Abstract
Purpose
This study was conducted to compare 18F-FDG PET/CT and electromyography (EMG) mapping in patients with primary cervical dystonia (PCD) to find dystonic superficial cervical muscles.
Methods
Ten consecutive patients with PCD (M:F = 5:5, age 44 ± 13 years) whose dystonic posture was not relieved with conventional muscle relaxant therapy were included. Target cervical muscles for the comparison between 18F-FDG PET/CT and EMG mapping were four representative superficial bilateral cervical muscles: splenius capitis muscle, sternocleidomstoid muscle, upper trapezius muscle, and leavator scapulae muscle. The diagnostic efficacy was compared between 18F-FDG PET/CT and EMG mapping using physical exam and measurement of rotation angle as the gold standard.
Results
Among 80 muscles evaluated, there were 21 (26%) dystonic superficial cervical muscles assessed with physical exam and motion analysis. The sensitivity, specificity, and accuracy for localizing dystonic muscles were 76, 92, and 88% for 18F-FDG PET/CT, and 95, 66, and 74% for EMG mapping, respectively. The sensitivity of EMG mapping was significantly higher than that of 18F-FDG PET/CT. In contrast, 18F-FDG PET/CT was significantly superior to EMG mapping for specificity and accuracy.
Conclusions
18F-FDG PET/CT is more specific and accurate than EMG mapping for finding superficial dystonic cervical muscles. The high sensitivity of EMG mapping suggests that 18F-FDG PET/CT and EMG mapping are complementary for finding dystonic superficial cervical muscles.
Keywords: Primary cervical dystonia, Spasmodic torticollis, 18F-FDG, PET/CT, Electromyography
Introduction
Primary (or idiopathic) cervical dystonia (PCD), sometimes referred to as spasmodic torticollis, is characterized by an abnormal head and neck posture due to involuntary tonic or phasic contractions of the neck muscles [1]. Various options are available for treating patients with PCD, and although no universally accepted treatment protocol exists, chemodenervation with botulinum toxin (BT) is regarded as a first-line treatment for PCD [1, 2]. However, 10–30% of patients do not respond to BT injection [2–4]. The poor localization of target muscles is a major cause of treatment failure [1, 5]. Once it is determined that dystonia is present, history, physical examination and, if necessary, laboratory tests must exclude secondary dystonia [1]. However, due to the lack of a specific diagnostic test, neurophysiologic techniques such as surface electromyography are recommended to document functional abnormalities in patients with dystonia and assist in differential diagnosis, evaluation of the pathophysiology, and directing treatment with botulinum toxin injections [6]. Mapping based on needle electromyography (EMG) is the most widely used method for identifying dystonic muscles in PCD. Needle EMG can detect the abnormal firing of the motor unit action potentials (MUAP) of dystonically contracting muscles [6, 7]. However, deep cervical muscles may be beyond reach with a conventional 37-mm needle electrode for EMG [8]. Also, the use of motion analysis systems has allowed the extraction of kinematics parameters in a quantitative and reliable way [9–12].
Application of PET images to evaluate muscular metabolic activity has been reported to be a useful strategy [13–16]. A recent study showed 18F-fluorodeoxyglucose (FDG) PET/CT could be effectively applied to localize dystonic muscles in PCD patients [17], however, 18F-FDG PET/CT was not compared with the EMG mapping data in this study. Comparison of 18F-FDG PET/CT with EMG mapping to localize dystonic superficial cervical muscles is needed. It is well-known that glucose metabolism and 18F-FDG uptake are enhanced in contracting skeletal muscles [13–15], and thus, it has been suggested that the degree of 18F-FDG uptake might be associated with the contraction strength of skeletal muscles [13–15]. Moreover, a recently developed integrated PET/CT method was found to provide both metabolic and anatomic information on hypermetabolic lesions. A recent report suggested that 18F-FDG PET/CT was potentially useful for identifying dystonic cervical muscles for BT therapy in patients with PCD [17]. However, there was no direct comparison between 18F-FDG PET/CT and EMG for localizing PCD to find dystonic superficial cervical muscles.
Materials and Methods
Subjects
The inclusion criteria used for this study were as follows: (1) All subjects had PCD without any evidence of an identifiable cause of secondary dystonia. (2) Extra-nuchal area was not affected by the dystonia. (3) Dystonic posture was not relieved despite conventional muscle relaxant therapy consisting of anticholinergics (trihexyphenidyl), benzodiazepines (clonazepam), and baclofen. Ten consecutive patients with PCD who visited our hospital met the study criteria and were included in this study. The ethics review committee at our institute approved the study protocol.
Determination of Dystonic Superficial Cervical Muscles
Target cervical muscles for the comparison between 18F-FDG PET/CT and EMG mapping were four representative superficial bilateral cervical muscles: splenius capitis muscle (SPC), sternocleidomstoid muscle (SCM), upper trapezius muscle (uTz), and leavator scapulae muscle (LS). The dystonic activities in these four superficial muscles could be objectively determined by physical exam and measurement of rotation angle, which were used as the gold standard in this study because they are considered the current best practical diagnostic tests to evaluate superficial cervical muscles in PCD [1, 8–12].
Physical examinations were performed by a single investigator. Bilateral SPC, SCM, uTz, and LS muscles were inspected and palpated. Hypertrophic muscles or muscles with visible or palpable contraction on inspection and palpation were defined as dystonic. Maximum rotation angles of dystonic postures were determined using a motion analysis system (Vicon 612 motion analysis system, Oxford Metrics, Oxford, UK). Patients sat on a chair, without a backrest, while clinical scores and rotation angles were determined. Patients were instructed not to resist abnormal head and neck postures. Rotation angles of dystonic posture were measured two dimensionally using six light-reflecting markers attached to the anterior/posterior and left/right sides of a helmet and to both acromioclavicular joints. A motion analysis system detected the positions of the markers while seated patients wore the helmet. Rotation angle was defined as the angle between the line connecting markers on bilateral acromioclavicular joints and the line connecting the anterior and posterior helmet markers (Fig. 1). Dystonic muscle was determined based on the results of both physical exam and measurement of rotation angle.
Fig. 1.
Measurement of rotation angle in dystonic posture. Six light-reflecting markers were located on the anterior/posterior and left/right sides of a helmet and on both acromioclavicular joints. The rotation angle was defined as the angle between the line connecting markers on bilateral acromioclavicular joints and the line connecting the anterior and posterior helmet markers
18F-FDG PET/CT
All patients were fasted for at least 6 h prior to the PET/CT study but were allowed water and muscle relaxant medications. To prevent nonspecific 18F-FDG uptake by muscles not related to PCD, muscle relaxants consisting of anticholinergics (trihexyphenidyl), benzodiazepines (clonazepam), and baclofen were administrated to all patients before PET/CT. PET/CT scans were performed using a Discovery LS PET/CT scanner (GE Healthcare, Milwaukee, WI). CT scanning from head to upper chest was performed using a continuous spiral technique on an 8-slice helical CT with 80 mAs, 140 KeV, at a 5 mm section width, and at a table feed rate of 5 mm per rotation. An emission scan was performed after CT scans from head to upper chest at 4 min per frame, 45 min after the administration of 370 MBq 18F-FDG i.v. Patients maintained a sitting position with head and neck in the adopted abnormal involuntary posture during the 18F-FDG uptake period without making any effort to correct this posture [17]. Attenuation-corrected PET images using CT data were reconstructed using an ordered subsets expectation maximization algorithm (28 subsets, two iterations). Using an Xeleris workstation, CT and PET scan data were accurately co-registered. Standardized uptake values (SUVs) were acquired using attenuation-corrected images, and the amount of 18F-FDG injected, patient body weight, and cross-calibration factors between PET and the dose calibrator were recorded. CT, PET, fused PET/CT, and maximum-intensity-projection (MIP) PET images were reviewed by one nuclear medicine physician. When increased 18F-FDG uptake was observed in bilateral SPC, SCM, uTz, and LS muscles, maximum SUV and the name of each hypermetabolic muscle were recorded.
EMG Mapping
The SPC, SCM, uTz, and LS muscles were studied bilaterally using a monopolar needle electrode (37 mm long, 27 gauge) and an EMG system (band pass 20–10,000 Hz, gain 200 µV/div, sweep speed 1 s/div) (Synergy, Oxford Instruments, Oxfordshire, UK). EMG electrode placements were made using standard anatomic landmarks. SCM muscle recordings were made at mid-belly of visible muscle. SPC recordings were made with the needle inserted 1–2 cm inferior and posterior to the SPC attachment to the mastoid process. LS recordings were made at mid-belly of the muscle after palpation during ipsilateral scapular elevation. To record the uTz muscle, the electrode was introduced at the front edge of the trapezius’s horizontal portion 2 cm removed from the medial angle of the shoulder. The electrode positions were tested by maximum innervation during rotation, flexion, extension, lateral flexion, and unilateral scapular elevation. No other cervical muscles were explored because it was difficult to place the needle tip accurately into targeted muscles. A single test was defined as a continuous 5 s monitoring period, and five tests were conducted on each muscle. A “turn” was defined as a reversal of slope separated from the previous reversal and the following reversal by an amplitude change of > 100 µV. The number of turns per second over the 5 s test periods was determined for each muscle studied, and a muscle was defined as displaying dystonic activity if the average number of turns per second exceeded 100 (Fig. 2). [7, 18–22]. EMG mapping was either carried out after 18F-FDG PET/CT scanning on the same day (eight patients) or within 4 days (two patients).
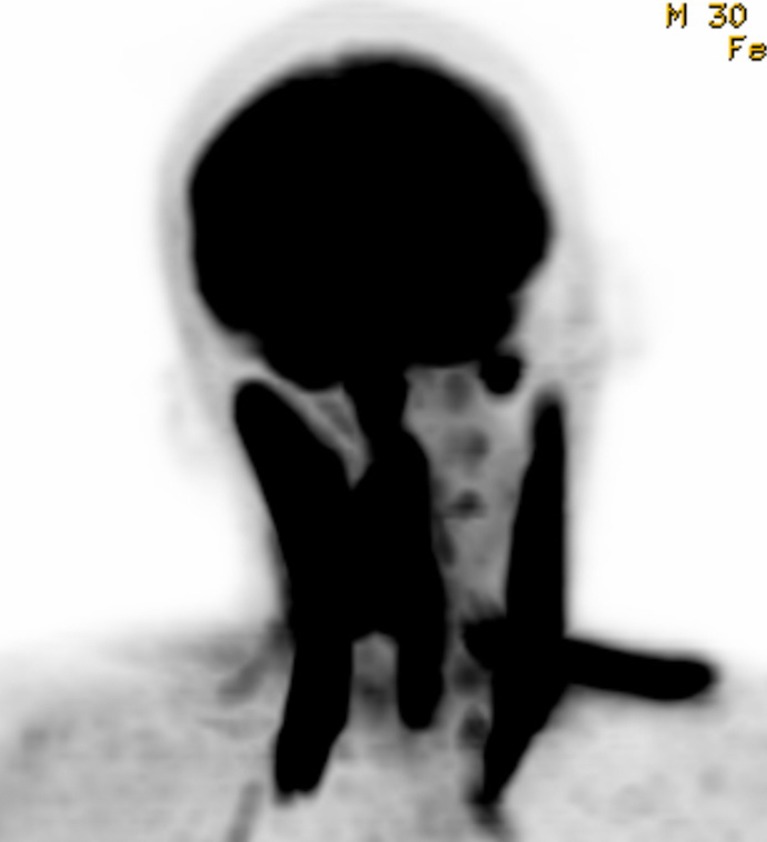
Fig. 2.
Maximum intensity projection volume-rendering PET image of a 30-year-old male patient with right PCD. Hypermetabolism was found in right splenius capitis muscle, right levator scapulae muscle, left trapezius muscle, and bilateral sternocleidomastoid muscles. All hypermetabolic muscles were dystonic based on physical exam and measurement of rotation angle
Statistical Analysis
To compare diagnostic efficacy between 18F-FDG PET/CT and EMG mapping, McNemar test was used. P < 0.05 was considered statistically significant.
Results
Clinical characteristics of patients are summarized in Table 1. Half of subjects were male. Mean age was 44 ± 13 years (range: 22–64 years). Among 80 muscles evaluated, there were 21 (26%) dystonic superficial cervical muscles assessed with physical exam and motion analysis (Table 2). Based on EMG mapping (Table 3), there were 40 dystonic muscles (50%). In contrast, there were 22 hypermetabolic muscles (28%) on 18F-FDG PET/CT (Table 4). Most commonly involved muscles were the ipsilateral splenius capitis and contralateral sternocleidomastoird muscles both on EMG and 18F-FDG PET/CT. False positive results of EMG analysis occurred most frequently in ipsilateral levator scapulae (25%) and contralateral upper trapezius (25%) muscles. Three out of five false positive results on 18F-FDG PET/CT occurred in the contralateral upper trapezius muscle. The sensitivity, specificity, and accuracy were 76, 92, and 88% for 18F-FDG PET/CT and 95, 66, and 74% for EMG mapping, respectively (Table 2). The sensitivity of EMG mapping was significantly higher than that of 18F-FDG PET/CT. In contrast, 18F-FDG PET/CT was superior to EMG mapping in terms of specificity and accuracy. Maximum SUVs in dystonic muscles as determined by 18F-FDG PET/CT fell in the range of 1.5 to 19.4.
Table 1.
Clinical characteristics of patients
| Characteristics | Values |
|---|---|
| Male/female | 5/5 |
| Age (years) | 44 ± 13 (22–64) |
| Age of onset (years) | 43 ± 12 (22–62) |
| Duration of dystonia (min) | 13 ± 17 (2–60) |
| Direction of rotation (right/left) | 7/3 |
Values are numbers or mean ± SD (range)
Table 2.
Result of electromyography mapping of each patient
| Patients (sex/age) | 1 (F/46) | 2 (F/22) | 3 (M/43) | 4 (M35) | 5 (M/30) | 6 (F/51) | 7 (M/63) | 8 (M/64) | 9 (F/42) | 10 (F48) |
|---|---|---|---|---|---|---|---|---|---|---|
| Ipsilaterala | ||||||||||
| SPC | Pc | P | P | P | P | P | P | P | P | P |
| SCM | N | N | N | P | P | N | N | N | N | N |
| LS | P | N | P | N | P | N | P | P | P | P |
| uTz | N | N | N | P | P | N | N | N | N | N |
| Contralateral | ||||||||||
| SPC | N | P | N | N | P | N | N | N | P | P |
| SCM | N | P | P | N | P | P | P | P | P | P |
| LS | N | P | N | N | P | N | N | N | P | P |
| uTz | P | P | N | P | P | P | N | P | N | N |
SPC Splenius capitis muscle, SCM sternocleidomastoid muscle, LS levator scapulae muscle, uTz upper trapezius muscle, P EMG positive dystonic muscle, N EMG negative nondystonic muscle
aThe side where the head is rotated
Table 3.
Standardized uptake values of each hypermetabolic dystonic muscle measured on 18F-FDG PET/CT
| Patient no. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Ipsilaterala | ||||||||||
| SPC | 5.7 | 6.8 | 4 | 3.4 | 19.4 | N | 3.6 | 2.7 | 6.6 | 4.6 |
| SCM | N | N | N | 4.4 | 6.4 | N | N | N | N | N |
| LS | 3.2 | N | N | N | 11.2 | N | N | N | N | N |
| uTz | 1.5 | N | N | 2.1 | N | N | N | N | N | N |
| Contralateral | ||||||||||
| SPC | N | N | N | N | N | N | N | N | N | N |
| SCM | N | N | N | N | 16.3 | 1.9 | 4.6 | N | 4.8 | 9 |
| LS | N | N | N | N | N | N | N | N | N | N |
| uTz | N | 1.6 | N | N | 10.9 | N | N | N | 5.6 | 1.6 |
SPC Splenius capitis muscle, SCM sternocleidomastoid muscle, LS levator scapulae muscle, uTz upper trapezius muscle, N18F-FDG PET/CT negative nondystonic muscle
aThe side where the head is rotated
Table 4.
Diagnostic efficacy for detecting of dystonic superficial cervical muscles between EMG mapping and 18F-FDG PET/CT
| Dystonic muscles (n) | 18F-FDG PET/CT | EMG mapping | |||||
|---|---|---|---|---|---|---|---|
| Sensitivity | Specificity | Accuracy | Sensitivity | Specificity | Accuracy | ||
| Ipsilaterala | |||||||
| SPC | 9 | 89% (8/9) | 100% (1/1) | 90% (9/10) | 100% (9/9) | 0% (0/1) | 90% (9/10) |
| SCM | 1 | 100% (1/1) | 89% (8/9) | 90% (9/10) | 100% (1/1) | 89% (8/9) | 90% (9/10) |
| LS | 2 | 100% (2/2) | 100% (8/8) | 100% (10/10) | 100% (2/2) | 38% (3/8) | 50% (5/10) |
| uTz | 0 | NA | 100% (10/10) | 100% (10/10) | NA | 100% (10/10) | 100% (10/10) |
| Contralateral | |||||||
| SPC | 0 | NA | 100% (10/10) | 100% (10/10) | NA | 60% (6/10) | 60% (6/10) |
| SCM | 8 | 50% (4/8) | 50% (1/2) | 50% (5/10) | 88% (7/8) | 50% (1/2) | 80% (8/10) |
| LS | 0 | NA | 100% (10/10) | 100% (10/10) | NA | 70% (7/10) | 70% (7/10) |
| uTz | 1 | 100% (1/1) | 60% (6/9) | 70% (7/10) | 100% (1/1) | 44% (4/9) | 50% (5/10) |
| Overall | 21 | 76% (16/21)* | 92% (54/59)* | 88% (70/80)* | 95% (20/21)* | 66% (39/59)* | 74% (59/80)* |
SPC Splenius capitis muscle, SCM sternocleidomastoid muscle, LS levator scapulae muscle, uTz upper trapezius muscle
*P < 0.05
aThe side where the head is rotated
Discussion
There are reliable clinical rating scale systems such as the Toronto Western Spasmodic Torticolis Rating Scale (TWSTRS). However, they may not be discriminating enough to estimate significant changes in cervical dystonia patients [12, 23]. As the availability of reasonably effective symptomatic treatment such as botulinum toxin injection has stimulated the development of various approaches to select candidate muscles, electrophysiologic and goniometric analysis has gained popularity. Although mapping based on needle electromyography (EMG) is the most widely used method for identifying dystonic muscles in PCD, the poor localization of target muscles is a major cause of treatment failure [1, 5]. Application of PET images to evaluate muscular metabolic activity has been reported to be a useful strategy [13–16]. A recent study showed 18F-FDG PET/CT could be effectively applied to localize dystonic muscles in PCD patients [17], however, 18F-FDG PET/CT was not compared with the EMG mapping data in this study.
In the present study, we compared 18F-FDG PET/CT with EMG mapping for the purpose of localizing dystonic superficial cervical muscles. 18F-FDG PET/CT showed higher specificity and accuracy than EMG mapping. High specificity is important to find dystonic cervical muscles because the permitted dose of BT injection is limited and BT is an expensive drug. The sensitivity was higher in EMG mapping than the 18F-FDG PET/CT. The specificity of EMG analysis can be compromised by intermittent symptoms, dystonia involving deep muscles that were not recorded by the surface electromyogram, or difficulties in reproducing the dystonic symptoms during the examination [24]. This relative lower specificity of EMG mapping suggests that the electrical abnormality of cervical muscles is not always correlated with dystonic muscles causing dystonic posture. In contrast, metabolic abnormality may be more correlated with clinically significant dystonic superficial muscles causing dystonic posture. This result suggests 18F-FDG/PET and EMG mapping can be reliably adopted as complementary tools for the detection of dystonic cervical muscles in PCD patients and as aids to establish an effective therapeutic plan. In this study, all patients underwent PET/CT while maintaining a sitting position during 18F-FDG uptake; this was shown to be the optimum posture for improving the sensitivity of PET/CT [17]. Also, all measurements were performed before BT therapy, which excludes the possible influence of muscle denervation owing to prior BT therapy.
This study had several limitations. First, the small number of subjects was a major limitation. Second, a comparison of the responses to BT therapy was not included. The response prediction for BT therapy is clinically more important. The detection of dystonic muscles in PCD patients with 18F-FDG PET/CT before and after therapy needs to be compared with EMG mapping, and feasibility as a therapy monitoring tool should also be determined. Therefore, this study presents preliminary results. Currently, we are undergoing clinical trials on a larger number of patients to substantiate the results of the present study, and we are including BT response data.
In conclusion, 18F-FDG PET/CT is more specific and accurate than EMG mapping for finding superficial dystonic cervical muscles. High sensitivity of EMG mapping suggests that 18F-FDG PET/CT and EMG mapping complement each other for finding dystonic superficial cervical muscles. Further comparative studies using a larger number of subjects and therapeutic response data are necessary.
Acknowledgments
This research was supported by the Nuclear R&D program of the Korea Science and Engineering Foundation funded by the Ministry of Education, Science, and Technology (2008-M20704000039-08M0400-03910).
References
- 1.Dauer WT, Burke RE, Greene P, Fahn S. Current concepts on the clinical features, aetiology and management of idiopathic cervical dystonia. Brain. 1998;121:547–560. doi: 10.1093/brain/121.4.547. [DOI] [PubMed] [Google Scholar]
- 2.Jankovic J, Schwartz K. Botulinum toxin injections for cervical dystonia. Neurology. 1990;40:277–280. doi: 10.1212/wnl.40.2.277. [DOI] [PubMed] [Google Scholar]
- 3.Greene P, Kang U, Fahn S, Brin M, Moskowitz C, Flaster E. Double-blind, placebo-controlled trial of botulinum toxin injections for the treatment of spasmodic torticollis. Neurology. 1990;40:1213–1218. doi: 10.1212/wnl.40.8.1213. [DOI] [PubMed] [Google Scholar]
- 4.Brans JW, de Boer IP, Aramideh M, Ongerboer de Visser BW, Speelman JD. Botulinum toxin in cervical dystonia: low dosage with electromyographic guidance. J Neurol. 1995;242:529–534. doi: 10.1007/BF00867425. [DOI] [PubMed] [Google Scholar]
- 5.Koller W, Vetere-Overfield B, Gray C, Dubinsky R. Failure of fixed-dose, fixed muscle injection of botulinum toxin in torticollis. Clin Neuropharmacol. 1990;13:355–358. doi: 10.1097/00002826-199008000-00011. [DOI] [PubMed] [Google Scholar]
- 6.Dressler D. Electromyographic evaluation of cervical dystonia for planning of botulinum toxin therapy. Eur J Neurol. 2000;7:713–718. doi: 10.1046/j.1468-1331.2000.00161.x. [DOI] [PubMed] [Google Scholar]
- 7.Van Gerpen JA, Matsumoto JY, Ahlskog JE, Maraganore DM, McManis PG. Utility of an EMG mapping study in treating cervical dystonia. Muscle Nerve. 2000;23:1752–1756. doi: 10.1002/1097-4598(200011)23:11<1752::AID-MUS12>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 8.Walker FO. Botulinum toxin therapy for cervical dystonia. Phys Med Rehabil Clin North Am. 2003;14:749–776. doi: 10.1016/s1047-9651(03)00045-7. [DOI] [PubMed] [Google Scholar]
- 9.Boccagni C, Carpaneto J, Micera S, Bagnato S, Galardi G. Motion analysis in cervical dystonia. Neurol Sci. 2008;29:375–381. doi: 10.1007/s10072-008-1033-z. [DOI] [PubMed] [Google Scholar]
- 10.Carpaneto J, Micera S, Galardi G, Micheli A, Carboncini MC, Rossi B, et al. A protocol for the assessment of 3D movements of the head in persons with cervical dystonia. Clin Biomech. 2004;19:659–663. doi: 10.1016/j.clinbiomech.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 11.Gregori B, Agostino R, Bologna M, Dinapoli L, Colosimo C, Accornero N, et al. Fast voluntary neck movements in patients with cervical dystonia: a kinematic study before and after therapy with botulinum toxin type A. Clin Neurophysiol. 2008;119:273–280. doi: 10.1016/j.clinph.2007.10.007. [DOI] [PubMed] [Google Scholar]
- 12.Salvia P, Champagne O, Feipel V, Rooze M, de Beyl DZ. Clinical and goniometric evaluation of patients with spasmodic torticollis. Clin Biomech. 2006;21:323–329. doi: 10.1016/j.clinbiomech.2005.11.011. [DOI] [PubMed] [Google Scholar]
- 13.Ohnuma M, Sugita T, Kokubun S, Yamaguchi K, Rikimaru H. Muscle activity during a dash shown by 18F-fluorodeoxyglucose positron emission tomography. J Orthop Sci. 2006;11:42–45. doi: 10.1007/s00776-005-0972-y. [DOI] [PubMed] [Google Scholar]
- 14.Pappas GP, Olcott EW, Drace JE. Imaging of skeletal muscle function using (18)FDG PET: force production, activation, and metabolism. J Appl Physiol. 2001;90:329–337. doi: 10.1152/jappl.2001.90.1.329. [DOI] [PubMed] [Google Scholar]
- 15.Tashiro M, Fujimoto T, Itoh M, Kubota K, Fujiwara T, Miyake M, et al. 18F-FDG PET imaging of muscle activity in runners. J Nucl Med. 1999;40:70–76. [PubMed] [Google Scholar]
- 16.Shinozaki T, Suzuki K, Yamaji T, Ichikawa A, Inoue T, Takagishi K, et al. Evaluation of muscle metabolic activity in the lower limb of a transfemoral amputee using a prosthesis by using (18)F-FDG PET imaging–an application of PET imaging to rehabilitation. J Orthop Res. 2004;22:878–883. doi: 10.1016/j.orthres.2003.10.018. [DOI] [PubMed] [Google Scholar]
- 17.Sung DH, Choi JY, Kim DH, Kim ES, Son YI, Cho YS, et al. Localization of dystonic muscles with 18F-FDG PET/CT in idiopathic cervical dystonia. J Nucl Med. 2007;48:1790–1795. doi: 10.2967/jnumed.107.044024. [DOI] [PubMed] [Google Scholar]
- 18.Comella CL, Jankovic J, Brin MF. Use of botulinum toxin type A in the treatment of cervical dystonia. Neurology. 2000;55:S15–S21. [PubMed] [Google Scholar]
- 19.Traba Lopez A, Esteban A. Botulinum toxin in motor disorders: practical considerations with emphasis on interventional neurophysiology. Neurophysiol Clin. 2001;31:220–229. doi: 10.1016/S0987-7053(01)00263-5. [DOI] [PubMed] [Google Scholar]
- 20.Ostergaard L, Fuglsang-Frederiksen A, Werdelin L, Sjo O, Winkel H. Quantitative EMG in botulinum toxin treatment of cervical dystonia. A double-blind, placebo-controlled study. Electroencephalogr Clin Neurophysiol. 1994;93:434–439. doi: 10.1016/0168-5597(94)90150-3. [DOI] [PubMed] [Google Scholar]
- 21.Buchman AS, Comella CL, Leurgans S, Stebbins GT, Goetz CG. The effect of changes in head posture on the patterns of muscle activity in cervical dystonia (CD) Mov Disord. 1998;13:490–496. doi: 10.1002/mds.870130320. [DOI] [PubMed] [Google Scholar]
- 22.Buchman AS, Comella CL, Stebbins GT, Tanner CM, Goetz CG. Quantitative electromyographic analysis of changes in muscle activity following botulinum toxin therapy for cervical dystonia. Clin Neuropharmacol. 1993;16:205–210. doi: 10.1097/00002826-199306000-00003. [DOI] [PubMed] [Google Scholar]
- 23.Tarsy D. Comparison of clinical rating scales in treatment of cervical dystonia with botulinum toxin. Mov Disord. 1997;12:100–102. doi: 10.1002/mds.870120117. [DOI] [PubMed] [Google Scholar]
- 24.Delval A, Krystkowiak P, Cassim F, Skrzypzak L, Destee A, Derambure P, et al. Usefulness and limitations of polymygraphic recordings in dystonia. Rev Neurol. 2004;160:547–553. doi: 10.1016/S0035-3787(04)70984-4. [DOI] [PubMed] [Google Scholar]