Abstract
Internal anal sphincter (IAS) tone plays an important role in rectoanal incontinence (RI). IAS tone may be compromised during aging, leading to RI in certain patients. We examined the influence of oxidative stress in the aging-associated decrease in IAS tone (AADI). Using adult (4–6 mo old) and aging (24–30 mo old) rats, we determined the effect of oxidative stress on IAS tone and the regulatory RhoA/ROCK signal transduction cascade. We determined the effect of the oxidative stress inducer LY83583, which produces superoxide anions (O2·−), on basal and stimulated IAS tone before and after treatment of intact smooth muscle strips and smooth muscle cells with the O2·− scavenger SOD. Our data showed that AADI was associated with a decrease in RhoA/ROCK expression at the transcriptional and translational levels. Oxidative stress with a LY83583-mediated decrease in IAS tone and relaxation of IAS smooth muscle cells was associated with a decrease in RhoA/ROCK signal transduction, which was reversible by SOD. In addition, LY83583 caused a significant decrease in IAS contraction produced by the RhoA activator and a known RhoA/ROCK agonist, U46619, that was also reversible by SOD. The inhibitory effects of LY83583 and the ROCK inhibitor Y27632 on the U46619-induced increase in IAS tone were similar. We conclude that an increase in oxidative stress plays an important role in AADI in the elderly and may be one of the underlying mechanisms of RI in certain aging patients.
Keywords: aging, internal anal sphincter, oxidative stress, rectoanal incontinence, RhoA/ROCK
although multifactorial, the intrinsic tone and fibroelastic properties of the internal anal sphincter (IAS) play major roles in maintaining rectoanal continence (4). The IAS exhibits spontaneous myogenic tone: it relaxes in response to the rectoanal inhibitory reflex, leading to expulsion of feces. Surgery, trauma, radiation, childbirth (4, 26), and aging are associated with a decrease in IAS tone and derangement of the fibroelastic properties of the IAS, leading to rectoanal incontinence (RI) (26, 42, 50). Molecular mechanisms underlying the aging-associated decrease in IAS tone (AADI) are not known.
Aging is characterized by a progressive decline in physiological function and increased susceptibility to disease. Although multiple theories have been proposed for aging-related bodily dysfunction, the “mitochondrial free radical theory of aging” is widely accepted (1, 16). It has been postulated that aging-related bodily dysfunction is the result of free radical-induced oxidative stress and the inability of the antioxidant defenses to counterbalance these changes.
It follows, therefore, that AADI may be associated with oxidative stress. In general, when examined individually, aging (6, 13, 47, 51) and oxidative stress (36, 46) have been reported to decrease overall propulsive activity of the gut because of degenerative changes at the neuronal and muscular levels. Neuromuscular degenerative changes have been reported in the aging IAS (49), and it has been suggested that a decrease in IAS tone and altered rectal sensation may be the leading causes of aging-associated RI (3, 13, 31, 42, 48). At the smooth muscle level (e.g., rat colon), aging-related changes in colonic motility have been suggested to be via decreases in phosphorylation of the myosin-binding subunit of myosin phosphatase target subunit 1 (MYPT1) and regulatory MLC (MLC20), secondary to RhoA-associated kinase (RhoA/ROCK) inactivation (41). Interestingly, no studies examining the role of oxidative stress during aging in gastrointestinal dysmotility, especially in AADI, have been published. Since the majority of basal IAS tone is a function of the integrity of myogenic properties (10, 21, 22, 28, 34), it is critical to examine the role of oxidative stress during aging in intact IAS smooth muscle and smooth muscle cells (SMCs).
Reactive oxygen species (ROS) are free radicals with a single unpaired electron (20, 44), mainly produced by mitochondria as by-products of O2 metabolism. The important ROS are superoxide anion (O2·−), H2O2, and the hydroxyl radical (ȮH) (44). While in physiological range, ROS play important roles in cell signaling processes, their overproduction leads to oxidative stress, causing damage to cellular constituents, including DNA, proteins, and lipids (14). Oxidative stress has been implicated in a large number of chronic degenerative diseases, such as atherosclerosis, pulmonary fibrosis, and cancer, and as a mechanism of senescence and aging (17). Also, it has been well established that oxidative stress leads to decreased contractility of gastrointestinal smooth muscle (9, 45). The body's antioxidant defense mechanisms, such as SOD, catalase, glutathione peroxidase, and heat shock proteins, play a crucial role in regulating ROS levels. While SOD detoxifies O2·− by dismutation into H2O2, catalase breaks down H2O2 into H2O and O2 (15).
Homeostasis between the production and deactivation (by antioxidants) of ROS, which is crucial for human physiology, is known to be disturbed during aging (27). The purpose of the present investigation is to determine the effect of oxidative stress on IAS tone in relation to aging, while monitoring the contractile signal transduction machinery RhoA/ROCK/phosphorylated MYPT1 (pMYPT1)/phosphorylated MLC20 (pMLC20). To induce and monitor oxidative stress, we used the O2·− generator LY83583 and specific dihydroethidium (DHE) staining strategy, respectively.
MATERIALS AND METHODS
Selection of animals for aging studies.
Sprague-Dawley rats of two distinct age groups, 4–6 mo (adult) and 24–30 mo (aging), were used.
IAS tissue sample preparation.
Rats were euthanized by decapitation, and their anal canals were quickly removed and transferred to oxygenated (95% O2-5% CO2) Krebs physiological solution (KPS) of the following composition (in mM): 118.07 NaCl, 4.69 KCl, 2.52 CaCl2, 1.16 MgSO4 1.01 NaH2PO2, 25 NaHCO3, and 11.10 glucose (37°C). The experimental protocols were approved by the Institutional Animal Care and Use Committee of Thomas Jefferson University and were in accordance with the recommendations of the American Association for the Accreditation of Laboratory Animal Care.
Force measurement.
IAS tissues were dissected from the anal canals of the adult and aging rats in KPS. The serosa, adventitia with blood vessels, longitudinal smooth muscle layer, and mucosa and submucosa were carefully removed, and ∼1 × 10-mm strips of the circular smooth muscle (CSM) layer were prepared and hung in 2-ml muscle baths containing oxygenated KPS. Initially, we applied a tension of 1.0 g and then allowed the strips to equilibrate for 60 min. To determine the amplitude of active basal tone in each strip, we determined the relaxing effects of Ca2+-free (0 Ca2+) KPS in the beginning and at the end of each experiment. All force data were monitored using force transducers (model FT03, Grass Instruments, Quincy, MA) and Chart 4.1.2 via a PowerLab/8SP data-acquisition system (ADInstruments, Colorado Springs, CO). We determined the effect of 3 × 10−5 M LY83583 (an oxidative stress inducer) on basal IAS tone and on U46619 (10−6 M)-induced contraction in the IAS before and after SOD. IAS smooth muscles were identified by development of steady basal tone that responded to immediate relaxation following electrical field stimulation (0.5–20 Hz, 0.5-ms pulse, 12 V, 4-s train duration).
Isolation of SMCs and cell culture.
SMCs from the IAS were isolated as described previously (38). Briefly, IAS tissues from the CSM layer of the anal canal from adult and aging rats were cut into ∼1-mm cubes, and the SMCs were dispersed using repeated short-term incubations with oxygenated KPS containing 0.1% collagenase type I and 0.01% soybean trypsin inhibitor at 37°C for 1 h. Finally, the cell suspension was filtered through a 500-μm Nitex mesh. The tissue trapped on the mesh was rinsed with 25 ml (5 × 5 ml) of collagenase-free KPS and incubated in collagenase-free KPS at 37°C. The filtrate containing the cells was centrifuged at 350 g for 10 min at room temperature. The cells in the pellet were resuspended on collagen-coated plates in DMEM with 5% fetal bovine serum, 5% penicillin-streptomycin, 50 μg/ml gentamicin, 2 μg/ml amphotericin B, and 50 μg/ml sodium ascorbate (5) in 100-mm tissue culture dishes (Corning) at 37°C and 5% CO2 in an incubator with regulated humidity.
Measurements of SMC length.
Freshly isolated SMCs were resuspended in oxygenated KPS (at 37°C) at a density of 3 × 104 cells/ml in 100-μl aliquots and exposed to different concentrations of LY83583 and RhoA activator for 3 min. SMCs were then fixed with acrolein (final concentration 1%) and transferred onto chrome-alum-coated glass slides (Fisher Scientific, Pittsburgh, PA). The studies were repeated in the presence of SOD and LY83583, respectively. Individual cell lengths were measured by micrometry using phase-contrast microscopy (19) with a custom-assembled microscope (Olympus, Tokyo, Japan) and a closed-circuit video camera (model MC-7, PULNiX America, Sunnyvale, CA) connected to a personal computer. Images of the cells were stored digitally, and cell lengths were measured by Image-Pro Plus version 4.0 (Media Cybernetics, Silver Spring, MD). Shortening of SMCs in each category of experiments was calculated as percentile of original cell length.
RT-PCR analysis.
mRNAs were isolated from the smooth muscle strips and the SMCs using the TRIzol method. The smooth muscle tissues were flash-frozen in liquid N2 and homogenized in 1 ml of TRIzol reagent (Sigma). SMCs were washed with PBS and lysed in 1 ml of TRIzol reagent. Then chloroform (200 μl) was added to tissue and SMC samples, and the samples were mixed vigorously. Solutions were centrifuged at 1,500 rpm for 5 min at room temperature, and the aqueous layer was collected into a separate tube. A 0.7 volume of isopropanol was added to the tube, the contents were mixed gently, and the solution was centrifuged for 12 min at 15,000 rpm. The supernatant was discarded carefully to ensure that the pellet was undisturbed. The pellet was then washed twice with 70% ethanol, and RNA was dissolved in diethyl pyrocarbonate-treated RNase-free distilled water. RNA quality was assessed on 0.8% agarose gel and quantified using a NanoDrop spectrophotometer as the ratio of fluorescence at 260 nm to fluorescence at 280 nm and the ratio of fluorescence at 260 nm to fluorescence at 230 nm.
RNA (1 μg) was reverse-transcribed using the Sensiscript RT kit for mRNA (Qiagen, Valencia, CA). RhoA, ROCK II, and GAPDH cDNAs were amplified using gene-specific forward and reverse primers (Table 1). PCRs were carried out with GoTaq Green Master Mix (Promega) using an Eppendorf Mastercycler personal thermocycler (Fisher, Allentown, PA). The PCR cycle consisted of 94°C for 5 min, 94°C for 30 s (denaturation phase), 60°C for 30 s (annealing phase), and 72°C for 1 min. PCR products were separated on a 1.5% (wt/vol) agarose gel containing ethidium bromide and visualized with UV light, and pictures were taken using the GelDoc-It gel documentation system (UVP). Relative densities of RhoA and ROCK II were calculated by normalization of the densities of each blot to the density of GAPDH.
Table 1.
Primers
| Sequence (5′-3′) | Position | Accession No. | |
|---|---|---|---|
| RhoA | NM_016802 | ||
| Forward | CAGCAAGGACCAGTTCCCAGA | 501–521 | |
| Reverse | TGCCATATCTCTGCCTTCTTCAGG | 847–870 | |
| ROCK II | NM_013022 | ||
| Forward | TAGAAGAACACCTTAGCAGTGAGGTACAAG | 1401–1430 | |
| Reverse | TGTCTCTTCTCCAGCTCTACTTTTGCT | 2034–2060 | |
| GAPDH | NM_017008.4 | ||
| Forward | GGCTCATGACCACAGTCCAT | 587–606 | |
| Reverse | CCCCTCCTGTTGTTATGGGG | 1203–1222 |
Western blot analysis.
IAS smooth muscle strips and SMCs from adult and aging rats, before and after treatment with LY83583, were flash-frozen in liquid N2, suspended in ice-cold homogenization buffer (10 mM Tris·HCl, pH 7.5, 5 mM MgCl2, 2 mM EDTA, 250 mM sucrose, 1 mM dithiothreitol, and 1% Triton X-100), and homogenized using an IKA Ultra-Turrax T8 tissue homogenizer (Werke). The extracts were centrifuged at 800 g for 10 min, and protein concentration in the resultant supernatant was determined using a bicinchoninic acid protein assay reagent kit (Pierce, Rockford, IL). Twenty micrograms of protein in 20 μl of lysates were mixed with 2× Laemmli sample buffer (with final concentrations of 62.5 mM Tris, 1% SDS, 15% glycerol, 0.005% bromophenol blue, and 2% mercaptoethanol) and placed in a boiling water bath for 5 min. Proteins in the samples were separated by SDS-polyacrylamide gel [7.5% gel for ROCK II, phosphorylated (Thr696) MYPT1, and MYPT1; 15% gel for RhoA, MLC20, and phosphorylated (Thr18/Ser19) MLC20] and then electrophoretically transferred onto polyvinylidene difluoride membranes using the iBlot dry blotting system (Invitrogen, Carlsbad, CA) at room temperature.
To block nonspecific antibody binding, the membranes were soaked for 1 h at room temperature in LI-COR Odyssey blocking buffer and then incubated with the specific primary antibodies (1:1,000 dilution of RhoA, ROCK II, pMYPT1, MYPT1, MLC20, pMLC20, and GAPDH) diluted in LI-COR buffer containing 0.1% Tween 20 for 1 h at room temperature. After three 10-min wash cycles in Tris-buffered saline-Tween 20, the membranes were incubated with the IRDye680- and IRDye800-conjugated secondary antibody (LI-COR Biosciences) in darkness [bovine anti-rabbit (1:10,000 dilution) for RhoA/ROCK II, MYPT1, pMYPT1, and MLC20; bovine anti-goat (1:5,000 dilution) for pMLC20]. After three more 10-min wash cycles in Tris-buffered saline-Tween 20, the membranes were kept in PBS buffer on a shaker for 10 min at room temperature in darkness and scanned using a LI-COR infrared scanner, and the integrated optical densities were determined using ImageJ software (National Institutes of Health, Bethesda, MD). The relative densities were calculated by normalization of the expression of each protein to that of GAPDH.
Oxidative stress quantification.
The oxidative stress measurement protocol was adopted from previously published studies (36). Briefly, a CSM layer from the IAS of adult and aging rats was carefully isolated, and unfixed preparations were incubated in 2 μM DHE at 37°C for 60 min in light-protected tubes. The whole mounts were washed three times with Krebs-Ringer HEPES solution (in mM: 130.0 NaCl, 1.3 KCl, 2.2 CaCl2, 1.2 MgSO4, 1.2 KH2PO4, 1.0 HEPES, and 0.09 glucose, pH 7.4; Sigma) and mounted on slides with Prolong Gold mounting medium (Invitrogen), and coverslips were applied. Microscopic images were taken using a Nikon Eclipse 80i microscope and an Olympus IX71 inverted microscope and analyzed using Nikon imaging software (NIS-Elements, version 3.1, Nikon Instruments, Melville, NY; Center for Translational Medicine, Department of Medicine, Thomas Jefferson University). Images were captured with identical exposure settings and relative intensities were calculated without adjustments for brightness or contrast.
For assessment of O2·− production in the SMCs, SMCs from the IAS of adult and aging rats were grown on coverslips or in 96-well plates. SMCs were treated with different agents for different time periods. SMCs were preloaded with DHE (5 μM) in DMEM for 30 min and then washed with medium. LY83583 (10−5 M) was added to the DHE-treated cells, which were then incubated at 37°C for 10 min. The effect of 200 U/ml SOD on DHE fluorescence was also determined.
To preserve the fluorescence signal, the cells were mounted with Prolong Gold mounting medium, and coverslips were applied. The slides were examined under the UV microscope, and photographs were taken under a Texas Red filter. Intensity of fluorescence (intensity per unit area, normalized to 1 in controls) was measured using Image-Pro software, and graphs were plotted using Prism version 5.1.
Chemicals and drugs.
Bovine erythrocyte SOD and 6-anilino-5,8-quinolinedione (LY83583) were obtained from Sigma; tin protoporyphyrin IX (SnPP IX) from Porphyrin Products (Logan, UT); U46619 and Y27632 from Biomol (Plymouth Meeting, PA); MLC20 antibodies from Sigma (St. Louis, MO); GAPDH, RhoA, ROCK II, MYPT1, phosphorylated (Thr696) MYPT1, and phosphorylated (Thr18/Ser19) MLC20 antibodies from Santa Cruz Biotechnology (Santa Cruz, CA); IRDye680- and IRDye800-conjugated mouse, goat, and rabbit secondary antibodies from LI-COR Biosciences; and RhoA activator (cell-permeable calpeptin, catalog no. CN01-A) from Cytoskeleton (Denver, CO).
Data analysis.
Values are means ± SE from at least four independent experiments and plotted using Prism 5.1 (GraphPad Software, La Jolla, CA). Student's unpaired t-test was used to compare two different groups. P < 0.05 was considered to be statistically significant.
RESULTS
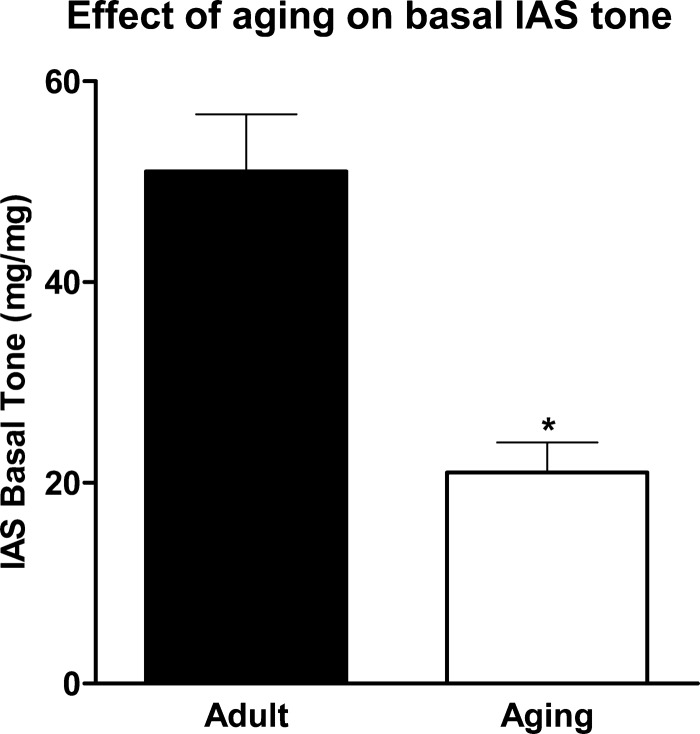
Effect of aging on basal IAS tone.
Basal IAS tone was significantly lower in aging than adult rats: 21.0 ± 3.0 vs. 51.0 ± 0.6 mg/mg tissue wt (P < 0.05, n = 4; Fig. 1).
Fig. 1.
Effect of aging on basal internal anal sphincter (IAS) tone. IAS tone was significantly decreased in aging compared with adult rats. Values are means ± SE of 4 animals. *P < 0.05.
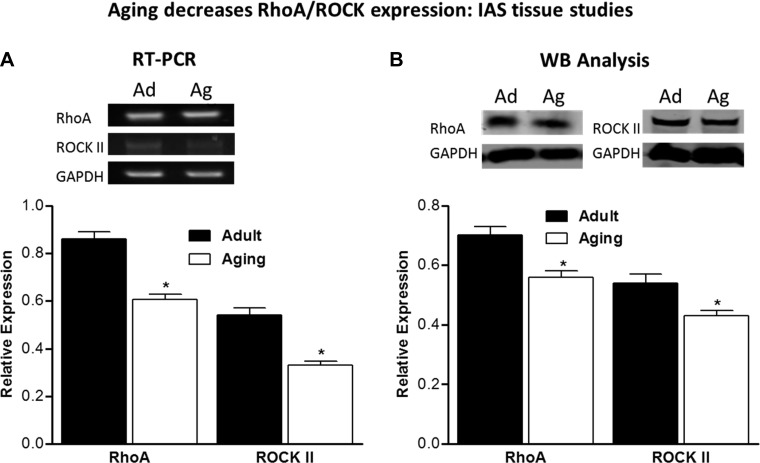
Effect of aging on RhoA/ROCK II expression.
RT-PCR data from IAS smooth muscle tissues showed significantly lower expression of RhoA (0.61 ± 0.021 vs. 0.86 ± 0.031, P < 0.05, n = 4) and ROCK II (0.33 ± 0.017 vs. 0.54 ± 0.033, P < 0.05, n = 4) in aging than adult rats. Similarly significant differences in IAS tissues between adult and aging rats were obtained using Western blot analysis for protein expression levels. RhoA levels were 0.70 ± 0.032 vs. 0.56 ± 0.02, while ROCK II levels were 0.54 ± 0.030 vs. 0.43 ± 0.017, in adult vs. aging rats, respectively (P < 0.05, n = 4; Fig. 2). These data suggest that the major molecular determinants of basal IAS tone, RhoA/ROCK (28, 29, 32, 33, 35), were significantly lower in IAS from aging rats.
Fig. 2.
A: RT-PCR data show significantly lower gene expression of RhoA and ROCK II in IAS tissues from aging (Ag) than adult (Ad) rats. Values are means ± SE of 4 animals. *P < 0.05. B: Western blot (WB) data show significantly lower expression of RhoA and ROCK II in IAS from aging than adult rats. Values are means ± SE of 4 animals. *P < 0.05.
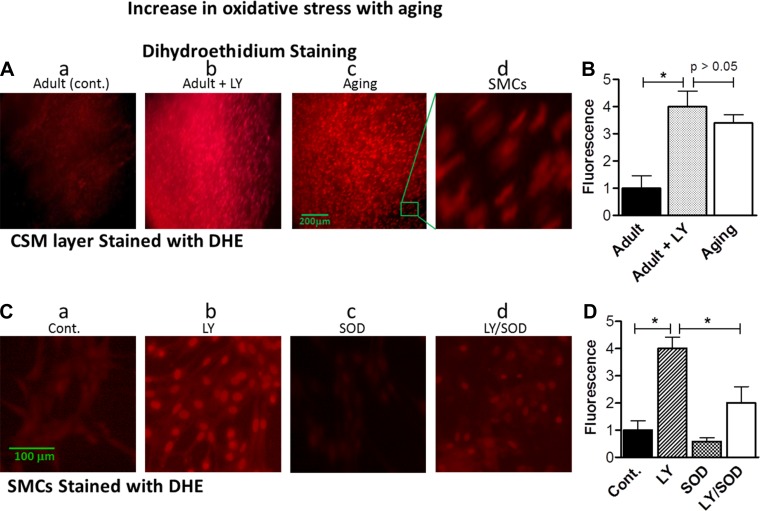
Increase in oxidative stress in IAS tissues and SMCs from aging rats.
We monitored oxidative stress in intact smooth muscles and SMCs via DHE staining, which is known to specifically stain intracellular ROS (12). Data revealed significantly higher levels of DHE fluorescence in SMCs from aging than adult rats (4.0 ± 0.67 vs. 1.0 ± 0.56, P < 0.05, n = 4; Fig. 3, A and B). These data suggest significantly higher levels of O2·− in the IAS during aging.
Fig. 3.
A: immunofluorescent images of dihydroethidium (DHE)-stained IAS circular smooth muscle (CSM) layer from adult and aging rats. B: quantitative data from images in A. LY83583 (LY) causes a significant increase in oxidative stress in the adult rats, comparable to that in aging rats. Values are means ± SE of 4 animals. *P < 0.05. C: immunofluorescent images of IAS smooth muscle cells (SMCs) from adult rats. D: quantitative data from images in C. Pretreatment with LY83583 caused a significant increase in oxidative stress that was significantly attenuated by pretreatment with 200 U/ml SOD. Changes in O2·− (as a measure of oxidative stress) were monitored by DHE fluorescence, which shows an increase in O2·− by LY83583 and during aging and attenuation of O2·− by SOD (reflecting a decrease in oxidative stress by conversion of O2·− to H2O2). Cont, control. Values are means ± SE of 4 animals. *P < 0.05.
The oxidative stress inducer LY83583 mimics aging-related decrease in basal IAS tone.
LY83583 has been widely used to induce an increase in O2·− production in SMCs (8). Treatment of IAS smooth muscle strips from adult rats with 10 μM LY83583 significantly increased O2·− production, as validated by a significant increase in DHE staining (from 1.0 ± 0.50 to 4.0 ± 0.45, P < 0.05, n = 4; Fig. 3, A and B). These values in IAS from adult rats pretreated with LY83583 were not significantly different from values in tissues from aging rats (P > 0.05; Fig. 3, A and B). Similar increases in DHE fluorescence were observed in SMCs treated with LY83583 (Fig. 3, C and D). The specificity of increases in oxidative stress following LY83583 was confirmed by significant blockade of DHE staining by pretreatment of cells with SOD (P < 0.05, n = 4; Fig. 3, C and D).
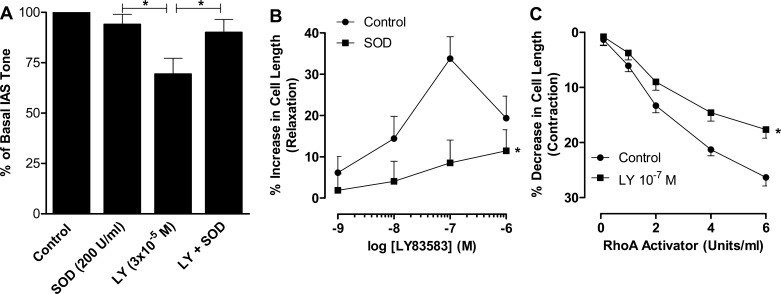
Decrease in basal IAS tone by LY83583 and reversal by SOD.
These studies performed in adult animals revealed that LY83583 (3 × 10−5 M) produced a significant decrease in basal tone of 31.0 ± 8.0% (from 100% of basal to 69.3 ± 7.8%, P < 0.05, n = 4; Fig. 4A). To determine the specificity of the inhibitory effect of LY83583 in IAS smooth muscle, we examined the rescuing effect of LY83583 by SOD. Data revealed that SOD significantly reversed the inhibitory effect of LY83583 to levels (90.0 ± 6.5%) not significantly different from the basal levels (P > 0.05, n = 4; Fig. 4A). In the same series of experiments, we found that SOD by itself had no significant effect on basal tone. These data revealed that the inhibitory effect of LY83583 on IAS tone was primarily via an increase in oxidative stress by increased production of O2·−.
Fig. 4.
A: LY83583 (3 × 10−5 M) produces a significant decrease in basal IAS tone in adult rats (n = 5) that is significantly attenuated by pretreatment with 200 U/ml SOD (n = 4). Values are means ± SE. *P < 0.05. B: LY83583 causes a concentration-dependent increase in IAS SMC length that is significantly attenuated by SOD. Values are means ± SE of 30–40 cells pooled from 4 animals for each set of experiments. *P < 0.05. C: RhoA activator results in a decrease in SMC length that is significantly blocked by LY83583. Values are means ± SE of 4 animals. *P < 0.05.
A direct effect of LY83583 on the decrease in IAS tone was confirmed by its inhibitory effect on IAS SMCs. LY83583 (10−9–10−6 M) caused a concentration-dependent increase in IAS SMC length; 10−7 M LY83583 was the maximal inhibitory concentration (Fig. 4B). In addition, this relaxant effect of LY83583 was significantly attenuated by pretreatment of the cells with SOD (P < 0.05; Fig. 4B).
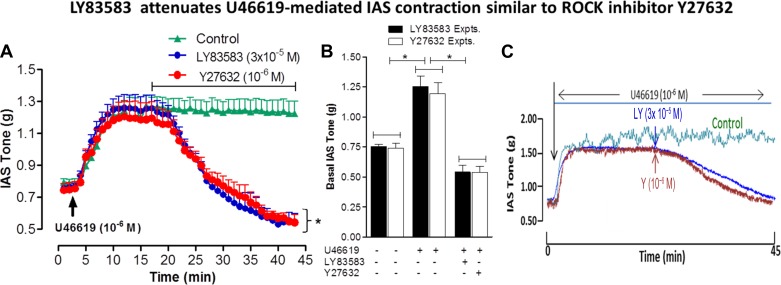
Inhibitory effects of LY83583 and Y27632 on the increase in IAS tone caused by Rho activator and U46619.
LY83583 (10−7 M) caused significant attenuation of the SMC contraction produced by RhoA activator (P < 0.05; Fig. 4C). Previously published studies from our laboratory in the IAS (11) and those of others using different smooth muscles (43) showed that U46619 produces sustained and concentration-dependent contraction via RhoA/ROCK activation. The present data show that, following sustained IAS contraction with U46619 (10−6 M), LY83583 caused an immediate and precipitous decrease in IAS tone (Fig. 5A); the U46619-induced increase in IAS tone lasts >45 min. This inhibitory effect of LY83583 on IAS tone following sustained contraction with U46619 was similar to that with the ROCK inhibitor Y27632 (Fig. 5A). Quantitative data in Fig. 5B show the absolute changes in IAS tone in the basal state following maximal sustained contraction at 10 min after U46619 and at peak relaxation with LY83583 and Y27632. Typical traces in Fig. 5C show the inhibitory effects of LY83583 and Y27632 on IAS contraction caused by U46619.
Fig. 5.
A: U46619 causes an increase in basal IAS tone that peaks at 10 min following its administration. This increase in IAS tone is significantly and similarly inhibited by LY83583 and Y27632 within 5 min of their administration. Values are means ± SE of 4 animals. *P < 0.05. B: increase in IAS tone by U46619 is similarly inhibited by pretreatment with LY83583 or Y27632. C: traces showing inhibitory effect of LY83583 and Y27632 (Y2) on the U46619-induced increase in IAS tone.
Inhibition of RhoA/ROCK and downstream signal transduction in the IAS by LY83583.
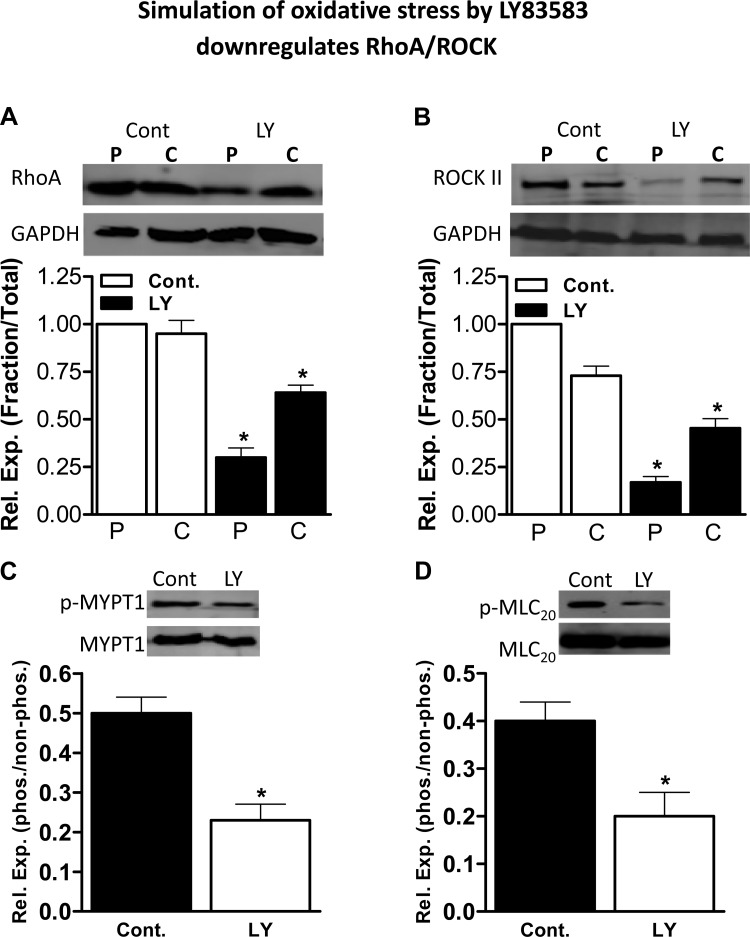
In these experiments using IAS smooth muscle strips, LY83583 significantly decreased the membranous expression levels of RhoA (from a normalized control value of 1.0 to 0.30 ± 0.05, P < 0.05, n = 4) and ROCK II (from a normalized control value of 1.0 to 0.17 ± 0.03, P < 0.05, n = 4; Fig. 6, A and B).
Fig. 6.
Western blots and corresponding quantitative data [relative expression (Rel Exp)] for the effect of LY83583 on particulate (P) and cytosolic (C) fractions of RhoA and ROCK II in the IAS. A and B: particulate RhoA and ROCK II levels were significantly decreased in IAS smooth muscle tissue. Values are means ± SE of 4 animals. *P < 0.05. C and D: phosphorylated (Thr696) myosin phosphatase target subunit 1 (pMYPT1) and phosphorylated (Thr18/Ser19) regulatory myosin light chain (pMLC20) were significantly decreased in the IAS following LY83583. Values are means ± SE of 4–6 animals. *P < 0.05. These data suggest significant inhibition of the RhoA/ROCK signal transduction cascade with LY83583.
Similarly, RhoA/ROCK downstream signal transduction data reveal that inhibitory effects of LY83583 on IAS tone are associated with significant decreases in the levels of phosphorylated (Thr696) MYPT1 and phosphorylated (Thr18/Ser19) MLC20 (P < 0.05, n = 4; Fig. 6, C and D). The above-described data showing decreases in RhoA/ROCK at the membrane level and decreases in pMYPT1 and pMLC20 are similar to data previously obtained with Y27632 in rat and human IAS (28, 35).
Together, these data suggest that an increase in oxidative stress (mimicked by LY83583) causes a decrease in IAS tone by its action directly at the SMC and via RhoA/ROCK inhibition.
DISCUSSION
These studies, for the first time, demonstrate the role of oxidative stress on the aging-associated decrease in IAS tone and the underlying control mechanisms, as depicted in the model in Fig. 7. On the basis of the present findings, we conclude that 1) AADI is associated with an increase in oxidative stress, 2) oxidative stress is, in part, responsible for the AADI that corresponds with disruption of the underlying molecular control mechanisms RhoA/ROCK and the downstream signaling cascade, and 3) the antioxidant SOD (a O2·− scavenger) successfully reverses oxidative damage in the IAS.
Fig. 7.
Simplified model for the mechanism of oxidative stress in the decrease in IAS tone during aging. An increase in ROS is a consequence of imbalance between levels of antioxidants (e.g., SOD, catalases, and heme oxygenase-1) and oxidants (e.g., O2·−, hydroxyl ions, ferryl ions, and heme) that may cause oxidative damage within IAS SMCs, leading to downregulation of the RhoA/ROCK and downstream signal transduction cascade. The net effect of these events leads to decreased IAS tone. We have used LY83583 to mimic generation of ROS and SOD to attenuate their damaging effect.
Rectoanal or fecal incontinence (RI) is known to have a devastating impact on quality of life (24). Although the etiology of RI is multifactorial, a recent study demonstrates that age is an independent predictor of RI after adjustment for the number of chronic illnesses, overall health status, and physical activity (50). Pathophysiology of aging-associated RI is not clearly understood. Knowledge of the molecular mechanisms and factors leading to AADI may hold the key in the therapeutic management of RI, at least in a certain category of patients with hypotensive IAS. Using a multipronged approach of functional and molecular biology, we evaluated the role of aging-associated oxidative stress in the IAS in adult (4–6 mo old) and aging (24–30 mo old) rats.
The present data suggest that aging leads to a decrease in the basal tone of the IAS due to downregulation of the RhoA/ROCK signal transduction cascade. Previously published studies from our laboratory show the critical role of the RhoA/ROCK pathway in maintaining the basal tone of the IAS in humans and rats (28, 33, 35). Accordingly, spontaneously active RhoA leads to autophosphorylation and activation of ROCK. The latter leads to phosphorylation of MYPT1, thus inhibiting MLC phosphatase, and an increase in the levels of pMLC20. Therefore, it follows that low levels of RhoA/ROCK in aging may unleash MLCP, causing dephosphorylation of pMLC20 and, thus, a reduction of basal IAS tone. In support of our studies, data from rat colon have shown reduced colonic motility associated with downregulation of the RhoA/ROCK signaling cascade with aging (40, 41). The present data, however, add a new dimension to the existing knowledge; i.e., these aging-associated changes in basal tone are due to increased oxidative stress during aging. Additionally, it is conceivable that aging may also adversely affect neurally mediated rectoanal inhibitory reflex-induced relaxation of the IAS; this aspect, however, is not within the scope of the present investigation.
Oxidative stress has been implicated in many diseases, including the aging-associated decrease in gastrointestinal smooth muscle tone (17, 37). The increase in oxidative stress is a consequence of imbalance between an increase in ROS production and a decrease in the body's antioxidant mechanisms (1, 16). Evidence in support of a relationship between oxidative stress and AADI emerges from our experiments with LY83583, which successfully induced oxidative stress in IAS smooth muscle tissues and SMCs, as demonstrated by the increase in DHE fluorescence intensity. The ability of LY83583 to generate O2·− (25) is confirmed by nearly complete reversal of the LY83583-induced increase in DHE fluorescence intensity by SOD. The data further suggest that an increase in oxidative stress in LY83583-treated adult rats is similar to that in untreated aging rats. These data suggest that induction of oxidative stress in adult animals mimics aging-induced effects on the IAS.
Functional data showing a significant decrease in IAS tone following pretreatment of the smooth muscles with LY83583 and its rescue by SOD further confirm the roles of oxidative stress and free radicals in the decreased IAS tone during aging. The present studies provide novel data that the oxidative stress-induced decrease in IAS tone is associated with downregulation of the RhoA/ROCK pathway within IAS SMCs. This is revealed by the similarities between the effects of LY83583 and Y27632 (a ROCK inhibitor) on basal IAS tone and on the precontracted IAS by a RhoA activator per se, as well as via Rho/ROCK activation via U46619. Additional data including the effect of LY83583 on the IAS SMC confirm concentration-dependent relaxation of the SMC that is rescued by pretreatment of the cells with SOD.
Western blot analysis of LY83583-treated IAS smooth muscles showing reduced protein expression of RhoA/ROCK and its downstream targets pMYPT1 and pMLC20 compared with control samples further confirms involvement of the RhoA/ROCK pathway in oxidative stress. This was shown by a significant shift in the levels of RhoA/ROCK from the membrane to the cytosol in IAS SMCs treated with LY83583. This demonstrates a significant decrease in the activation of RhoA/ROCK, as it has been shown that basal IAS tone is primarily dependent on constitutively active RhoA/ROCK (29).
The role of oxidative stress in the decrease in IAS smooth muscle tone is in agreement with data from other regions of the gastrointestinal tract (37, 52). Similar data (a decrease in IAS tone; not shown) were obtained following pretreatment of IAS smooth muscle strips and SMCs with other superoxide generators, 2,2′-azobis (2-amidinopropane) and 1,1-diphenyl-2-picryl-hydrazyl (2, 18, 30). These data from the gastrointestinal smooth muscle are, however, different from data from certain vascular smooth muscles, which showed superoxide-mediated contraction via RhoA activation (7, 23). The exact reason for the differences remains to be determined. A possible mechanism for this contraction is ROS-mediated inhibition of nitric oxide effects (23, 25, 39), since the basal state of smooth muscle contractility in those tissues is speculated to be a consequence of a net balance between multiple intracellular excitatory and inhibitory pathways. This implies that, in those tissues, nitric oxide synthase (NOS) activation plays an important role in the equilibrium for the basal tone, and NOS inhibition subsequent to oxidative stress may tilt the balance toward raising the smooth muscle tone. In support of this concept, we have observed that, in some experiments, LY83583 produces an increase in IAS tone that is converted to a decrease following treatment with the NOS inhibitor N-nitro-l-arginine. Conversely, in the human IAS, none of the IAS smooth muscle experiments displayed such a decrease in IAS tone (data not shown). Therefore, it is possible that differences in the responses to ROS may be tissue-specific and may depend on the redox status of the tissues and the type of maneuvers used to generate ROS. It is of interest that the effect of oxidative stress has not been examined in the spontaneously tonic smooth muscle tissues such as the IAS, which are driven by upregulated RhoA/ROCK signal transduction machinery (28, 29, 32, 33, 35), where NOS plays a limited role in the spontaneous tone.
In summary, the present data suggest that oxidative stress during aging plays a significant role in the mediation of reduced basal IAS tone and that antioxidants such as SOD have an important role in reversing the aging-associated oxidative damage in IAS. In addition, these studies suggest a therapeutic potential of such antioxidants in reversing the effect of aging-associated oxidative stress in the hypotensive IAS and, in turn, in RI.
GRANTS
This work was supported by National Institute of Diabetes and Digestive and Kidney Diseases Grant RO1 DK-035385 and an institutional grant from Thomas Jefferson University.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
J.S., S.K., and C.V.K. performed the experiments; J.S. and C.V.K. analyzed the data; J.S. and S.K. prepared the figures; J.S., S.K., C.V.K., and S.R. drafted the manuscript; J.S., S.K., C.V.K., and S.R. approved the final version of the manuscript; S.R. is responsible for conception and design of the research; S.R. interpreted the results of the experiments; S.R. edited and revised the manuscript.
REFERENCES
- 1.Barja G. Updating the mitochondrial free radical theory of aging: an integrated view, key aspects, and confounding concepts. Antioxid Redox Signal 19: 1420–1445, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Berrougui H, Martin-Cordero C, Khalil A, Hmamouchi M, Ettaib A, Mahuenda E, Herrera MD. Vasorelaxant effects of harmine and harmaline extracted from Peganum harmala L. seeds in isolated rat aorta. Pharmacol Res 54: 150–157, 2006 [DOI] [PubMed] [Google Scholar]
- 3.Bharucha AE. Outcome measures for fecal incontinence: anorectal structure and function. Gastroenterology 126: S90–S98, 2004 [DOI] [PubMed] [Google Scholar]
- 4.Bharucha AE, Rao SS. An update on anorectal disorders for gastroenterologists. Gastroenterology 146: 37–45, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bi D, Nishimura J, Niiro N, Hirano K, Kanaide H. Contractile properties of the cultured vascular smooth muscle cells: the crucial role played by RhoA in the regulation of contractility. Circ Res 96: 890–897, 2005 [DOI] [PubMed] [Google Scholar]
- 6.Bitar KN, Greenwood-Van Meerveld B, Saad R, Wiley JW. Aging and gastrointestinal neuromuscular function: insights from within and outside the gut. Neurogastroenterol Motil 23: 490–501, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Broughton BR, Jernigan NL, Norton CE, Walker BR, Resta TC. Chronic hypoxia augments depolarization-induced Ca2+ sensitization in pulmonary vascular smooth muscle through superoxide-dependent stimulation of RhoA. Am J Physiol Lung Cell Mol Physiol 298: L232–L242, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cheng JC, Cheng HP, Tsai IC, Jiang MJ. ROS-mediated downregulation of MYPT1 in smooth muscle cells: a potential mechanism for the aberrant contractility in atherosclerosis. Lab Invest 93: 422–433, 2013 [DOI] [PubMed] [Google Scholar]
- 9.Choi K, Chen J, Mitra S, Sarna SK. Impaired integrity of DNA after recovery from inflammation causes persistent dysfunction of colonic smooth muscle. Gastroenterology 141: 1293–1301, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Culver PJ, Rattan S. Genesis of anal canal pressures in the opossum. Am J Physiol Gastrointest Liver Physiol 251: G765–G771, 1986 [DOI] [PubMed] [Google Scholar]
- 11.De Godoy MA, Rattan N, Rattan S. Arachidonic acid metabolites follow the preferential course of cyclooxygenase pathway for the basal tone in the internal anal sphincter. Am J Physiol Gastrointest Liver Physiol 296: G727–G734, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Demel SL, Dong H, Swain GM, Wang X, Kreulin DL, Galligan JJ. Antioxidant treatment restores prejunctional regulation of purinergic transmission in mesenteric arteries of deoxycorticosterone acetate-salt hypertensive rats. Neuroscience 168: 335–345, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Firth M, Prather CM. Gastrointestinal motility problems in the elderly patient. Gastroenterology 122: 1688–1700, 2002 [DOI] [PubMed] [Google Scholar]
- 14.Freeman BA, Crapo JD. Biology of disease: free radicals and tissue injury. Lab Invest 47: 412–426, 1982 [PubMed] [Google Scholar]
- 15.Fridovich I. Superoxide radical and superoxide dismutases. Annu Rev Biochem 64: 97–112, 1995 [DOI] [PubMed] [Google Scholar]
- 16.Gemma C, Vila J, Bachstetter A, Bickford PC. Oxidative stress and the aging brain: from theory to prevention. In: Brain Aging, edited by Riddle DR. Boca Raton, FL: CRC, 2007 [PubMed] [Google Scholar]
- 17.Halliwell B, Gutteridge JM, Cross CE. Free radicals, antioxidants, and human disease: where are we now? J Lab Clin Med 119: 598–620, 1992 [PubMed] [Google Scholar]
- 18.Hernandez L, Grasa L, Fagundes DS, Gonzalo MP, Arruebo MP, Plaza MA, Murillo MD. Role of potassium channels in rabbit intestinal motility disorders induced by 2,2′-azobis (2-amidinopropane) dihydrochloride (AAPH). J Physiol Pharmacol 61: 279–286, 2010 [PubMed] [Google Scholar]
- 19.Hillemeier C, Bitar KN, Sohn U, Biancani P. Protein kinase C mediates spontaneous tone in the cat lower esophageal sphincter. J Pharmacol Exp Ther 277: 144–149, 1996 [PubMed] [Google Scholar]
- 20.Jiang F, Zhang Y, Dusting GJ. NADPH oxidase-mediated redox signaling: roles in cellular stress response, stress tolerance, and tissue repair. Pharmacol Rev 63: 218–242, 2011 [DOI] [PubMed] [Google Scholar]
- 21.Jones OM, Moore JA, Brading AF, Mortensen NJ. Botulinum toxin injection inhibits myogenic tone and sympathetic nerve function in the porcine internal anal sphincter. Colorectal Dis 5: 552–557, 2003 [DOI] [PubMed] [Google Scholar]
- 22.Khaikin M, Bashankaev B, Sands D, Weiss EG, Zbar A, Wexner SD. The effect of topical anal captopril on resting anal pressure in healthy volunteers: the first human pilot study. Tech Coloproctol 18: 139–143, 2013 [DOI] [PubMed] [Google Scholar]
- 23.Knock GA, Snetkov VA, Shaifta Y, Connolly M, Drndarski S, Noah A, Pourmahram GE, Becker S, Aaronson PI, Ward JP. Superoxide constricts rat pulmonary arteries via Rho-kinase-mediated Ca2+ sensitization. Free Radic Biol Med 46: 633–642, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Koughnett JA, Wexner SD. Current management of fecal incontinence: choosing amongst treatment options to optimize outcomes. World J Gastroenterol 19: 9216–9230, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kumagai Y, Midorikawa K, Nakai Y, Yoshikawa T, Kushida K, Homma-Takeda S, Shimojo N. Inhibition of nitric oxide formation and superoxide generation during reduction of LY83583 by neuronal nitric oxide synthase. Eur J Pharmacol 360: 213–218, 1998 [DOI] [PubMed] [Google Scholar]
- 26.Lazarescue A, Turnbull GK, Vanner S. Investigating and treating fecal incontinence: when and how. Can J Gastroenterol 23: 301–308, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Macias B, Gomez-Pinilla PJ, Camello-Almaraz C, Pascua P, Tresguerres JA, Camello PJ, Pozo MJ. Aging impairs Ca2+ sensitization pathways in gallbladder smooth muscle. Age 34: 881–893, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Patel CA, Rattan S. Spontaneously tonic smooth muscle has characteristically higher levels of RhoA/ROK compared with the phasic smooth muscle. Am J Physiol Gastrointest Liver Physiol 291: G830–G837, 2006 [DOI] [PubMed] [Google Scholar]
- 29.Patel CA, Rattan S. Cellular regulation of basal tone in internal anal sphincter smooth muscle by RhoA/ROCK. Am J Physiol Gastrointest Liver Physiol 292: G1747–G1756, 2007 [DOI] [PubMed] [Google Scholar]
- 30.Peluso I, Campolongo P, Valeri P, Romanelli L, Palmery M. Intestinal motility disorder induced by free radicals: a new model mimicking oxidative stress in gut. Pharmacol Res 46: 533–538, 2002 [DOI] [PubMed] [Google Scholar]
- 31.Rao SS. Pathophysiology of adult fecal incontinence. Gastroenterology 126: S14–S22, 2004 [DOI] [PubMed] [Google Scholar]
- 32.Rattan S, Benjamin P, Maxwell IV. RhoA/ROCK-kinase: pathophysiologic and therapeutic implications in gastrointestinal smooth muscle tone and relaxation. Gastroenterology 138: 13–18, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rattan S, De Godoy MA, Patel CA. Rho kinase as a novel molecular therapeutic target for hypertensive internal anal sphincter. Gastroenterology 131: 108–116, 2006 [DOI] [PubMed] [Google Scholar]
- 34.Rattan S, Singh J. Basal internal anal sphincter tone, inhibitory neurotransmission, and other factors contributing to the maintenance of high pressures in the anal canal. Neurogastroenterol Motil 23: 3–7, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rattan S, Singh J. RhoA/ROCK pathway is the major molecular determinant of basal tone in intact human internal anal sphincter. Am J Physiol Gastrointest Liver Physiol 302: G664–G675, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Roberts JA, Durnin L, Sharkey KA, Mutafova-Yambolieva VN, Mawe GM. Oxidative stress disrupts purinergic neuromuscular transmission in the inflamed colon. J Physiol 591: 3725–3737, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shi XZ, Lindholm PF, Sarna SK. NF-κB activation by oxidative stress and inflammation suppresses contractility in colonic circular smooth muscle cells. Gastroenterology 124: 1369–1380, 2003 [DOI] [PubMed] [Google Scholar]
- 38.Singh J, Maxwell IV, Rattan S. Immunocytochemical evidence for PDBu-induced activation of RhoA/ROCK in human internal anal sphincter smooth muscle cells. Am J Physiol Gastrointest Liver Physiol 301: G317–G325, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Snetkov VA, Smirnov SV, Kua J, Aaronson PI, Ward JP, Knock GA. Superoxide differentially controls pulmonary and systemic vascular tone through multiple signalling pathways. Cardiovasc Res 89: 214–224, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Somara S, Bashllari D, Gilmont RR, Bitar KN. Real-time dynamic movement of caveolin-1 during smooth muscle contraction of human colon and aged rat colon transfected with caveolin-1 cDNA. Am J Physiol Gastrointest Liver Physiol 300: G1022–G1032, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Somara S, Gilmont RR, Martens JR, Bitar KN. Ectopic expression of caveolin-1 restores physiological contractile response of aged colonic smooth muscle. Am J Physiol Gastrointest Liver Physiol 293: G240–G249, 2007 [DOI] [PubMed] [Google Scholar]
- 42.Speakman CT, Hoyle CH, Kamm MA, Swash M, Henry MM, Nicholls RJ, Chir M, Burnstock G. Abnormal internal anal sphincter fibrosis and elasticity in fecal incontinence. Dis Colon Rectum 38: 407–410, 1995 [DOI] [PubMed] [Google Scholar]
- 43.Strittmatter F, Gratzke C, Weinhold P, Steib CJ, Hartmann AC, Schlenker B, Andersson KE, Hedlund P, Stief CG, Hennenberg M. Thromboxane A2 induces contraction of human prostate smooth muscle by Rho kinase- and calmodulin-dependent mechanisms. Eur J Pharmacol 650: 650–655, 2011 [DOI] [PubMed] [Google Scholar]
- 44.Thannickal VJ, Fanburg BL. Reactive oxygen species in cell signaling. Am J Physiol Lung Cell Mol Physiol 279: L1005–L1028, 2000 [DOI] [PubMed] [Google Scholar]
- 45.Van der Vliet A, Tunistra TJ, Bast A. Modulation of oxidative stress in the gastrointestinal tract and effect on rat intestinal motility. Biochem Pharmacol 38: 2807–2818, 1989 [DOI] [PubMed] [Google Scholar]
- 46.Voukali E, Shotton HR, Lincoln J. Selective responses of myenteric neurons to oxidative stress in diabetic stimuli. Neurogastroenterol Motil 23: 964-e411, 2011 [DOI] [PubMed] [Google Scholar]
- 47.Wade PR. Aging and neural control of GI tract. I. Age-related changes in enteric nervous system. Am J Physiol Gastrointest Liver Physiol 283: G489–G495, 2002 [DOI] [PubMed] [Google Scholar]
- 48.Wald A. Faecal incontinence in the elderly: epidemiology and management. Drugs Aging 22: 131–139, 2005 [DOI] [PubMed] [Google Scholar]
- 49.Wang C, Houghton MJ, Gamage PP, Collins HE, Patel BA, Yeoman MS, Ranson RN, Saffrey MJ. Changes in the innervation of the mouse internal anal sphincter during aging. Neurogastroenterol Motil 25: e469–e477, 2013 [DOI] [PubMed] [Google Scholar]
- 50.Whitehead WE, Borrud L, Goode PS, Meikle S, Mueller ER, Tuteja A, Weidner A, Weinstein M, Ye W. Fecal incontinence in US adults: epidemiology and risk factors. Gastroenterology 137: 512–517, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wiley JW. Aging and neural control of the GI tract. III. Senescent enteric nervous system: lessons from extraintestinal sites and nonmammalian species. Am J Physiol Gastrointest Liver Physiol 283: G1020–G1026, 2002 [DOI] [PubMed] [Google Scholar]
- 52.Winston JH, Li Q, Sarna SK. Paradoxical regulation of ChAT and nNOS expression in animal models of Crohn's colitis and ulcerative colitis. Am J Physiol Gastrointest Liver Physiol 305: G295–G302, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]