Abstract
A 20-year-old woman, who presented with a several-week history of abdominal pain, was referred for magnetic resonance imaging (MRI) and 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET)/computed tomography (CT) after an ultrasound showed complex cystic masses arising from both ovaries. The MRI and 18F-FDG PET/CT imaging characteristics of the ovarian masses were strongly suspicious for malignancy, and the masses were surgically removed. Histopathological evaluation revealed a bilateral tubo-ovarian abscess, with no evidence of malignancy. This case highlights a potentially serious pitfall in the evaluation of suspicious pelvic masses by 18F-FDG PET/CT, whereby a complex bilateral tubo-ovarian abscess may mimic the PET/CT imaging characteristics of an ovarian or pelvic malignancy.
Keywords: Tubo-ovarian abscess, Ovarian cancer, Fluorodeoxyglucose, FDG, PET
Introduction
Tubo-ovarian abscess (TOA) is a serious complication of acute pelvic inflammatory disease (PID), which most commonly results from the spread of bacteria from the lower genital tract. It is a polymicrobial infection typically occurring in young sexually active women [1]. Predisposing factors in the development of TOA include sexual activity, multiple sexual partners, nulliparity, previous episodes of PID, lower socioeconomic status and the use of intrauterine devices [2]. Transvaginal ultrasound is the initial imaging modality of choice in the diagnosis of TOA, due to its convenience and cost-effectiveness. It allows detailed visualization of pelvic structures, although its findings are sometimes indeterminate due to limited tissue contrast, resolution and field of view [3]. Atypical or equivocal cases often require further imaging with computed tomography (CT) or magnetic resonance imaging (MRI); however, CT and MRI findings can also be non-specific and TOA has been mistaken for ovarian tumors on CT as well as MRI [4]. 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT has been successfully used to image a wide variety of infectious processes, including abscesses [5]; however, the PET/CT literature on TOA is very limited. It is important for PET/CT readers to be aware of this benign differential diagnosis of bilateral FDG-avid adnexal masses, even in the context of CT or MRI findings suspicious for malignancy, as this may impact clinical management with consideration to important patient-related issues, such as maintaining fertility in premenopausal women.
Case Report
A 20-year-old woman presented to emergency with a several-week history of diffuse abdominal pain, but no history of fever or chills. On physical examination, the patient complained of mild bilateral adnexal pain on manual vaginal examination and mild, diffuse lower abdominal pain on palpation. Laboratory investigations showed a mildly elevated white blood cell (WBC) count of 12.2 × 109/L (normal range 4.8-10.8) and significantly elevated C-reactive protein at 98.5 mg/L (normal range 0.0-5.0). Her tumor markers were not elevated: carcinoembryonic antigen (CEA) was 0.4 μg/L (normal range 0–4.9), and cancer antigen 125 (CA-125) was 14 U/ml (normal range 0–35). Microscopic analysis of a urine sample showed elevated WBCs, red blood cells (RBCs) and bacteria (2+) suggestive of urinary tract infection. The patient was referred for an abdominal, pelvic and endovaginal ultrasound with Doppler, which showed a 5.1-cm solid hypoechoic mass behind the uterus with bridging vessels in the myometrium (suspected to be a pedunculated fibroid), an adjacent 4-cm cystic mass in the right adnexa, and two complex cystic masses measuring 4 and 2.8 cm in the left adnexa. Possible diagnoses, such as hemorrhagic cysts and endometrioma, were suggested; however, due to the complex nature of the cysts/masses, the patient was referred for an MRI and subsequently an 18F-FDG PET/CT to rule out malignancy.
Gadolinium-enhanced MRI of the pelvis was performed (Figs. 1a, 2a). There were two large complex mixed cystic and solid masses identified in the pelvis. The right adnexal mass measured 6.4 × 5.2 × 4.3 cm with a solid component measuring 4.3 × 2.5 cm, with low signal on T1- and T2-weighted images, along with a few adjacent cystic components (possibly hydrosalpinx). The left adnexal mass measured 5.8 × 5.4 × 5.8 cm, with smaller solid components measuring 2.4 × 1.2 cm, along with a focus of hemorrhage measuring 1.1 × 0.7 cm. The two masses abutted each other in the midline just above the anteverted uterus. Dynamic images showed diffuse enhancement of both complex masses, with evidence of invasion in the sigmoid bowel wall and possible myometrial invasion from the left ovarian mass. There was a minimal amount of free fluid in the pelvis. These findings were suspicious for malignancy, with possible diagnoses of bilateral mixed germ-cell tumors, endometrioid carcinoma, or even Krukenberg tumors. The patient was referred for a contrast enhanced CT and an 18F-FDG PET/CT to evaluate for distant disease.
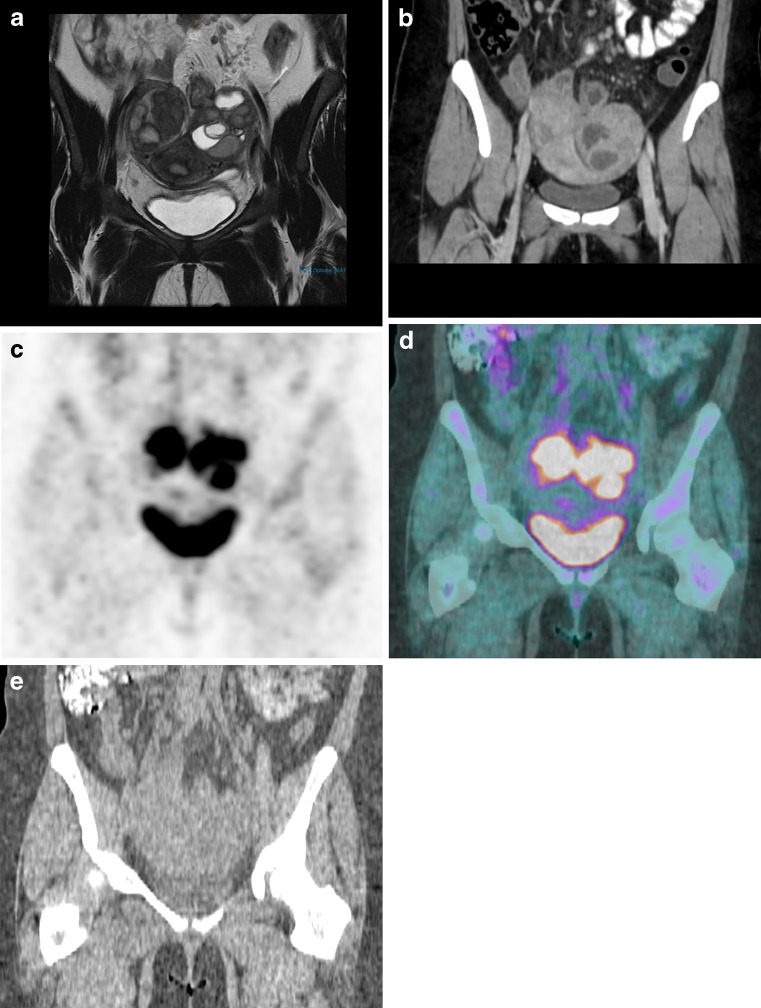
Fig. 1.
Coronal views of the pelvis on (a) T2-weighted FSE (fast spin echo) MRI, (b) contrast-enhanced CT, and corresponding 18F-FDG PET/CT with (c) PET, (d) PET/CT fusion, and (e) the CT portion of the PET/CT images show two large complex mixed cystic and solid masses that were suspicious for ovarian or pelvic malignancy, but were histologically shown to be bilateral TOA. The PET/CT showed intense 18F-FDG uptake in the two pelvic masses, with SUVmax of 13.3 in the 3.5-cm right adnexal mass, and SUVmax of 15.6 in the 5-cm left adnexal mass
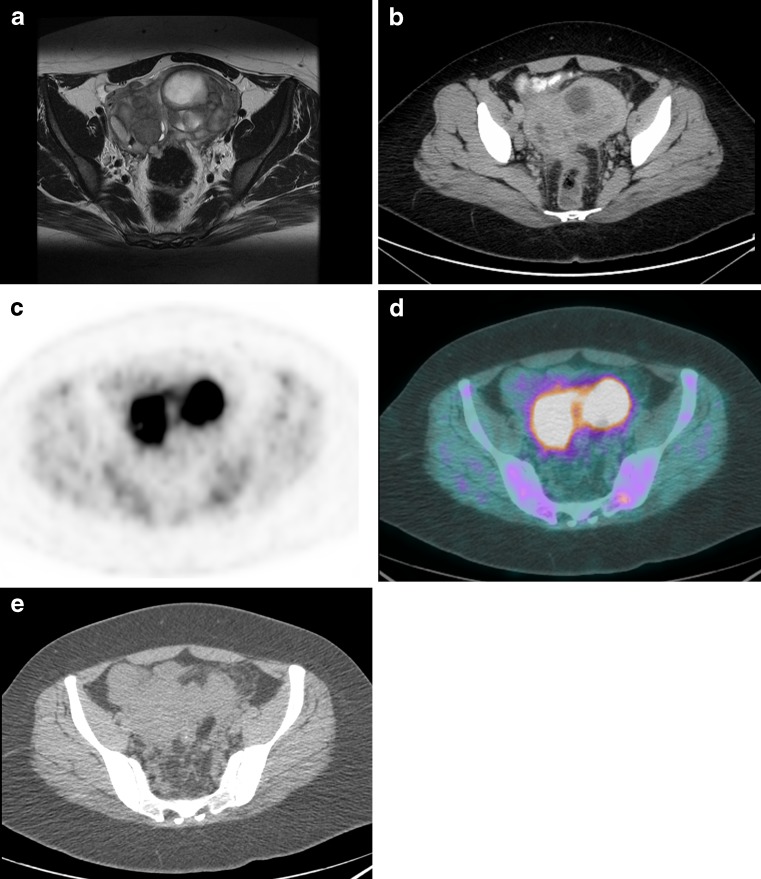
Fig. 2.
Transaxial views of the pelvis on (a) T2-weighted FSE MRI, (b) contrast-enhanced CT, and corresponding 18F-FDG PET/CT with (c) PET, (d) PET/CT fusion, and (e) the CT portion of the PET/CT images show the two large complex pelvic masses with evidence of invasion into the sigmoid bowel wall
A contrast-enhanced CT of the abdomen and pelvis (Figs. 1b, 2b) was performed to assess for possible pelvic lymphadenopathy. In addition to the known bilateral large complex adnexal masses, there was thickening of the anterior mesorectal fascia with numerous presacral, retroperitoneal, bilateral common iliac and internal obturator lymph nodes (the largest measuring 9 mm). There was some minimal mesorectal fat stranding. Several superior mesenteric nodes were also seen, the largest measuring 1 cm.
An 18F-FDG PET/CT (Discovery ST, GE Healthcare, Montreal, Canada) was performed 4 weeks following the MRI (Figs. 1c–e, 2c–e). The patient was fasted overnight prior to the examination, and waited in a quiet, dark room the morning of the scan. No muscle relaxants were given and no urinary bladder catheterization was performed. Oral contrast (barium) was administered. An 18F-FDG emission scan extending from the base of the skull to mid thighs was obtained 60 min after intravenous injection of 0.22 mCi/kg of 18F-FDG. Emission scans were acquired for 5 min per field of view, each covering 15 cm, at an axial sampling thickness of 3.75 mm/slice. The 16-slice helical CT acquisition was performed prior to a full-ring dedicated PET scan of the same axial image. The CT component was operated with an X-ray tube voltage peak of 140 kVp, 80 mA, a 1.75:1 pitch, a slice thickness of 3.75 mm and a rotational speed of 0.8 s per rotation. The patient was allowed to breathe normally during the PET and CT acquisitions. PET images were reconstructed with CT-derived attenuation correction using ordered subset expectation maximization (OSEM) software. Only the maximum standardized uptake value (SUVmax) was reported, corrected for body weight.
The PET/CT showed intense 18F-FDG uptake in the two pelvic masses, with an SUVmax of 13.3 in a 3.5-cm right adnexal mass, and SUVmax of 15.6 in a 5-cm left adnexal mass (Figs. 1c–e, 2c–e). There were no other FDG-avid abnormalities in the remainder of the scan, as can be seen on the whole body maximum intensity projection (MIP) images (Fig. 3). The PET/CT findings were suspicious for an ovarian or pelvic malignancy.
Fig. 3.
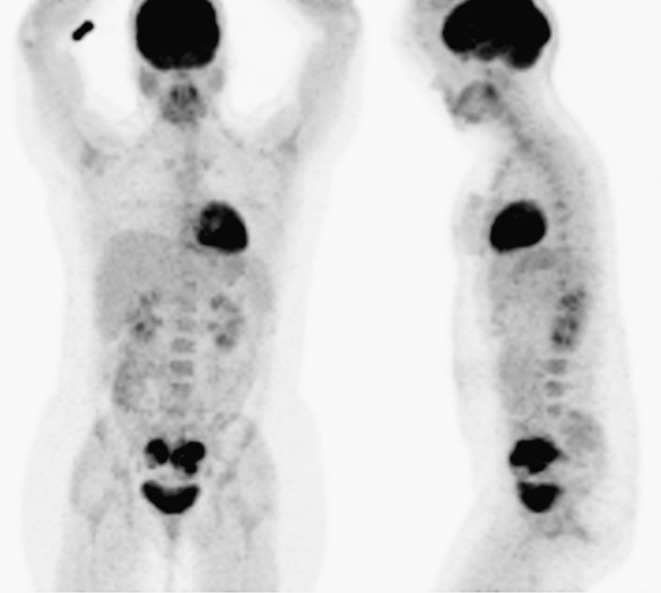
18F-FDG PET/CT whole-body maximum-intensity projection (MIP) images with anterior and left lateral views show the two intensely FDG-avid pelvic masses with no other FDG-avid abnormalities in the remainder of the body
Given the suspicious MRI and PET/CT findings, the patient had surgical removal of both ovarian masses as well as a wedge of the posterior aspect of the uterine body. Histopathological evaluation revealed a TOA with fragments of tissue with severe acute and chronic inflammation composed of neutrophils, lymphocytes, plasma cells, eosinophils and histiocytes. There was no evidence of malignancy. Micro-organism staining showed 1+ Gram-positive cocci, 2+ WBCs and 4+ RBCs. The culture of the abscess tissue grew Bacteroides fragilis and Streptococcus mitis (susceptible to clindamycin and resistant to erythromycin).
Discussion
Primary TOA is one of the most severe complications of PID and can lead to significant morbidity and occasional mortality. PID is a polymicrobial infection characterized by inflammation of the upper genital tract, including endometritis, salpingitis and pelvic peritonitis [6–9]. Secondary TOA can rarely arise from inflammatory bowel disease, diverticulitis, appendicitis, pelvic surgery, pelvic malignancy, or ovarian hyperstimulation [10, 11]. Only 50% of women with TOA present with fever and chills, symptoms that were absent in our patient. Other symptoms include abdominal pain, nausea, vaginal discharge and abnormal vaginal bleeding. Although most patients have an elevated WBC count, up to one quarter of patients can have a normal WBC count. The most common pathogens cultured from TOA include a mixed polymicrobial infection with a high prevalence of anaerobes such as Bacteroides species (as was present in our case) [9].
The most common CT finding of TOA is a pelvic mass with uniform thickened walls, internal septations, and indistinct margins. The mass contains fluid collections and the density of the fluid within these cystic structures is usually greater than fluid in the urinary bladder, representing pyogenic or hemorrhagic material [12, 13]. Fluid-filled tubular lesions with enhancing, thick walls are findings of pyosalpinx and are frequently seen adjacent to or in a portion of TOAs [14–16]. CT is generally not indicated for differential diagnosis of adnexal masses because of poor soft tissue discrimination (except for fatty tissue and calcification) and the disadvantages of irradiation [17].
On MRI, TOA usually appears as a pelvic mass with low signal intensity on T1-weighted images and heterogeneous high signal intensity on T2-weighted images. The signal intensity of the abscess content depends on its viscosity and protein content [18–20]. Tukeva et al. [12] showed the superior sensitivity and specificity of MRI for TOA of 100% and 90% respectively, compared with transvaginal ultrasound (sensitivity of 56% and specificity of 86%). Despite the significantly higher sensitivity and specificity of MRI compared with CT or ultrasound, there are occasional cases of TOA that have been reported to mimic benign or malignant tumors of the ovaries or pelvis. Unusual causes of TOA such as actinomycosis, tuberculosis and xanthogranulomatous inflammation are more frequently misdiagnosed as ovarian malignancies on CT and MRI. TOA from actinomycosis frequently has a solid appearance with linear, solid, well-enhancing lesions extending directly from the mass on CT and MRI. Tuberculous TOAs usually mimic peritoneal carcinomatosis from ovarian cancers [4]. The rectosigmoid colon and ureter are the most common organs that can be involved by TOA [21–23]. Rectal involvement was present in our case, which contributed to the suspicion of malignancy.
18F-FDG PET/CT has been used in the evaluation of a large variety of infectious processes, including abscesses [5]. On PET/CT, abscesses often exhibit a hypometabolic center surrounded by an intensely FDG-avid rim [24, 25], although they can also exhibit solid uniformly intense FDG uptake throughout, as in our case [26]. There is very little PET/CT literature on the imaging characteristics of TOA, with two reports mentioning elevated 18F-FDG uptake [27, 28]. In our case, the SUVmax of 15.6 was significantly higher than any that had been previously reported in the literature for TOA (SUVmax not reported in a FDG positive unilateral TOA [27], and SUVmax of 8.4 in a bilateral TOA [28]). This contributed to the strong suspicion of malignancy. Bilateral adnexal 18F-FDG uptake is rare, although it has been described as a normal physiologic uptake in premenopausal women [29, 30], in disseminated tuberculosis [31], ovarian cancer [32], ovarian lymphoma [33], and ovarian metastases from another primary malignancy [34, 35].
In conclusion, PET/CT readers should be aware of the imaging appearance of an intensely 18F-FDG-avid bilateral TOA, as a potentially serious pitfall in the evaluation of ovarian malignancies by 18F-FDG PET/CT, especially in the context of transvaginal ultrasound, CT or MRI findings suspicious for malignancy. Being aware of this potential benign differential diagnosis may influence how such a patient is subsequently managed, with regards to such important patient-related issues as maintaining fertility in the premenopausal woman.
References
- 1.Banikarim C, Chacko MR. Pelvic inflammatory disease in adolescents. Semin Pediatr Infect Dis. 2005;16:175–80. doi: 10.1053/j.spid.2005.04.006. [DOI] [PubMed] [Google Scholar]
- 2.Luedders DW, Chalvatzas N, Banz C, Horneman A, Diedrich K, Kavallaris A. Tubo-ovarian abscess in woman with an intrauterine device forgotten for 22 years. Gynecol Surg. 2010;7:181–4. doi: 10.1007/s10397-009-0550-y. [DOI] [Google Scholar]
- 3.Lee DC, Swaminathan AK, Sensitivity of ultrasound for the diagnosis of tubo-ovarian abscess: a case report and literature review. J Emerg Med 2011;40(2):170-5. [DOI] [PubMed]
- 4.Kim SH, Kim SH, Yang DM, Kim KA. Unusual causes of tubo-ovarian abscess: CT and MR imaging findings. Radiographics. 2004;24:1575–89. doi: 10.1148/rg.246045016. [DOI] [PubMed] [Google Scholar]
- 5.Stumpe KD, Dazzi H, Schaffner A, von Schulthess GK. Infection imaging using whole-body FDG-PET. Eur J Nucl Med. 2000;27:822–32. doi: 10.1007/s002590000277. [DOI] [PubMed] [Google Scholar]
- 6.Halperin R, Levinson O, Yaron M, Bukovsky I, Schneider D. Tubo-ovarian abscess in older women: is the woman’s age a risk factor for failed response to conservative treatment? Gynecol Obstet Invest. 2003;55:211–15. doi: 10.1159/000072076. [DOI] [PubMed] [Google Scholar]
- 7.Kamprath S, Merker A, Kühne-Heid R, Schneider A. Abdominal actinomycosis with IUD. Zentralbl Gynakol. 1997;119:21–4. [PubMed] [Google Scholar]
- 8.Lareau SM, Beigi RH. Pelvic inflammatory disease and tubo-ovarian abscess. Infect Dis Clin North Am. 2008;22:693–708. doi: 10.1016/j.idc.2008.05.008. [DOI] [PubMed] [Google Scholar]
- 9.Landers DV, Sweet RL. Tubo-ovarian abscess: contemporary approach to management. Rev Infect Dis. 1983;5:876–84. doi: 10.1093/clinids/5.5.876. [DOI] [PubMed] [Google Scholar]
- 10.Govaerts I, Devreker F, Delbaere A, Revelard P, Englert Y. Short-term medical complications of 1500 oocyte retrievals for in vitro fertilization and embryo transfer. Eur J Obstet Gynecol Reprod Biol. 1998;77:239–43. doi: 10.1016/S0301-2115(97)00263-7. [DOI] [PubMed] [Google Scholar]
- 11.Gjelland K, Ekerhovd E, Granberg S. Transvaginal ultrasound-guided aspiration for treatment of tubo-ovarian abscess: a study of 302 cases. Am J Obstet Gynecol. 2005;193:1323–30. doi: 10.1016/j.ajog.2005.06.019. [DOI] [PubMed] [Google Scholar]
- 12.Tukeva TA, Aronen HJ, Karjalainen PT, Molander P, Paavonen T, Paavonen J. MR imaging in pelvic inflammatory disease: comparison with laparoscopy and US. Radiology. 1999;210:209–16. doi: 10.1148/radiology.210.1.r99ja04209. [DOI] [PubMed] [Google Scholar]
- 13.Kalish GM, Patel MD, Gunn ML, Dubinsky TJ. Computed tomographic and magnetic resonance features of gynecologic abnormalities in women presenting with acute or chronic abdominal pain. Ultrasound Q. 2007;23:167–75. doi: 10.1097/RUQ.0b013e31815202df. [DOI] [PubMed] [Google Scholar]
- 14.Wilbur AC, Aizenstein RI, Napp TE. CT findings in tuboovarian abscess. AJR Am J Roentgenol. 1992;158:575–9. doi: 10.2214/ajr.158.3.1738998. [DOI] [PubMed] [Google Scholar]
- 15.Alonso RC, Nacenta SB, Martinez PD, Maria NI, Sanz LI, Galindez EZ. Role of multidetector CT in the management of acute female pelvic disease. Emerg Radiol. 2009;16:453–72. doi: 10.1007/s10140-009-0808-8. [DOI] [PubMed] [Google Scholar]
- 16.Du TQ, Xu QY, Dong J, Ding BZ, CT differential diagnosis between tubovarian abscess and benign ovarian tumors. Chinese J Med Imaging 2007; 06.
- 17.Togashi K. Ovarian cancer: the clinical role of US, CT and MRI. Eur Radiol. 2003;13(Suppl 4):L87–104. doi: 10.1007/s00330-003-1964-y. [DOI] [PubMed] [Google Scholar]
- 18.Ha HK, Lim GY, Cha ES, Lee HG, Ro HJ, Kim HS, et al. MR imaging of tubo-ovarian abscess. Acta Radiol. 1995;36:510–14. [PubMed] [Google Scholar]
- 19.Ueda H, Togashi K, Kataoka ML, Koyama T, Fujiwara T, Fujii S, et al. Adnexal masses caused by pelvic inflammatory disease: MR appearance. Magn Reson Med Sci. 2002;15:207–15. doi: 10.2463/mrms.1.207. [DOI] [PubMed] [Google Scholar]
- 20.Takeshita T, Ninoi T, Doh K, Hashimoto S, Inoue Y, Diffusion-weighted magnetic resonance imaging in tubo-ovarian abscess: a case report. Osaka City Med 2009; J 55:109–14. [PubMed]
- 21.Wilbur A. Computed tomography of tuboovarian abscesses. J Comput Assist Tomogr. 1990;14:625–8. doi: 10.1097/00004728-199007000-00021. [DOI] [PubMed] [Google Scholar]
- 22.Sam JW, Jacobs JE, Birnbaum BA. Spectrum of CT findings in acute pyogenic pelvic inflammatory disease. Radiographics. 2002;22:1327–34. doi: 10.1148/rg.226025062. [DOI] [PubMed] [Google Scholar]
- 23.Prakash RK, Shah N, Ferguson DR. A tubo-ovarian abscess that perforates the sigmoid colon. Clin Gastroenterol Hepatol. 2010;8(6):A26. doi: 10.1016/j.cgh.2009.11.008. [DOI] [PubMed] [Google Scholar]
- 24.Ichiya Y, Kuwabara Y, Sasaki M, Yoshida T, Akashi Y, Murayama S, et al. FDG-PET in infectious lesions: the detection and assessment of lesion activity. Ann Nucl Med. 1996;10:185–91. doi: 10.1007/BF03165391. [DOI] [PubMed] [Google Scholar]
- 25.Shreve PD, Anzai Y, Wahl RL. Pitfalls in oncologic diagnosis with FDG PET imaging: physiologic and benign variants. Radiographics. 1999;19:61–77. doi: 10.1148/radiographics.19.1.g99ja0761. [DOI] [PubMed] [Google Scholar]
- 26.Bleeker-Rovers CP, Warris A, Drenth JP, Corstens FH, Oyen WJ, Kullberg BJ. Diagnosis of candida lung abscesses by 18F-fluorodeoxyglucose positron emission tomography. Clin Microbiol Infect. 2005;11:493–5. doi: 10.1111/j.1469-0691.2005.01155.x. [DOI] [PubMed] [Google Scholar]
- 27.Ho KC, Lai CH, Wu TI, Ng KK, Yen TC, Lin G, et al. 18F-fluorodeoxyglucose positron emission tomography in uterine carcinosarcoma. Eur J Nucl Med Mol Imaging. 2008;35:484–92. doi: 10.1007/s00259-007-0533-z. [DOI] [PubMed] [Google Scholar]
- 28.Kim Y, Kim S, Lee JW, Lee SM, Kim TS. Ovarian mass mimicking malignancy: a case report. Nucl Med Mol Imaging. 2010;44:290–3. doi: 10.1007/s13139-010-0045-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lerman H, Metser U, Grisaru D, Fishman A, Lievshitz G, Even-Sapir E. Normal and abnormal 18F-FDG endometrial and ovarian uptake in pre- and postmenopausal patients: assessment by PET/CT. J Nucl Med. 2004;45:266–71. [PubMed] [Google Scholar]
- 30.Kim SK, Kang KW, Roh JW, Sim JS, Lee ES, Park SY. Incidental ovarian 18F-FDG accumulation on PET: correlation with the menstrual cycle. Eur J Nucl Med Mol Imaging. 2005;32:757–63. doi: 10.1007/s00259-005-1771-6. [DOI] [PubMed] [Google Scholar]
- 31.Das CJ, Kumar R, Balarishnan VB, Chawla M, Malhotra A. Disseminated tuberculosis masquerading as metastatic breast carcinoma on PET-CT. Clin Nucl Med. 2008;33:359–61. doi: 10.1097/RLU.0b013e31816a858e. [DOI] [PubMed] [Google Scholar]
- 32.Yoshida Y, Kurokawa T, Kawahara K, Tsuchida T, Okazawa H, Fujibayashi Y, et al. Incremental benefits of FDG positron emission tomography over CT alone for the preoperative staging of ovarian cancer. AJR Am J Roentgenol. 2004;182:227–33. doi: 10.2214/ajr.182.1.1820227. [DOI] [PubMed] [Google Scholar]
- 33.Komoto D, Nishiyama Y, Yamamoto Y, Monden T, Sasakawa Y, Toyama Y, et al. A case of non-Hodgkin’s lymphoma of the ovary: usefulness of 18F-FDG PET for staging and assessment of the therapeutic response. Ann Nucl Med. 2006;20:157–60. doi: 10.1007/BF02985629. [DOI] [PubMed] [Google Scholar]
- 34.Ho L, Quan V, Henderson R. Bilateral ovarian metastases from breast carcinoma on FDG PET-CT. Clin Nucl Med. 2007;32:935–6. doi: 10.1097/RLU.0b013e318159663f. [DOI] [PubMed] [Google Scholar]
- 35.Henley T, Reddy MP, Ramaswamy MR, Lilien DL. Bilateral ovarian metastases from colon carcinoma visualized on F-18 FDG PET scan. Clin Nucl Med. 2004;29:322–3. doi: 10.1097/01.rlu.0000124010.98447.47. [DOI] [PubMed] [Google Scholar]