Abstract
Limited cell growth and the resulting low volumetric productivity of ethanologenic Escherichia coli KO11 in mineral salts medium containing xylose have been attributed to inadequate partitioning of carbon skeletons into the synthesis of glutamate and other products derived from the citrate arm of the anaerobic tricarboxylic acid pathway. The results of nuclear magnetic resonance investigations of intracellular osmolytes under different growth conditions coupled with those of studies using genetically modified strains have confirmed and extended this hypothesis. During anaerobic growth in mineral salts medium containing 9% xylose (600 mM) and 1% corn steep liquor, proline was the only abundant osmolyte (71.9 nmol ml−1 optical density at 550 nm [OD550] unit−1), and growth was limited. Under aerobic conditions in the same medium, twice the cell mass was produced, and cells contained a mixture of osmolytes: glutamate (17.0 nmol ml−1 OD550 unit−1), trehalose (9.9 nmol ml−1 OD550 unit−1), and betaine (19.8 nmol ml−1 OD550 unit−1). Two independent genetic modifications of E. coli KO11 (functional expression of Bacillus subtilis citZ encoding NADH-insensitive citrate synthase; deletion of ackA encoding acetate kinase) and the addition of a metabolite, such as glutamate (11 mM) or acetate (24 mM), as a supplement each increased the intracellular glutamate pool during fermentation, doubled cell growth, and increased volumetric productivity. This apparent requirement for a larger glutamate pool for increased growth and volumetric productivity was completely eliminated by the addition of a protective osmolyte (2 mM betaine or 0.25 mM dimethylsulfoniopropionate), consistent with adaptation to osmotic stress rather than relief of a specific biosynthetic requirement.
Conversion of lignocellulose to fuel ethanol and other chemicals typically derived from petroleum offers the opportunity to decrease dependence on imported oil (2). The sugar constituents of lignocellulose can be effectively metabolized by Escherichia coli, a commercially important biocatalyst for the production of amino acids and recombinant proteins (1, 11). This organism has recently been engineered for the production of a variety of bulk chemicals, such as organic acids (4, 8, 18, 20, 36, 47, 53), diols (17, 35, 44), and fuel ethanol (22). Ethanologenic E. coli KO11 was previously constructed by the chromosomal integration of Zymomonas mobilis genes encoding a pyruvate decarboxylase with a low Km (pdc) and alcohol dehydrogenase II (adhB) (37). This strain ferments hexoses and pentoses to ethanol with high efficiency in rich laboratory media (22, 29, 37). However, volumetric productivity was limited by low cell mass (low biocatalyst concentration) when fermentations were conducted with minerals and less expensive commercial nutrients (29, 51, 52). Similar problems have been reported for the production of other products using genetically engineered strains as biocatalysts (3, 9, 10, 53).
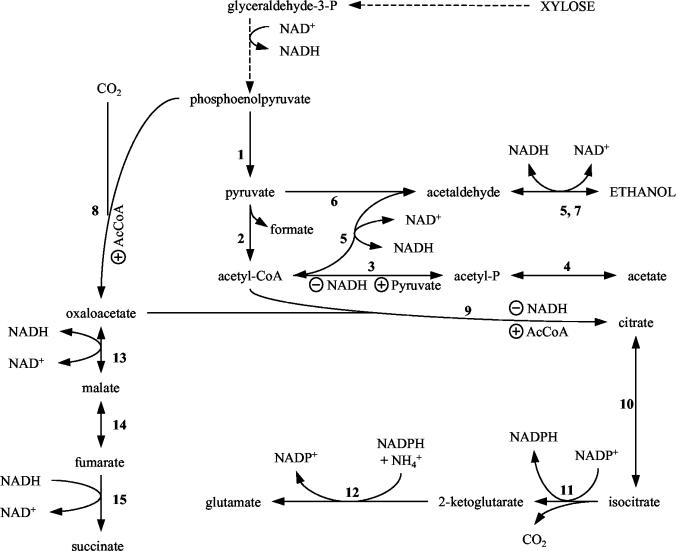
The limited cell growth and low volumetric productivity of ethanol in CSL medium (containing mineral salts, 10 g of corn steep liquor liter−1, and 90 g of xylose liter−1) have been attributed to inadequate partitioning of carbon skeletons into the synthesis of glutamate and other products derived from the citrate arm of the anaerobic tricarboxylic acid (TCA) pathway (45, 46) (Fig. 1). Supplementing fermentations with potassium glutamate (2 g liter−1) or expressing Bacillus subtilis citZ encoding an NADH-insensitive citrate synthase stimulated growth and increased volumetric ethanol production (45). Similar benefits were also observed from the deletion of acetate kinase, a mutation that increased the availability of acetyl coenzyme A (acetyl-CoA) (46). Acetyl-CoA is an antagonist of the NADH-mediated inhibition of native E. coli citrate synthase (49). All three approaches are presumed to increase the synthesis of compounds such as glutamate that are derived from the citrate arm of the TCA pathway (Fig. 1).
FIG. 1.
Central carbon metabolism in E. coli KO11 during xylose fermentation. Unless noted otherwise, enzymes listed are native to E. coli. Enzymes: 1, pyruvate kinase (pykA, pykF); 2, pyruvate formate-lyase (pflB); 3, phosphotransacetylase (pta); 4, acetate kinase (ackA); 5, alcohol or aldehyde dehydrogenase (adhE); 6, Z. mobilis pyruvate decarboxylase (pdc); 7, Z. mobilis alcohol dehydrogenase II (adhB); 8, phosphoenolpyruvate carboxylase (ppc); 9, citrate synthase (gltA); 10, aconitase (acn); 11, isocitrate dehydrogenase (icd); 12, glutamate dehydrogenase (gdhA); 13, malate dehydrogenase (mdh); 14, fumarase (fumB); 15, fumarate reductase (frdABC). Abbreviations: P, phosphate; AcCoA, acetyl-CoA. Symbols: ⊕, allosteric activation; ⊖, allosteric inhibition.
Glutamate is the most abundant free amino acid in the cytoplasm of E. coli and related organisms during aerobic growth in mineral salts media (14, 27, 33). In addition to essential roles in nitrogen assimilation and biosynthesis, glutamate also serves as a protective intracellular osmolyte during adaptation to osmotic stress (13-15, 32, 50). The high osmolarity of CSL medium used for ethanol production (∼600 mM xylose plus other compounds) may represent a significant stress for E. coli and increase the cellular requirement for glutamate. Ethanol, the primary product from fermentation by E. coli KO11, is freely permeable, does not plasmolyze cells, and does not contribute to osmotic stress (13).
In this paper, we have used nuclear magnetic resonance (NMR) to investigate intracellular pools of compatible solutes in E. coli KO11. Changes in pool sizes and compositions have been correlated with final cell density and volumetric productivity in the presence and absence of medium supplements and in genetically modified strains.
MATERIALS AND METHODS
Microorganisms and media.
Ethanologenic E. coli KO11 [frd cat pfl+ pfl::(Z. mobilis pdc+ adhB+)] (37) and SU102 (KO11 ΔackA) (46) are prototrophic and were grown in CSL medium containing the following ingredients (per liter): 90 g of xylose, 10 g of corn steep liquor (∼50% dry weight), 1 g of KH2PO4, 0.5 g of K2HPO4, 3.1 g of (NH4)2SO4, 0.4 g of MgCl2 · 6H2O, and 20 mg of FeCl3 · 6H2O (45). Solid medium contained 1.5% agar, 20 g of xylose liter−1, and chloramphenicol (40 to 600 mg liter−1). Working cultures were transferred daily on solid medium. Stock cultures were stored frozen at −75°C in 40% glycerol. No antibiotics were added to broth cultures.
Stock solutions of glutamate, acetate, proline, betaine, dimethylsulfoniopropionate, and taurine were dissolved in deionized water, neutralized as required (NaOH, H2SO4, or NaHCO3) and sterilized by filtration. All osmoprotectants were purchased from Sigma (St. Louis, Mo.) except dimethylsulfoniopropionate (TCI America, Portland, Oreg.). Corn steep liquor was purchased from the Grain Processing Corporation (Muscatine, Iowa).
Aerobic growth studies.
Colonies were transferred from solid medium to baffled flasks (1 liter) containing 250 ml of CSL medium (initial cell concentration of ∼3 mg [dry weight] of cells liter−1) and incubated at 35°C (220 rpm).
Fermentation.
Seed cultures and fermentations were performed in CSL medium at 35°C (pH 6.5, 120 rpm) as described previously (34, 45). Fermentations were inoculated to provide an initial cell density of 33 mg (dry weight) liter−1.
13C NMR.
Intracellular osmolytes were analyzed by NMR as previously described (37). Cells were harvested in late exponential phase. Cell pellets from 700 ml of broth (∼24 h for anaerobic; ∼12 h for aerobic) were washed twice in the mineral salts component of CSL medium containing additional NaCl (600 mM) and extracted with 3 volumes of 95% ethanol. Suspensions were rocked gently for 16 to 24 h at 4°C. Debris was then removed by centrifugation (4°C, 10,000 × g, 30 min). Extracts were dried under vacuum twice to remove all traces of ethanol, dissolved in 4.0 ml of 25% D2O, and filtered (0.2-μm-pore-size filter). Acetone (10 μl) was added as an internal reference. NMR spectra were obtained using a modified Nicolet NT300 spectrometer operating in the Fourier transform mode as follows: 75.46 MHz; excitation pulse width, 25 μs; pulse repetition delay, 40 s; spectral width, 18 kHz; and broadband (bilevel) decoupling of protons (>1,000 scans) (38). Osmolyte pools were estimated using a standard curve for each compound with acetone as the internal standard. Intracellular pools are reported in nanomoles per milliliter per optical density at 550 nm [OD550] unit as described by Csonka et al. (14).
Analytical methods.
For fermentation studies, cell mass was estimated from OD550 using a Bausch & Lomb Spectronic 70 spectrophotometer and a weight calibration curve (1 OD550 unit = 0.33 g [dry weight] of cells liter−1). Ethanol was measured by gas chromatography (Varian 3400CX) (34).
RESULTS AND DISCUSSION
Comparison of intracellular osmolytes in E. coli KO11 during aerobic and anaerobic growth.
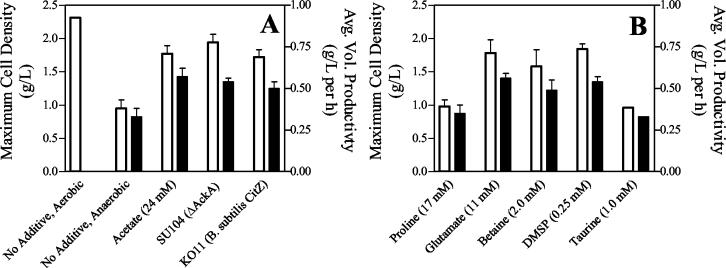
During aerobic growth in CSL medium (9% xylose, 600 mM), E. coli KO11 grew to more than twice the cell density of cultures grown anaerobically in the same medium with or without additional minerals and trace metals (Fig. 2A) (45). ATP production does not appear to limit anaerobic growth in 9% xylose (45, 46), since cell densities achieved with xylose (<0.5 ATP molecule per glucose molecule) were equivalent to those with glucose (>2.0 ATP molecules per glucose molecule). The lower cell mass during anaerobic growth limited volumetric ethanol productivity, increasing the time required to complete fermentation (45).
FIG. 2.
Fermentation of xylose by E. coli KO11 in CSL medium containing various supplements. (A) Effects of conditions and mutations that promote glutamate synthesis. (B) Effects of protective osmolytes on E. coli KO11. Unless indicated otherwise, all cultures were anaerobic. Maximum cell mass (white bars) and average volumetric productivity of ethanol (black bars) (initial 72-h period) are shown. Values are averages ± standard deviations (error bars) of two or more experiments. Abbreviation: DMSP, dimethylsulfoniopropionate.
An NMR comparison of major intracellular osmolytes during aerobic and anaerobic growth revealed striking differences (Table 1; Fig. 3A and B). During aerobic growth, high levels of glutamate (17.0 nmol ml−1 OD550 unit−1), trehalose (9.9 nmol ml−1 OD550 unit−1), and betaine (19.8 nmol ml−1 OD550 unit−1) were present, consistent with adaptation to the highly osmotic environment in unmodified strains of E. coli (5, 27). Although E. coli is incapable of de novo betaine synthesis, this protective osmolyte can be synthesized under aerobic conditions (only) by oxidizing choline (26), which is present in corn steep liquor (Traders' guide to fermentation media formulation, Traders Protein, Memphis, Tenn.). Choline cannot be oxidized to betaine during fermentative growth.
TABLE 1.
Intracellular accumulation of protective osmolytes by E. coli KO11 during growth in CSL medium
| Fermentation conditions | Intracellular osmolyte (nmol ml−1 OD550 unit−1)a
|
|||||
|---|---|---|---|---|---|---|
| Proline | Glutamate | Betaine | DMSPb | Trehalose | Total osmolytesc | |
| Aerobic | ||||||
| No additive | <4.0 | 17.0 | 19.8 | <4.0 | 9.9 | 46.7 |
| Anaerobic | ||||||
| No additive | 71.9 ± 1.4 | <4.0 | <4.0 | <4.0 | <4.0 | 71.9 ± 1.4 |
| With acetate (24 mM) | 42.7 | 14.7 | <4.0 | <4.0 | <4.0 | 57.4 |
| SU102 (ΔackA) | 43.5 | 19.7 | <4.0 | <4.0 | <4.0 | 64.2 |
| KO11 (B. subtilis citZ) | 47.1 | 17.5 | <4.0 | <4.0 | <4.0 | 64.6 |
| Avgd | 44.4e ± 2.3 | 17.3e ± 2.5 | <4.0 | <4.0 | <4.0 | 62.1 ± 4.0 |
| With additive | ||||||
| Proline (17 mM) | 81.9 | <4.0 | <4.0 | <4.0 | <4.0 | 81.9 |
| Glutamate (11 mM) | 45.6 | 16.2 | <4.0 | <4.0 | <4.0 | 61.8 |
| Betaine (2.0 mM) | <4.0 | <4.0 | 55.4 | <4.0 | <4.0 | 55.4 |
| DMSP (0.25 mM) | 21.4 | <4.0 | <4.0 | 28.0 | <4.0 | 49.4 |
| Taurine (1.0 mM) | 68.8 | <4.0 | <4.0 | <4.0 | <4.0 | 68.8 |
Detection limit for intracellular osmolytes under our conditions, 4.0 nmol ml−1 OD550 unit−1.
DMSP, dimethylsulfoniopropionate.
Average of total osmolytes for all strains and conditions, 63.8 ± 10.3 nmol ml−1 OD550 unit−1.
Average for approaches designed to increase glutamate biosynthesis.
Significantly different (P < 0.01) from the value for anaerobic growth of KO11 without additives.
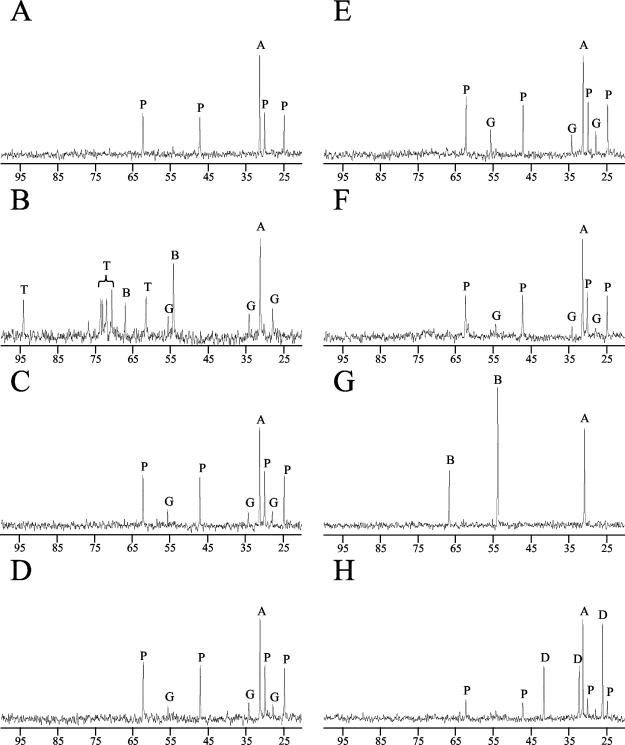
FIG. 3.
13C NMR spectra of intracellular osmolytes from ethanologenic E. coli KO11. (A) KO11 during fermentation without additives; (B) KO11 during aerobic growth without additives; (C) KO11 supplemented with 11 mM (2 g liter−1) potassium glutamate during fermentation; (D) KO11 supplemented with 24 mM (2 g liter−1) sodium acetate during fermentation; (E) SU102 (ΔackA) fermentation without additives; (F) KO11 expressing CitZ, an NADH-insensitive citrate synthase from B. subtilis; (G) KO11 supplemented with 2 mM betaine during fermentation; (H) KO11 supplemented with 0.25 mM dimethylsulfoniopropionate during fermentation. Peaks are labeled as follows: A, acetone; P, proline; G, glutamate; B, betaine; D, dimethylsulfoniopropionate; and T, trehalose. The scale is in parts per million.
During anaerobic growth, proline (71.9 nmol ml−1 OD550 unit−1) was the only osmolyte above our limit of detection (≤4.0 nmol ml−1 OD550 unit−1). Since allosteric regulation prevents the biosynthesis of proline as an osmoprotectant (12, 41, 42), it is likely that corn steep liquor is also the source of intracellular proline. Supplementing CSL medium with additional proline (17 mM) had no effect on cell growth or volumetric productivity despite a 14% increase in the intracellular pool. Although proline accumulation is presumed to be beneficial as an osmolyte, it is typically less effective than betaine as an adaptation to osmotic stress (30, 40). The absence of trehalose (<4.0 nmol ml−1 OD550 unit−1) during anaerobic growth in CSL medium is also noteworthy. The relatively low ATP yield from xylose catabolism (0.4 ATP molecule per pyruvate molecule [43, 45]) may serve to restrict gluconeogenesis and trehalose production during fermentation.
Supplementing CSL medium with glutamate during fermentation increased the concentration of cells by 87% (Fig. 2B) and increased the glutamate pool (16.2 nmol ml−1 OD550 unit−1) to that present during aerobic growth (17.0 nmol ml−1 OD550 unit−1), with a corresponding decrease in proline (Table 1; Fig. 3C). These results demonstrated that both limited cell growth and low volumetric productivity correlate with a small intracellular pool of glutamate, consistent with a deficiency of glutamate for osmoprotection (45, 46).
Citrate synthase flux limits the intracellular accumulation of glutamate.
Insufficient carbon partitioning into 2-ketoglutarate, the immediate precursor of glutamate (Fig. 1), has been proposed to limit E. coli KO11 cell mass (and volumetric productivity) during anaerobic growth (high NADH/NAD+ ratio) in CSL medium, consistent with a small glutamate pool (Table 1). Citrate synthase is a key allosteric enzyme at the committed branch point for 2-ketoglutarate synthesis (48) (Fig. 1). Allosteric inhibition of this native enzyme by high NADH levels such as those associated with anaerobic growth (16) can be antagonized by acetyl-CoA (49). Two independent approaches that have been previously used to increase the availability of acetyl-CoA (and thereby promote cell growth) were investigated for their effects on the glutamate pool during fermentations in the CSL medium: (i) supplementing with 24 mM acetate (25, 46) and (ii) inactivating the primary anaerobic acetate production pathway (ΔackA) (25, 46). Both approaches increased the glutamate pool to levels similar to that observed during aerobic growth (Table 1; Fig. 3D and E), consistent with an increase in citrate synthase activity in vivo. Cell mass and average volumetric productivity were increased by approximately twofold (Fig. 2A).
The primary citrate synthase in B. subtilis (CitZ) is insensitive to NADH and is allosterically regulated by ATP (24). Expression of B. subtilis citZ also doubled cell mass, increased volumetric productivity (Fig. 3A), and increased the intracellular glutamate pool (17.5 nmol ml−1 OD550 unit−1) (Table 1; Fig. 3F). Thus, all approaches that increased the intracellular pool of glutamate were also beneficial for cell growth and ethanol production. The beneficial effects of a large intracellular pool of glutamate are consistent with the adaptation to osmotic stress as previously characterized in unmodified E. coli (13).
Deletion of ackA also eliminated ATP production by this pathway and improved growth, providing further evidence that cell growth is not ATP limited under our conditions.
Added osmoprotectants supplant the glutamate requirement.
If the primary role of a large glutamate pool (resulting in increased cell growth and higher volumetric productivity) is osmoprotection, alternative protective osmolytes should provide a similar benefit and eliminate the need to alter carbon partitioning. To test this hypothesis, four different osmolytes were investigated (Table 1; Fig. 2B). The addition of taurine and proline, relatively poor osmoprotectants for E. coli (31), had no effect on final cell density or volumetric productivity. Taurine was not accumulated in cells, and proline levels were essentially the same with or without a proline supplement. Betaine and dimethylsulfoniopropionate, both excellent osmoprotectants for E. coli (7, 40), accumulated in the cytoplasm, with a corresponding reduction in proline (Fig. 2G and H). Both additives increased final cell density and volumetric productivity (Fig. 2). Since neither compound is known to be metabolized by E. coli, betaine and dimethylsulfoniopropionate are presumed to act directly as osmoprotectants.
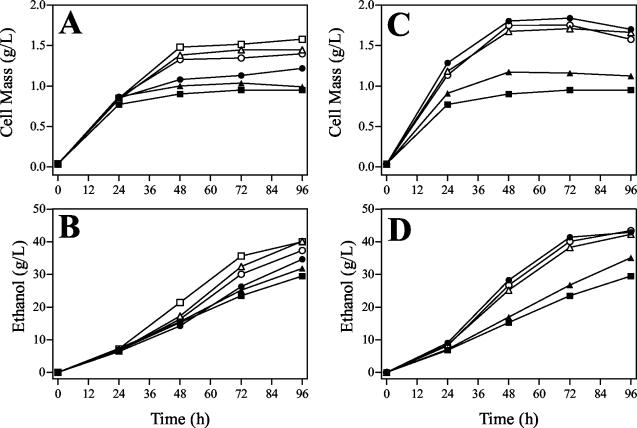
Cell mass and volumetric productivity were dose dependent for each additive with optima of approximately 2.0 mM for betaine and 0.25 mM for dimethylsulfoniopropionate (Fig. 4). Thus, dimethylsulfoniopropionate acts as a more effective osmolyte than betaine during xylose fermentation. In contrast, previous studies have identified betaine as the most effective osmolyte for salt stress (7, 40). Under aerobic conditions, the effectiveness of betaine in promoting growth has been reported to vary in response to osmotic stress with different sugars (19). The relative effectiveness of specific protective osmolytes appears to be dependent on the nature of the stressing agent (sugars and salts) and the growth conditions.
FIG. 4.
Effects of betaine and dimethylsulfoniopropionate on E. coli KO11 growth and ethanol production. (A and B) Effects of betaine on growth (A) and ethanol production (B). (C and D) Effects of dimethylsulfoniopropionate on growth (C) and ethanol production (D). Symbols: ▪, no addition; ▴, 0.1 mM; •, 0.25 mM; ▵, 0.5 mM; ○, 1.0 mM; □, 2.0 mM.
Betaine was the only osmolyte detected in cells from fermentations supplemented with betaine (Fig. 3G; Table 1), consistent with a low Km for betaine transport (6, 28, 39). Both betaine and proline are concentrated from the medium by the same transporters, ProP and ProU (28). The high level of betaine in the medium (2.0 mM) coupled with the low Km of the primary betaine transport pathway (ProU) is consistent with exclusion by the preferential transport of betaine. Added taurine was not accumulated in cells and may be similarly excluded by the preferential transport of proline in corn steep liquor.
Cells from fermentations supplemented with 0.25 mM dimethylsulfoniopropionate accumulated both proline and dimethylsulfoniopropionate. Although little is known about dimethylsulfoniopropionate transport, the osmotically activated transporters (ProP and ProU) are known to be promiscuous, transporting proline, taurine, ectoine, betaine, and other structurally analogous compounds (21, 23, 28, 31, 39). Presumably, dimethylsulfoniopropionate is also transported by ProU and ProP. The lower extracellular concentration of dimethylsulfoniopropionate (0.25 mM) compared to betaine (2.0 mM) appears to allow the accumulation of intracellular pools of both proline and dimethylsulfoniopropionate.
Glutamate was not detected in cells grown in CSL medium with either betaine or dimethylsulfoniopropionate as a supplement (Table 1; Fig. 3G and H), confirming that a large glutamate pool per se is not required to increase final cell density and volumetric productivity. Thus, the apparent requirement for additional glutamate during fermentation in CSL medium can be directly attributed to a requirement for osmotic adaptation. Under these conditions, cell growth is limited by insufficient levels of an effective osmolyte, rather than a nutritional requirement for glutamate during biosynthesis.
Conclusions.
The limited cell growth and accompanying low volumetric productivity of ethanologenic E. coli KO11 during xylose fermentation in CSL medium can be attributed to osmotic stress and inadequate levels of protective osmolytes. Inactivation of acetate kinase (ΔackA), supplementing with acetate, supplementing with glutamate, and expression of B. subtilis citZ were each shown to increase the partitioning of carbon into the citrate arm of the anaerobic TCA pathway, as evidenced by an increase in the intracellular glutamate concentration. All approaches that increased the intracellular pool of glutamate also increased final cell density and volumetric productivity. The apparent requirement for glutamate was completely eliminated by supplementing with alternative osmoprotectants (dimethylsulfoniopropionate or betaine), demonstrating that this requirement is related to osmotic stress rather than to a specific biosynthetic need.
Acknowledgments
This research was supported in part by grants from the U.S. Department of Agriculture (01-35504-10669 and 00-52104-9704), the U.S. Department of Energy (FG02-96ER20222), and the Florida Agricultural Experiment Station.
Footnotes
Florida Agricultural Experiment Station publication no. R-09759.
REFERENCES
- 1.Akesson, M., P. Hagander, and J. P. Axelsson. 2001. Avoiding acetate accumulation in Escherichia coli cultures using feedback control of glucose feeding. Biotechnol. Bioeng. 73:223-230. [DOI] [PubMed] [Google Scholar]
- 2.Arntzen, C. E., and B. E. Dale. 1999. Biobased industrial products, priorities for research and commercialization. National Academy Press, Washington, D.C. [PubMed]
- 3.Bunch, P. K., F. Mat-Jan, N. Lee, and D. P. Clark. 1997. The ldhA gene encoding the fermentative lactate dehydrogenase of Escherichia coli. Microbiology 143:187-195. [DOI] [PubMed] [Google Scholar]
- 4.Causey, T. B., S. Zhou, K. T. Shanmugam, and L. O. Ingram. 2003. Engineering the metabolism of Escherichia coli W3110 for the conversion of sugar to redox-neutral and oxidized products: homoacetate production. Proc. Natl. Acad. Sci. USA 100:825-832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cayley, S., B. A. Lewis, H. J. Guttman, and M. T. Record, Jr. 1991. Characterization of the cytoplasm of Escherichia coli K-12 as a function of external osmolarity. Implications for protein-DNA interactions in vivo. J. Mol. Biol. 222:281-300. [DOI] [PubMed] [Google Scholar]
- 6.Cayley, S., B. A. Lewis, and M. T. Record, Jr. 1992. Origins of the osmoprotective properties of betaine and proline in Escherichia coli K-12. J. Bacteriol. 174:1586-1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chambers, S. T., C. M. Kunin, D. Miller, and A. Hamada. 1987. Dimethylthetin can substitute for glycine betaine as an osmoprotectant molecule for Escherichia coli. J. Bacteriol. 169:4845-4847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chang, D., S. Shin, J. Rhee, and J. Pan. 1999. Homofermentative production of d- or l-lactate in metabolically engineered Escherichia coli RR1. Appl. Environ. Microbiol. 65:1384-1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chao, Y., and J. C. Liao. 1994. Metabolic responses to substrate futile cycling in Escherichia coli. J. Biol. Chem. 269:5122-5126. [PubMed] [Google Scholar]
- 10.Chao, Y., R. Patnaik, W. D. Roof, R. F. Young, and J. C. Liao. 1993. Control of gluconeogenic growth by pps and pck in Escherichia coli. J. Bacteriol. 175:6939-6944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chotani, G., T. Dodge, A. Hsu, M. Kumar, R. LaDuca, D. Trimbur, W. Weyler, and K. Sanford. 2000. The commercial production of chemicals using pathway engineering. Biochim. Biophys. Acta 1543:434-455. [DOI] [PubMed] [Google Scholar]
- 12.Csonka, L. N. 1988. Regulation of cytoplasmic proline levels in Salmonella typhimurium: effect of osmotic stress on synthesis, degradation, and cellular retention of proline. J. Bacteriol. 170:2374-2378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Csonka, L. N. 1989. Physiological and genetic responses of bacteria to osmotic stress. Microbiol. Rev. 53:121-147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Csonka, L. N., T. P. Ikeda, S. A. Fletcher, and S. Kustu. 1994. The accumulation of glutamate is necessary for optimal growth of Salmonella typhimurium in media of high osmolality but not induction of the proU operon. J. Bacteriol. 176:6324-6333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Csonka, L. N., and A. D. Hanson. 1991. Prokaryotic osmoregulation: genetics and physiology. Annu. Rev. Microbiol. 45:469-606. [DOI] [PubMed] [Google Scholar]
- 16.de Graef, M. R., S. Alexeeva, J. L. Snoep, and M. J. Teixiera de Mattos. 1999. The steady-state internal redox state (NADH/NAD) reflects the external redox state and is correlated with catabolic adaptation in Escherichia coli. J. Bacteriol. 181:2351-2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Diaz-Torres, M., N. S. Dunn-Coleman, M. W. Chase, and D. Trimbur. August. 2000. Method for the recombinant production of 1,3-propanediol. U.S. patent 6,136,576.
- 18.Dien, B. S., N. N. Nichols, and R. J. Bothast. 2001. Recombinant Escherichia coli engineered for the production of L-lactic acid from hexose and pentose sugars. J. Ind. Microbiol. Biotechnol. 27:259-264. [DOI] [PubMed] [Google Scholar]
- 19.Dulaney, E. L., D. D. Dulaney, and E. L. Rickes. 1968. Factors in yeast extract which relieve growth inhibition of bacteria in defined medium of high osmolarity. Dev. Ind. Microbiol. 9:260-269. [Google Scholar]
- 20.Gokarn, R. R., M. A. Eiteman, and E. Altman. 2000. Metabolic analysis of Escherichia coli in the presence of carboxylating enzymes phosphoenolpyruvate carboxylase and pyruvate carboxylase. Appl. Environ. Microbiol. 60:1844-1850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Grothe, S., R. L. Krogsrud, D. J. McClellan, J. L. Milner, and J. M. Wood. 1986. Proline transport and osmotic stress response in Escherichia coli K-12. J. Bacteriol. 166:253-259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ingram, L. O., H. C. Aldrich, A. C. C. Borges, T. B. Causey, A. Martinez, F. Morales, A. Saleh, S. A. Underwood, L. P. Yomano, S. W. York, J. Zaldivar, and S. Zhou. 1999. Enteric bacterial catalyst for fuel ethanol production. Biotechnol. Prog. 15:855-866. [DOI] [PubMed] [Google Scholar]
- 23.Jebbar, M., R. Talibert, K. Gloux, T. Bernard, and C. Blanco. 1992. Osmoprotection of Escherichia coli by ectoine: uptake and accumulation characteristics. J. Bacteriol. 174:5027-5035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jin, S., and A. L. Sonenshein. 1996. Characterization of the major citrate synthase of Bacillus subtilis. J. Bacteriol. 178:3658-3660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kirkpatrick, C., L. M. Maurer, N. E. Oyelakin, Y. N. Yoncheva, R. Maurer, and J. L. Slonczewski. 2001. Acetate and formate stress: opposite responses in the proteomes of Escherichia coli. J. Bacteriol. 183:6466-6477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Landfald, B., and A. R. Strøm. 1986. Choline-glycine betaine pathway confers a high level of osmotic tolerance in Escherichia coli. J. Bacteriol. 165:849-855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lewis, B. A., S. Cayley, S. Padmanabhan, V. M. Kolb, V. Brushaber, C. F. Anderson, and M. T. Record, Jr. 1990. Natural abundance 14N and 13C NMR of glycine betaine and trehalose as probes of the cytoplasm of Escherichia coli K-12. J. Magn. Reson. 90:612-617. [Google Scholar]
- 28.Lucht, J. H., and E. Bremer. 1994. Adaptation of Escherichia coli to high osmolarity environments: osmoregulation of the high-affinity glycine betaine transport system ProU. FEMS Microbiol. Rev. 14:3-20. [DOI] [PubMed] [Google Scholar]
- 29.Martinez, A., S. W. York, L. P. Yomano, V. L. Pineda, F. C. Davis, J. C. Shelton, and L. O. Ingram. 1999. Biosynthetic burden and plasmid burden limit expression of chromosomally integrated heterologous genes (pdc, adhB) in Escherichia coli. Biotechnol. Prog. 15:891-897. [DOI] [PubMed] [Google Scholar]
- 30.Mason, T. G., and G. Blunden. 1989. Quaternary ammonium and tertiary sulphonium compounds of algal origin as alleviators of osmotic stress. Bot. Mar. 32:313-316. [Google Scholar]
- 31.McLaggan, D., and W. Epstein. 1991. Escherichia coli accumulates the eukaryotic osmolyte taurine at high osmolarity. FEMS Microbiol. Lett. 81:209-214. [DOI] [PubMed] [Google Scholar]
- 32.McLaggan, D., J. Naprstek, E. T. Buurman, and W. Epstein. 1994. Interdependence of K+ and glutamate accumulation during osmotic adaptation of Escherichia coli. J. Biol. Chem. 269:1911-1917. [PubMed] [Google Scholar]
- 33.Measures, J. C. 1975. Role of amino acids in osmoregulation of non-halophilic bacteria. Nature 257:398-400. [DOI] [PubMed] [Google Scholar]
- 34.Moniruzzaman, M., and L. O. Ingram. 1998. Ethanol production from dilute acid hydrolysate of rice hulls using genetically engineered Escherichia coli. Biotechnol. Lett. 20:943-947. [Google Scholar]
- 35.Nakamura, C. E., A. A. Gatenby, A. K.-H. Hsu, R. D. La Reau, S. L. Haynie, M. Diaz-Torres, D. E. Trimbur, G. M. Whited, V. Nagarajan, M. S. Payne, S. K. Picataggio, and R. V. Nair. January. 2000. Method for the production of 1,3-propanediol by recombinant microorganisms. U.S. patent 6,013,494.
- 36.Niu, W., K. M. Draths, and J. W. Frost. 2002. Benzene-free synthesis of adipic acid. Biotechnol. Prog. 18:201-211. [DOI] [PubMed] [Google Scholar]
- 37.Ohta, K., D. S. Beall, J. P. Mejia, K. T. Shanmugam, and L. O. Ingram. 1991. Genetic improvement of Escherichia coli for ethanol production of chromosomal integration of Zymamonas mobilis genes encoding pyruvate decarboxylase and alcohol dehydrogenase II. Appl. Environ. Microbiol. 57:893-900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Park, Y. I., M. L. Buszko, and J. E. Gander. 1997. Utilization of phosphocholine from extracellular complex polysaccharide as a source of cytoplasmic choline derivatives in Penicillium fellutanum. J. Bacteriol. 179:1186-1192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Perroud, B., and D. Le Rudulier. 1985. Glycine betaine transport in Escherichia coli: osmotic modulation. J. Bacteriol. 161:393-401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Randall, K., M. Lever, B. A. Peddie, and S. T. Chambers. 1995. Competitive accumulation of betaines by Escherichia coli K-12 and derivative strains lacking betaine porters. Biochim. Biophys. Acta 1245:116-120. [DOI] [PubMed] [Google Scholar]
- 41.Smith, C. J., A. H. Deutch, and K. E. Rushlow. 1984. Purification and characteristics of a γ-glutamyl kinase involved in Escherichia coli proline biosynthesis. J. Bacteriol. 157:545-551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Smith, L. T. 1985. Characterization of a γ-glutamyl kinase from Escherichia coli that confers proline overproduction and osmotic tolerance. J. Bacteriol. 164:1088-1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tao, H., R. Gonzales, A. Martinez, M. Rodriguez, L. O. Ingram, J. F. Preston, and K. T. Shanmugam. 2001. Use of expression arrays to investigate the basis for increased glycolytic flux (xylose) in ethanologenic Escherichia coli KO11. J. Bacteriol. 183:2979-2988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tong, I., H. H. Liao, and D. C. Cameron. 1991. 1,3-Propanediol production by Escherichia coli expressing genes from the Klebsiella pneumoniae DHA regulon. Appl. Environ. Microbiol. 57:3541-3546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Underwood, S. A., M. L. Buszko, K. T. Shanmugam, and L. O. Ingram. 2002. Flux through citrate synthase limits the growth of ethanologenic Escherichia coli KO11 during xylose fermentation. Appl. Environ. Microbiol. 68:1071-1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Underwood, S. A., S. Zhou, T. B. Causey, L. P. Yomano, K. T. Shanmugam, and L. O. Ingram. 2002. Genetic changes to optimize carbon partitioning between ethanol and biosynthesis in ethanologenic Escherichia coli. Appl. Environ. Microbiol. 68:6263-6272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vemuri, G. N., M. A. Eiteman, and E. Altman. 2002. Effects of growth mode and pyruvate carboxylase on succinic acid production by metabolically engineered strains of Escherichia coli. Appl. Environ. Microbiol. 68:1715-1727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Walsh, K., and D. E. Koshland, Jr. 1985. Characterization of rate-controlling steps in vivo by use of an adjustable expression vector. Proc. Natl. Acad. Sci. USA 82:3577-3581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Weitzman, P. D. J. 1966. Regulation of citrate synthase activity in Escherichia coli. Biochim. Biophys. Acta 128:213-215. [DOI] [PubMed] [Google Scholar]
- 50.Yan, D. L., T. P. Ikeda, A. E. Shauger, and S. Kustu. 1996. Glutamate is required to maintain the steady-state potassium pool in Salmonella typhimurium. Proc. Natl. Acad. Sci. USA 93:6527-6531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.York, S. W., and L. O. Ingram. 1996. Soy-based medium for ethanol production by Escherichia coli KO11. J. Ind. Microbiol. 16:374-376. [DOI] [PubMed] [Google Scholar]
- 52.York, S. W., and L. O. Ingram. 1996. Ethanol production by recombinant Escherichia coli KO11 using crude yeast autolysate as a nutrient supplement. Biotechnol. Lett. 18:683-688. [Google Scholar]
- 53.Zhou, S. D., K. T. Shanmugam, and L. O. Ingram. 2003. Functional replacement of the Escherichia coli d-(−)-lactate dehydrogenase gene (ldhA) with the l-(+)-lactate dehydrogenase gene (ldhL) from Pediococcus acidilactici. Appl. Environ. Microbiol. 69:2237-2244. [DOI] [PMC free article] [PubMed] [Google Scholar]