Abstract
The production and consumption of the greenhouse gases (GHGs) methane (CH4), carbon dioxide (CO2) and nitrous oxide (N2O) in soil profile are poorly understood. This work sought to quantify the GHG production and consumption at seven depths (0–30, 30–60, 60–90, 90–150, 150–200, 200–250 and 250–300 cm) in a long-term field experiment with a winter wheat-summer maize rotation system, and four N application rates (0; 200; 400 and 600 kg N ha−1 year−1) in the North China Plain.
The gas samples were taken twice a week and analyzed by gas chromatography. GHG production and consumption in soil layers were inferred using Fick’s law. Results showed nitrogen application significantly increased N2O fluxes in soil down to 90 cm but did not affect CH4 and CO2 fluxes. Soil moisture played an important role in soil profile GHG fluxes; both CH4 consumption and CO2 fluxes in and from soil tended to decrease with increasing soil water filled pore space (WFPS). The top 0–60 cm of soil was a sink of atmospheric CH4, and a source of both CO2 and N2O, more than 90% of the annual cumulative GHG fluxes originated at depths shallower than 90 cm; the subsoil (>90 cm) was not a major source or sink of GHG, rather it acted as a ‘reservoir’. This study provides quantitative evidence for the production and consumption of CH4, CO2 and N2O in the soil profile.
Introduction
Atmospheric concentrations of carbon dioxide (CO2), methane (CH4) and nitrous oxide (N2O) have increased considerably since the industrial revolution, and are still increasing annually by about 0.5%, 1.1% and 0.3%, respectively [1]. Worldwide concerns about the increased greenhouse gases (GHGs) concentrations in the atmosphere and its effects on our future environment require a better understanding of the cause of these emissions [2]. Agricultural lands occupy 37% of the earth’s land surface; about 13.5% of global anthropogenic GHG was emitted from agricultural production [1]. It was estimated that 84% of N2O and 52% of CH4 emitted from agriculture activities [3]. In China, agriculture tends to produce more emissions than the global average over the last 30 years due to increased chemical and manure N inputs. Gaining a better understanding of GHG production and emission processes, and developing methods for mitigating emissions from agroecosystems are essential steps in order to mitigate climate change [4].
Agricultural soils are main sources and sinks of GHG emissions, depending on their characteristics and management. Many studies have been conducted to quantify the net fluxes of CO2, CH4, and N2O across the soil/atmosphere interface [3], [5]–[9]. These studies provide an integrative estimate of the net production and consumption of CH4, CO2 and N2O in the soil, but do not provide information on the depth-distribution of CH4, CO2 and N2O production-consumption patterns within soil profiles. It has been suggested that subsurface processes exert a significant control on carbon (C) and nitrogen (N) dynamics and hence on CO2, CH4, and N2O emissions from soil [10], but few studies have elucidated the role of the subsoil so far. Understanding these processes might also provide a better insight into the possibilities and effectiveness of measures to reduce GHG emissions. For example, a temporary accumulation of GHG in the soil profile influences GHG flux patterns at the soil surface over time, and thereby may confuse empirical relationships between agricultural activities and measured GHG emissions [11]. Thus, measurements of CH4, CO2 and N2O concentration profiles may be helpful for increasing the understanding of the net exchanges of these gases between soil and atmosphere.
Though few studies have examined the production and consumption of CO2 [12], [13], CH4 [14] and N2O [15] within individual soil horizons and their transports between soil horizons so far, very few studies have made combined measurements of the dynamics of CO2, CH4 and N2O production and emission processes in soil profiles in agro-ecosystems, especially in China [16]. It has been well-established that N fertilizer applications increase crop growth and N2O emissions, and tend to decrease CH4 emissions into the atmosphere, but there is little information about the combined effects of N fertilizer application and irrigation on subsoil N2O, CO2 and CH4 production, consumption and transport.
Recently, Wang et al [16] presented bi-weekly measured CH4, CO2 and N2O concentration profiles down to a depth of 300 cm in a winter wheat-summer maize rotation in the North China Plain, with four N application rates (0; 200; 400 and 600 kg N ha−1). Here, we build on the results of that study, and present calculated subsurface fluxes of CH4, CO2 and N2O over a whole-year period. The purpose of this study is to evaluate the effects of seasonal cropping, N applications, irrigation, soil temperature, and soil moisture on net subsurface transport of CO2, N2O and CH4.
Materials and Methods
Site description
The study was conducted at Luancheng Agroecosystem Experimental Station (37°53′N, 114°41′E, elevation 50 m), Chinese Academy of Sciences. This area is at the piedmont of the Taihang Mountains, in the North China Plain. Mean annual precipitation is about 480 mm, 70% of which is in the period from July to September. Annual average air temperature is 12.5 °C. The dominant cropping system in the region is a winter wheat-summer maize double-cropping system (two crops harvested in a single year) without fallow between the crops.
Field experimental design
The field experiment with a randomized complete block design was laid down in a winter wheat (Triticum aestivum L. Wheat variety Kenong 199)-summer maize (Zea mays L. Maize variety Xianyu 335) double-cropping system in 1998. It had four N fertilizer (urea) application treatments in triplicate: 0 (N0), 200 (N200), 400 (N400) and 600 (N600) kg N ha−1 year−1. Plot size was 7 m×10 m. Results of the present study refer to the period March 2007 to January 2008. Details on fertilizer application and crop management activities are presented in Table 1. Crops are flood-irrigated with pumped groundwater about five times per year, depending on rainfall distribution.
Table 1. Fertilization treatments (A); and timing of crop management activities (B).
| (A) Treatments | Basal fertilization, applied at wheat sowing (kg·ha−1) | Supplementary N fertilization (kg·ha−1) | |||
| N | P2O5 | K2O | Wheat (in April) | Maize (in July) | |
| N0 | 0 | 65 | 0 | 0 | 0 |
| N200 | 50 | 65 | 0 | 50 | 100 |
| N400 | 100 | 65 | 0 | 100 | 200 |
| N600 | 150 | 65 | 0 | 150 | 300 |
Soil sampling, analysis and climate data collection
The soil has a silt-loam texture in the upper 90 cm and clay-loam to clay texture at depth of 90–300 cm (Table 2). All soil samples were collected from different depths of the soil profile before the GHG measurements (on 5 December 2006); soil samples were mixed to make a specific representative soil sample for each depth; and all analyses of soil chemical properties in Table 2 were based on the standard methods for soil analyses described by Sparks [17]. Soil bulk density was determined using the cutting ring method. Soil particle size analysis was done by the Bouyoucos Hydrometer Method [18]. Soil pH was measured in a suspension of 5 g soil with 25 ml distilled water after 1 h after shaking.
Table 2. Soil characteristics at the experimental site in 2007.
| Depth (cm) | pH (H2O) | Sand (%) | Silt (%) | Clay (%) | Textural classa | Dry bulk Density (g cm−3) | Total organic matter (g kg−1) | Total nitrogen (g N kg−1) | Available nitrogen (mg N kg−1) | Available phosphorus (mg P kg−1) | Available potassium (mg K kg−1) |
| 0–30 | 8.5 | 25 | 58 | 17 | SSL | 1.47 | 16.7 | 1.40 | 148 | 4.1 | 79.3 |
| 30–60 | 7.74 | 22 | 60 | 18 | L | 1.40 | 10.9 | 0.85 | 72.0 | 2.1 | 54.4 |
| 60–90 | 7.78 | 31 | 55 | 14 | L | 1.45 | 7.8 | 0.64 | 70.1 | 0.44 | 36.9 |
| 90–150 | 7.76 | 15 | 59 | 26 | SCL | 1.57 | 6.5 | 0.38 | 49.8 | 0.17 | 27.8 |
| 150–200 | 7.74 | 18 | 47 | 35 | GCL | 1.43 | 5.4 | 0.28 | 38.9 | 0.13 | 21.9 |
| 200–250 | 7.77 | 15 | 35 | 50 | C | 1.51 | 4.2 | 0.15 | 30.1 | 0.09 | 13.9 |
| 250–300 | 7.75 | 12 | 35 | 53 | C | 1.50 | 3.0 | 0.86 | 25.9 | 0.04 | 7.1 |
SSL: Silty sandy loam; L: Loam; SCL: Silty clay loam: GCL: Gravely clay loam; C = Clay.
Soil core samples were collected from different depths (0–30, 30–60, 60–90, 90–150, 150–200, 200–250, 250–300 cm) of the soil profile in the farmland described above on 5 December 2006 (before the GHG measurements), 16 June 2007 (after the winter wheat harvest) and 11 October 2007 (after the summer maize harvest), respectively. Three different sub-samples, taken from a cross-section around the soil auger (3 meter in length), were mixed to make a specific representative soil sample for each depth from each point. The soil profile samples were sealed in dark plastic bags immediately after sampling and stored at 4°C until NO3 extraction. Samples of soil NO3-N were extracted with 1 M KCl solution (1∶5 w/v) by shaking for 1 h. The extracts were then filtered and the concentrations of NO3-N in the soil extracts were measured colorimetrically using a UV spectrophotometer (UV-2450, Shimadzu, Japan). Each measurement was replicated three times.
Soil temperature was measured using seven CS107b soil temperature probes (Cambell Scientific Inc., Logan, UT) installed at depths of 30, 60, 90, 150, 200, 250 and 300 cm. Three-meter neutron access tubes were installed at each plot. Soil moisture at seven depths (30, 60, 90, 150, 200, 250 and 300 cm.) was measured using a neutron moisture meter when gas samples were collected. Soil temperature and water content were used to explore the relationships between calculated CO2, N2O and CH4 fluxes and soil water-filled pore space (WFPS) and soil temperature at various depths. Daily rainfall was recorded at a weather station on the experimental site.
Soil gas sampling and measurements
Measurements of CO2, N2O and CH4 concentrations in soil started in March 2007, i.e., 9 years after the start of the field experiment, assuming that by then the CO2, N2O and CH4 production-consumption dynamics in the subsoil had been adjusted to the experimental treatments. Seven subsurface soil air equilibration tubes were installed at each site with sampling ports at 30, 60, 90, 150, 200, 250 and 300 cm in December 2006 (for more details, see reference 16). Soil-air samples were taken twice a week between 9:00 AM and 11:00 AM, using 100 ml plastic syringes connected to the tubes via the three-way stopcocks at the surface. The surface air was concurrently sampled at a height of 5 cm above the soil surface. The gas samples were analyzed by gas chromatography (Agilent GC-6820, Agilent Technologies Inc. Santa Clara, California, US) with separate electron capture detector (ECD at 330°C) for N2O determination and flame ionization detector (FID at 200°C) for CH4 and CO2 determinations.
Calculations of gas fluxes
The basic method of our study followed that of Campbell [19]. It was assumed that the soil conditions are uniform in horizontal direction, and that the gas diffusion in soil is in one-dimensional vertical flow, that fundamentally follows Fick’s law [20], [21]:
| (1) |
Where q is the gas flux density (g gas m−2 soil s−1), Dp is the soil-gas diffusivity (m3 soil air m−1 soil s−1), 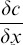 is the concentration gradient between two soil layers (g gas m−3 soil air m−1 soil).
is the concentration gradient between two soil layers (g gas m−3 soil air m−1 soil).
Dp was derived from the following equation [22], [23]:
| (2) |
Where, D0 is the gas diffusivity (m2 air s−1). We estimated the diffusion coefficient D0 of CH4, CO2 and N2O at 298 K and 1 kPa at 1.79×10−5, 1.32×10−5, 1.29×10−5 m2 s−1, respectively, by using a semiempirical equation by Gilliland et al [24]. Parameter ε is the soil air filled porosity (m3 air m−3 soil), and E is the soil porosity (m3 voids m−3 soil).
The Millington-Quirk model was used to compute ε and E [25]:
| (3) |
| (4) |
Where ρb is the dry bulk density (g m−3) at each soil depth (Table 2), ρs is the average bulk density of surface soil (2.65 g m−3); θ is the volumetric soil water content which was measured using a neutron moisture meter at each depth.
Calculations of annual cumulative gas fluxes
The annual cumulative emissions were obtained by multiplying the average daily flux from two consecutive measurements within a week by the number of days between the measurements, and then summing the fluxes of these periods to an accumulative flux for the whole year [26]:
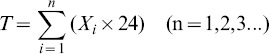 |
(5) |
Where: T (kg ha−1), Xi (kg ha−1 h−1) and i are the accumulative GHG emission, the average daily GHG emission rate, and the number of days, respectively.
Data analyses
All data were subjected to statistical analysis (SPSS 13.0). Differences between treatments were analyzed using ANOVA, followed by LSD at the 0.05 probability level. Regression analysis was used to identify relationships between CH4, CO2 and N2O fluxes and the climatic variables.
Results
Concentrations of CH4, CO2 and N2O
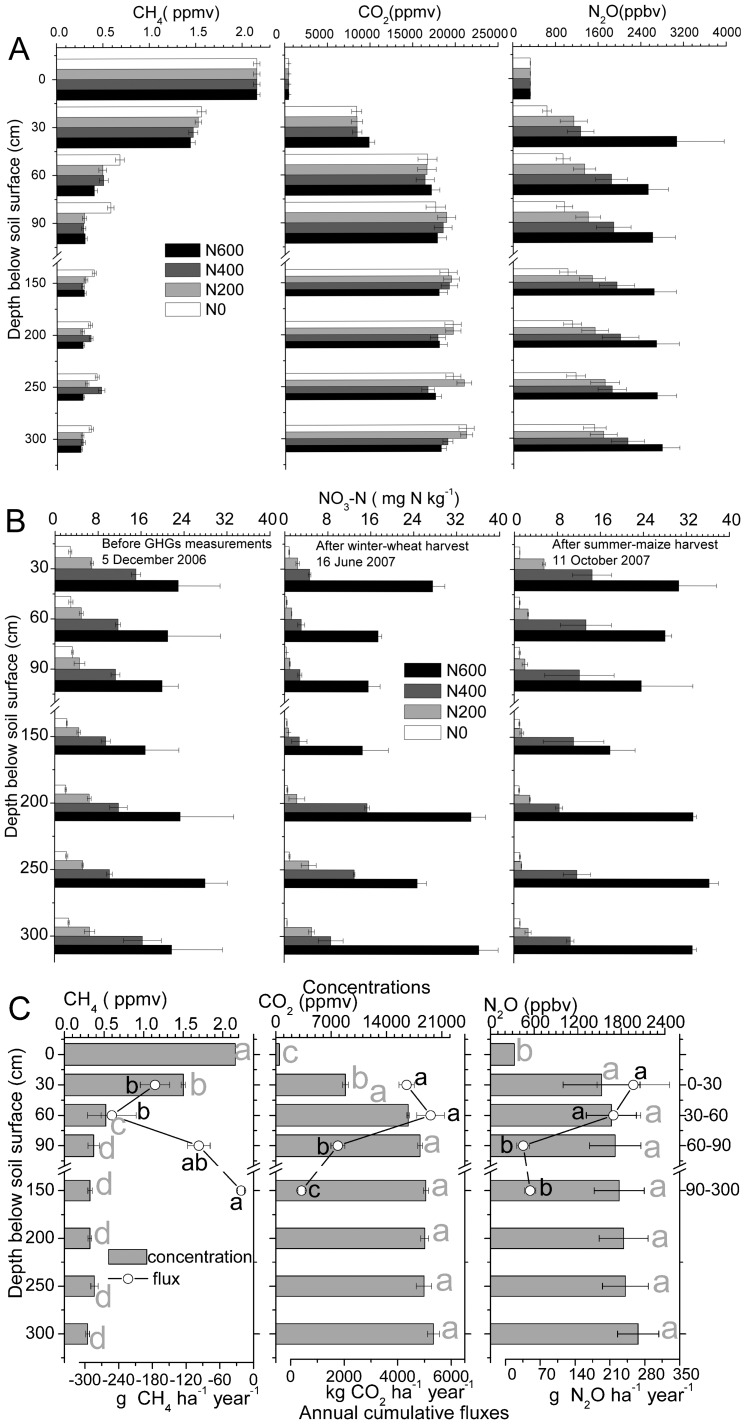
Mean concentrations and its standard deviations of CH4, CO2 and N2O at each depth are shown in Figure 1 A, as function of N application rates. Mean CH4 concentration decreased with soil depth. Ambient air CH4 concentration in the area was about 2.2 ppmv. At a depth of 30 cm, CH4 concentration ranged between 1.4 and 1.6 ppmv and at depth of 60 to 300 cm between 0.3 and 0.6 ppmv. There were no clear effects of N fertilizer application on the mean CH4 concentration (Figure 1 A). Mean CH4 concentrations decreased significantly at soil depths of 0, 30, 60 and 90 cm (P<0.05); changes in mean concentration below a depth of 90 cm were not significant (Figure 1 C).
Figure 1. CH4, CO2 and N2O concentrations (mean ± standard deviations, n = 3) in soil air at various soil depths in a winter wheat–summer maize double cropping rotation receiving 0, 200, 400 and 600 kg of N ha−1 year−1, in 2007–2008 (A); NO3-N contents (mean ± standard deviations, n = 3) at various soil depths as function of N fertilizer application rate, in 2007–2008 (B); Profiles of concentration and annual cumulative flux of CH4, CO2 and N2O, in 2007–2008 (mean ± standard deviations, n = 4).
Same letters next to the bars indicated no significant differences between slope positions (P<0.05). (C). Note the differences in X-axes.
Mean CO2 concentration increased with soil depth. At a depth of 30 cm, CO2 concentration ranged between 8400 and 9900 ppmv and at depth of 60 to 300 cm between 16000 and 21000 ppmv. Mean CO2 concentrations increased significantly at soil depths of 0, 30 and 60 cm (P<0.05); changes in mean concentration below a depth of 60 cm were not significant (Figure 1 C). There were no clear effects of N fertilizer application on the mean CO2 concentrations.
Concentrations of N2O were strongly influenced by agricultural management activities such as N application and irrigation. Fertilizer N applications increased the mean N2O concentrations. Mean N2O concentrations at depth of 30 to 300 cm ranged from 600 to 1500, 1100 to 1700, 1600 to 2100 and 2500 to 3000 ppbv for the N0, N200, N400 and N600 treatments, respectively (Figure 1 A). Mean N2O concentrations increased significantly from soil surface to a depth of 30 cm (P<0.05), but changes in mean concentration below a depth of 30 cm were not significant (Figure 1 C). Fertilizer N application increased soil NO3-N content; differences in mean N2O concentrations were correlated with differences in mean NO3-N contents in the four fertilizer N treatments (Figure 1 B).
Fluxes of CH4 in soil
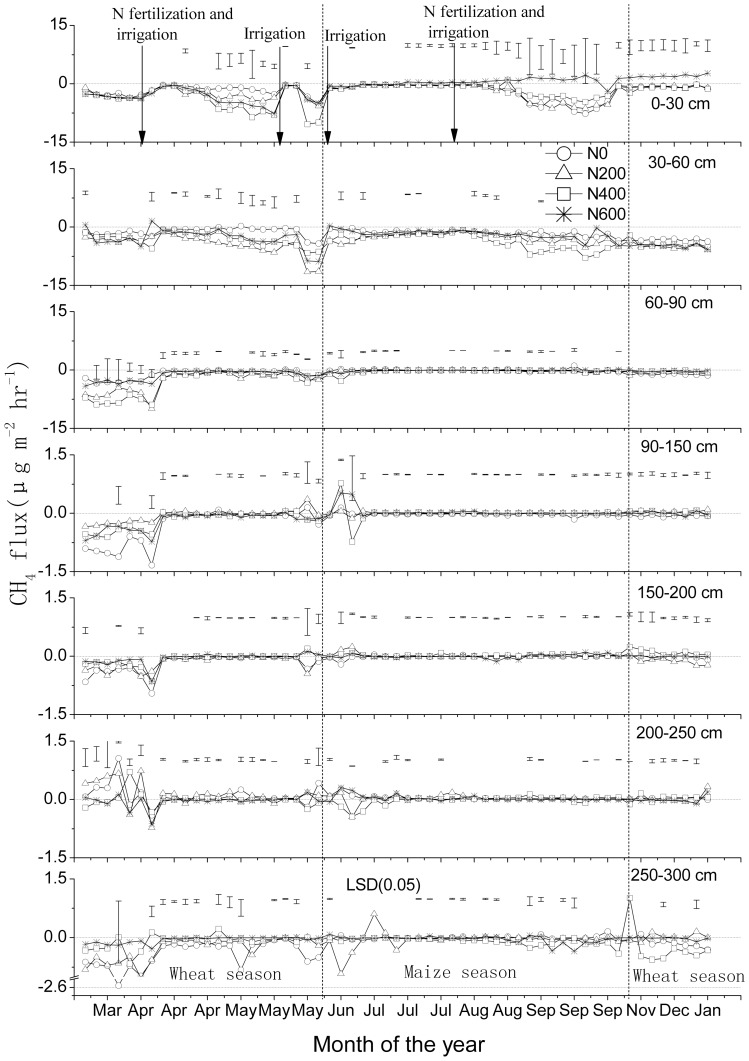
Diffusive fluxes between soil layers and between soil and atmosphere were calculated from the concentration gradients, using equation 1. There was a net influx of atmospheric CH4 into the top 0–60 cm (Figure 2), suggesting consumption of CH4 by methanotropic bacteria. Interestingly, the calculated fluxes into the soil were rather similar for the 0–30 and 30–60 cm soil layers, suggesting similar CH4 uptake rates. Uptake of CH4 apparently also occurred in the layers 60–90, 90–150 and 150–200 cm during the first one or two months of the measurement period (Figure 2). However, we cannot exclude the possibility that this apparent uptake of CH4 in the subsoil during the first two months is an artifact related to the installation of the samplers when atmospheric CH4 may have diffused into the subsoil. Fluxes between soil layers were negligible small during most of the maize growing season (Figure 2).
Figure 2. CH4 flux rates (means ± standard deviations, n = 3) at various soil depths in a winter wheat-summer maize double cropping rotation receiving 0, 200, 400 and 600 kg of N ha−1 year−1, in 2007–2008.
Vertical dashed lines indicate a change in crop. Bars in figures indicate 1 standard deviation (n = 3). Note the differences in Y-axes.
Annual cumulative fluxes of CH4 for all soil layers and N fertilizer treatments are shown in Table 3 A. Evidently, the influx of atmospheric CH4 decreased with soil depth. During the study period, mean calculated uptake was about 176 g CH4 per ha by the top 30 cm, 252 g CH4 per ha by the soil layer 30–60 cm, 98 g CH4 per ha by the layer 60–90 cm and 22 g CH4 per ha below a depth of 90 cm; mean calculated uptakes in the layers 0–30 and 30–60 cm were both significantly higher than that in the layer 90–300 cm (P<0.05) (Figure 1 C). Annual cumulative CH4 uptake in the layer 0–90 cm (526 g CH4 per ha per year) contributed about 96% to that in the layer 0–300 cm (547 g CH4 per ha per year). Annual cumulative uptake in 0–30 cm layer is relatively low compared to literature data [4]–[6], [27].
Table 3. Annual cumulative emissions of CH4 and N2O (in g ha−1 yr−1) and of CO2 (in kg ha−1 yr−1) between soil layers.
| (A) CH4 | ||||||||
| Treatments | 0–30 cm | 30–60 cm | 60–90 cm | 90–150 cm | 150–200 cm | 200–250cm | 250–300 cm | 0–300cm |
| N0 | −167 (12) b | −138 (13) a | −63 (2) a | −15 (0.5) c | −9 (0.1) d | 5 (0.04) a | −21 (0.3) d | −408 |
| N200 | −201 (9) bc | −322 (10) c | −123 (3) b | −2 (0.07) a | −7 (0.5) c | 5 (0.08) a | −12 (0.6) c | −662 |
| N400 | −228 (3) c | −318 (8) c | −140 (10) b | −6 (0.5) b | −1 (0.1) a | −1 (0.06) b | −10 (0.4) b | −704 |
| N600 | −106 (15) a | −231 (5) b | −64 (7) a | −6 (0.5) b | −2 (0.1) b | −1 (0.05) b | −4 (0.1) a | −414 |
Values (means with SE in the brackets) followed by the same letter are not significantly different within columns (one-way ANOVA with LSD; P<0.05)
Fluxes of CO2 in soil
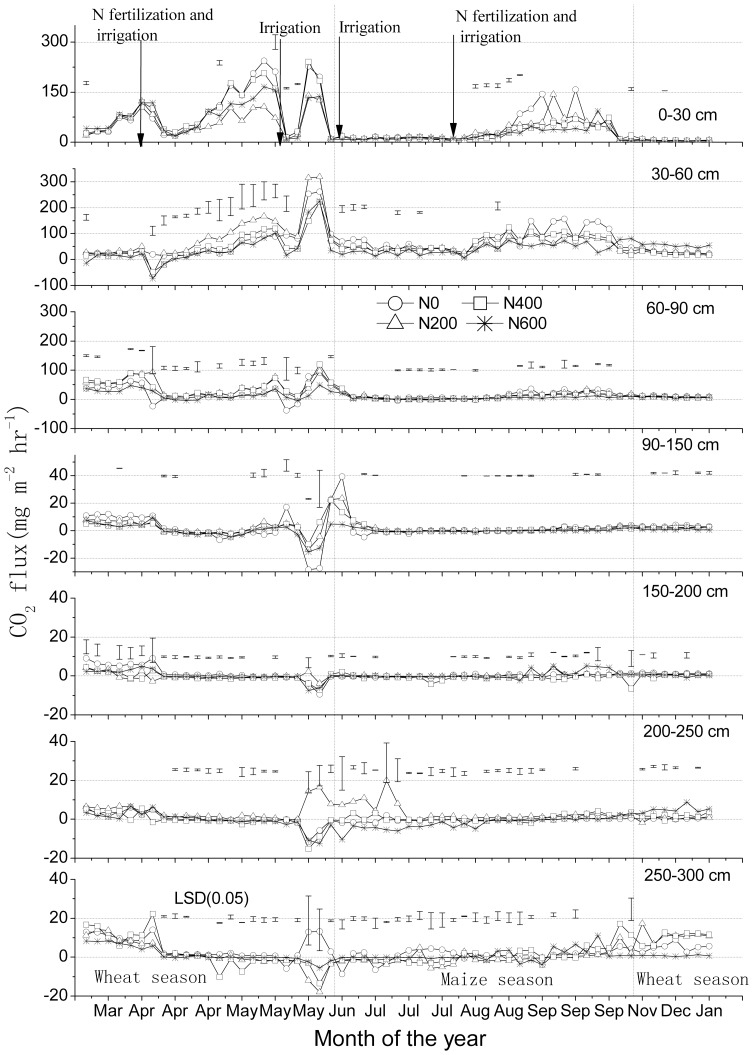
There was a large efflux of CO2 from the top 30 cm of soil to the atmosphere from March till June, i.e., during the second half of the wheat growing season, and from August till October, i.e., during the second half of the maize growing season (Figure 3). The same holds for the upward flux from the layer 30–60 to the layer 0–30 cm. These patterns were related to the crop growing seasons of wheat and maize, and to the changes in water filled pore space (WFPS) and soil temperature. There were no clear relationships between N treatments and CO2 fluxes. Treatment N200 had the smallest flux from the layer 0–30 cm to the atmosphere, but the largest from 30–60 cm to 0–30 cm from April to May. Upward fluxes from the layers 60–90 cm and especially below this layer were much smaller. There were small but significant changes in fluxes in the subsoil at the transition of the winter-wheat growing season to the summer-maize growing season (Figure 3).
Figure 3. CO2 flux rates (means ± standard deviations, n = 3) at various soil depths in a winter wheat-summer maize double cropping rotation receiving 0, 200, 400 and 600 kg of N ha−1 year−1, in 2007–2008.
Vertical dashed lines indicate a change in crop. Bars in figures indicate 1 standard deviation (n = 3). Note the differences in Y-axes.
Surprisingly, annual cumulative fluxes of CO2 tended to decrease with increasing N fertilizer application rates (Table 3 B). Moreover, cumulative upward fluxes were somewhat larger from the layer 30–60 cm (mean 5,227; range 3,800–6,000 kg CO2 per ha) than from the layer 0–30 cm to the atmosphere (mean 4,331; range 3,800–5,000 kg CO2 per ha) (Figure 1 C; Table 3 B). This suggests that a relatively large portion of total respiration in soil took place in the layer 30–60 cm. However, we can not exclude the possibility that the calculated CO2 efflux from the top layer is underestimated, because the concentration gradient in the upper 0–30 cm soil layer was averaged, and soil diffusivity may be higher in the top few cm than the bulk of the top 30 cm of soil [28]. Annual cumulative CO2 flux in the layer 0–90 cm (11,327 kg CO2 per ha per year) contributed about 97% to that in the layer 0–300 cm (11,744 kg CO2 per ha per year). Mean calculated fluxes in the layers 0–30, 30–60 and 60–90 cm were all significantly higher than that in the layer 90–300 cm (P<0.05); mean annual cumulative fluxes from the soil below 90 cm were very small and in upwards direction (Figure1 C; Table 3 B).
Fluxes of N2O in soil
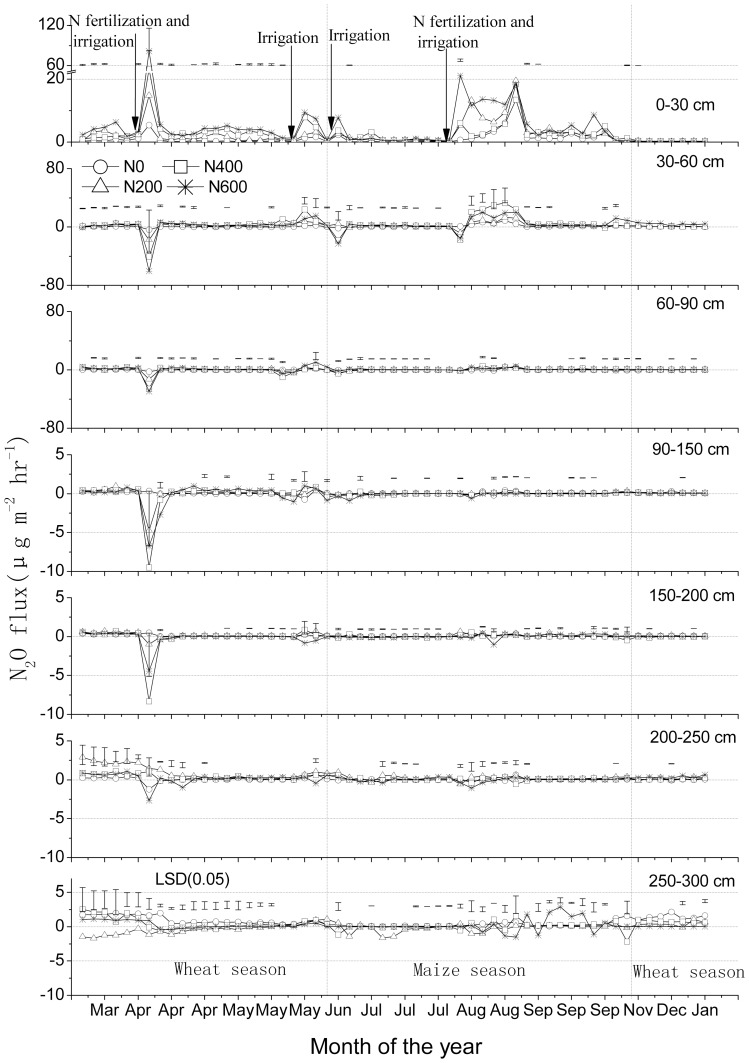
Fertilizer application, irrigation and precipitation events triggered an efflux of N2O from the topsoil to the atmosphere (Figure 4). The peak efflux, associated with the supplemental N fertilizer application and flooding in early April (wheat growing season), was accompanied with significant downward directed fluxes below the topsoil layer (0–30 cm). There was another relatively large efflux of N2O into the atmosphere during the relatively moist and warm August summer month (maize growing season) (Figures 4 and 5), but this peak was not accompanied with significant downward directed fluxes below the topsoil layer. In the subsoil, fluxes were relatively small and directions variable (Figure 4). Essentially all seasonal fluctuations of N2O flux rates in the subsoil (60–200 cm) seem to be related to fertilizer application, irrigation and rainfall events and changes in WFPS; therefore, there was no clear evidence of N2O production in the subsoil after excluding these influence of interfering factors [16] .
Figure 4. N2O flux rates (means ± standard deviations, n = 3) at various soil depths in a winter wheat-summer maize double cropping rotation receiving 0, 200, 400 and 600 kg of N ha−1 year−1, in 2007–2008.
Vertical dashed lines indicate a change in crop. Bars in figures indicate 1 standard deviation (n = 3). Note the differences in Y-axes.
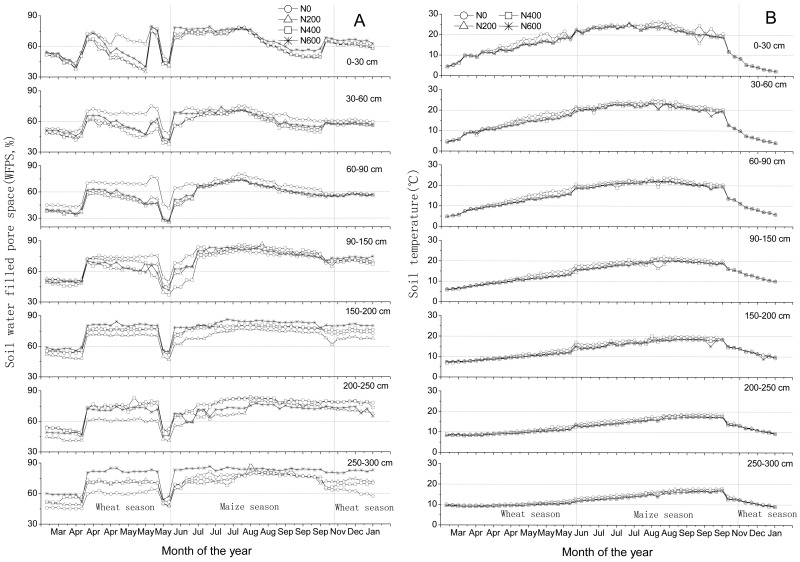
Figure 5. Water-filled pore space (WFPS) at various soil depths in a winter wheat-summer maize double cropping rotation receiving 0, 200, 400 and 600 kg of N ha−1 year−1, in 2007–2008.
Bars in figures indicate 1 standard deviation (n = 3). (A); Soil temperatures at various soil depths in winter wheat-summer maize double cropping rotation receiving 0, 200, 400 and 600 kg of N ha−1 year−1, in 2007–2008.(B)
Annual cumulative fluxes of N2O increased with increasing N fertilizer application rates; calculated total emissions at the soil surface were 93, 226, 263 and 447 g N2O per ha for the N0, N200, N400 and N600 treatments, respectively; net upward fluxes from the 30–60 cm layer were almost as large (90, 199, 358 and 222 g N2O per ha for the N0, N200, N400 and N600 treatments, respectively) as the fluxes from the 0–30 cm layer to the atmosphere (Table 3 C). Mean calculated fluxes in the layers 0–30 and 30–60 cm were both significantly higher than those in the layers 60–90 and 90–300 cm (P<0.05) (Figure 1 C); mean annual cumulative fluxes from the soil below 90 cm were small but mostly in upwards direction, suggesting that the subsoil was a small source of N2O, and/or that accumulated N2O from the previous season contributed to the net upward directed fluxes.
Relations between WFPS and temperature and CH4, CO2 and N2O fluxes
Linear regression relationships between WFPS and CH4 fluxes (positive) and between WFPS and CO2 fluxes (negative) were statistically significant (p<0.05) for almost all soil layers (Table 4). Uptake of CH4 by the soil was relatively high when WFPS was relatively low, probably because the diffusion rate of CH4 into the soil was high when soil was dry, and vice versa [6]. Similarly, the upward transport of CO2 was low when WFPS was high, and vice versa. This indicates that soil moisture exerted a dominant control on CH4 and CO2 fluxes. The linear relationship between WFPS and N2O flux was also significant for the soil layers 200–250 and 250–300 cm (p<0.05), but not for the other layers. Significant downward directed fluxes below the topsoil were only observed down to a deep of 200 cm (Figure 4); note that N2O fluxes were very low in 200–300 cm soil layer. Apparently, in 200–300 cm deep soil profile, soil moisture exerted a dominant control on nitrification and denitrification processes. But in 0–200 cm soil layer, WFPS was not the dominant controlling factor for the diffusive N2O flux, likely the combination of WFPS, ammonia, nitrate and metabolizable carbon, because these factors commonly control nitrification and denitrification processes.
Table 4. Linear regressions for the relationship between climatic variables and GHG fluxes.
| Climatic variable | Soil depth (cm) | CH4 (μg m−2 hr−1) | CO2(mg m−2 hr−1) | N2O (μg m−2 hr−1) |
| Soil water filled pore space | 0–30 | 0.853** | −0.775** | −0.149 |
| 30–60 | 0.645** | −0.372** | 0.084 | |
| 60–90 | 0.787** | −0.852** | 0.067 | |
| 90–150 | 0.637** | −0.447** | 0.093 | |
| 150–200 | 0.771** | −0.268* | 0.084 | |
| 200–250 | −0.146 | −0.289* | −0.692** | |
| 250–300 | 0.763** | −0.532** | −0.579** | |
| Soil temperature | 0–30 | −0.001 | 0.042 | 0.104 |
| 30–60 | 0.270* | 0.356** | 0.211 | |
| 60–90 | 0.635** | −0.348** | 0.093 | |
| 90–150 | 0.620** | −0.225 | 0.062 | |
| 150–200 | 0.575** | −0.266* | −0.075 | |
| 200–250 | −0.122 | −0.299* | −0.493** | |
| 250–300 | 0.473** | −0.323* | −0.263* |
Pearson's correlation coefficient, 2-tailed tests of significance.
**Significant correlation at a <0.01.
*Significant correlation at a <0.05.
Relationships between soil temperature and CH4, CO2 and N2O fluxes showed relatively large scatter (Table 4). Evidently, high temperatures are associated with the summer season, which is relatively moist (Figure 5). The significant relationships (p<0.05) between soil temperature and CH4 fluxes at depth of 60–300 cm may be the result in part of the covariance between WFPS and soil temperature. Fluxes of N2O in soil were not significantly related to soil temperature (Table 4).
Discussion
Fluxes of CH4, CO2 and N2O at the interface of soil and atmosphere are the net result of production, consumption and transport in the soil [11]. In this study, we inferred fluxes in the soil profile from changes in concentrations with depth and over time, so as to identify soil horizons of CH4, CO2 and N2O production and consumption, and thereby to increase the understanding of the dynamics of the net fluxes at the interface of soil and atmosphere. The study is unique in the sense that the inference of subsurface fluxes of CH4, CO2 and N2O in 300 cm deep soil profiles at high temporal resolution over a full year has not been reported before in such comprehensive manner.
Though the C and N cycles are intimately linked in the biosphere, there were significant differences in the dynamics of CH4, CO2 and N2O production, consumption and transport in the studied soil. The concentration profiles have distinct characteristics (Figure 1 A); the seasonal dynamics were much larger in the topsoil than subsoil. Moreover, the seasonal dynamics in inferred fluxes occurred during distinct periods (Figures 2, 3 and 4), and these were related to changes in WFPS (Figure 5 A), following rainfall and irrigation events. Fertilizer N application affected N2O fluxes greatly, but not those of CH4 and CO2. The soil under the winter wheat-summer maize double cropping system was a net sink of atmospheric CH4 and a net source of N2O. It was also a large source of CO2 but it is unknown whether the efflux compensated the influx of C into the soil via plant growth, as the latter influx was not measured.
The inferred fluxes at the soil-atmosphere interface (Figure 1 C; Table 3) were relatively small compared to those observed in other studies [5], [6], [29]. Our estimated soil surface fluxes are very likely underestimates because the depth resolution of the gas samplers in the top soil was too low to capture the curvature of the concentration profile properly. Hence, our study may have underestimated the dynamics of the CH4, CO2 and N2O fluxes in the top soil. The depth resolution of the sampling below 90 cm appeared to be adequate. Below, we discuss the dynamics of the CH4, CO2 and N2O fluxes in the soil profile in more detail.
CH4 flux
Application of fertilizer N has been shown to inhibit CH4 oxidation in soil [30], [31], and several studies noted that non amended soils act as sink of CH4 [32]–[34]. In our study, seasonal mean emission rates and annual cumulative fluxes of CH4 for all soil layers and N fertilizer treatments were consistently directed downward (Figures 1 C and 2; Table 3 A). Though statistical significant differences in cumulative CH4 fluxes between fertilizer N treatments were observed (Table 3 A), there was no clear trend that an increase in total N application decreased CH4 uptake by soil. Inferred uptake was higher in the N200 and N400 treatments than in the N0 and N600 treatments at depth of 30 to 90 cm.
The magnitude of methane uptake by soils is largely controlled by diffusion of atmospheric methane into the soil [35], which in turn is strongly influenced by soil moisture [28]. The rate of diffusion of CH4 in soil was high when WFPS was low. Our results showed a significant negative linear correlation between CH4 uptake rate and WFPS for almost all layers; and the highest CH4 uptake rates took place when WFPS was under 70% (Figures 2 and 5 A; Table 4). This is in agreement with the studies by Guo et al, Wu et al and Wang et al [4], [6], [16]. Inferred downward CH4 fluxes decreased with depth (Figures 1 C and 2; Table 3 A). It has been reported that methanotrophic activity is most pronounced in the top soil [4], [27], but our study suggests that significant uptake took place up to depths of 60 to 90 cm; below 90 cm, inferred fluxes of CH4 were negligibly small (Figure 1 C).
CO2 flux
Application of 200 kg fertilizer N per ha per year and more roughly doubled grain yields relative to the control treatment [16], but did not have statistical significant effects on the CO2 efflux from the soil and the diffusive flux in the soil profile (Figure 3; Table 3 B). Apparently, fertilizer N application affected predominantly aboveground biomass production, and not so much underground biomass production and respiration. Yet, we may have missed some of the topsoil dynamics, also because the incorporation of the stubbles by ploughing was in the top 15 cm of soil only. A relatively large portion of total respiration in soil took place in the layer 30–60 cm; and the total respiration in the layer was significantly higher than those in the layers 60–90 and 90–300 cm (P<0.05) (Figure 1 C).
When soil WFPS ranged between 40 and 70% and soil temperature was >10 °C (Figure 5), highest CO2 fluxes took place at depth of 0–60 cm during the second half of the growing seasons of wheat and maize, i.e., from mid-April to mid-May and from mid-August to mid-September (Figures 3 and 5 A). These elevated CO2 emissions are attributed to root respiration and to enhanced mineralization of soil organic matter by increased microbial activity [36], but also to changes in the stability and formation of soil aggregates and in the microbial community structure [37]. Sufficient soil moisture is needed to allow and support substrate diffusion to the sites of microbial activity. However, if soil moisture values exceed certain thresholds (which do depend on soil properties such as porosity, bulk density and SOC content) microbial soil respiration can get O2 limited due to diffusion constrains [6]. In a saturated soil, air is pushed out of soil pore spaces and root respiration further depletes O2 in the soil air [38], [39]. In our study, a significant negative linear correlation was found between CO2 flux rate and WFPS (40–70%) in all layers (Table 4); and low CO2 fluxes took place when WFPS exceeded 70%, especially after irrigation or heavy rainfall events, i.e., from June to August (Figures 3 and 5 A). This assertion is also supported by results presented by Davidson et al, Jassal et al and Fang et al [38]–[40].
Generally, the CO2 evolution from soil is directly correlated with soil temperature, though within a certain temperature range [41], [42], and depending on the presence of active roots [43]. In our study, the relationship of soil temperature and CO2 fluxes was variable, likely because of the dominant effect of WFPS (Table 4).
N2O flux
Nitrogen application and irrigation/rainfall are main triggers for increased N2O concentrations in a soil profile and for increased emissions [15], [16]. The top soil was the source of N2O production. The combined urea applications and irrigations in early April and by the end of July 2007 strongly increased NO3 − (Figure 1 B), NH4 + (data not shown) contents and WFPS (Figure 5 A) in soil, and induced large upward directed fluxes in the upper 0–30 cm soil layer; interestingly, relatively large downward directed fluxes in the subsoil only took place in April (up to the depths of 60 and 90 cm) (Figure 4), we cannot exclude the possibility that the apparent downward directed peaks during the first two months probably related to the soil structure disturbance that resulted from the installation of the samplers in December 2006.
In soil, N2O is mainly produced by nitrification and denitrification processes. The most important factors controlling these processes are NH4 + and NO3 − contents, O2 partial pressure, and available carbon to fuel heterotrophic denitrification [44], [45]. The rapid increases of WFPS (Figure 5 A), NO3-N content (Figure 1 B) and N2O productions in the subsoil (Figures 1 A and 4) would suggest that convective transport contributed to the downward transport of water and solutes (especially in the maize growing season), which is in line with other observations [2], [46]. For instance, significant upward directed fluxes of N2O took place during the relatively moist and warm August (maize growing season) than in the preceding wheat season in 30–60 cm soil layer (Figure 4). Several studies have demonstrated that higher values of soil moisture and temperature result in higher N2O fluxes [44]–[46] Also, Li et al found while carrying out a three-year field experiment at the same study sites that significant NO3- leaching events occur predominantly during August to October (maize growing season) [47]. Zhu et al found while carrying out a four-year field experiment in a hillslope cropland that soil NO3- concentrations in the subsurface soil (15–30 cm) were higher than in the topsoil (0–15 cm) during most of the maize season, indicating a rapid and effective transport of NO3- to the subsurface soil following over irrigation or rainfall events [48]. Our results indicate that NO3-N contents in soil layers after the maize harvest were higher than after the wheat harvest (Figure 1 B); but due to missing measurements, we can not fully rule out that high NH4 + content [47] may have contributed to relatively high N2O emissions in the warm and wet maize season. Although N2O concentration increased with soil depth, changes in inferred N2O flux below a depth of 60 cm were relatively small (Figures 1 A and 4). It may be related to the variation of the vertical N2O concentration gradient; changes in mean N2O concentration below a depth of 30 cm were not significant (Figure 1 C).
It has been frequently observed that high rates of N2O emissions take place when WFPS ranges between 30 and 70% [49]. According to Zou et al the N2O production in dry land soil of Northern China is mostly driven by nitrification [50]. Wang et al suggests that nitrification is likely a main source of the N2O production in the soil profile when WFPS varied between 45 and 70% at the study site [16]. N2 starts being emitted through denitrification at a WFPS of 70%, and is the main N gas emitted when WFPS exceeds 75% [51]. This may explain the relatively high inferred N2O flux from late April to mid-May (WFPS, 40–70%) and the very low flux from late June to late July (before applying nitrogen) (WFPS, >70%) in the layer 0–30 cm (Figures 4 and 5 A).
The accumulated N2O fluxes were significantly related to N application rate. This was most apparent in the top 30 to 90 cm of soil (Table 3 C). Annual cumulative N2O flux in the layer 0–90 cm (511 g N2O per ha per year) contributed about 90% to that in the layer 0–300 cm (560 g N 2O per ha per year). The 90 cm thick cinnamon top soil overlays the so-called Shajiang layer (90–140 cm) with silty clay loam texture [52]. The Shajiang layer has no crop roots, contains many iron-manganese nodules and has high bulk density (Table 2). This compacted subsoil may explain that fertilizer application and irrigation mainly affected N2O fluxes down to 90 cm (Figure 1 C; Table 3 C). In this study, calculated total emissions in the layer 0–90 cm were 206, 449, 644 and 743 g N2O per ha for the N0, N200, N400 and N600 treatments, respectively; these fluxes translate into fertilizer-derived emissions of 0.14, 0.10 and 0.07% for the N200, N400 and N600 treatments, respectively. The fertilizer induced emission factors (0.07–0.14%) were lower than the 0.30–0.39% measured by Ding et al [53] over the maize-wheat rotation year in a long-term mineral nitrogen addition field experiments (150–300 kg N ha−1 year−1, over 20-years) in the North China Plain. A reason for the lower fertilizer induced emission factor is probably related to the likely underestimates of soil surface N2O fluxes, because the concentration gradient in the upper 0–30 cm soil layer was averaged, and soil diffusivity may be higher in the top few cm than the bulk of the top 30 cm of soil [28]. Furthermore, due to missing measurements we can not fully rule out the indirect N2O emissions from leaching and atmospheric deposition [47], [53].
Conclusions
Our study is one of few that inferred CH4, CO2 and N2O transport between soil layers from changes in CH4, CO2 and N2O concentrations in the upper 300 cm of soil, measured at (bi)-weekly time intervals for one year in a winter wheat-summer maize double crop rotation. The top 30 to 60 cm of soil was a sink of atmospheric CH4, and a source of both CO2 and N2O. There was little or no evidence that the subsoil (>90 cm) acted as a sink or source of GHG; rather it acted as “reservoir”.
Nitrogen fertilizer application increased N2O fluxes but did not affect CH4 and CO2 fluxes. The fertilizer-derived N2O flux was small, likely because our sampling design may have missed N2O production in the top 15 cm of soil. This holds as well for the CH4 consumption by soil and the CO2 emissions from soil; both are likely underestimated. Soil moisture (WFPS) was found to play an important regulating role for CH4, CO2 and N2O fluxes in soil and between soil and atmosphere. Both CH4 consumption and CO2 fluxes in and from soil all tended to decrease with increasing WFPS.
More than 90% of the annual cumulative GHG fluxes originated at depths shallower than 90 cm. Mostly because the productive soil of our study site in the North China Plain had two distinct layers (0–90 and >90 cm), with different texture and bulk density. These differences showed up in characteristic differences in GHG concentration profiles and fluxes.
Acknowledgments
The authors would like to thank Luancheng Agroecosystem Experimental Station, Chinese Academy of Sciences for tireless efforts with maintaining the long-term fertilizer experiments. The authors would also like to thank two anonymous reviewers, whose suggestions and comments greatly improved the manuscript.
Funding Statement
This research is supported by the “Strategic Priority Research Program of Chinese Academy of Sciences” (Grant No. XDA0505050202 and XDA05050601) and the “National Science & Technology Pillar Program” (Grant No. 2012BAD14B07-5). It is also supported by the “National Basic Research Program of China” (Grant No. 2010CB833501); the “National Natural Science Foundation of China” (Grant No. 30970534) and the “Main Direction Program of Knowledge Innovation of Chinese Academy of Sciences” (Grant No. KSCX2-EW-J-5). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.IPCC (2007) Agriculture. In: Metz, B., D.O.R., Bosch P.R. (Eds.), Climate Change 2007: Mitigation, Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge. United Kingdom and New York, NY, USA.
- 2. Jassal RS, Black TA, Trofymow AJ, Roy R, Nesic Z (2010) Forest-floor CO2 and N2O flux dynamics in a nitrogen-fertilized Pacific Northwest Douglas-fir stand. Geoderma 157(3–4): 118–125. [Google Scholar]
- 3. Smith P, Martino D, Cai ZC, Gwary D, Janzen H, et al. (2008) Greenhouse gas mitigation in agriculture. Philosophical Transactions of the Royal Society B: Biological Sciences 363: 789–813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Guo JP, Zhou CD (2007) Greenhouse gas emissions and mitigation measures in Chinese agroecosystems. Agricultural and Forest Meteorology 142: 270–277. [Google Scholar]
- 5. Kim Y, Ueyama M, Nakagawa F, Tsunogai U, Harazono Y, et al. (2007) Assessment of winter fluxes of CO2 and CH4 in boreal forest soils of central Alaska estimated by the profile method and the chamber method: a diagnosis of methane emission and implications for the regional carbon budget. Tellus 59B: 223–233. [Google Scholar]
- 6. Wu X, Yao Z, Brüggemann N, Shen ZY, Wolf B, et al. (2010) Effects of soil moisture and temperature on CO2 and CH4 soil-atmosphere exchange of various land use/cover types in a semi-arid grassland in Inner Mongolia, China. Soil Biology & Biochemistry 42: 773–787. [Google Scholar]
- 7. Banger K, Tian HQ, Lu CQ (2012) Do nitrogen fertilizers stimulate or inhibit methane emissions from rice fields? Global Change Biology 18: 3259–3267. [DOI] [PubMed] [Google Scholar]
- 8. Sanz-Cobena A, Sánchez-Martín L, García-Torres L, Vallejo A (2012) Gaseous emissions of N2O and NO and NO3− leaching from urea applied with urease and nitrification inhibitors to a maize (Zea mays) crop. Agriculture, Ecosystems and Environment 149: 64–73. [Google Scholar]
- 9. Sanz-Cobena A, García-Marco S, Quemada M, Gabriel JL, Almendros P, et al. (2014) Do cover crops enhance N2O, CO2 or CH4 emissions from soil in Mediterranean arable systems? Science of the Total Environment 466–467: 164–174. [DOI] [PubMed] [Google Scholar]
- 10. Valentini R, Matteucci G, Dolman AJ, Schulze ED, Rebmann C, et al. (2000) Respiration as the main determinant of carbon balance in European forests. Nature 404: 861–865. [DOI] [PubMed] [Google Scholar]
- 11. Bowden WB, Bormann FH (1986) Transport and loss of nitrous oxide in soil water after forest cutting. Science 233: 867–869. [DOI] [PubMed] [Google Scholar]
- 12. Tang JW, Baldocchi DD, Qi Y, Xu LK (2003) Assessing soil CO2 efflux using continuous measurements of CO2 profiles in soils with small solid-state sensors. Agricultural and Forest Meteorology 118: 207–220. [Google Scholar]
- 13. Fierer N, Chadwick OA, Trumbore SE (2005) Production of CO2 in Soil Profiles of a California Annual Grassland. Ecosystems 8: 412–429. [Google Scholar]
- 14. Gebert J, Röer IU, Scharff H, Roncato CDL, Cabral AR (2011) Can soil gas profiles be used to assess microbial CH4 oxidation in landfill covers? Waste Management 31: 987–994. [DOI] [PubMed] [Google Scholar]
- 15. Reth S, Graf W, Gefke O, Schilling R, Seidlitz HK, et al. (2008) Whole-year-round Observation of N2O Profiles in Soil: A Lysimeter Study. Water Air Soil Pollut: Focus 8: 129–137. [Google Scholar]
- 16. Wang YY, Hu CS, Ming H, Zhang YM, Li XX, et al. (2013) Concentration profiles of CH4, CO2 and N2O in soils of a wheat–maize rotation cosystem in North China Plain, measured weekly over a whole year. Agriculture, Ecosystems & Environment 164: 260–272. [Google Scholar]
- 17.Sparks DL (1996) Methods of soil analysis, part 3–chemical methods. In: Sparks, D.L. (Eds.), SSSA book series, No. 5. SSSA, Inc and American Society of Agronomy, Inc. Madison, WI. pp. 475–1185.
- 18.Gee GW, Bauder JW (1986) Particle-size analysis. In: Klute, A. (Ed.), Methods of Soil Analysis Part 1-Physical and Mineralogical Methods. American Society of gronomy, Madison, WI. pp. 383–409.
- 19.Campbell GS (1985) Soil Physics with BASIC. Elsevier Science Publishers BV, Amsterdam. Soil Science Society of America Book Series: 5 Methods of Soil Analysis Part I-Physical and Mineralogical Methods.
- 20. Marshall TJ (1959) The diffusion of gas through porous media. J Soil Sci 10: 79–82. [Google Scholar]
- 21.Rolston DE (1986) 47 Gas Flux, 1103–1109. American Society of Agronomy, Inc. Soil Science Society of America, Inc. Publisher. Madison, Wisconsin USA.
- 22. Sallam A, Jury WA, Letey J (1984) Measurement of gas-diffusion coefficient under relatively low air-filled porosity. Soil Sci Soc Am J 48: 3–6. [Google Scholar]
- 23.Jury WA, Gardner WR, Gardner WH (1991) Soil Physics, fifth ed. Wiley, New York.
- 24. Gilliland E, Baddour R, Perkinson G, Sladek KJ (1974) Diffusion on surfaces. I. Effect of concentration on the diffusivity of physically adsorbed gases. Ind Eng Chem Fundam 13(2): 95–100. [Google Scholar]
- 25. Millington R, Quirk JP (1961) Permeability of porous solids. Trans Faraday Soc 57: 1200–1207. [Google Scholar]
- 26. Wang YY, Hu CS, Zhu B, Xiang HY, He XH (2010) Effects of wheat straw application on methane and nitrous oxide emissions from purplish paddy fields. Plant, Soil and Environ 56 (1): 16–22. [Google Scholar]
- 27. Stiehl-Braun PA, Powlson DS, Poulton PR, Niklaus PA (2011) Effects of N fertilizers and liming on the micro-scale distribution of soil methane assimilation in the long-term Park Grass experiment at Rothamsted. Soil Biology and Biochemistry 43(5): 1034–1041. [Google Scholar]
- 28. Shrestha BM, Sitaula BK, Singh BR, Bajracharya RM (2004) Fluxes of CO2 and CH4 in soil profiles of a mountainous watershed of Nepal as influenced by land use, temperature, moisture and substrate Addition. Nutrient Cycling in Agroecosystems 68: 155–164. [Google Scholar]
- 29. Chu H, Hosen Y, Yagi K (2004) Nitrogen oxide emissions and soil microbial properties as affected by N-fertilizer management in a Japanese Andisol. Soil Science and Plant Nutrition 50: 287–292. [Google Scholar]
- 30. Steudler PA, Bowden RD, Melillo JM, Aber JD (1989) Influence of nitrogen fertilisation on methane uptake in temperate forest soils. Nature 341: 314–316. [Google Scholar]
- 31. Kravchenko I, Boeckx P, Galchenko V, Van Cleemput O (2002) Shortand medium-term effects of NH4 +on CH4 and N2O fluxes in arable soils with a different texture. Soil Biology & Biochemistry 34: 669–678. [Google Scholar]
- 32. Flessa H, Beese F (2000) Laboratory estimates of trace gas emissions following surface application and injection of cattle slurry. J Environ Qual 29: 262–268. [Google Scholar]
- 33. Sherlock RR, Sommer SG, Khan RZ, Wood CW, Guertal EA, et al. (2002) Emissions of ammonia, methane and nitrous oxide from pig slurry applied to a pasture in New Zealand. J Environ Qual 31: 1491–1501. [DOI] [PubMed] [Google Scholar]
- 34. Rodhe L, Pell M, Yamulki S (2006) Nitrous oxide, methane and ammonia emissions following slurry spreading on grassland. Soil Use Manage 22: 229–237. [Google Scholar]
- 35. Koschorreck M, Conrad R (1993) Oxidation of atmospheric methane in soil: measurements in the field, in soil cores and in soil samples. Global Biogeochemical Cycles 7: 109–121. [Google Scholar]
- 36. Borken W, Matzner E (2009) Reappraisal of drying and wetting effects on C and N mineralization and fluxes in soils. Global Change Biology 15: 808–824. [Google Scholar]
- 37. Denef K, Six J, Bossuyt H, Frey SD, Elliott ET, et al. (2001) Influence of dry-wet cycles on the interrelationship between aggregate, particulate organic matter, and microbial community dynamics. Soil Biology and Biochemistry 33: 1599–1611. [Google Scholar]
- 38. Davidson EA, Belk E, Boone RD (1998) Soil water content and temperature as independent or confounded factors controlling soil respiration in a temperate mixed hardwood forest. Global Change Biol 4: 217–227. [Google Scholar]
- 39. Jassal RS, Black TA, Drewitt GB, Novak MD, Gaumont-Guay D, et al. (2004) A model of the production and transport of CO2 in soil: predicting soil CO2 concentrations and CO2 efflux from a forest floor. Agric For Meteorol 124: 219–236. [Google Scholar]
- 40. Fang YT, Gundersen P, Zhang W, Zhou GY, Christiansen JR, et al. (2009) Soil-atmosphere exchange of N2O, CO2 and CH4 along a slope of an evergreen broad-leaved forest in southern China. Plant and Soil 319: 37–48. [Google Scholar]
- 41. Bajracharya RM, Lal R, Kimble JM (2000) Erosion effect on carbon dioxide concentration and carbon flux from an Ohio Alfisol. Soil Sci. Soc. Am. J 64: 694–700. [Google Scholar]
- 42. Fang C, Moncrieff JB (2001) The dependence of soil CO2 efflux on temperature. Soil Biology & Biochemistry 33: 155–165. [Google Scholar]
- 43. Kelting DL, Burger JA, Edwards GS (1998) Estimating root respiration, microbial respiration in the rhizosphere, and root-free soil respiration in forest soils. Soil Biology and Biochemistry 30: 961–968. [Google Scholar]
- 44. Clough TJ, Sherlock RR, Kelliher FM (2003) Can liming mitigate N2O fluxes from a urine-amended soil? Aust J Soil Res 41: 439–457. [Google Scholar]
- 45. Clough TJ, Kelliher FM, Sherlock RR, Ford CD (2004) Lime and soil moisture effects on nitrous oxide emissions from a Urine Patch. Soil Sci Soc A J 68: 1600–1609. [Google Scholar]
- 46. Grandy SA, Robertson PG (2006) Initial cultivation of a temperate-region soil immediately accelerates aggregate turnover and CO2 and N2O fluxes. Global Change Biology 12: 1507–1520. [Google Scholar]
- 47. Li XX, Hu CS, Delgado JA, Zhang YM, Ouyang ZY (2007) Increased nitrogen use efficiencies as a key mitigation alternative to reduce nitrate leaching in north China plain. Agricultural Water Management 89: 137–147. [Google Scholar]
- 48. Zhu B, Wang T, Kuang F, Luo Z, Tang J, et al. (2009) Measurements of nitrate leaching from a hillslope cropland in the Central Sichuan Basin, China. Soil Sci Soc Am J 73: 1419–1426. [Google Scholar]
- 49. Dobbie KE, Smith KA (2003) Nitrous oxide emission factors for agricultural soils in Great Britain: the impact of soil water-filled pore space and other controlling variables. Global Change Biology 9: 204–218. [Google Scholar]
- 50. Zou GY, Zhang FS, Chen XP, Li XH (2001) Nitrification–denitrification and N2O emission from arable soil. Soil Environ Sci 10 ((4)) 273–276 (in Chinese).. [Google Scholar]
- 51. Davidson EA (1992) Sources of nitric oxide and nitrous oxide following wetting of dry soil. Soil Sci Soc Am J 56: 95–102. [Google Scholar]
- 52.Zhu HJ, He YG eds (1992) Soil Geography. Higher Education Press, Beijing, China (In Chinese).
- 53. Ding WX, Luo JF, Li J, Yu HY, Fan JL, et al. (2013) Effect of long-term compost and inorganic fertilizer application on background N2O and fertilizer-induced N2O emissions from an intensively cultivated soil. Science of the Total Environment 465: 115–124. [DOI] [PubMed] [Google Scholar]