Abstract
We studied the combined effect of sesamin (1:1 mixture of sesamin and episesamine) and soybean phospholipid on lipid metabolism in rats. Male rats were fed diets supplemented with 0 or 2 g/kg sesamin, and containing 0 or 50 g/kg soybean phospholipid, for 19 days. Sesamin and soybean phospholipid decreased serum triacylglycerol concentrations and the combination of these compounds further decreased the parameter in an additive fashion. Soybean phospholipid but not sesamin reduced the hepatic concentration of triacylglycerol. The combination failed to cause a strong decrease in hepatic triacylglycerol concentration, presumably due to the up-regulation of Cd36 by sesamin. Combination of sesamin and soybean phospholipid decreased the activity and mRNA levels of hepatic lipogenic enzymes in an additive fashion. Sesamin strongly increased the parameters of hepatic fatty acid oxidation enzymes. Soybean phospholipid increased hepatic activity of 3-hydroxyacyl-CoA dehydrogenase although it failed to affect the activity of other enzymes involved in fatty acid oxidation. Sesamin strongly increased hepatic concentration of carnitine. Sesamin and soybean phospholipid combination further increased this parameter, accompanying a parallel increase in mRNA expression of carnitine transporter. These changes can account for the strong decrease in serum triacylglycerol in rats fed a diet containing both sesamin and soybean phospholipid.
Keywords: sesamin, soybean phospholipid, hepatic lipogenesis, hepatic fatty acid oxidation, carnitine
Introduction
Sesamin is one of the most abundant lignans in sesame seeds and is epimerized during acid-clay bleaching in the oil-refining process to form episesamin;(1) therefore, sesamin preparations obtained as a byproduct of the oil-refining process contain sesamin and episesamin at about an equivalent ratio. It has been demonstrated that this sesamin preparation exerts a serum lipid-lowering effect on experimental animals(2,3) and humans.(4) We previously found that the sesamin preparation profoundly and dose-dependently increased the activity and gene expression of fatty acid oxidation enzymes in rat liver.(2) Also, the sesamin preparation decreased hepatic activity and mRNA levels of enzymes involved in fatty acid synthesis.(3) A later study showed that episesamin is more effective than sesamin in increasing the activity and gene expression of fatty acid oxidation enzymes.(5)
Serum and liver lipid-lowering effects of dietary soybean phospholipid have long been recognized in animal and human studies.(6–10) In relation to this, we previously demonstrated that soybean phospholipid profoundly and dose-dependently decreased the activity and mRNA levels of lipogenic enzymes in rat liver.(7,8,10)
It is apparent that sesamin and soybean phospholipid are dietary factors that profoundly affect hepatic fatty acid metabolism and hence lower the serum lipid level. It is expected that the combination of these compounds in the diet would more profoundly affect hepatic fatty acid metabolism, and hence be effective in reducing lipid levels in serum and tissues and the incidence of atherosclerosis. In these contexts, here we studied the combined effect of sesamin and soybean phospholipid on lipid metabolism in rats.
Materials and Methods
Animals and diets
Male Sprague-Dawley rats obtained from Charles River Japan (Kanagawa, Japan) at 4 weeks of age were housed individually in animal cages in a room with controlled temperature (20–22°C), and lighting (lights on from 07:00 to 19:00), and fed a commercial diet (Type NMF; Oriental Yeast Co., Tokyo, Japan). After 7 days of acclimatization, rats were fed purified experimental diets supplemented in 1 kg with 0 or 2 g sesamin (1:1 mixture of sesamin and episesamin; gift from Takemoto Oil Co., Aichi, Japan), and containing in 1 kg either 31 g soybean oil (Nacalai Tesque, Inc., Kyoto, Japan) or 50 g soybean phospholipid (gift from Taiyo Kagaku Co., Yokkaichi, Japan) for 19 days. These dietary lipids served comparative amounts of fatty acids in diets (30.2 g/kg and 30.3 g/kg, respectively). In addition, all the experimental diets contained 7% coconut oil. The basal composition of the purified experimental diets was (in g/kg): casein, 200; coconut oil, 70; corn starch, 150; cellulose, 20; mineral mixture,(11) 35; vitamin mixture,(11) 10; L-cystine, 3; choline bitartrate, 2.5 and sucrose to 1 kg. Sesamin, soybean oil and soybean phospholipid were added to experimental diets in lieu of sucrose. Phospholipid and fatty acid compositions of soybean phospholipid as well as fatty acid composition of soybean oil are shown in Table 1. Animals had free access to the diets and water during the experimental period. This study was approved by the review board of animal ethics of our university and we followed the university’s guidelines in the care and use of laboratory animals.
Table 1.
Phospholipid and fatty acid compositions of dietary lipids
| Dietary lipids |
||
|---|---|---|
| Soybean oil | Soybean phospholipid | |
| Phosphorous content (µmol/g) | — | 877 |
| Phospholipid classes (mol%) | ||
| Lysophosphatidylcholine | — | 10.8 |
| Phosphatidylcholine | — | 34.2 |
| Phosphatidylinositol | — | 18.1 |
| Phosphatidylethanolamine | — | 37.3 |
| Fatty acid content (mg/g) | 975 | 605 |
| Fatty acid composition (weight%) | ||
| 16:0 | 8.5 | 16.4 |
| 16:1 | 0.1 | 0.1 |
| 18:0 | 3.5 | 3.6 |
| 18:1 | 24.8 | 7.5 |
| 18:2 | 54.2 | 63.4 |
| 18:3 | 5.6 | 6.2 |
| 20:0 | 0.5 | 0.0 |
| 22:0 | 0.2 | 0.1 |
| 24:0 | 0.3 | 0.3 |
Enzyme assays
The activity of lipogenic enzymes was measured spectrophotometrically using 15,000 × g supernatant of the liver homogenate as an enzyme source.(3,12) Activity levels of various hepatic fatty acid oxidation enzymes were measured spectrophotometrically using the whole liver homogenate as an enzyme source. The peroxisomal palmitoyl-CoA oxidation rate and acyl-CoA oxidase activities were measured using palmitoyl-CoA as a substrate. Carnitine acyltransferase activities were measured using octanoy-CoA and palmitoyl-CoA as substrates. We used crotonyl-CoA in assaying enoyl-CoA hydratase. The activity of 3-hydroxyacyl-CoA dehydrogenase was assayed in both reverse and forward reactions. When activity was assayed in the reverse reaction, the rate of acetoacetyl-CoA-dependent oxidation of NADH was taken as the activity of this enzyme. The forward reaction of this enzyme was assayed measuring the rate of increase in the optical density at 303 nm following the addition of dl-3-hydroxybutyric acid or dl-3-hydroxydecanoyl-CoA in the presence of NAD, pyruvate, and lactate dehydrogenase. 3-Keotacyl-CoA thiolase activities were measured using acetoacetyl-CoA as a substrate. The methods used in assaying the activities of various enzymes involved in fatty acid oxidation were all the same as those employed in our previous studies.(12,13)
RNA analysis
RNA in the liver was extracted, and mRNA abundance was analyzed by quantitative real-time PCR, as detailed elsewhere.(14) mRNA abundance was calculated as a ratio to the β-actin level in each cDNA sample and expressed as a fold-change, assigning a value of 1 for rats fed a diet free of sesamin and soybean phospholipid.
Analyses of serum and liver components
Serum triacylglycerol, cholesterol, phospholipid, and glucose concentrations were measured using commercial enzyme kits (Wako Pure Chem., Osaka, Japan). The serum concentration of β-hydroxybutyrate was analyzed by HPLC.(15) Liver triacylglycerol,(16) phospholipid(17) and cholesterol(18) concentrations were determined as described previously. The hepatic concentration of carnitine was analyzed by the method of Pearson et al.(19) Serum and liver concentrations of sesamin and episesamin were analyzed by HPLC as described previously.(20)
Statistical analysis
Microsoft Excel add-in software (Excel Statistics 2010; Social Survey Research Information Co., Tokyo, Japan) was used for statistical analysis. The data were analyzed by two-way ANOVA to establish the effect of soybean phospholipid and sesamin or any interaction between these two factors. When the interaction was significant, the data were reanalyzed by one-way ANOVA and Tukey’s post-hoc test. The data for serum and liver concentrations of sesamin and episesamin were analyzed by Student’s t test. Differences were considered significant when p<0.05.
Results
Animal growth and liver weight
The average daily food intake during the 0–19 day experimental period was the same among four groups of rats (17.2–17.9 g/day). Also, sesamin and phospholipid did not affect the growth of animals during the 19 day feeding period and the body weights at the time of killing were 262 ± 4 g and 261 ± 6 g, and 274 ± 5 g and 270 ± 6 g for rats fed a sesamin-free or sesamin-supplemented (2 g/kg) diet containing 0 or 50 g/kg soybean phospholipid, respectively. Sesamin significantly increased the liver weight of animals but soybean phospholipid decreased this parameter. The values were 5.33 ± 0.12 and 4.94 ± 0.09 g/100 g body weight, and 5.95 ± 0.09 and 5.69 ± 0.17 g/100 g body weight for rats fed a sesamin-free or sesamin-supplemented (2 g/kg) diet containing 0 or 50 g/kg soybean phospholipid, respectively.
Effect of sesamin and soybean phospholipid on hepatic fatty acid synthesis
Both sesamin and soybean phospholipid significantly reduced the activities of lipogenic enzymes except for malic enzyme (Fig. 1). The diet containing 2 g/kg sesamin compared to a diet containing 50 g/kg soybean phospholipid was somewhat competent in reducing the activity levels of fatty acid synthase, ATP-citrate lyase, glucose 6-phosphate dehydrogenase, 6-phosphogluconate dehydrogenase, and pyruvate kinase. The combination of sesamin and soybean phospholipid effectively reduced the activity levels of these enzymes in an additive fashion. Meanwhile, soybean phospholipid but not sesamin significantly reduced the activity of malic enzyme.
Fig. 1.
Effect of sesamin and soybean phospholipid on the activities of lipogenic enzymes in rat liver. Values are the means ± SEM, n = 7. The data were analyzed by two-way ANOVA. When the interaction of two factors (sesamin and soybean phospholipid) was significant, the data were reanalyzed by one-way ANOVA and Tukey’s post-hoc test. Means without a common letter differ (p<0.05). PL, phospholipid; ns, not significant.
Fig. 2 shows mRNA levels of proteins related to lipogenesis. There are two types of acetyl-CoA carboxylase, i.e., alpha and beta. The alpha but not beta form appears to be involved in fatty acid synthesis in cytosol.(21) Mammalian tissues contain 3 distinct isoforms of malic enzyme (malic enzyme 1, 2 and 3). Malic enzyme 1 appears to be involved in the regulation of lipogenesis.(22) There are four isoforms of pyruvate kinase in mammals. L-pyruvate kinase is an enzyme expressed in the liver.(23) Adiponutrin is a protein presumed to be involved in the regulation of lipogenesis.(24) We also analyzed mRNA expressions of enzymes involved in the desaturation of fatty acids, i.e., stearoyl-CoA desaturase 1, and Δ5- and Δ6-desaturases.
Fig. 2.
Effect of sesamin and soybean phospholipid on mRNA levels of lipogenic enzymes, adiponutrin, fatty acid desaturases and SREBP-1c in rat liver. Values are the means ± SEM, n = 7. The data were analyzed by two-way ANOVA. When the interaction of two factors (sesamin and soybean phospholipid) was significant, the data were reanalyzed by one-way ANOVA and Tukey’s post-hoc test. Means without a common letter differ (p<0.05). PL, phospholipid; ns, not significant.
Consistent with the observations made on enzyme activity, both sesamin and soybean phospholipid significantly reduced mRNA levels of acetyl-CoA carboxylase α, fatty acid synthase, ATP-citrate lyase, glucose 6-phosphate dehydrogenase, 6-phosphogluconate dehydrogenase and L-pyruvate kinase. Combination of sesamin and soybean phospholipid further decreased the mRNA levels of these enzymes. The values in rats fed a diet simultaneously containing sesamin and soybean phospholipid were 11–32% of those observed with a diet free of these compounds. Both sesamin and soybean phospholipid also significantly lowered mRNA expression of adiponutrin, spot14 and stearoyl-CoA desaturase 1. Marked changes in mRNA expression of adiponutrin due to feeding with sesamin and soybean phospholipid were observed. The levels in rats fed a diet solely containing either sesamin or soybean phospholipid were 7% and 29%, respectively, of the value observed on the control diet. The value was extremely low with the diet containing both sesamin and soybean phospholipid (0.4% of the value observed on a control diet). Consistent with the observations made on enzyme activity, soybean phospholipid but not sesamin lowered the mRNA expression of malic enzyme 1. The level observed with a diet containing sesamin and soybean phospholipid in combination was the same as the value observed with a diet solely containing soybean phospholipid. The responses to sesamin and soybean phospholipid of mRNA expression of Δ5- and Δ6-desaturases resembled that observed with malic enzyme 1. Sterol regulatory element binding protein-1c (SREBP-1c) is a transcription factor involved in the regulation of the gene expression of many lipogenic enzymes. In spite of the fact that both sesamin and soybean phospholipid were effective in reducing the activity and mRNA levels of many lipogenic enzymes, soybean phospholipid but not sesamin significantly lowered mRNA levels of this transcription factor.
Effect of sesamin and soybean phospholipid on hepatic fatty acid oxidation
Consistent with the observations made in previous studies,(2,5) sesamin significantly increased the activities of many enzymes involved in hepatic fatty acid oxidation (Table 2). Soybean phospholipid did not affect the activity of various enzymes involved in hepatic fatty acid oxidation except 3-hydroxyacyl-CoA dehydrogenase. Both sesamin and soybean phospholipid increased the activity of this enzyme in an additive fashion. This was confirmed in the enzyme activity measured in both reverse and forward reactions using acetoacetyl-CoA substrate for the former and 3-hydroxybutyryl-CoA and 3-hydroxydecanoyl-CoA substrates for the latter, respectively.
Table 2.
Effect of sesamin and soybean phospholipid on the activity and mRNA levels of hepatic fatty acid oxidation enzymes
| Sesamin (g/kg) |
Two-way ANOVA (p value) |
||||||
|---|---|---|---|---|---|---|---|
| 0 |
2 |
Sesamin | Soybean PL | Sesamin × Soybean PL | |||
| Soybean phospholipid (g/kg) | 0 | 50 | 0 | 50 | |||
| Enzyme activity (nmol/min per mg protein) | |||||||
| Peroxisomal palmitoyl-CoA oxidation | 2.17 ± 0.13a | 2.44 ± 0.09a | 7.65 ± 0.53b | 8.80 ± 0.54b | <0.01 | <0.05 | ns |
| Acyl-CoA oxidase | 1.09 ± 0.07 | 1.16 ± 0.07 | 5.11 ± 0.22 | 5.54 ± 0.24 | <0.01 | ns | ns |
| Carnitine acyltransferase | |||||||
| Octanoyl-CoA substrate | 6.62 ± 0.38 | 6.34 ± 0.40 | 22.5 ± 1.9 | 27.2 ± 2.4 | <0.01 | ns | ns |
| Palmitoyl-CoA substrate | 4.80 ± 0.16a | 4.39 ± 0.26a | 10.6 ± 0.2b | 11.7 ± 0.6b | <0.01 | ns | <0.05 |
| Enoyl-CoA hydratase | 4,340 ± 459 | 4,101 ± 218 | 11,536 ± 666 | 11,588 ± 797 | <0.01 | ns | ns |
| 3-Hydroxyacyl-CoA dehydrogenase | |||||||
| Acetoacetyl-CoA substrate | 426 ± 15a | 843 ± 15b | 1,135 ± 58c | 1,834 ± 47d | <0.01 | <0.01 | <0.01 |
| 3-Hydroxybutyryl-CoA substrate | 111 ± 4a | 254 ± 6b | 255 ± 16c | 437 ± 4d | <0.01 | <0.01 | <0.01 |
| 3-Hydroxydecanoyl-CoA substrate | 157 ± 5 | 257 ± 5 | 365 ± 13 | 470 ± 17 | <0.01 | <0.01 | ns |
| 3-Ketoacyl-CoA thiolase | 107 ± 4a | 102 ± 3a | 237 ± 8b | 260 ± 8b | <0.01 | ns | <0.05 |
| mRNA level (fold change) | |||||||
| Peroxisomal enzyme | |||||||
| Carnitine O-octanoyltransferase | 1.00 ± 0.16 | 1.05 ± 0.13 | 6.90 ± 0.60 | 11.5 ± 1.8 | <0.01 | ns | ns |
| Acyl-CoA oxidase 1 | 1.00 ± 0.07 | 0.98 ± 0.07 | 4.61 ± 0.23 | 6.26 ± 0.81 | <0.01 | ns | ns |
| Enoyl-CoA hydratase/3-hydroxyacyl CoA dehydrogenase | 1.00 ± 0.07a | 0.83 ± 0.05a | 18.7 ± 1.8b | 29.0 ± 6.3b | <0.01 | ns | <0.05 |
| Acetyl-CoA acyltransferase 1A | 1.00 ± 0.12 | 0.93 ± 0.14 | 5.41 ± 0.93 | 7.64 ± 1.51 | <0.01 | ns | ns |
| Mitohondrial enzyme | |||||||
| Carnitine palmitoyltransferase 1a | 1.00 ± 0.11 | 0.82 ± 0.15 | 1.58 ± 0.14 | 1.31 ± 0.17 | <0.01 | ns | ns |
| Carnitine palmitoyltransferase 2 | 1.00 ± 0.09 | 0.94 ± 0.06 | 3.34 ± 0.15 | 4.24 ± 0.56 | <0.01 | ns | ns |
| Hydroxyacyl-CoA dehydrogenase/3-ketoacyl-CoA thiolase/enoyl-CoA hydratase, α subunit | 1.00 ± 0.02 | 1.18 ± 0.20 | 2.77 ± 0.19 | 3.16 ± 0.3 | <0.01 | ns | ns |
| Hydroxyacyl-CoA dehydrogenase/3-ketoacyl-CoA thiolase/enoyl-CoA hydratase, β subunit | 1.00 ± 0.13 | 1.00 ± 0.10 | 3.13 ± 0.25 | 3.45 ± 0.33 | <0.01 | ns | ns |
| Hydroxyacyl-CoA dehydrogenase | 1.00 ± 0.17 | 1.13 ± 0.13 | 2.27 ± 0.20 | 2.76 ± 0.42 | <0.01 | ns | ns |
| Acetyl-CoA acyltransferase 2 | 1.00 ± 0.05 | 1.19 ± 0.12 | 3.78 ± 0.27 | 4.66 ± 0.62 | <0.01 | ns | ns |
| Perxisome proliferator activated receptor α | 1.00 ± 0.09 | 1.18 ± 0.16 | 1.51 ± 0.18 | 1.58 ± 0.13 | <0.01 | ns | ns |
| Cd36 | 1.00 ± 0.16a | 0.56 ± 0.05a | 2.89 ± 0.23b | 6.23 ± 2.07b | <0.01 | ns | <0.01 |
Values are the means ± SEM, n = 7. Means in a row with superscripts without a common letter differ (p<0.05). PL, phospholipid; ns, not significant.
Consistent with the observations made on enzyme activities, sesamin increased mRNA levels of many enzymes involved in fatty acid oxidation. In spite of the fact that soybean phospholipid increased the activity of 3-hydroxyacyl-CoA dehydrogenase, this dietary factor was totally irrelevant in affecting mRNA levels of proteins which exhibit 3-hydroxyacyl-CoA dehydrogenase activity (peroxisomal bifuntional enzyme having enoyl-CoA, hydratase/3-hydroxyacyl CoA dehydrogenase activities, subunits of mitochondrial trifuncitonal enzyme having 3-hydroxyacyl-CoA dehydrogenase/3-ketoacyl-CoA thiolase/enoyl-CoA hydratase activities and mitochondrial 3-hydroxyacyl-CoA dehydrogenase. Sesamin caused about 1.5-fold increase in mRNA level of perxisome proliferator activated receptor α (PPARα), a transcription factor involved in the regulation of gene expression of many fatty acid oxidation enzymes. However, soybean phospholipid was totally irrelevant in modulating this parameter. In addition to the mRNA levels of various fatty acid oxidation enzymes and PPARα, we also measured the mRNA level of Cd36 which, mediates the transport of fatty acids from the blood stream into hepatocytes and hence is involved in the regulation of fatty acid metabolism in the liver. Consistent with a previous observation,(25) sesamin significantly increased the mRNA level of this protein but soybean phospholipid did not affect this parameter.
Effect of sesamin and soybean phospholipid on mRNA levels of proteins involved in the regulation of carnitine metabolism in the liver
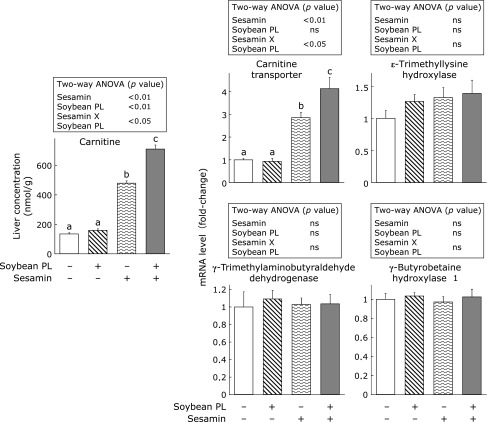
Consistent with previous observations,(25,26) sesamin greatly increased the carnitine concentration and mRNA level of carnitine transporter in the liver (Fig. 3). Soybean phospholipid did not affect these values when the diet was free of sesamin. Interestingly, however, hepatic carnitine concentration was much higher in rats fed a diet containing sesamin and soybean phospholid in combination than in the animals fed a diet solely containing sesamin. This was accompanied by a parallel increase in the mRNA level of carnitine transporter. Changes in the rate of hepatic carnitine synthesis appear to be one factor altering the concentration of carnitine in this tissue.(27,28) We therefore analyzed mRNA levels of hepatic enzymes involved in carnitine biosynthesis (ε-trimethyllysine hydroxylase, γ-trimethylaminobutyraldehyde dehydrogenase and γ-butyrobetaine hydroxylase 1) in the present study. However, sesamin and soybean phospholipid were totally ineffective in altering these values.
Fig. 3.
Effect of sesamin and soybean phospholipid on carnitine concentration, and mRNA levels of carnitine transporter and enzymes involved in carnitine biosynthesis in rat liver. Values are the means ± SEM, n = 7. The data were analyzed by two-way ANOVA. When the interaction of two factors (sesamin and soybean phospholipid) was significant, the data were reanalyzed by one-way ANOVA and Tukey’s post-hoc test. Means without a common letter differ (p<0.05). PL, phospholipid; ns, not significant.
Effect of sesamin and soybean phospholipid on serum and liver lipids
Sesamin and soybean phospholipid significantly lowered serum concentrations of triacylglycerol in an additive fashion, and the level observed with a diet containing both of these dietary factors was the lowest among the groups (Table 3). Sesamin and soybean phospholipid solely added to experimental diets at the levels of 2 g/kg and 50 g/kg, respectively, were equally effective in reducing serum lipid concentrations of cholesterol and phospholipid; however, these dietary factors when added in combination did not cause additive decreases in these parameters. As expected, sesamin significantly increased serum concentrations of β-hydroxybutyrate. Soybean phospholipid when added solely to the diet was in no way effective in increasing this parameter. Interestingly, however, the value became significantly higher in rats given sesamin and soybean phospholipid in combination than in the animals fed a diet containing sesamin alone.
Table 3.
Effect of sesamin and soybean phospholipid on the concentrations of serum and liver lipids and lignans, and serum β-hydroxybutyrate
| Sesamin |
Two-way ANOVA (p value) |
||||||
|---|---|---|---|---|---|---|---|
| 0 |
2 |
Sesamin | Soybean PL | Sesamin × Soybean PL | |||
| Soybean PL (g/kg) | 0 | 50 | 0 | 50 | |||
| Serum components | |||||||
| Triacylglycerol (mmol/L) | 3.47 ± 0.38 | 2.48 ± 0.30 | 1.91 ± 0.21 | 0.99 ± 0.09 | <0.01 | <0.01 | ns |
| Cholesterol (mmol/L) | 2.58 ± 0.09b | 1.96 ± 0.07a | 1.94 ± 0.08a | 1.79 ± 0.09a | <0.01 | <0.01 | <0.01 |
| Phospholipid (mmol/L) | 2.94 ± 0.11b | 2.23 ± 0.13a | 2.35 ± 0.12a | 2.24 ± 0.15a | <0.05 | <0.01 | <0.05 |
| β-Hydroxybutyrate (mmol/L) | 0.108 ± 0.008a | 0.104 ± 0.005a | 0.149 ± 0.007b | 0.186 ± 0.008c | <0.01 | <0.05 | <0.05 |
| Lignan | |||||||
| Sesamin (mmol/L) | — | — | 0.089 ± 0.011 | 0.086 ± 0.005 | |||
| Episesamin (mmol/L) | — | — | 0.716 ± 0.061b | 0.434 ± 0.064a | |||
| Total (mmol/L) | — | — | 0.805 ± 0.072b | 0.521 ± 0.068a | |||
| Liver components | |||||||
| Triacylglycerol (µmol/g) | 65.2 ± 5.4c | 11.3 ± 1.1a | 62.8 ± 10.4c | 29.0 ± 1.6b | <0.01 | <0.01 | <0.01 |
| Cholesterol (µmol/g) | 6.03 ± 0.22c | 4.03 ± 0.12a | 5.07 ± 0.07b | 3.76 ± 0.08a | <0.01 | <0.01 | <0.05 |
| Phospholipid (µmol/g) | 39.1 ± 1.1 | 41.4 ± 1.0 | 48.0 ± 0.9 | 51.8 ± 1.3 | <0.01 | <0.01 | ns |
| Lignan | |||||||
| Sesamin (nmol/g) | — | — | 4.40 ± 0.53 | 3.15 ± 0.40 | |||
| Episesamin (nmol/g) | — | — | 12.9 ± 1.1b | 8.91 ± 0.90a | |||
| Total (nmol/g) | — | — | 26.2 ± 3.1 | 18.0 ± 2.4 | |||
Values are means ± SEM, n = 7. Means in a row with superscripts without a common letter differ (p<0.05). PL, phospholipid; ns, not significant.
Soybean phospholipid solely added to the diet strongly reduced the hepatic triacylglycerol concentration; however, sesamin did not affect this parameter. The value was significantly lower in the animals fed both soybean phospholipid and sesamin than in those fed a diet solely containing sesamin and a diet free of these compounds, but was significantly higher than in those fed a diet solely containing soybean phospholipid. Both soybean phospholipid and sesamin significantly reduced hepatic concentration of cholesterol, the former being more competent than the latter in this respect. The value was significantly lower in rats fed both of these components than in the animals fed a diet only containing sesamin, but was comparable to the value in those fed only containing soybean phospholipid. Both soybean phospholipid and sesamin significantly increased the hepatic concentration of phospholipid, the latter being more competent in this respect.
Sesame lignans were detected in both the serum and liver in rats fed sesamin-containing diets but not in the animals fed sesamin-free diets. Although the sesamin preparation used in this study contained both sesamin and episesamin in equal amounts, episesamin predominated in both the serum and liver. Total lignan and episesamin but not sesamin concentrations in serum were significantly lower in rats fed soybean phospholipid-containing diets than in the animals fed diets free of this compound. Episesamin but not sesamin and total lignin concentrations in liver were significantly lower in the former than in the latter.
Discussion
Combined effect of sesamin and soybean phospholipid on hepatic lipogenesis
Many lipogenic enzymes are under the control of SREBP-1. This transcription factor also regulates the gene expression of enzymes involved in the desaturation of fatty acid (stearoyl-CoA desaturase 1, and Δ5- and Δ6-desaturases).(29,30) It has been reported that the expressions of the genes of malic enzyme 1(31) as well as fatty acid desaturases(29,30) are dually regulated by SREBP-1 and PPARα. Our previous studies showed that both sesamin(3,26) and soybean phospholipid(7,8,10) decreased the activity and mRNA levels of many enzymes involved in hepatic lipogenesis. These previous findings were also confirmed in the present study (Fig. 1 and 2). Also, the experimental diet containing both of these compounds effectively decreased many of these parameters in an additive fashion. However, the responses to sesamin and soybean phospholipid of mRNA levels of malic enzyme 1, and of Δ5- and Δ6-desaturases were considerably different from those of other enzymes (Fig. 2). Sesamin appears to be an agonist of PPARα,(2) and hence is expected to stimulate gene expressions of these enzymes, and at the same time, the lignan should conversely down-regulate their mRNA expression through a SREBP-1-dependent mechanism.(3) These effects will cause only a moderate change in the mRNA expression of these genes, as actually observed in rats fed sesamin in the current study. In spite of the fact that the gene of stearoyl-CoA desaturase 1 is also under the control of both SREBP-1 and PPARα, sesamin and soybean phospholipid are almost comparable in their abilities to reduce mRNA expression of this gene, and a combination of these compounds reduced the parameter in an additive fashion (Fig. 2). Therefore, the sensitivities to SREBP-1 and PPARα of these fatty acid desaturase genes may be mutually different. As expected, soybean phospholipid, unlike sesamin, decreased mRNA expressions not only of the various lipogenic enzymes, including malic enzyme 1, but also various fatty acid desaturases. The combination of this compound with sesamin results in the lowering of the expression of malic enzyme 1 and Δ5- and Δ6-desaturases to the levels observable in rats fed a diet solely containing soybean phospholipid. It was suggested that the combination of sesamin and soybean phospholipid potentiated the down-regulation of the SREBP-1 signaling pathway and overcame the sesamin-mediated stimulation of PPAR action on Δ5- and Δ6-fatty acid desaturase genes and the malic enzyme 1 gene.
It has been well demonstrated that many enzymes involved in lipogenesis are under the control of SREBP-1. It is reasonable to consider that both sesamin and soybean phospholipid decreased hepatic lipogenesis through a SREBP-1-dependent mechanism. However, the mRNA level of SREBP-1c did not parallel those of lipogenic genes (Fig. 2). This is not surprising because the protein levels of the mature active form of SREBP-1 do not necessarily parallel the mRNA levels of this transcription factor.(3) Determination of the protein levels of mature form of SREBP-1 is required to clarify this point.
Combined effect of sesamin and soybean phospholipid on hepatic fatty acid oxidation
In the present study, we confirmed previous findings that sesamin increased the hepatic activity and mRNA levels of various fatty acid oxidation enzymes.(2,25,26) It is reasonable that sesamin is a ligand and activator of PPARα and hence increases hepatic fatty acid oxidation. We observed in the present study that sesamin increased mRNA levels of PPARα (Table 2). Therefore, an increase in gene expression of this transcription factor may also be involved in the sesamin-dependent increase in hepatic fatty acid oxidation. We found that not only sesamin but also soybean phospholipid caused a considerable increase in the activities of 3-hydoxyacyl-CoA dehydrogenase (Table 2). This unexpected finding was confirmed using various substrates. However, the phospholipid was not effective in altering the activities of other enzymes, except for a slight increase in the peroxisomal palmitoyl-CoA oxidation rate. Three enzyme molecules have so far been identified to possess 3-hydoxyacyl-CoA dehydrogenase activities, i.e., peroxisomal bifunctional enzyme having enoyl-CoA hydratase/3-hydroxyacyl CoA dehydrogenase activities (Ehhadh), mitochondrial trifunctional enzyme having 3-hydroxyacyl-CoA dehydrogenase/3-ketoacyl-CoA thiolase/enoyl-CoA hydratase activities (this enzyme is composed of two subunits, Hadha and Hadhb) and mitochondrial monofunctional 3-hydroxyacyl-CoA dehydrogenase (Hadh). Soybean phospholipid, however, did not affect mRNA levels of all these enzymes (Table 2). It is therefore uncertain which enzyme molecule is responsible for the soybean phospholipid-dependent change in 3-hydroxyacyl-CoA dehydrogenase activity. There is a possibility that up-regulation of enzyme molecules that have not hitherto been identified is responsible. Alternatively, soybean phospholipid may stimulate the gene expression of known enzymes at the level of translation without affecting mRNA levels of the enzymes.
In relation to the physiological activity of sesamin and soybean phospholipid affecting hepatic fatty acid oxidation, we confirmed a previous finding that sesamin strongly increased the hepatic concentration of carnitine (Fig. 3).(25,26) Moreover, we found that soybean phospholipid increased hepatic carnitine concentration when it was added to a sesamin-containing diet. As the changes in hepatic carnitine concentration well paralleled those in mRNA levels of carnitine transporter (Fig. 3), alteration in the gene expression of this transporter may be primarily responsible for the changes in hepatic carnitine concentration. mRNA expression of carnitine transporter is under the control of PPARα;(27,28) therefore, it is likely that sesamin activated PPARα and hence up-regulated mRNA expression of the transporter. However, soybean phospholipid-dependent changes in carnitine transporter mRNA levels can not be accounted for by a PPARα-mediated mechanism because soybean phospholipid failed to increase mRNA expressions of various fatty acid oxidation enzymes many of which are targeted by PPARα. The mechanism underlying the up-regulation of mRNA expression of carnintine transporter in rats given a diet containing both sesamin and soybean phospholipid is currently unknown. A diet containing both sesamin and soybean phospholipid compared to a diet solely containing sesamin significantly increased hepatic activity of 3-hydroxyacyl-CoA dehydrogenase and carnitine concentration. Therefore, it is plausible that the former increased β-oxidation in the liver more than the latter. In fact, serum concentration of β-hydroxybutyrate was significantly higher in rats given a diet containing both sesamin and soybean phospholipid than in the animals given a diet solely containing sesamin.
Combined effect of sesamin and soybean phospholipid on serum and hepatic lipid levels
Alterations in hepatic fatty acid synthesis(32) and oxidation(33) modify the availability of fatty acids for the synthesis of triacylglycerol, and in turn alter very-low-density lipoprotein production by the liver; therefore, a change in the rate of these metabolic processes is crucial to determine serum lipid concentrations. In the current study, alterations by dietary sesamin and soybean phospholipid of hepatic fatty acid synthesis and oxidation were accompanied by large decreases in serum triacylglycerol concentration (Table 3). Apparently, these compounds decreased this parameter in an additive fashion, and the level observed in the animals fed a diet containing both of these compounds was very low. However, this additive effect was not observed in the serum concentration of cholesterol and phospholipid. This is not surprising because very-low-density lipoproteins are rich in triacylglycerol, but contain less cholesterol and phospholipid, and these lipids are mainly distributed in low-density and high-density lipoproteins. It is suggested that the combination of sesamin and soybean phospholipid decreased the production of very-low-density lipoproteins by the liver and hence decreased triacylglycerol concentrations in serum, but exerted a different effect on the metabolism of low- and high-density lipoproteins and hence did not decrease concentrations of cholesterol and phospholipid in serum. In relation to this, LeBlanc et al.(34) reported that soybean phospholipid decreased the serum concentration of very-low-density lipoprotein, but increased the value of low-density lipoprotein and did not affect the value of high-density lipoprotein in rats. With regard to the effect of sesame lignan, Ogawa et al.(35) reported that both sesamin and episesamin significantly decreased serum concentrations of very-low-density lipoproteins but increased the values of high-density lipoproteins. These results support the hypothesis that the combination of sesamin and phospholipid profoundly decreases the concentration of very-low-density lipoproteins but rather increases the values of low- and high-density lipoproteins. Analysis of serum lipoprotein composition is required to clarify this point.
A diet solely containing soybean phospholipid profoundly decreased the hepatic concentration of triacylglycerol; however, a diet solely containing sesamin failed to do so (Table 3). The value was lower in animals fed both of these compounds than in animals fed a diet solely containing sesamin and a diet free of sesamin and phospholipid, but was higher than the value in animals fed a diet solely containing soybean phospholipid. In relation to this, sesamin profoundly increased hepatic mRNA expression of Cd36 (Table 2). It has been indicated that Cd36 gene is targeted by both PPARα and PPARγ2.(36,37) Therefore, both PPARα and PPARγ2 agonists increase mRNA expression of Cd36 in tissues.(36) Sesamin appears to be an agonist to activate PPARα.(2) Therefore, it is reasonable that this lignan increases mRNA expression of Cd36 in the liver. Moreover, our recent study(26) raised the possibility that sesamin increases mRNA expression of hepatic Cd36 through up-regulation of gene expression of PPARγ2. Recent studies in mice(14,38,39) indicated that Cd36 plays a crucial role in the transport of fatty acid in hepatocytes and hence regulates hepatic triacylglycerol concentration. Therefore, up-regulation of Cd36 can account for the failure of sesamin to decrease hepatic triacylglycerol, although this compound reduced hepatic lipogenesis and increased fatty acid oxidation.
Phospholipids are crucial in the emulsification of lipophilic compounds. There is the possibility that dietary soybean phospholipid helps emulsification and in turn stimulates intestinal absorption of sesamin, a lipophilic compound. This may contribute to the physiological effects of a diet containing both sesamin and soybean phospholipid on hepatic fatty acid metabolism observed in the present study. The observation that dietary phospholipid rather decreased serum and liver levels of the sesame lignan (Table 3) does not support this consideration.
In conclusion, sesamin and soybean phospholipid effectively decreased the serum triacylglycerol concentration and the combination of these compounds further decreased this parameter in an additive fashion. Strong additive reduction of hepatic lipogenesis may be primarily responsible. In addition, there is the possibility that not only sesamin but also soybean phospholipid increased hepatic fatty acid oxidation. Therefore, the additive increase in hepatic fatty acid oxidation by these dietary factors may also account for the observed change in serum triacylglycerol levels. The situation is considerably different in the liver. Soybean phospholipid but not sesamin strongly decreased hepatic triacylglycerol concentration. The addition of sesamin to a diet containing soybean phospholipid rather increased this parameter. Up-regulation by sesamin of Cd36 may account for this consequence.
Acknowledgments
This study was supported by a grant-in-aid for scientific research (Scientific Research C, No. 22580143) from the Japan Society for the Promotion of Science. Expert technical assistances by Ms. Masako Katsurai, Ms. Natsuki Sato, Ms. Kana Sindo, Ms. Kurumi Masaki, Ms. Kyoko Yamazaki, and Ms. Masumi Yamada are greatly appreciated.
Conflict of Interest
No potential conflicts of interest were disclosed.
References
- 1.Fukuda Y, Nagata M, Osawa T, Namiki M. Contribution of lignan analogues to antioxidative activity of refined unroasted sesame seed oil. J Am Oil Chem Soc. 1986;63:1027–1031. [Google Scholar]
- 2.Ashakumary L, Rouyer IA, Takahashi Y, et al. Sesamin, a sesame lignan, is a potent inducer of hepatic fatty acid oxidation in the rat. Metabolism. 1999;48:1303–1313. doi: 10.1016/s0026-0495(99)90272-x. [DOI] [PubMed] [Google Scholar]
- 3.Ide T, Ashakumary L, Takahashi Y, Kushiro M, Fukuda N, Sugano M. Sesamin, a sesame lignan, decreases fatty acid synthesis in rat liver accompanying the down-regulation of sterol regulatory element binding protein-1. Biochim Biophys Acta. 2001;1534:1–13. doi: 10.1016/s1388-1981(01)00167-6. [DOI] [PubMed] [Google Scholar]
- 4.Hirata F, Fujita K, Ishikura Y, Hosoda K, Ishikawa T, Nakamura H. Hypocholesterolemic effect of sesame lignan in humans. Atherosclerosis. 1996;122:135–136. doi: 10.1016/0021-9150(95)05769-2. [DOI] [PubMed] [Google Scholar]
- 5.Kushiro M, Masaoka T, Hageshita S, Takahashi Y, Ide T, Sugano M. Comparative effect of sesamin and episesamin on the activity and gene expression of enzymes in fatty acid oxidation and synthesis in rat liver. J Nutr Biochem. 2002;13:289–295. doi: 10.1016/s0955-2863(01)00224-8. [DOI] [PubMed] [Google Scholar]
- 6.Cohn JS, Wat E, Kamili A, Tandy S. Dietary phospholipids, hepatic lipid metabolism and cardiovascular disease. Curr Opin Lipidol. 2008;19:257–262. doi: 10.1097/MOL.0b013e3282ffaf96. [DOI] [PubMed] [Google Scholar]
- 7.Ide T, Hirabayashi S, Kano S, Sugano M. Soybean phospholipid dependent reductions in triacylglycerol concentration and synthesis in the liver of fasted-refed rats. Biochim Biophys Acta. 1992;1124:163–170. doi: 10.1016/0005-2760(92)90093-b. [DOI] [PubMed] [Google Scholar]
- 8.Ide T, Murata M. Depressions by dietary phospholipids of soybean and egg yolk origins of hepatic triacylglycerol and fatty acid synthesis in fasted-refed rats. Ann Nutr Metab. 1994;38:340–348. doi: 10.1159/000177832. [DOI] [PubMed] [Google Scholar]
- 9.Mori T, Kondo H, Hase T, Murase T. Dietary phospholipids ameliorate fructose-induced hepatic lipid and metabolic abnormalities in rats. J Nutr. 2011;141:2003–2009. doi: 10.3945/jn.111.143602. [DOI] [PubMed] [Google Scholar]
- 10.Rouyer IA, Takahashi Y, Ide T. Dietary phospholipid-dependent reductions in gene expression and activity of liver enzymes in fatty acid synthesis in fasted-refed rats. J Nutr Sci Vitaminol (Tokyo) 1999;45:287–302. doi: 10.3177/jnsv.45.287. [DOI] [PubMed] [Google Scholar]
- 11.Reeves PG, Nielsen FH, Fahey GC., Jr AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr. 1993;123:1939–1951. doi: 10.1093/jn/123.11.1939. [DOI] [PubMed] [Google Scholar]
- 12.Ide T, Takahashi Y, Kushiro M, Tachibana M, Matsushima Y. Effect of n-3 fatty acids on serum lipid levels and hepatic fatty acid metabolism in BALB/c.KOR-Apoeshl mice deficient in apolipoprotein E expression. J Nutr Biochem. 2004;15:169–178. doi: 10.1016/j.jnutbio.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 13.Ide T, Kobayashi H, Ashakumary L, et al. Comparative effects of perilla and fish oils on the activity and gene expression of fatty acid oxidation enzymes in rat liver. Biochim Biophys Acta. 2000;1485:23–35. doi: 10.1016/s1388-1981(00)00026-3. [DOI] [PubMed] [Google Scholar]
- 14.Ide T. Interaction of fish oil and conjugated linoleic acid in affecting hepatic activity of lipogenic enzymes and gene expression in liver and adipose tissue. Diabetes. 2005;54:412–423. doi: 10.2337/diabetes.54.2.412. [DOI] [PubMed] [Google Scholar]
- 15.Ide T. Enzymatic-HPLC method to analyze D-3-hydroxybutyric acid in rat serum. Biosci Biotechnol Biochem. 2010;74:1578–1582. doi: 10.1271/bbb.100167. [DOI] [PubMed] [Google Scholar]
- 16.Fletcher MJ. A colorimetric method for estimating serum triglycerides. Clin Chim Acta. 1968;22:393–397. doi: 10.1016/0009-8981(68)90041-7. [DOI] [PubMed] [Google Scholar]
- 17.Rouser G, Fkeischer S, Yamamoto A. Two dimensional then layer chromatographic separation of polar lipids and determination of phospholipids by phosphorus analysis of spots. Lipids. 1970;5:494–496. doi: 10.1007/BF02531316. [DOI] [PubMed] [Google Scholar]
- 18.Ide T, Oku H, Sugano M. Reciprocal responses to clofibrate in ketogenesis and triglyceride and cholesterol secretion in isolated rat liver. Metabolism. 1982;31:1065–1072. doi: 10.1016/0026-0495(82)90153-6. [DOI] [PubMed] [Google Scholar]
- 19.Pearson DJ, Chase JFA, Tubbs PK. The assay of (–)-carnitine and its O-acyl derivatives. Methods Enzymol. 1969;14:612–622. [Google Scholar]
- 20.Kushiro M, Takahashi Y, Ide T. Species differences in the physiological activity of dietary lignan (sesamin and episesamin) in affecting hepatic fatty acid metabolism. Br J Nutr. 2004;91:377–386. doi: 10.1079/BJN20031067. [DOI] [PubMed] [Google Scholar]
- 21.Abu-Elheiga L, Matzuk MM, Abo-Hashema KA, Wakil SJ. Continuous fatty acid oxidation and reduced fat storage in mice lacking acetyl-CoA carboxylase 2. Science. 2001;291:2613–2616. doi: 10.1126/science.1056843. [DOI] [PubMed] [Google Scholar]
- 22.Loeber G, Infante AA, Maurer-Fogy I, Krystek E, Dworkin MB. Human NAD(+)-dependent mitochondrial malic enzyme. cDNA cloning, primary structure, and expression in Escherichia coli. J Biol Chem. 1991;266:3016–3021. [PubMed] [Google Scholar]
- 23.Noguchi T, Iritani N, Tanaka T. Molecular mechanism of induction of key enzymes related to lipogenesis. Proc Soc Exp Biol Med. 1992;200:206–209. doi: 10.3181/00379727-200-43419. [DOI] [PubMed] [Google Scholar]
- 24.Jenkins CM, Mancuso DJ, Yan W, Sims HF, Gibson B, Gross RW. Identification, cloning, expression, and purification of three novel human calcium-independent phospholipase A2 family members possessing triacylglycerol lipase and acylglycerol transacylase activities. J Biol Chem. 2004;279:48968–48975. doi: 10.1074/jbc.M407841200. [DOI] [PubMed] [Google Scholar]
- 25.Ide T, Lim JS, Odbayar TO, Nakashima Y. Comparative study of sesame lignans (sesamin, episesamin and sesamolin) affecting gene expression profile and fatty acid oxidation in rat liver. J Nutr Sci Vitaminol (Tokyo) 2009;55:31–43. doi: 10.3177/jnsv.55.31. [DOI] [PubMed] [Google Scholar]
- 26.Ide T, Azechi A, Kitade S, Kunimatsu Y, Suzuki N, Nakajima C. Combined effect of sesamin and α-lipoic acid on hepatic fatty acid metabolism in rats. Eur J Nutr. 2013;52:1015–1027. doi: 10.1007/s00394-012-0408-3. [DOI] [PubMed] [Google Scholar]
- 27.Koch A, König B, Stangl GI, Eder K. PPARα mediates transcriptional upregulation of novel organic cation transporters-2 and -3 and enzymes involved in hepatic carnitine synthesis. Exp Biol Med (Maywood) 2008;233:356–365. doi: 10.3181/0706-RM-168. [DOI] [PubMed] [Google Scholar]
- 28.van Vlies N, Ferdinandusse S, Turkenburg M, Wanders RJ, Vaz FM. PPARα-activation results in enhanced carnitine biosynthesis and OCTN2-mediated hepatic carnitine accumulation. Biochim Biophys Acta. 2007;1767:1134–1142. doi: 10.1016/j.bbabio.2007.07.001. [DOI] [PubMed] [Google Scholar]
- 29.Matsuzaka T, Shimano H, Yahagi N, et al. Dual regulation of mouse Δ5- and Δ6-desaturase gene expression by SREBP-1 and PPARα. J Lipid Res. 2002;43:107–114. [PubMed] [Google Scholar]
- 30.Paton CM, Ntambi JM. Biochemical and physiological function of stearoyl-CoA desaturase. Am J Physiol Endocrinol Metab. 2009;297:E28–E37. doi: 10.1152/ajpendo.90897.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Castelein H, Gulick T, Declercq PE, Mannaerts GP, Moore DD, Baes MI. The peroxisome proliferator activated receptor regulates malic enzyme gene expression. J Biol Chem. 1994;269:26754–26758. [PubMed] [Google Scholar]
- 32.Windmueller HG, Spaeth AE. De novo synthesis of fatty acid in perfused rat liver as a determinant of plasma lipoprotein production. Arch Biochem Biophys. 1967;122:362–369. doi: 10.1016/0003-9861(67)90206-8. [DOI] [PubMed] [Google Scholar]
- 33.Ide T, Ontko JA. Increased secretion of very low density lipoprotein triglyceride following inhibition of long chain fatty acid oxidation in isolated rat liver. J Biol Chem. 1981;256:10247–10255. [PubMed] [Google Scholar]
- 34.LeBlanc MJ, Brunet S, Bouchard G, et al. Effects of dietary soybean lecithin on plasma lipid transport and hepatic cholesterol metabolism in rats. J Nutr Biochem. 2003;14:40–48. doi: 10.1016/s0955-2863(02)00253-x. [DOI] [PubMed] [Google Scholar]
- 35.Ogawa H, Sasagawa S, Murakami T, Yoshizumi H, Shimizu S, Yamada H. Effects of sesamin on serum lipoprotein metabolism in normocholesterolemic and hypercholesterolemic stroke-prone SHR. Ann N Y Acad Sci. 1993;676:338–339. doi: 10.1111/j.1749-6632.1993.tb38748.x. [DOI] [PubMed] [Google Scholar]
- 36.Motojima K, Passilly P, Peters JM, Gonzalez FJ, Latruffe N. Expression of putative fatty acid transporter genes are regulated by peroxisome proliferator-activated receptor alpha and gamma activators in a tissue- and inducer-specific manner. J Biol Chem. 1998;273:16710–16714. doi: 10.1074/jbc.273.27.16710. [DOI] [PubMed] [Google Scholar]
- 37.Sato O, Kuriki C, Fukui Y, Motojima K. Dual promoter structure of mouse and human fatty acid translocase/CD36 genes and unique transcriptional activation by peroxisome proliferator-activated receptor alpha and gamma ligands. J Biol Chem. 2002;277:15703–15711. doi: 10.1074/jbc.M110158200. [DOI] [PubMed] [Google Scholar]
- 38.Degrace P, Moindrot B, Mohamed I, et al. Upregulation of liver VLDL receptor and FAT/CD36 expression in LDLR-/- apoB100/100 mice fed trans-10,cis-12 conjugated linoleic acid. J Lipid Res. 2006;47:2647–2655. doi: 10.1194/jlr.M600140-JLR200. [DOI] [PubMed] [Google Scholar]
- 39.Yamazaki T, Nakamori A, Sasaki E, Wada S, Ezaki O. Fish oil prevents sucrose-induced fatty liver but exacerbates high-safflower oil-induced fatty liver in ddy mice. Hepatology. 2007;46:1779–1790. doi: 10.1002/hep.21934. [DOI] [PubMed] [Google Scholar]