1. Introduction
Depression is frequent in cancerology. Despite its clear impact on patients, it continues to be under-diagnosed and inadequately treated. There are many reasons for this, ranging from the underestimation of depressive symptoms by clinicians, their widespread presence in the context of cancer, the entanglement of depressive symptoms with those associated with the cancer and its treatment, or, indeed, the difficulty of clinicians in exploring emotional symptoms [1,2]. Beyond the fact that depression causes mental suffering that is not taken into consideration, even though it can be extremely intense in nature, this situation has a major impact on both morbidity and mortality through a number of different mechanisms [3,4]:
-
•
Deterioration of quality of life [5].
-
•
Increased sensitivity to pain [6].
-
•
Difficulties observing treatment [7].
-
•
Difficulties communicating with carers, friends and family.
-
•
Significant burden placed on close relatives [8].
-
•
Increased risk of suicide [9].
-
•
Longer periods of hospitalisation [10].
-
•
Reduced expectation of survival [11].
Depression also results in additional medico-economic costs, the extent of which we are only just beginning to understand [12,13].
There is also a risk of over-treatment, with antidepressants being systematically administered for what may only be an intense feeling of sadness, which may nevertheless be appropriate in the context and temporary in duration.
2. What is the prevalence of depression in cancerology?
Although depression is frequently observed in cancerology, the figures reported by the various related studies differ owing to the variability of the clinical forms of depressive disorders – major depressive episode (MDE), dysthymia, adjustment disorders with depressed mood, etc. – and the way in which the diagnosis is performed: clinical interview, questionnaires or self-report questionnaires which may be specific to depression (BDI, CES-D and Zung scale) or more general (HADS, POMS and SCL-90 etc.), variability of the cut-offs, etc. The figures also vary depending on the sample and, in particular, on the patient’s medical status, type of cancer, location, stage, treatment during or after cancer, etc. There may also be a bias associated with the selection of the patients (patients who have agreed to a clinical psychiatric assessment, convenience sample etc.).
On average, studies report a prevalence of MDE of 5–10%, two to three times higher than that in the general population. In a recent meta-analysis of 70 studies (n = 10,071) conducted by Mitchell et al. [14], the prevalence of depressions of all types (ICD, DSM criteria) was 16.3%, with the prevalence of MDE reaching 6%. The corresponding values rose to 24.6–29% and 14.3–16.5% in the analysis of 24 studies (n = 4007) of palliative-phase patients.
The way in which depressive disorders develop during the period of cancer care is still poorly understood because of a lack of longitudinal studies.
A depressive episode may be isolated or may have been preceded by other such episodes. In this latter case, the disorder is a recurrent unipolar depression. The patient’s antecedents may also include one or more phases of hypomanic or manic excitation, or he or she may have presented a hypomanic reaction when under antidepressants. In this case, a bipolar mood disorder is probable. Referral to a psychiatrist will then be necessary for diagnosis and identification of the relevant treatment.
3. What are the predictive factors?
Some of the risk factors for depression are known and must be identified at an early stage [15,16].
Personal risk factors:
-
•
Recent personal history of negative or stressful life events (bereavement, succession of losses).
-
•
Personal psychiatric antecedents (depression, suicide attempt, drug addiction and alcoholism) or familial antecedents (depression, suicide attempt and suicide) [17].
-
•
Personality traits – tendency not to express one’s emotions, tendency to consider life events as uncontrollable and inevitable, low self-esteem and poor emotional support and tendency to pessimism [18,19].
-
•
Unlike the data obtained in the general population, the most recent meta-analysis conducted by Mitchell et al. [14] suggests that gender is not predictive, possibly owing to the weight of the factors associated with the cancer itself.
-
•
The role of age (<50 years) remains controversial [14,20].
Social risk factors:
-
•
Being alone (single, divorced, separated and widowed);
-
•
Social isolation [21].
-
•
Low socio-economic status [21].
-
•
Belonging to an ethnic minority [22].
-
•
Perceived lack of social support.
Risk factors associated with the cancerous disease or its treatment:
-
•
Type of cancer (pancreas, head and neck) [23].
-
•
Advanced stage or metastatic disease.
-
•
Critical phases involving the disclosure of the diagnosis, disclosure of recurrence or aggravation (metastases) and palliative care.
-
•
Limitation of treatments.
-
•
Presence of uncontrolled physical symptoms: firstly pain (RR increased by 2–4 times) [6], as well as nausea, vomiting, fatigue etc. [24,25].
-
•
Functional handicap and loss of autonomy (neurological sequelae, stomata and impairment of general condition etc.).
Treatment-related risk factors:
-
•
Immunotherapy – interferon (IFN), interleukin 2 (IL2)
-
•
Long-term corticotherapy [1].
-
•
Certain antipileptics (such as levetiracetam).
-
•
Certain neurotoxic chemotherapies (vinblastine, procarbazine).
-
•
The thymic toxicity of certain targeted therapies which cross the hematoencephalic barrier has given rise to a number of case reports.
-
•
Despite a number of case reports, cohort studies or observations obtained during double-blind tests have not revealed any depressogenic impact of tamoxifen [26].
Organic causes:
-
•
Neurological anomalies (primary brain tumour or cerebral metastases).
-
•
Endocrine disorders (hyper- or hypothyroidism, adrenal insufficiency).
-
•
Metabolic disorders (vitamin B12, folate deficiency).
4. How should depression be diagnosed?
A depressive syndrome is the association of depressive symptoms that persist over time. The diagnosis of depression is already a complex affair, even outside of the context of physical pathology, since it consists in the lasting association of a variety of subjective symptoms (at least five, see below) [27], none of which is specific [28]. In most cases, these symptoms are difficult to reveal (for example, social withdrawal that could be explained by cancer treatments), and the most highly visible symptoms (such as tearfulness) are the least specific.
In the case of cancer patients, this complexity is further aggravated by the potentially adapted nature of the depressive emotion in the context of the multiple bereavements that accompany cancer. One important area will therefore be to differentiate between ‘normal’ sadness, on the one hand, and adjustment disorders or depressive episode on the other.
For depression to be diagnosed, it is therefore necessary for least five symptoms to be present nearly all day for at least 15 consecutive days. In practical terms, the diagnostic approach should consist of the following steps:
-
•
Use of two simple questions to attempt to identify one of the major symptoms, namely mental suffering and lack of interest (absence of desire for anything, affective indifference which can lead to social withdrawal and a loss of commitment to contact with friends and family) [29].
-
•
If one of these two symptoms is present, identification of the time criterion – almost every day, almost all day, for a minimum of 15 days.
-
•
Then attempt to identify associated symptoms (at least three or four from the list below).
-
o
Feelings of worthlessness or inappropriate guilt (in particular a painful vision of the past, for example: ‘for my family, I’m now nothing more than a burden’, ‘I’ve been a failure with my children’).
-
o
Recurrent thoughts of death or suicide.
-
o
Significant weight loss or gain (>5% of body weight), or increase or decrease in appetite.
-
o
Insomnia or hypersomnia.
-
o
Psychomotor agitation or retardation.
-
o
Fatigue or loss of energy.
-
o
Diminished concentration or indecisiveness.
A change in character with the appearance of irritability or aggressiveness that is inconsistent with earlier behaviour is also possible.
If five symptoms from the above list, including mental pain and/or absence of pleasure, are present then the patient is very probably depressed, and antidepressant treatment is recommended.
If not, the patient is exhibiting either a temporary depressive reaction (if the criterion of persistence over time is not met) or an adjustment disorder with depressed mood (if insufficient symptoms are present: for example, sadness on its own without feelings of worthlessness, sleep disorders or anhedonia). Antidepressants are not indicated a priori.
Another difficulty affecting the diagnosis of depression is associated with the distinction between its somatic manifestations (fatigue, anorexia and/or loss of weight, cognitive or sleep disorders, loss of libido) and the physical symptoms associated with the cancerous disease or its treatment [30]. A number of ways of overcoming this difficulty have been described:
-
•
An inclusive approach in which all the symptoms are taken into account, irrespective of whether or not they are attributable to the cancer (which clearly leads to a risk of over-diagnosis).
-
•
An exclusive approach in which the somatic symptoms are systematically excluded from the diagnostic criteria.
-
•
A substitution method in which these very same somatic symptoms are replaced by affective substitute symptoms [31].
When the exclusive and substitution approaches are used, the risk that the diagnosis may be underestimated is high, in particular in the case of patients who conceal or repress their emotions.
In practice, all manifestations that are not clearly linked to another cause (physical or iatrogenic causes) should be considered as contributing to the diagnosis of the depressive syndrome. The chronology of the disorders is particularly useful in this context.
It is therefore necessary to pay special attention to the cognitive and affective symptoms that are not directly linked to the somatic state, together with all their possible nuances:
-
•
Self-devaluation and a painful vision of the past: feelings of uselessness, worthlessness and guilt (invasive, generalised).
-
•
Loss of interest and pleasure: affective anaesthesia, indifference.
-
•
Feeling of worthlessness coupled with feelings of helplessness and hopelessness.
-
•
Desire for death and thoughts of suicide must be systematically explored; far from inducing suicidal behaviour, putting such ideas into words is an opportunity for the patient to become aware of their pathological nature and their link, not to a rational perception of reality, but to his or her own depressive suffering.
-
•
Pathological pessimism.
A final difficulty lies in the fact that patients rarely spontaneously express those depressive symptoms that are primarily purely affective in nature (emotional withdrawal, for example), and that depressive symptoms themselves (psychomotor retardation, social withdrawal and shame) and lack of knowledge or minimisation of depressive symptoms lower this expression. Simple, open questions should therefore be favoured during the interview (‘How’s your mood? And everyday life? Are there some good times? etc.’). It is important to avoid moralizing phraseology (‘face up to’, ‘keep going’) and closed questions (‘Are you in a good mood? Are you coping?’) which risk blocking the expression of the experienced feelings and increasing the sensation of guilt due the patient’s perception of his or her depressed state. The availability of the carer, whether verbal or non-verbal, is vital for facilitating emotional expression.
The diagnosis must take into account the time of the assessment. The assessment of depressive symptoms is difficult during the days immediately following the announcement of bad news or in the case of an evolutive, uncontrolled somatic symptomatology, in particular if this is associated with the experience of pain. In such cases, the patient should be reassessed at a distance or after the symptoms have been treated.
Finally, the idea of a break with earlier behaviour is also important. The clinician must remain attentive to changes in the patient’s functioning, whether this is revealed by the patient or his or her friends and family. Helping patients become aware of this change may also make it easier for them to grasp the pathological nature of their condition.
5. How to screen for depression in cancerology?
Simple, validated tools can be used, and the aim for screening purposes is to select short tools taking no more than 5 min to complete. A recent review of the various potential tools for the screening of distress undertaken by Mitchell [32,33] identified 35 short tools for the screening of depression, consisting of 1–14 items. Although some of these specifically assess depression and have been validated for use in cancerology, most of them were not specifically designed for administration to patients suffering from a somatic pathology. This fact can be problematic when the deterioration of the patient’s general condition aggravates scores on items that evaluate the respondent’s somatic state; this criticism has been made with regard to the BDI (Beck Depression Inventory), for example [34]. Others, such as HADS (Hospital Anxiety and Depression Scale), assess emotional distress at a more general level but have the advantage of excluding somatic items. The following tools have been validated for use with cancer patients: single verbal item, PHQ1, PHQ2 (and PHQ2 + help question), two verbal items BCFD (Bried Case Find for Depression), Edinburgh Postnatal Depression Scale (EPDS, and Brief EPDS), Hornheide Screening Instrument (and Hornheide Short Form), General Health Questionnaire 9, BDI Short Form, HADS (and HADS depression subscale).
The sensitivity and specificity of the different scales vary as a function of the cut-offs used and of the patient clinical condition. None of the tools used today can be claimed to be irrefutably preferable to any other. It should nevertheless be noted that in most studies the sensitivity and specificity of ultra-short tools (consisting of just one or two questions, ‘low mood’ and ‘loss of interest or pleasure’) have been found to be at least as good as those of longer tools. In the meta-analysis of screening and case-finding tools for depression in cancer conducted by the Depression in Cancer Care consensus group [29], 56 diagnostic validity studies (n = 10,009) were reviewed. For case-finding, one stem question, two stem questions and the BDI-II all had level 2 evidence. For screening, two stem questions had level 1b evidence. As they are highly acceptable, the stem questions are thus recommended (grade B recommendation). For every 100 people screened, the two questions would accurately detect 18 cases in advanced cancer settings (one missed and seven falsely identified), and 17 in non-palliative settings (two missed, and 11 falsely identified as cases).
By contrast, clinical appraisals that are not guided by at least one or two questions are not recommended, in particular if the clinicians are not trained [35].
In practice, it is important to remember that the diagnosis of depression is too complex for any screening tool, used in isolation, to be able to provide absolute certainty. Nevertheless, their use is recommended and can be incorporated within a more global assessment of the patient’s symptoms (as in the case of the Edmonton Symptom Assessment System, ESAS), supportive care needs or expectations [32]. The main value of these tools – and in particular of those which permit an assessment based on two questions – is that they make it possible to identify with a high degree of certainty those patients who are not depressed, and consequently focus the efforts of the oncologist, carer or psychologist/psychiatrist on obtaining a more in-depth assessment of those individuals who have screened positively.
However, systematic screening for depression has not as yet proved its effectiveness in significantly improving patients’ psychological outcomes [36]. Far from casting doubt on its usefulness, this fact emphasises the vital need to include this type of screening in an overall treatment process that extends through to the proposal and acceptance of appropriate pharmacological and/or psychotherapeutic care.
6. Depression and the risk of suicide
Despite the variability that can be found in the literature, owing to problems of both methodology and definition, it is possible to estimate the risk of suicide in the oncological context at 1.95–2.8 times higher than in the general population [9,37]. The wish to die is present in 17% of patients in an advanced phase of cancer.
Certain suicide risk factors are known [20,37]
-
•
Poorly controlled symptoms (pain, fatigue, etc.).
-
•
Masculine gender (relative risk of 1.7 in men and 1.4 in women compared with the general population [38]).
-
•
Disclosure phase (first year following the diagnosis) [37].
-
•
Site of the tumour (head and neck, lung, gastrointestinal, brain tumours [9]).
-
•
Existence of a psychopathological disorder (depression, hopelessness, delirium).
-
•
Familial antecedents of suicide and/or psychiatric illness, previous personal suicide attempts.
-
•
Pathological impulsivity (personality disorder).
-
•
Substance abuse (alcohol etc.).
-
•
Recent loss (bereavement, for example).
-
•
Situation of hopelessness, loss of control or autonomy [1].
It is interesting to note that most of these factors are found in cases where patients ask to accelerate their death (hastening death requests are associated at a significant level with depressive states, hopelessness, low level of social support, very poor physical condition and a lower level of recourse to spirituality) [1]. Whenever patients make this type of request, one should systematically and carefully search for an emotional disorder that may indicate pathological suicidal ideation.
7. Treatment plan and overall patient care
The treatment of depression has to start at an early stage [39]. It can take a long time to make patients aware of the psychological nature of their difficulties, encourage them to request a psychological consultation or to accept the prescription of psychotropic or, in particular, antidepressant medicaments. The more intense the psychological distress the longer this delay may be. It is the systematic attention paid by clinicians to their patients’ psychological states that permits early diagnoses and the effective elaboration of an appropriate treatment plan.
The treatment of depression must form part of an overall care context. First of all, it is necessary to take account of the patient’s somatic condition, the associated symptoms and any comorbidities that may be present (pain, fatigue and sexual problems). It is not possible, for example, to treat depression in patients with uncontrolled pain [40].
To start from the patients’ expectations – for example, by identifying with them the symptom that causes them the greatest distress, or discussing what they can expect from treatment – may help them to accept depression treatment. Certain patients, for example, who can no longer themselves perceive the loss of the ability to experience pleasure or are unaware of their psychomotor retardation, may primarily complain of sleep-related problems. An initial prescription of drugs that are effective in combating these sleep disorders will encourage subsequent adherence to therapy. The patient’s habitual mode of psychological functioning may also help the clinician guide him or her towards appropriate psychotherapeutic support. It is also necessary to try to identify the representations associated with depression, antidepressant treatments or psychotherapies, together with any possible prior intolerance of antidepressant treatment.
Patients must be given information about their socio-professional circumstances and psychosocial resources (family, work and environment). What psychosocial resources can they count on in their family or professional environments? And, conversely, what strains or obligations are imposed on them?
The next step is to check the level of information that patients possess regarding the cancer for which they are being treated, their understanding of the treatment plan and their adherence to it. The quality of the cooperation between the psychologist or psychiatrist, on the one hand, and the oncologist, on the other, may if necessary make it possible to revisit any information that has been inadequately understood.
Finally, therapeutic care for mental distress, in particular when drug-based treatments are used, must always be initiated by a stage during which the psychological nature of the disorders is explained and clear information about the prescribed psychotropic drug – its purpose and any possible side effects (risk of dependence associated with benzodiazepines or hypnotic substances, for example) – is provided. Nevertheless, given the specific symptoms that characterise depressive states, it can sometimes be difficult to secure the commitment of depressed patients to a course of treatment, in particular when they find it difficult to acknowledge the pathological nature of their condition or when the loss of hope or confidence in the future is significant. Memory and attention disorders may aggravate a patient’s reluctance to agree to psychotropic treatment and impair compliance with it, especially given that the anxiety that is frequently felt at the start of treatment may be associated with an increase in the real or feared side effects.
The discussion of the diagnosis is therefore the first step in the administration of treatment. The information provided to depressed patients enables them to better understand the nature of their distress and retain their independence. The empathy shown by carers in response to their psychological distress also constitutes an initial vital and beneficial experience for depressed patients, who often feel isolated due to the shame and guilt they experience (and withdraw themselves both from their friends and family and from their oncological team) [41].
To treat major depressive episodes it is necessary to prescribe both an antidepressant and, in certain cases, a course of psychotherapeutic care [1,15] or, at the very least, to ensure the availability of psychosocial support to facilitate adherence to treatment [42].
8. When and how to prescribe an antidepressant?
It is appropriate to prescribe an antidepressant for all major depressive episodes. This prescription does not always have to be accompanied by treatment by a psychiatrist except in the case of depression affecting patients with bipolar mood disorders who should be referred to a psychologist for diagnosis and treatment. In a recent Cochrane review [43] it was established that antidepressants outperform placebo level among patients with somatic diseases (OR 2.33, IC 1.8–3, P < 0.00001). This finding redresses a scientific shortcoming in the field of cancerology: in 2008, only 20 studies of the effectiveness of antidepressants had been conducted in this field and none of them had reached a sufficient level of proof [2]. However, we still need to gain a better understanding of the rationale underlying the pathogenesis of depression in the cancer field and, in particular, its links with the immune and inflammatory mechanisms.
A preventive treatment is not recommended except in some very specific cases, as in e.g. depression induced by high-dose interferon alpha [44].
Generally speaking, SRIs (serotonin reuptake inhibitors) (Table 1) tend to be the first drugs to be prescribed in the field of cancerology [45]. There is no reason to consider the effectiveness of any antidepressant to be formally superior to that of any other [46–48]. When administered at antidepressant dosage (which is considerably higher than the antalgic dosage), tricyclics (Table 1) are difficult to handle and should be reserved for more complex cases. Escitalopram might be the medicament with the best effectiveness-to-tolerance ratio among patients who have no contraindication for this drug [45]. Furthermore, the choice of an antidepressant is specific to the patient and is determined by individual factors: the side effects of the drug, the patient’s tolerance to it (which includes potential side effects in combination with other drugs being co-administered or that might be used in the future), reactions to earlier treatments, and the patient’s preferences [49]. In particular, it is especially important to make sure that no potential iatrogenics exacerbate the somatic or non-somatic difficulties that are already present (for example, sexual difficulties or asthenia).
Table 1.
Families of antidepressants.
| Family |
|---|
| SNRI : Venlafaxine, minalcipran and duloxetine |
| Nassa : Mianserine and mirtazapine |
| Melatonihergic : Agomelatine |
| Others : Tianeptine |
In the majority of cases, monotherapy is the rule; to combine two antidepressants is a priori pointless and potentially dangerous. Although exceptions exist, such prescriptions should only be made by specialists. Accompanying prescriptions – for example of anxiolytic or hypnotic drugs – should be restricted. These may be prescribed at the start of treatment but clinicians should attempt to reduce or eliminate them as treatment progresses.
The fact that a period of 2–3 weeks is required before any effect can be observed must be explained to the patient who must also be alerted to the possibility of undesirable side effects. It is important to remain vigilant with regard to the aggravated vulnerability of cancer patients (specific metabolic characteristics, associated treatments) and remain attentive to the identification of secondary effects which sometimes may not be easy to detect in a somatic context. Tolerance to antidepressants varies depending on the drug in question, and tolerance to tricyclic antidepressants is often poor since these drugs frequently produce side effects (orthostatic hypertension, weight gain, sedation, irregular heartbeat, confusion, epilepsy and potentially fatal in the event of an overdose), which is why they are not administered as the first line of treatment. The toxicity of serotonin reuptake inhibitors is less serious and is often restricted to self-limiting disorders (headaches, gastrointestinal disorders). All drugs may also lead to an increase in anxiety. Venlafaxine and duloxetine have also been reported to lead to increased blood pressure. Mirtazapine and mianserine often lead to weight gain and drowsiness, two effects that can lead to resistance, particularly on the part of female patients at the end of the period of treatment who are already exposed to weight gain owing to other mechanisms.
There are no restrictions relating to prescriptions for patients with suicidal thoughts. According to the review conducted by Möller [50], the risk that such patients will actually take their own lives under the influence of antidepressants appears to be small and decreases from the age of 30 years onwards. If possible, patients who express suicidal thoughts should rapidly be sent for psychological or psychiatric assessment. However, if these thoughts are associated with depression, this referral should not cause any delay to treatment. A drug with only a low stimulant effect (a pure serotoninergic drug such as citalopram or paroxetine, but no noradrenergic products) should be preferred and should only be prescribed for a short initial period (1 week), after which the patient should be reassessed.
If there is a risk of suicide, the introduction of the antidepressant treatment is an emergency, but not such as the organisation of the care that is to be received. While awaiting the results of psychiatric assessment, it is necessary to organise enhanced monitoring of the patient and inform his or her family of the suicidal risk, insofar as professional ethics permit this. Ensuring that the patient is accompanied as much as possible is an important factor in helping to prevent the occurrence of suicide.
In all cases, it may be of value to try to limit the patient’s impulsivity by prescribing a sedative and anxiolytic neuroleptic such as cyamemazine (e.g. 25–50 mg/day).
Antidepressants present no carcinogenic risk, at least in the case of SSRIs and tricyclics [51]. Nevertheless, vigilance is recommended in the case of the prolonged administration (more than 10 years) of serotonin–norepinephrine reuptake inhibitors (SNRIs), since for these recent products it has not yet been possible to study the associated long-term risks.
The risk of serotonin syndrome is also frequently cited. This takes the form of excessive serotonergic stimulation which causes motor excitation, hyperreflexia, trembling, myoclonia and dysfunctioning of the autonomous central nervous system and, in extreme cases, epilepsy, comas or death. These effects are not due to any idiopathic effect but are caused by the interaction of drugs sharing similar mechanisms or by overdoses [52]. It is extremely difficult to state their frequency since minor forms are probably not recognised and still less often reported [52].
Although a treatment can start to have an effect as of the first few days following prescription, it cannot be viewed as ineffective until 3–4 weeks of administration at an effective dosage level (6 weeks in the case of elderly subjects). The effective dosage can sometimes vary considerably (by a factor of 1–3 or even 1–4), which means that in the case of lack of efficacy it is necessary, providing that the patient tolerates the drug without difficulty, to progressively increase the dosage before moving on to a new drug. If a drug appears to be ineffective, it is also necessary to verify correct adherence (and therefore also tolerance to the correct dosage) on the part of the patient. The minimum period of treatment is 6 months in order to avoid any premature relapse. Treatment must not be interrupted suddenly given the risks associated with withdrawal which, with paroxetine, may appear in less than 48 hours. If oral administration is impossible, it is generally possible to replace the prescribed drug with injectable citalopram, the only SNRI available in this form. If administration has to be performed via a tube, then drugs available in soluble form should be preferred (citalopram, mirtazapine, fluoxetine and paroxetine).
Table 2 lists the main limitations to prescriptions and contraindications. This should be updated by the clinician in the light of the data available at the time of prescription.
Table 2.
Main limitations to antidepressant prescriptions and contraindications.
| Moderate renal or hepatic insufficiency | SSRI, generally not contraindicated; use the minimum effective dose and start off with half this dose |
| Severe hepatic insufficiency | Use tianeptine or minalcipran |
| Conduction disorders | Avoid tricyclics |
| Extension of QT, or co-prescription with drugs associated with this risk | Avoid SNRIs |
| Cardiac disorders in general | Prefer mianserine |
| Hyponatremia | Risk of aggravation in response to SNRIs: monitor |
| Convulsions or risk of convulsions (cerebral metastases) without anticonvulsive treatment | Only tianeptine does not reduce the threshold of epileptogenesis |
| Arterial hypertension | Avoid venlafaxine |
| Prostatic adenoma | Tricyclics are contraindicated. Caution is necessary with minalcipran and mirtazapine, SNRIs authorised but possibility of urination difficulties |
| Administration of tamoxifen | Paroxetine and fluoxetine are contraindicated. |
SSRI : selective serotonin reuptake inhibitors.
There are also risks of interactions with other drugs or of overdoses. Serotonin reuptake inhibitors are Cyt P450 substrates and may accumulate in individuals with slow metabolism or if Cyt P450 is inhibited. The SNRIs are also CytP450 inhibitors whose effect varies from drug to drug (in contact with different isoenzymes). There is therefore a risk of interaction. This is due as much to the accumulation of the substrate as to its ineffectiveness when in the form of an inactive prodrug. The important thing is to be aware of the possibility of interactions and to be vigilant [53]. Several internet sites provide regularly updated information that can be consulted if required.
Although the prescription of psychostimulant drugs (primarily methylphenidate or modafinil) may be considered in the case of depressed patients (but in most countries remains outside the scope of marketing authorisation), this should be done only following a specialist assessment; in any case, these products are nowadays subject to an initial psychiatric prescription. Their stimulant effect on vigilance and attention may be of particular value in cases of asthenia associated with the prescription of opiates to overcome pain. In some studies, these drugs have exhibited a rapid antidepressant effect which can be of value, in particular when the end of life is near at hand and the period of action required by antidepressants is therefore unacceptable or in order to obtain an immediate effect during this end-of-life period. However, high-quality studies, in particular involving adequate sample sizes, are still required in this area. Such psychostimulants should be prescribed only to patients for whom the experience of fatigue and the functional constraints that this imposes are perceived as an additional source of stress or an intolerable limitation to their quality of life. Their prescription should never be influenced by a desire for increased performance, in particular when made by friends and family distressed by the patient’s waning enthusiasm and drive and who would be reassured to see him or her recover the roles which he is no longer able to assume.
9. Is it necessary to prescribe a benzodiazepine?
These drugs should not be prescribed systematically. In practical terms, it is justifiable to prescribe a benzodiazepine or anxiolytic:
-
•
In the presence of a high level of anxiety, in particular in combination with somatic symptoms which might result in the poor tolerance of antidepressants (attribution to the drug of functional symptoms caused or exacerbated by anxiety).
-
•
If the patient is already being treated with a benzodiazepine – this may subsequently be interrupted; however, stopping it straight away may aggravate the clinical picture because of drug withdrawal (which, once again, risks being incorrectly attributed to poor tolerance of the antidepressant).
In depressed, impulsive patients it is often preferable to treat anxiety with low doses of a sedative neuroleptic (e.g. cyamemazine) rather than with a benzodiazepine.
10. What are the roles of the different actors in diagnosing and caring for depression in the field of cancerology?
Various organisational schemas for care administration in response to depressive disorders have been evaluated as a function of the actors responsible for screening, diagnosing or prescribing treatment [54].
It is not essential for the initial prescription to be made by a psychiatrist, in particular given the facts that there are not enough psychiatrists to provide care to all depressed patients, and that some of these patients are reluctant to consult a mental health specialist. Systematic screening followed by the diagnosis and the prescription of treatment by the oncological team itself appears to be the most efficient way of providing effective treatment [42] if it includes the arrangement of – at the very least – support services (coordinating nurse, possibly communicating by telephone etc.) or other psychotherapeutic services. A psychiatrist may assist in the task of prescribing in the most complex cases. This approach has been validated in North America and seems to be suitable for use in the European context. It allows issue of an initial prescription without waiting for the psychiatrist, and emphasises the value, in addition to the prescribed antidepressant [1], of providing to the patient individual support by a psychologist or a non-psychologist.
It is advisable to obtain a psychiatric opinion from the outset in the case of patients with schizophrenia, a unipolar or bipolar mood disorder (manic-depressive disorder), a severe personality disorder or suicidal thoughts.
A psychiatric opinion should also be sought as a second line of treatment in the event of resistance to treatment even after an increase in dose, or in the case of any doubt concerning the diagnosis.
Even in the absence of a psychiatric opinion, the question of hospitalising the patient should be discussed if there is a risk of suicide or, in the case of refusal of treatment, agitation, extreme anxiety or delirium. It might also be considered in the case of extremely isolated patients.
All authors have emphasised the value of offering psychological support to patients, either in the form of psychosocial support or in the form of psychotherapy. In major depressive disorders, combining the administration of an antidepressant with the provision of psychotherapeutic care improves the effectiveness of treatment. In a meta-analysis of depression in a non-cancerological environment (n = 1843), patients treated with a combination of psychotherapy (of all types) and antidepressants exhibited a considerably higher level of improvement than patients treated with drugs only (OR 1.86, 95%CI 1.38–2.52). Furthermore, the beneficial effect of this combination increased in the case of treatments lasting for 3 months or longer (OR 2.21, 95%CI, 1.22–4.03) [55]. In this context, initial prescription by a psychiatrist would be ideal in order to improve the diagnosis and ensure, from the outset, that the prescription forms part of a coordinated therapeutic approach, possibly accompanied by a psychotherapeutic element [56]. However, this solution is only rarely available.
Various types of psychotherapy have been proposed, sometimes in combination:
-
•
Psychoeducation.
-
•
Relaxation training.
-
•
Problem-solving therapies.
-
•
Cognitive behavioural therapies.
-
•
Interpersonal therapies.
-
•
Supportive expressive therapy.
The degree to which these therapies have been validated is sometimes limited and, in both psycho-oncology and psychiatry, the question of the evaluation of psychotherapies and psychosocial interventions continues to be a complex subject. The differing natures of the cancer patients, the approaches and therapeutic goals, the training and experience of the therapists, and the multiplicity of assessment scales, as well as the different periods studied during the health care circuit, all further complicate such analyses and relativise the results [57–59]. In this field, the very idea of randomised trials can be called into question [60] since they do not make it possible to appraise the subtle effects and individual benefits brought about by psychotherapy.
Beyond the above reservations, it is accepted that psychotherapies have a beneficial impact on anxiety, depression, psychological distress and quality of life [61,62]. Although it also seems to be the most depressed patients who gain the greatest benefits [63,64], this is at the same time the population for which the lowest level of evidence is available in the field of cancerology (only three well-conducted studies involving CBT and, in the case of metastatic patients, supportive–expressive therapies) [65].
It is important to identify the psychotherapeutic technique that is best suited for each patient as a function of their personality, their expressive capacities, their psychosocial situation, their concern for others and the time at which oncological care is administered. Thus, patients at the start of treatment might more readily accept a cognitive or psycho-educational approach, whereas those suffering from a recurrence of the disease or confronted with a serious development in its course might request a more existential form of psychotherapeutic support. Psychotherapies inspired by psychoanalytical and/or psychocorporal techniques could ideally be offered to all patients who request them provided that their psychotherapists are able, during preliminary interviews, to assess their ability to commit themselves to such activities and benefit from them.
Fig. 1 summarises the main stages during diagnosis and the prescription of an antidepressant.
Fig. 1.
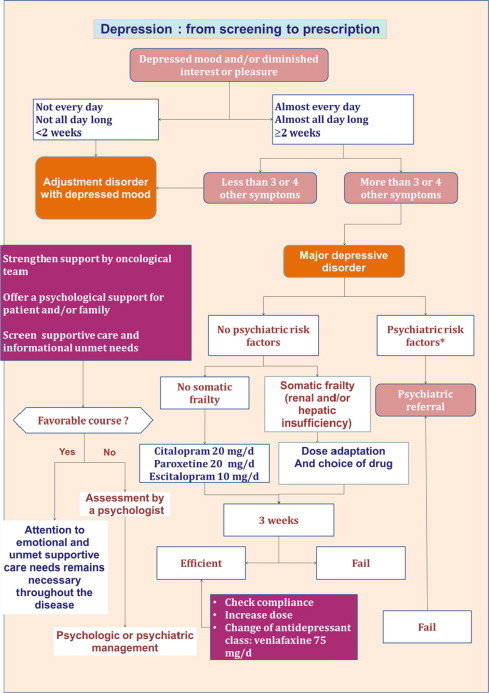
Summary of the main stages during diagnosis and the prescription of an antidepressant.
11. Conclusion
Depression remains highly prevalent in cancer patients, and appears to have a great impact on their quality of life as well as on certain cancer outcomes, even if probably by the means of its impact on compliance, physical activity, social support etc. This broad impact justifies carrying out systematic screening that can be performed by standardised tools but also by one or two simple questions. To be useful, this screening must be followed by an adequate clinical diagnosis that relies on a precise identification of emotional and cognitive symptoms of depression. Considering the prevalence of depressed patients, the oncological teams are expected to do the depression diagnosis and make the first antidepressants prescription by themselves.
To be efficient, depression care must be part of a comprehensive care plan, including treatment of somatic symptoms, and an adequate response to information needs and unmet needs. When possible, and if accepted by the patient, the help of a psychologist is highly appreciated. In all cases, patients should benefit from an accompaniment that can be ensured by a nurse or a social worker.
More research is still needed on factors that may cause varying rates of depression and that predict which patients are mostly at risk. An adequate collaborative care process ranging from depression screening to effective treatment has to be implemented and assessed. Longitudinal studies are still needed to understand the evolution of depressive symptoms. Randomised controlled trials should also help to differentiate between the effectiveness of types of psychosocial interventions. Newer antidepressants and stimulants also should be studied in this population.
Conflict of interest
None declared.
References
- 1.Chochinov H. Depression in cancer patients. Lancet Oncol. 2001;2:499–505. doi: 10.1016/S1470-2045(01)00456-9. [DOI] [PubMed] [Google Scholar]
- 2.Fann J.R., Thomas-Rich A.M. Major depression after breast cancer: a review of epidemiology and treatment. Gen Hosp Psychiatry. 2008;30:112–126. doi: 10.1016/j.genhosppsych.2007.10.008. [DOI] [PubMed] [Google Scholar]
- 3.Hopko D.R., Bell J.L. The phenomenology and screening of clinical depression in cancer patients. J Psychosoc Oncol. 2008;26:31–51. doi: 10.1300/j077v26n01_03. [DOI] [PubMed] [Google Scholar]
- 4.Spiegel D., Giese-Davis J. Depression and cancer: mechanisms and disease progression. Biol Psychiatry. 2003;54:269–282. doi: 10.1016/s0006-3223(03)00566-3. [DOI] [PubMed] [Google Scholar]
- 5.Skarstein J., Aass N., Fossa S.D. Anxiety and depression in cancer patients: relation between the hospital anxiety and depression scale and the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire. J Psychosom Res. 2000;49:27–34. doi: 10.1016/s0022-3999(00)00080-5. [DOI] [PubMed] [Google Scholar]
- 6.Gagliese L., Gauthier L.R. Cancer pain and depression: a systematic review of age-related patterns. Pain Res Manage. 2007;12:205–211. doi: 10.1155/2007/150126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Colleoni M., Mandala M. Depression and degree of acceptance of adjuvant cytotoxic drugs. Lancet. 2000;356:1326–1327. doi: 10.1016/S0140-6736(00)02821-X. [DOI] [PubMed] [Google Scholar]
- 8.Pitceathly C., Maguire P. The psychological impact of cancer on patients’ partners and other key relatives: a review. Eur J Cancer. 2003;39(1517–24):1532. doi: 10.1016/s0959-8049(03)00309-5. [DOI] [PubMed] [Google Scholar]
- 9.Misono S., Weiss N.S., Fann J.R., Redman M., Yueh B. Incidence of suicide in persons with cancer. J Clin Oncol. 2008;26:4705–4707. doi: 10.1200/JCO.2007.13.8941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Prieto J.M., Blanch J., Atala J. Psychiatric morbidity and impact on hospital length of stay among hematologic cancer patients receiving stem-cell transplantation. J Clin Oncol. 2002;20:1907–1917. doi: 10.1200/JCO.2002.07.101. [DOI] [PubMed] [Google Scholar]
- 11.Satin J.R., Linden W., Phillips M.J. Depression as a predictor of disease progression and mortality in cancer patients.A meta-analysis. Cancer. 2009;115:5349–5361. doi: 10.1002/cncr.24561. [DOI] [PubMed] [Google Scholar]
- 12.Chiles J., Lambert M., Hatch A. The impact of psychological interventions on medical cost offset: a meta-analytic review. Clin Psych Sci Pract. 1999;6:204–220. [Google Scholar]
- 13.Carlson L.E., Bultz B.D. Efficacy and medical cost offset of psychosocial interventions in cancer care: making the case for economic analyses. Psycho-Oncol. 2004;13:837–849. doi: 10.1002/pon.832. [DOI] [PubMed] [Google Scholar]
- 14.Mitchell A.J. Prevalence of depression, anxiety, and adjustment disorder in oncological, haematological, and palliative-care settings: a meta-analysis of 94 interview-based studies. Lancet Oncol. 2011;12:160–174. doi: 10.1016/S1470-2045(11)70002-X. [DOI] [PubMed] [Google Scholar]
- 15.Pirl W.F. Evidence report on the occurrence, assessment ant treatment of depression in cancer patients. JNCI. 2004;32:32–39. doi: 10.1093/jncimonographs/lgh026. [DOI] [PubMed] [Google Scholar]
- 16.Raison C.L., Miller A.H. Depression in cancer: new developments regarding diagnosis and treatment. Biol Psychiatry. 2003;54:283–294. doi: 10.1016/s0006-3223(03)00413-x. [DOI] [PubMed] [Google Scholar]
- 17.Costanzo E.S., Lutgendorf S.K. Adjusting to life after treatment: distress and quality of life following treatment for breast cancer. Br J Cancer. 2007;97:1625–1631. doi: 10.1038/sj.bjc.6604091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Montazeri A., Jarvandi S. Anxiety and depression in breast cancer patients before and after participation in a cancer support group. Patient Educ Couns. 2001;45:195–198. doi: 10.1016/s0738-3991(01)00121-5. [DOI] [PubMed] [Google Scholar]
- 19.Wong-Kim E.C., Bloom J.R. Depression experienced by young women newly diagnosed with breast cancer. Psychooncology. 2005;14:564–573. doi: 10.1002/pon.873. [DOI] [PubMed] [Google Scholar]
- 20.Rasic D.T., Belik S.L. Cancer, mental disorders, suicidal ideation and attempts in a large community sample. Psychooncology. 2008;17:660–667. doi: 10.1002/pon.1292. [DOI] [PubMed] [Google Scholar]
- 21.Dalton S.O., Schüz J. Social inequality in incidence of and survival from cancer in a population-based study in Denmark, 1994–2003: summary of findings. Eur J Cancer. 2008;44:2074–2085. doi: 10.1016/j.ejca.2008.06.018. [DOI] [PubMed] [Google Scholar]
- 22.Ell K., Sanchez K. Depression, correlates of depression, and receipt of depression care among low-income women with breast or gynecologic cancer. J Clin Oncol. 2005;23:3052–3060. doi: 10.1200/JCO.2005.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brintzenhofe-Szoc K.M., Levin T.T., Li Y., Kissane D.W., Zabora J.R. Mixed anxiety/depression symptoms in a large cancer cohort: prevalence by cancer type. Psychosomatics. 2009;50:383–391. doi: 10.1176/appi.psy.50.4.383. [DOI] [PubMed] [Google Scholar]
- 24.Ciaramella A., Poly P. Assessment of depression among cancer patients: the role of pain, cancer type and treatment. Psychooncology. 2001;10:156–165. doi: 10.1002/pon.505. [DOI] [PubMed] [Google Scholar]
- 25.So W.K., Marsh G., Ling W.M. The symptom cluster of fatigue, pain, anxiety, and depression and the effect on the quality of life of women receiving treatment for breast cancer: a multicenter study. Oncol Nurs Forum. 2009;36:205–214. doi: 10.1188/09.ONF.E205-E214. [DOI] [PubMed] [Google Scholar]
- 26.Lee K.C., Ray G.T. Tamoxifen treatment and new-onset depression in breast cancer patients. Psychosomatics. 2007;48:205–210. doi: 10.1176/appi.psy.48.3.205. [DOI] [PubMed] [Google Scholar]
- 27.American Psychiatric Association . Fourth ed. Revised; American Psychiatric Association, Washington, DC: 1994. Diagnostic and statistical manual of mental disorders (DSM IV-R) [Google Scholar]
- 28.Reuter K., Raugust S. Depressive symptom patterns and their consequences for diagnosis of affective disorders in cancer patients. Support Care Cancer. 2004;12:864–870. doi: 10.1007/s00520-004-0694-2. [DOI] [PubMed] [Google Scholar]
- 29.Mitchell A.J. Meta-analysis of screening and case finding tools for depression in cancer: evidence based recommendations for clinical practice on behalf of the depression in cancer care consensus group. J Affect Disord. 2012;140:149–160. doi: 10.1016/j.jad.2011.12.043. [DOI] [PubMed] [Google Scholar]
- 30.Mitchell A.J., Lord K., Symons P. Which symptoms are indicative of DSMIV depression in cancer settings? An analysis of the diagnostic significance of somatic and non-somatic symptoms. J Affect Disord. 2012;140:137–148. doi: 10.1016/j.jad.2011.11.009. [DOI] [PubMed] [Google Scholar]
- 31.Endicott J. Measurement of depression in patients with cancer. Cancer 1984; 53: (Suppl):2243–9. [DOI] [PubMed]
- 32.Mitchell A.J. Short screening tools for cancer-related distress: a review and diagnostic validity meta-analysis. JNCCN. 2010;8:487–494. doi: 10.6004/jnccn.2010.0035. [DOI] [PubMed] [Google Scholar]
- 33.Mitchell A.J. Pooled results from 38 analyses of the accuracy of distress thermometer and other ultra-short methods of detecting cancer-related mood disorders. J Clin Oncol. 2007;25:4670–4681. doi: 10.1200/JCO.2006.10.0438. [DOI] [PubMed] [Google Scholar]
- 34.Wedding U., Koch A. Requestioning depression in patients with cancer: contribution of somatic and affective symptoms to Beck’s depression inventory. Ann Oncol. 2007;18:1875–1881. doi: 10.1093/annonc/mdm353. [DOI] [PubMed] [Google Scholar]
- 35.Mitchell A.J., Hussain N., Grainger L., Symonds P. Identification of patient-reported distress by clinical nurse specialists in routine oncology practice: a multicentre UK study. Psychooncology. 2011;20:1076–1083. doi: 10.1002/pon.1815. [DOI] [PubMed] [Google Scholar]
- 36.Mitchell A.J. Screening for cancer-related distress: when is implementation successful and when is it unsuccessful? Acta Oncol. 2013;52:216–224. doi: 10.3109/0284186X.2012.745949. [DOI] [PubMed] [Google Scholar]
- 37.Hem E., Loge J.H., Haldorsen T., Ekeberg Ø. Suicide risk in cancer patients from 1960 to 1999. J Clin Oncol. 2004;22:4209–4216. doi: 10.1200/JCO.2004.02.052. [DOI] [PubMed] [Google Scholar]
- 38.Yousaf U., Christensen M.L., Engholm G., Storm H.H. Suicides among Danish cancer patients 1971–1999. Br J Cancer. 2005;92:995–1000. doi: 10.1038/sj.bjc.6602424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fisch M. Treatment of depression in cancer. J Natl Cancer Inst Monogr. 2004;32:105–111. doi: 10.1093/jncimonographs/lgh011. [DOI] [PubMed] [Google Scholar]
- 40.Lloyd-Williams M., Dennis M. A prospective study to determine the association between physical symptoms and depression in patients with advanced cancer. Palliat Med. 2004;18:558–563. doi: 10.1191/0269216304pm923oa. [DOI] [PubMed] [Google Scholar]
- 41.Montgomery C., Lydon A. Psychological distress among cancer patients and informed consent. J Psychosom Res. 1999;46:241–245. doi: 10.1016/s0022-3999(98)00089-0. [DOI] [PubMed] [Google Scholar]
- 42.Ell K. Randomized controlled trial of collaborative care management of depression among low-income patients with cancer. J Clin Oncol. 2008;26:4488–4496. doi: 10.1200/JCO.2008.16.6371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rayner L, et al. Antidepressants for depression in physically ill people. Cochrane review; 2010: issue 4. [DOI] [PMC free article] [PubMed]
- 44.Musselman D.L., Lawson D.H. Paroxetine for the prevention of depression induced by high-dose interferon alfa. N Engl J Med. 2001;344:961–966. doi: 10.1056/NEJM200103293441303. [DOI] [PubMed] [Google Scholar]
- 45.Cipriani A. Comparative efficacy and acceptability of 12 new-generation antidepressants: a multiple-treatments meta-analysis. Lancet. 2009;373:746–758. doi: 10.1016/S0140-6736(09)60046-5. [DOI] [PubMed] [Google Scholar]
- 46.Holland J.C., Romano S.J. A controlled trial of fluoxetine and desipramine in depressed women with advanced cancer. Psychooncology. 1998;7:291–300. doi: 10.1002/(SICI)1099-1611(199807/08)7:4<291::AID-PON361>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 47.Pezzella G., Moslinger-Gehmayr R. Treatment of depression in patients with breast cancer: a comparison between paroxetine and amitriptyline. Breast Cancer Res Treat. 2001;70:1–10. doi: 10.1023/a:1012518831494. [DOI] [PubMed] [Google Scholar]
- 48.Musselman D.L., Somerset W.I. A double-blind, multicenter, parallel-group study of paroxetine, desipramine, or placebo in breast cancer patients (stages I, II, III, and IV) with major depression. J Clin Psychiatry. 2006;67:288–296. doi: 10.4088/jcp.v67n0217. [DOI] [PubMed] [Google Scholar]
- 49.Rodin G., Lloyd N. The treatment of depression in cancer patients: a systematic review. Support Care Cancer. 2007;15:123–136. doi: 10.1007/s00520-006-0145-3. [DOI] [PubMed] [Google Scholar]
- 50.Möller H.J. Do SSRIs or antidepressant in general increase suicidality? Eur Arch Psychiatry Clin Neurosci. 2008;258(Suppl 3):3–23. doi: 10.1007/s00406-008-3002-1. [DOI] [PubMed] [Google Scholar]
- 51.Coogan P. Review of 2000 the epidemiological literature on antidepressant use and breast cancer risk. Expert Rev Neurother. 2006;6:1363–1374. doi: 10.1586/14737175.6.9.1363. [DOI] [PubMed] [Google Scholar]
- 52.Boyer E.W., Shannon M. The serotonin syndrome. N Engl J Med. 2005;352:1112–1120. doi: 10.1056/NEJMra041867. [DOI] [PubMed] [Google Scholar]
- 53.Yap K.Y.-L., Tay W.L., Chui W., Chan A. Clinically relevant drug interactions between anticancer drugs and psychotropic agents. Eur J Cancer Care. 2011;20:6–32. doi: 10.1111/j.1365-2354.2009.01113.x. [DOI] [PubMed] [Google Scholar]
- 54.Gilbody S., Bower P. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes. Arch Intern Med. 2006;166:2314–2321. doi: 10.1001/archinte.166.21.2314. [DOI] [PubMed] [Google Scholar]
- 55.Pampallona S., Bollini P., Tibaldi G., Kupelnick B., Munizza C. Combined pharmacotherapy and psychological treatment for depression: a systematic review. Arch Gen Psychiatry. 2004;61:714–719. doi: 10.1001/archpsyc.61.7.714. [DOI] [PubMed] [Google Scholar]
- 56.Pasquini M., Biondi M. Depression in cancer patients: a critical review. Clin Pract Epidemiol Ment Health. 2007;3:2. doi: 10.1186/1745-0179-3-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Stanton A.L. How and for whom? Asking questions about the utility of psychosocial interventions for individuals diagnosed with cancer. J Clin Oncol. 2005;23:4818–4820. doi: 10.1200/JCO.2005.01.913. [DOI] [PubMed] [Google Scholar]
- 58.Kissane D.W., Grabsch B. Supportive-expressive group therapy for women with metastatic breast cancer: survival and psychosocial outcome from a randomized controlled trial. Psychooncology. 2007;16:277–286. doi: 10.1002/pon.1185. [DOI] [PubMed] [Google Scholar]
- 59.Boesen E.H., Johansen C. Impact of psychotherapy on cancer survival: time to move on? Curr Opin Oncol. 2008;20:372–377. doi: 10.1097/CCO.0b013e3283021690. [DOI] [PubMed] [Google Scholar]
- 60.Cunningham A.J., Edmonds C.V. A randomized controlled trial of the effects of group psychological therapy on survival in women with metastatic breast cancer. Psychooncology. 1998;7:508–517. doi: 10.1002/(SICI)1099-1611(199811/12)7:6<508::AID-PON376>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 61.Sheard T., Maguire P. The effect of psychological interventions on anxiety and depression in cancer patients: results of two meta-analyses. Br J Cancer. 1999;80:1770–1780. doi: 10.1038/sj.bjc.6690596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Osborn R.L., Demoncada A.C., Feuerstein M. Psychosocial interventions for depression, anxiety, and quality of life in cancer survivors: meta-analyses. Int J Psychiatry Med. 2006;36:13–34. doi: 10.2190/EUFN-RV1K-Y3TR-FK0L. [DOI] [PubMed] [Google Scholar]
- 63.Blake-Mortimer J., Gore-Felton C. Improving the quality and quantity of life among patients with cancer: a review of the effectiveness of group psychotherapy. Eur J Cancer. 1999;35:1581–1586. doi: 10.1016/s0959-8049(99)00194-x. [DOI] [PubMed] [Google Scholar]
- 64.Goodwin P.J., Leszcz M. The effect of group psychosocial support on survival in metastatic breast cancer. N Engl J Med. 2001;345:1719–1726. doi: 10.1056/NEJMoa011871. [DOI] [PubMed] [Google Scholar]
- 65.Li M., Fitzgerald P., Rodin G. Evidence-based treatment of depression in patients with cancer. J Clin Oncol. 2012;30:1187–1196. doi: 10.1200/JCO.2011.39.7372. [DOI] [PubMed] [Google Scholar]


