Abstract
Purpose
We compared alternative routes for 18F-fluorodeoxyglucose (FDG) administration, such as the retroorbital (RO), intraperitoneal (IP) and per oral (PO) routes, with the intravenous (IV) route in normal tissues and tumors of mice.
Materials and Methods
CRL-1642 (ATCC, Lewis lung carcinoma) cells were inoculated in female BALB/c-nu/nu mice 6 to 10 weeks old. When the tumor grew to about 9 mm in diameter, positron emission tomography (PET) scans were performed after FDG administration via the RO, IP, PO or IV route. Additional serial PET scans were performed using the RO, IV or IP route alternatively from 5 to 29 days after the tumor cell injection.
Results
There was no significant difference in the FDG uptake in normal tissues at 60 min after FDG administration via RO, IP and IV routes. PO administration, however, showed delayed distribution and unwanted high gastrointestinal uptake. Tumoral uptake of FDG showed a similar temporal pattern and increased until 60 min after FDG administration in the RO, IP and IV injection groups. In the PO administration group, tumoral uptake was delayed and reduced. There was no statistical difference among the RO, IP and IV administration groups for additional serial PET scans.
Conclusion
RO administration is an effective alternative route to IV administration for mouse FDG PET scans using normal mice and tumor models. In addition, IP administration can be a practical alternative in the late phase, although the initial uptake is lower than those in the IV and RO groups.
Keywords: Administration route, Intraperitoneal, Retroorbital, Per oral, Intravenous, Small animal PET, FDG
Introduction
Small animal positron emission tomography/computed tomography (PET/CT) is widely used in neuroscience studies, oncology, basic pharmacology, hypoxic imaging and stem cell tracking [1–5]. Semiquantification using the standard uptake value (SUV) in preclinical study is one of the strengths of small animal PET/CT. This semiquantitative information, however, can be influenced by many aspects in animal PET/CT studies. Most importantly, the experimental condition, which is expected to be controlled, could influence the results of experiments with living animals. The route of administration or mode of the radiotracer is the cornerstone for experiments that use radiopharmaceuticals, and it is the step that is most vulnerable to experimental errors because the routes and the velocity of administration could influence the resorption and final distribution.
Radiopharmaceuticals are administered via bolus intravenous (IV) injection, and the tail vein is most commonly use in mice. As the tail vein of the mouse is fragile and small to cannulate, leakage or failure of injection is not rare, even for experienced researchers. Furthermore, if the experiment needs repetitive cannulations with short intervals, it is not easy to succeed in every successive experiment. In spite of these difficulties, repetitive PET imaging is mandatory in many studies that are monitoring tumor response, and these studies are important for testing the efficacy of new drugs and for other types of therapeutic management.
Alternative administration routes of radiopharmaceuticals, such as intraperitoneal (IP), retroorbital (RO) and per oral (PO) administration, are technically easier, much faster and more reproducible without any complications related to its repetition. Because of the potential difference in pharmacokinetics or the temporal and regional biodistribution, however, it is mandatory to carefully compare these routes when using these alternative methods instead of intravenous administration.
As far as we know, there has been no comprehensive comparative study of these different administration routes in terms of both normal tissue uptake and tumoral uptake of any radiopharmaceuticals or FDG.
The aim of this study was to compare alternative routes of administration, such as RO, IP and PO, with IV for assessing tumoral glucose metabolism in a typical preclinical mouse model.
Materials and Methods
Preparation of the Radiopharmaceuticals and Animals
FDG was produced by a standard method using our on-site cyclotron (RDS-111, Siemens) and automatic radiosynthesis modules (CPCU, Siemens). BALB/c-nu/nu female mice were used for all the experiments. For the tumor-bearing model, mouse Lewis lung carcinoma cells (ATCC CRL-1642TM) were cultured with RPMI in the rapidly growing phase (confluence less than 80%). The cells were collected with a rapid preparation procedure. Eighty million cells were inoculated subcutaneously in the shoulder region of the mice (6 to 10 weeks old, mean weight: about 20 g), which were anesthetized with 2% isoflurane in 100% oxygen. After all the experiments, the mice were killed using a CO2 chamber.
PET/CT Scanning
All mice were fasted for at least 6 h before the F-18 FDG scans, and only water was supplied without other caloric food materials. The mouse’s body temperature was controlled before and after each administration, throughout all the imaging processes, using a heating lamp and a heating pad. The blood glucose levels were measured before every FDG injection, and this was verified to be below 100 g/ml of glucose for all the experiments. Anesthesia was administered using 2% isoflurane in 100% oxygen. Fifteen MBq of FDG was administrated through the IV, IP, RO or PO administration routes according to the experimental plans.
The FDG PET/CT scans were performed with the three-dimensional acquisition mode (eXplore Vista PET/CT, GE). For PET scans, normalization was applied, but scatter correction and attenuation correction were not. The PET scans were acquired for 9 min per bed position for all the studies. The CT scans were used for anatomical reference. For the CT scans, the X-ray sources were used at 300 μA and 40 kV for 6 min (1 shot per projection). The CT resolution was 200 μm, and the number of acquired projections was 360.
The images were normalized as SUVs using the formula: SUV = decay of the corrected mean tissue activity concentration (in Bq/ml)/{injected dose (in Bq) × body weight (in g)}. A volume of interest (VOI) was manually drawn around each organ of interest (liver, kidneys, lungs, brain, heart, brown fat, muscle, retroorbital and acromio-humeral joint), and the VOIs of the tumors were drawn automatically according to an isointensity curve with an SUV over 1 g/ml (PMOD; University Hospital Zurich, Zurich, Switzerland). The maximal SUV and mean SUV were calculated for the corresponding VOIs. The total regional FDG accumulations (TRFA) of the selected VOI were calculated by multiplying the volume and the mean SUV of the VOI in order to assess how much FDG had accumulated in the VOI.
Administration Routes
For the IV route, the tail was warmed with warm water before cannulation, if necessary. A volume of 50 μl of FDG was administered through the tail vein using a 30-gauge needle of an insulin syringe. The volume and dose of FDG were less than 50 μl and 15 MBq, respectively.
For the RO route, we injected 50 μl (15 MBq) of FDG into the retroorbital area using a 30-gauge needle of an insulin syringe, applying gentle pressure with two fingers in the periorbital area to allow the egression of the bulb.
IP administration was done at the left lower abdominal area. We injected a volume of 50 μl with an insulin syringe. The volume and radioactivity of FDG were less than 50 μl and 15 MBq, respectively.
For the PO route, 15 MBq of FDG in 1 ml of water was administered using gavage, and the gavage was washed three times with 1 ml of water. To verify the net injected dose, the residual dose of radiopharmaceuticals in the syringe was measured after the injection.
Study Design for Comparing the Extents of Accumulation in the Normal Organs and Tumor According to the Routes of Administration
In order to assess and compare the normal regional distributions of FDG, the mice were divided into three groups. Each group of mice was allocated to the IV vs. IP group (n = 7), the IV vs. RO group (n = 7) and the IV vs. PO group (n = 4), respectively. PET/CT scans were taken twice per each mouse with a 24-h interval using the two given different administration routes (Table 1). Serial PET/CT scans were performed at the time points of 10, 20, 30, 40, 50, 60, 120 and 180 min after administration.
Table 1.
Design of the study for the F-18 FDG distribution in the normal mice
| No. of mice | Date of scan | ||
|---|---|---|---|
| D1 | D2 | ||
| IV vs. IP (n = 7) | 4 | IV | IP |
| 3 | IP | IV | |
| IV vs. RO (n = 7) | 4 | IV | RO |
| 3 | RO | IV | |
| IV vs. PO (n = 4) | 2 | IV | PO |
| 2 | PO | IV | |
Tumoral FDG accumulation was evaluated using two experimental protocols. First, we compared the tumoral FDG uptakes according to the administration route groups by performing a cross-sectional uptake study at 2 weeks after inoculation (Table 2). The tumor grew to about 10 mm in diameter 2 weeks after inoculation. Eighteen mice were selected, in which the size of tumors ranged from 8–12 mm in diameter. We allocated the 18 mice into three groups: IV vs. IP (n = 7), IV vs. RO (n = 7) and IV vs. PO (n = 4). Serial PET/CT scans were performed at 10, 20, 30, 40, 50 and 60 min after administration using one administration method (day 14), and another set of PET/CT scans was done with the other paired administration method the next day (day 15). On day 17, multiple PET/CT scans were performed at 60, 120 and 180 min after administration with the same administration method as on day 14, and on the next day (day 18) another set of PET/CT scans was done with the same administration method as on day 15.
Table 2.
Design of the study for the F-18 FDG distribution in a xenograft mouse tumor model
| No. of mice | Days after inoculation | |||||
|---|---|---|---|---|---|---|
| D14 | D15 | D16 | D17 | D18 | ||
| IV vs. IP (n = 7) | 4 | IV | IP | IV | IP | |
| 3 | IP | IV | IP | IV | ||
| IV vs. RO (n = 7) | 4 | IV | RO | IV | RO | |
| 3 | RO | IV | RO | IV | ||
| IV vs. PO (n = 4) | 2 | IV | PO | IV | PO | |
| 2 | PO | IV | PO | IV | ||
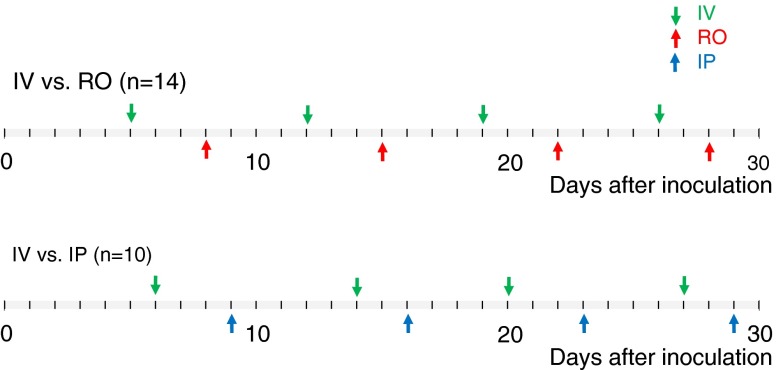
Second, we compared the tumoral FDG uptakes successively over 4 weeks using IV, IP and RO administration for a long-term follow-up study from 5 to 29 days after inoculation (Fig. 1). In order to minimize other influencing factors related to the inter-object tumoral inhomogeneity, such as the size difference, several PET/CT scans were performed alternatively with paired administration routes in the same mouse for up to 29 days. The mice were allocated into two groups: the IV vs. RO group (n = 14) and IV vs. IP group (n = 10). All the PET/CT scans were done 60 min after FDG injection.
Fig. 1.
Design of the study for long-term F-18 FDG distribution in a xenograft mouse tumor model
Statistical Analysis
The data were presented as means ± 1 standard deviation (SD). The values at the selected time, such as the SUV values of the normal or tumoral tissue at 60 min, were compared using the paired sample t-test (MedCalc for Windows, version 9.4.2.0, MedCalc Software, Mariakerke, Belgium).
In order to compare the temporal FDG accumulation changes from 10 to 180 min after FDG administration, the areas under the curve (AUC) of the time-uptake curve from the four groups were calculated and compared among the groups using the paired sample t-test. Because we could not obtain the input curve of FDG administration because of technical problems in the experiment using small animals and we could not conduct a compartmental analysis, the time to the FDG accumulation curve was fitted to a linear line, a third order polynomial curve, a one-phase exponential decay curve or an exponential decay curve. The goodness of fit was obtained (Graph Pad Software, Inc., San Diego, CA), and P values less than 0.05 were considered statistically significant.
Results
FDG Accumulations in the Typical Organs of Normal Mouse
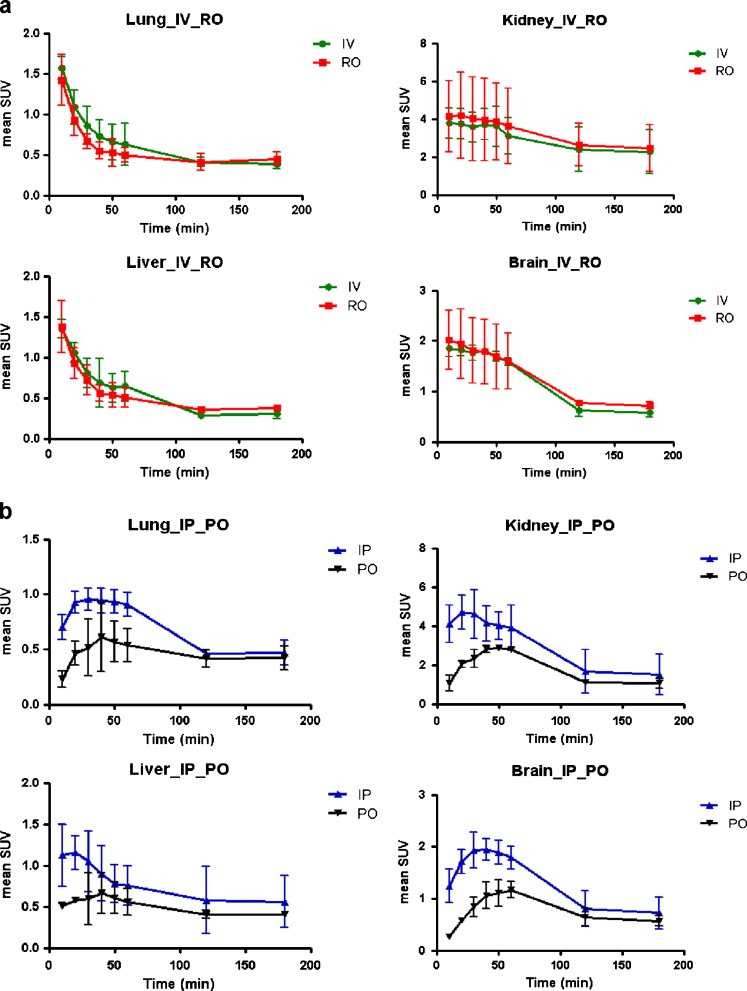
In all four groups, all the normal tissues showed that the FDG uptakes were substantially low, and they did not mimic the pathologic uptake of the tumor. In the late phase (60–180 min after administration), the liver, lung, brain and muscle did not exceed the maximum SUV of 1 g/ml in all four administration groups. In all four groups, the FDG uptakes in the early phase were high, and these early uptakes decreased or were excreted with time. However, the detailed temporal patterns of the FDG uptake in the normal tissue of the four groups were different, especially during the initial phase. In the IV and RO groups, the maximum SUV reached the peak values in 10 min, and this was followed by rapid excretion. In the IP and PO groups, however, the maximum SUV reached the peak values after 20 min, and then there was relatively slow excretion (Fig. 2). The extent of FDG uptake in the heart varied widely regardless of the administration routes. The curves of FDG uptake in the IV and RO groups were fitted to an exponential decay curve, whereas those in the IP and PO groups were not. The maximum FDG accumulations or retentions were seen at between 30 and 60 min in terms of the organs. The area under the curve (AUC) of the IP group was greater than that of the PO group in the lung, liver, kidney, brain, muscle and joint regions. The FDG uptake of normal tissue in the PO group was decreased and delayed compared to that of the IP group. The times of the peak of the IP were 30 min in the lung, 20 min in the liver, 20 min in the kidney, 30 min in brain, 20 min in muscle and 60 min in the joints, and those of the PO were 40, 40, 50, 60, 40 and 60 min, respectively.
Fig. 2.
FDG accumulations of the typical organs on dynamic PET scans. a The time to tissue uptake curve of the IV and RO groups, and b those of the IP and PO groups. IV: intravenous, RO: retroorbital, IP: intraperitoneal, PO: per oral
The FDG accumulations between the IV and IP groups and between the IV and RO groups were not significantly different in the liver, lung, brain, heart, kidney and joint regions at 60 min after administration (Table 3). Although the accumulation of the PO group seemed to be less than that of the IV group grossly, it was not statistically different, except in muscle.
Table 3.
F-18 FDG uptakes by the administration groups and regions 60 min after administration
| Mode of injection, and acquisition (SUV) | Liver | Kidneys | Lungs | Brain | Heart | Muscle (hind limb) | Joint (hind limb) | Retro-orbital region | Tail | Whole abdomen |
|---|---|---|---|---|---|---|---|---|---|---|
| IV injection (n = 7) | ||||||||||
| Mean | 0.58 | 3.56 | 0.60 | 2.04 | 7.59 | 0.47 | 1.42 | 3.78 | 1.33 | 0.69 |
| SD | 0.29 | 0.90 | 0.29 | 0.57 | 5.77 | 0.29 | 0.60 | 3.70 | 0.30 | 0.18 |
| IP injection (n = 7) | ||||||||||
| Mean | 0.65 | 4.40 | 0.58 | 2.17 | 6.73 | 0.32 | 1.54 | 1.78 | 0.34 | 0.96 |
| SD | 0.29 | 2.41 | 0.25 | 0.45 | 4.55 | 0.06 | 0.46 | 0.90 | 0.04 | 0.32 |
| p | 0.66 | 0.44 | 0.92 | 0.64 | 0.76 | 0.19 | 0.38 | 0.18 | 0.01 | 0.09 |
| IV injection (n = 7) | ||||||||||
| Mean | 0.51 | 3.28 | 0.33 | 1.69 | 2.76 | 0.31 | 0.87 | 4.21 | 1.06 | 0.54 |
| SD | 0.27 | 0.62 | 0.09 | 0.52 | 1.68 | 0.06 | 0.25 | 2.58 | 0.46 | 0.09 |
| RO injection (n = 7) | ||||||||||
| Mean | 0.49 | 6.03 | 0.43 | 2.66 | 5.76 | 0.34 | 2.63 | 20.07 | 0.35 | 0.68 |
| SD | 0.15 | 4.90 | 0.12 | 1.41 | 3.12 | 0.07 | 3.80 | 16.03 | 0.03 | 0.15 |
| p | 0.84 | 0.17 | 0.11 | 0.09 | 0.12 | 0.36 | 0.27 | 0.05 | 0.06 | 0.01 |
| IV injection (n = 4) | ||||||||||
| Mean | 0.41 | 4.24 | 0.37 | 2.07 | 3.06 | 0.37 | 1.01 | 5.43 | 1.25 | 0.63 |
| SD | 0.14 | 1.32 | 0.13 | 0.52 | 3.24 | 0.05 | 0.24 | 2.59 | 0.57 | 0.11 |
| PO administration (n = 4) | ||||||||||
| Mean | 0.50 | 4.21 | 0.39 | 1.44 | 3.39 | 0.27 | 0.73 | 1.75 | 0.29 | 1.54 |
| SD | 0.18 | 2.17 | 0.10 | 0.71 | 1.79 | 0.09 | 0.20 | 0.56 | 0.04 | 0.42 |
| P | 0.25 | 0.99 | 0.87 | 0.35 | 0.86 | 0.04 | 0.26 | 0.09 | 0.04 | 0.01 |
| IV (n = 18) | 0.52 | 3.60 | 0.44 | 1.91 | 4.71 | 0.39 | 1.12 | 4.31 | 1.22 | 0.62 |
| IP (n = 7) | 0.65 | 4.40 | 0.58 | 2.17 | 6.73 | 0.32 | 1.54 | 1.78 | 0.34 | 0.96 |
| PO (n = 4) | 0.50 | 4.21 | 0.39 | 1.44 | 3.39 | 0.27 | 0.73 | 1.75 | 0.29 | 1.54 |
| RO (n = 7) | 0.49 | 6.03 | 0.43 | 2.66 | 5.76 | 0.34 | 2.63 | 20.07 | 0.35 | 0.68 |
Standard uptake value: SUV, standard deviation: SD, intravenous: IV, intraperitoneal: IP, per oral, PO, retroorbital: RO
FDG Accumulations in the Xenograft Mouse Tumor Model
All the tumors were successfully visualized using all four administration routes, and there were no notable problems from the viewpoint of the qualitative or visual characteristics. From the quantitative point of view, however, there were some differences among the groups.
In all four groups, the tumoral uptake of FDG increased during the initial 60 min, and this reached a plateau after approximately 60–120 min in all four groups (Fig. 3a and b). In the initial phase, the IV, RO and IP curves were fitted to the one phase exponential association curve, but the PO curves were not. Analysis using AUC showed that the IV curve and RO curve were not different, but the IV curve was different from the IP and PO curves (p < 0.05). The IP curve was different from the PO curve (p < 0.05). In the late phase at 120 and 180 min, no statistical differences among the four groups were noted. The initial uptakes in tumors were slightly delayed in the IP group, but they were substantially delayed in the PO group. For example, at 60 min, the uptake of the IP group was equal to that of the IV and RO groups, but the uptake of the PO group was only 60 ± 35% of the uptake of the IV group (Table 4).
Fig. 3.
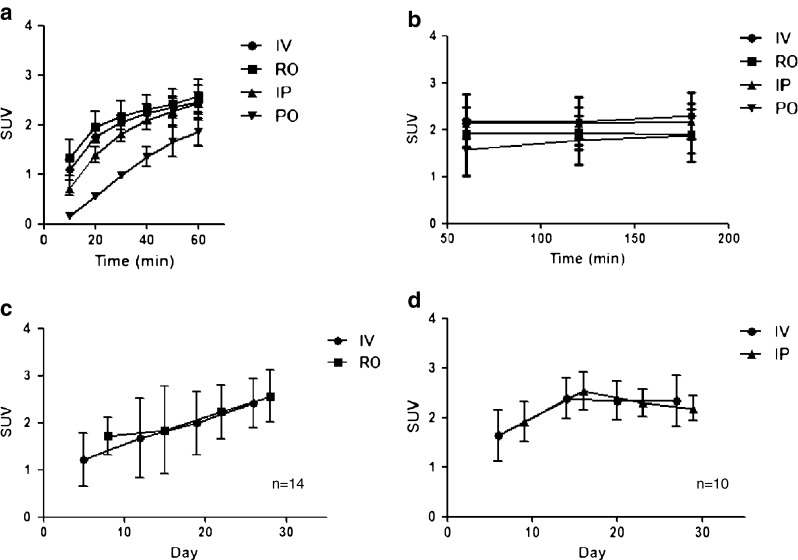
FDG accumulations according to the administration route in the tumor model. a, b Multiple static FDG PET scans were undertaken in 18 tumor-bearing mice at 10, 20, 30, 40, 50, 60, 120 and 180 min using the IV, RO, IP and PO methods. A volume of interest was drawn on the tumor, and the mean SUV was calculated. 8 × 107 CRL-1642 cells per mouse were inoculated in 24 mice. c, d Serial FDG PET scans were done 60 min after FDG administration during 4 weeks with the IV and RO routes by turns, and then with the IV and IP routes by turns. A volume of interest was drawn on the tumor, and the mean SUV was calculated. IV: intravenous, RO: retroorbital, IP: intraperitoneal, PO: per oral
Table 4.
Relative tumor uptake at 60 min after F-18 FDG administration
| RO/IV | IP/IV | PO/IV | |
|---|---|---|---|
| SUVmax | 1.16 ± 0.27 | 1.39 ± 0.85 | 0.60 ± 0.35 |
| SUVmean | 1.12 ± 0.35 | 1.08 ± 0.31 | 0.64 ± 0.27 |
On the long-term follow-up study, the FDG uptakes at 60 min after administration in the IV, RO and IP groups were similar to each other. Serial PET scans were undertaken using the RO, IV or IP route alternatively from 5 days to 29 days after tumor cell inoculation. The tumor increased in size as time passed, and the three administration groups all faithfully reflected the tumor growth. The time versus FDG accumulation curves were not statistically different in the case of IV vs. RO and IV vs. IP (Fig. 3c and d).
Discussion
Many factors can influence the robustness, reproducibility and quantification of small animal PET studies using FDG. These include the fasting duration, inter-species differences, technical aspects and extent of defluorination [6]. Among them, precise administration of radiopharmaceuticals is essential, but this is not always easy to control. When small animals are used, it is not always easy and possible to administer an exact dose of radiopharmaceuticals, and if this is repeated several times with short time intervals, then it is much more difficult. For example, considering the most common technique, the intravenous injection technique, even well-trained researchers could not succeed in always injecting radiopharmaceuticals without any leakage. Therefore, if we plan experiments that need repeated injections and scans, the probability of accomplishing the entire experiment without any injection-related problems is low.
IV administration is the standard administration route, and its characteristics and pharmacokinetics are well known. IP, RO and PO administrations are the alternative routes to IV for drug delivery and euthanasia [7–12]. A few previous studies in the literature have dealt with comparing these routes in terms of radiopharmaceutical administration. These studies mainly focused on the accumulation pattern of normal tissue. Higashi and coworkers [7] compared IV and PO administration in normal mice with various solutions: hypo-, iso- and hypertonic. Fueger and coworkers [8] demonstrated that the distribution of FDG with intraperitoneal administration was comparable to that with intravenous administration at 60 min post-injection, and Nanni and coworkers [9] suggested that RO administration was an effective route for FDG administration in normal mice. As far as we know, however, there has been no study that has comprehensively compared various alternative routes with IV administration in an animal tumor model, which is essential for preclinical experiments on drug development, and IV administration is the most typical type used for animal PET/CT.
Steel and coworkers [10] compared IV and RO administration for drug delivery in a tumor model, and they concluded that both were similarly effective, but RO administration is less stressful. Schoell and coworkers insisted that RO administration be considered a technically simple and superior alternative for mouse euthanasia [11]. Because the RO area is far away from the typical xenografted tumor sites, such as the shoulder or flank, RO administration could be the suitable administration route for an animal tumor model. The RO area, however, is close to the brain, and so the RO area might not be suitable for brain studies. IP administration is more useful for brain imaging [12]. According to our results, the total residual activity around the RO area was not as high as we expected. The pharmacokinetics and the extent of uptake of RO administration were similar with those of IV administration in both the initial and late phases of administration and for both normal mice and the tumor model. It could be thought that RO administration is almost identical to IV administration. We can speculate that FDG is absorbed intravenously through the retroorbital venous plexus with RO administration.
The peritoneal wall is also rich in vessels, so it is used for the administration of drugs and performing peritoneal dialysis. Glucose is also freely absorbable through the peritoneum. Although RO administration is easier than IV administration, it also needs some technique and experience. IP administration is very easy and safe to learn, perform and repeat; therefore, IP administration is the most suitable candidate as an alternative to IV administration. Of the four administration routes, IP administration is technically the easiest, and IP administration is even easier than PO administration, in which gavage should be used. According to our results, however, IP administration is not identical to IV administration for the aspects of pharmacokinetics, and some cases revealed that the residual radioactivity of the peritoneum might hinder assessing diseased organs in the abdominal cavity, such as hepatic metastasis or another orthotopic model. Most cases, however, did not reveal problem-causing high FDG accumulation in the abdominal peritoneal cavity region. Although we could not obtain the blood input function using IP administration, we can speculate that the tissue uptake curve and input peak may be low and the time to the peak may be delayed. The initial FDG uptake was slightly delayed, but the initial tumoral uptake did not significantly show this delay, and in the late phase, there was no difference with IV administration in the normal and tumoral tissue, which was comparable with the findings of other studies [8]. If we consider this, IP administration could be a practical alternative to IV administration in the late phase after 60 min, whereas we should be conservative about using IP administration in the initial phase.
Glucose can be nearly completely absorbed through the gastrointestinal tract [13]. Because FDG is an analogue of glucose, it can be well absorbed through the gastrointestinal tract. Per oral administration is a technically non-invasive and robust method if we administer FDG using gavage under anesthesia, but as this requires gavage and anesthesia, it was not as easy as expected. Moreover, PO administration was not comparable with IV administration, which is the de facto standard, until 60 min, although our result after 120 min showed similar uptake with IV administration. We cannot recommend PO administration as an alternative to IV administration. There was much more nonspecific gastrointestinal uptake in the PO group than in the IV, IP and RO groups. These intense abdominal accumulations and the slower pharmacokinetic characteristics could hinder evaluating the FDG uptake of the abdominal region of interest and quantifying the tumoral uptake.
Our experimental protocol has some limitations. We could not compare all four methods in the same mice, but we think our protocol is practically the most ideal. FDG PET study is difficult or impossible to repeat in a day. For this reason, we could not perform the four methods in the same mouse, which would have required at least 4 days’ successive PET scans, too long an anesthetic time and too long a fasting time. Mice cannot endure such harsh experimental conditions. For the tumor uptake experiment, it is biologically impossible to perform FDG PET scans in similar tumors because although the tumors are similar in size, the FDG accumulations vary widely because of many factors, such as the extent of tumor necrosis and the local tumoral milieu. Some tumor grows too rapidly; it is even difficult to synchronize the size of the tumors when performing experiments with several mice. Because of these problems, we tried to normalize the methods by pairing two routes in the same mouse with as short a time interval as possible (24 h in the cross-sectional study) and performing repeated PET scans with alternation of the administration routes in the same mouse for several weeks in the long-term follow-up study. This could be a practical method to minimize the variance from many factors that can be difficult to control.
In conclusion, retroorbital administration is an effective alternative route to intravenous administration when performing mouse FDG PET scans using normal mice and tumor models. In addition, intraperitoneal administration could be a practical alternative in the late phase, although the initial uptake is lower than that of IV and RO. Researchers could consider their circumstances and needs, and select the appropriate administration routes among these alternatives.
Acknowledgements
This research was supported by the Pioneer Research Center Program through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (2010-0002209).
References
- 1.Endepols H, Sommer S, Backes H, Wiedermann D, Grat R, Hauber W. Effort-based decision making in the rat: an [18 F] fluorodeoxyglucose micro positron emission tomography study. J Neurosci. 2010;30:9708–14. doi: 10.1523/JNEUROSCI.1202-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liang Z, Cho HT, Williams L, Zhu A, Liang K, Huang K, et al. Potential biomarker of L-type amino acid transporter 1 in breast cancer progression. Nucl Med Mol Imaging. 2011;45:93–102. [DOI] [PMC free article] [PubMed]
- 3.Lancelot S, Zimmer L. Small-animal positron emission tomography as a tool for neuropharmacology. Trends Pharmacol Sci. 2010;31:411–7. doi: 10.1016/j.tips.2010.06.002. [DOI] [PubMed] [Google Scholar]
- 4.Frangioni JV, Hajjar RJ. In vivo tracking of stem cells for clinical trials in cardiovascular disease. Circulation. 2004;110:3378–83. doi: 10.1161/01.CIR.0000149840.46523.FC. [DOI] [PubMed] [Google Scholar]
- 5.Wyss MT, Honer M, Schubiger PA, Ametamey SM. NanoPET imaging of [18F] fluoromisonidazole uptake in experimental mouse tumours. Eur J Nucl Med Mol Imaging. 2006;33:311–8. doi: 10.1007/s00259-005-1951-4. [DOI] [PubMed] [Google Scholar]
- 6.Jacobson MS, Dankwart HR, Mahoney DW. Radiolysis of 2-[18F]fluoro-2-deoxy-D-glucose ([18F]FDG) and the role of ethanol and radioactive concentration. Appl Radiat Isot. 2009;67:990–9957. doi: 10.1016/j.apradiso.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 7.Higashi T, Fisher SJ, Nakada K, Romain DJ, Whal RL. Is enteral administration of fluorine-18-fluorodeoxyglucose (F-18 FDG) a palatable alternative to IV injection? Pre-clinical evaluation in normal rodents. Nucl Med Biol. 2002;29:363–73. doi: 10.1016/S0969-8051(01)00312-2. [DOI] [PubMed] [Google Scholar]
- 8.Fueger BJ, Czernin J, Hildebrandt I. Impact of animal handling on the results of 18 F-FDG PET studies in mice. J Nucl Med. 2006;47:999–1006. [PubMed] [Google Scholar]
- 9.Nanni C, Pettinato C, Ambrosini V, Spinelli A, Trespidi S, Rubello D, et al. Retro-orbital injection is an effective route for radiopharmaceutical administration in mice during small-animal PET studies. Nucl Med Commun. 2007;28:547–53. doi: 10.1097/MNM.0b013e3281fbd42b. [DOI] [PubMed] [Google Scholar]
- 10.Steel CD, Stephen AL, Hahto SM, Singletery SJ, Ciavarra RP. Comparison of the lateral tail vein and the retro-orbital venous sinus as routes of intravenous drug delivery in a transgenic mouse model. Lab Anim. 2008;37:26–32. doi: 10.1038/laban0108-26. [DOI] [PubMed] [Google Scholar]
- 11.Schoell AR, Heyde BR, Weir DE, Chiang PC, Hu Y, Tung DK. Euthanasia method for mice in rapid time-course pulmonary pharmacokinetic studies. J Am Assoc Lab Anim Sci. 2009;48:506–11. [PMC free article] [PubMed] [Google Scholar]
- 12.Schiffer WK, Mirrione MM, Deway SL. Optimizing experimental protocols for quantitative behavioral imaging with 18F-FDG in rodent. J Nucl Med. 2007;48:277–87. [PubMed] [Google Scholar]
- 13.Atkinson RM, Parsons BJ, Smyth DH. The intestinal absorption of glucose. J Physiol. 1957;135:581–9. doi: 10.1113/jphysiol.1957.sp005732. [DOI] [PMC free article] [PubMed] [Google Scholar]