Google Scholar]
- 31.Lawinger P, Venugopal R, Guo ZS, et al. The neuronal repressor REST/NRSF is an essential regulator in medulloblastoma cells. Nat Med. 2000;6:826–831. doi: 10.1038/77565. [DOI] [PubMed] [Google Scholar]
- 32.Su X, Gopalakrishnan V, Stearns D, et al. Abnormal expression of REST/NRSF and Myc in neural stem/progenitor cells causes cerebellar tumors by blocking neuronal differentiation. Mol Cell Biol. 2006;26:1666–1678. doi: 10.1128/MCB.26.5.1666-1678.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McCord AM, Jamal M, Shankavarum UT, et al. Physiologic Oxygen Concentration Enhances the Stem-Like Properties of CD133+ Human Glioblastoma Cells In vitro. Molecular Cancer Research. 2009;7:489–497. doi: 10.1158/1541-7786.MCR-08-0360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Martin V, Xu J, Pabbisetty SK, et al. Tie2-mediated multidrug resistance in malignant gliomas is associated with upregulation of ABC transporters. Oncogene. 2009;28:2358–2363. doi: 10.1038/onc.2009.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang W, Xia Y, Ji H, et al. Nuclear PKM2 regulates [bgr]-catenin transactivation upon EGFR activation. Nature. 2011 doi: 10.1038/nature10598. advance online publication. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jiang H, Gomez-Manzano C, Aoki H, et al. Examination of the Therapeutic Potential of Delta-24-RGD in Brain Tumor Stem Cells: Role of Autophagic Cell Death. Journal of the National Cancer Institute. 2007;99:1410–1414. doi: 10.1093/jnci/djm102. [DOI] [PubMed] [Google Scholar]
- 37.Zhang N, Wei P, Gong A, et al. FoxM1 Promotes beta-Catenin Nuclear Localization and Controls Wnt Target-Gene Expression and Glioma Tumorigenesis. Cancer Cell. 2011;20:427–442. doi: 10.1016/j.ccr.2011.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liu M, Dai B, Kang SH, et al. FoxM1B is overexpressed in human glioblastomas and critically regulates the tumorigenicity of glioma cells. Cancer Res. 2006;66:3593–3602. doi: 10.1158/0008-5472.CAN-05-2912. [DOI] [PubMed] [Google Scholar]
- 39.Galli R, Binda E, Orfanelli U, et al. Isolation and characterization of tumorigenic, stem-l ike neural precursors from human glioblastoma. Cancer Res. 2004;64:7011–7021. doi: 10.1158/0008-5472.CAN-04-1364. [DOI] [PubMed] [Google Scholar]
- 40.Lee J, Kotliarova S, Kotliarov Y, et al. Tumor stem cells derived from glioblastomas cultured in bFGF and EGF more closely mirror the phenotype and genotype of primary tumors than do serum-cultured cell lines. Cancer Cell. 2006;9:391–403. doi: 10.1016/j.ccr.2006.03.030. [DOI] [PubMed] [Google Scholar]
- 41.Lal S, Lacroix M, Tofilon P, et al. An implantable guide-screw system for brain tumor studies in small animals. J Neurosurg. 2000;92:326–333. doi: 10.3171/jns.2000.92.2.0326. [DOI] [PubMed] [Google Scholar]
- 42.Graham V, Khudyakov J, Ellis P, et al. SOX2 functions to maintain neural progenitor identity. Neuron. 2003;39:749–765. doi: 10.1016/s0896-6273(03)00497-5. [DOI] [PubMed] [Google Scholar]
- 43.Suh H, Consiglio A, Ray J, et al. In vivo fate analysis reveals the multipotent and self-renewal capacities of Sox2+ neural stem cells in the adult hippocampus. Cell Stem Cell. 2007;1:515–528. doi: 10.1016/j.stem.2007.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Guardavaccaro D, Frescas D, Dorrello NV, et al. Control of chromosome stability by the [bgr]-TrCP-REST-Mad2 axis. Nature. 2008;452:365–369. doi: 10.1038/nature06641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Westbrook TF, Hu G, Ang XL, et al. SCF[bgr]-TRCP controls oncogenic transformation and neural differentiation through REST degradation. Nature. 2008;452:370–374. doi: 10.1038/nature06780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chen ZF, Paquette AJ, Anderson DJ. NRSF/REST is required in vivo for repression of multiple neuronal target genes during embryogenesis. Nat Genet. 1998;20:136–142. doi: 10.1038/2431. [DOI] [PubMed] [Google Scholar]
- 47.Immaneni A, Lawinger P, Zhao Z, et al. REST-VP16 activates multiple neuronal differentiation genes in human NT2 cells. Nucleic Acids Res. 2000;28:3403–3410. doi: 10.1093/nar/28.17.3403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Griffero F, Daga A, Marubbi D, et al. Different Response of Human Glioma Tumor-initiating Cells to Epidermal Growth Factor Receptor Kinase Inhibitors. J. Biol. Chem. 2009;284:7138–7148. doi: 10.1074/jbc.M807111200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Aboody KS, Brown A, Rainov NG, et al. Neural stem cells display extensive tropism for pathology in adult brain: evidence from intracranial gliomas. Proc Natl Acad Sci U S A. 2000;97:12846–12851. doi: 10.1073/pnas.97.23.12846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ng HH, Surani MA. The transcriptional and signalling networks of pluripotency. Nat Cell Biol. 2011;13:490–496. doi: 10.1038/ncb0511-490. [DOI] [PubMed] [Google Scholar]
- 51.Kim J, Chu J, Shen X, et al. An extended transcriptional network for pluripotency of embryonic stem cells. Cell. 2008;132:1049–1061. doi: 10.1016/j.cell.2008.02.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Marson A, Levine SS, Cole MF, et al. Connecting microRNA genes to the core transcriptional regulatory circuitry of embryonic stem cells. Cell. 2008;134:521–533. doi: 10.1016/j.cell.2008.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure 1. REST regulates self-renewal of GSCs over multiple passages. Rest knockdown in both HR-GSC1 (A) and HR-GSC2 (B) with shRest, as compared to control shNT, caused decreased self-renewal over multiple passages (P1-P3).
Supplementary Figure 2. REST gain-of-function experiments. Expression of exogenous REST in LR-GSCs (expression of exogenous GFP served as a control) caused increased expression of Sox2. Actin was used as an internal control.
Supplementary Figure 3. REST regulates survival in mice bearing GSC-mediated brain tumors. Survival of mice bearing tumors generated by 5×103 cells of the stable lines of HR-GSC1 expressing either shRest or shNT (compare Fig. 4C for tumors with 5×104 cells) (n=7 mice per treatment group).
Supplementary Figure 4. REST regulates cell-proliferation of GSCs in culture. BrdU-incorporation assays indicated that knockdown of REST by shRest in both HR-GSC1 and HR-GSC2 lowered cell-proliferation ability when compared to shNT control.
Supplementary Figure 5. REST regulates invasion of GSC in vitro. Invasion chamber assays indicated decreased invasion property of both the two HR-GSC lines upon REST knock down using shRest treatment (shNT-treatment served as a control).