Abstract
Previous studies have suggested a potential link between histamine H3 receptors (H3R) signaling and anxiolytic-like and antidepressant-like effects. The aim of this study was to investigate the acute effects of ST-1283, a novel H3R antagonist, on anxiety-related and depression-related behaviors in comparison with those of diazepam and fluoxetine. The effects of ST-1283 were evaluated using the elevated plus maze test, open field test, marbles burying test, tail suspension test, novelty suppressed feeding test, and forced swim test in male C57BL/6 mice. The results showed that, like diazepam, ST-1283 (7.5 mg/kg) significantly modified all the parameters observed in the elevated plus maze test. In addition, ST-1283 significantly increased the amount of time spent in the center of the arena without altering general motor activity in the open field test. In the same vein, ST-1283 reduced the number of buried marbles as well as time spent digging in the marbles burying test. The tail suspension test and forced swim test showed that ST-1283 was able to reduce immobility time, like the recognized antidepressant drug fluoxetine. In the novelty suppressed feeding test, treatment with ST-1283 decreased latency to feed with no effect on food intake in the home cage. Importantly, pretreatment with the H3R agonist R-α-methylhistamine abrogated the anxiolytic and antidepressant effects of ST-1283. Taken together, the present series of studies demonstrates the novel effects of this newly synthesized H3R antagonist in a number of preclinical models of psychiatric disorders and highlights the histaminergic system as a potential therapeutic target for the treatment of anxiety-related and depression-related disorders.
Keywords: anxiety, depression, histamine, H3 receptor, R-α-methylhistamine, ST-1283
Introduction
Anxiety and depression belong to neurobehavioral disorders which are considered by their diagnostic measures into obsessive-compulsive, panic, social phobia, and post-traumatic stress disorders. Benzodiazepines are the most commonly prescribed anxiolytic drugs, being efficacious against a spectrum of anxiety disorders, whereas the major classes of antidepressants are selective serotonin reuptake inhibitors, tricyclic antidepressants, and serotonin noradrenaline reuptake inhibitors, and are known as antidepressants in patients with a wider spectrum of anxiety and depressive disorders.1,2 However, accumulating evidence shows that there are issues with addiction, tolerance, and dependence/withdrawal, as well as adverse effects, including sedation, cognitive and psychomotor impairment, and anterograde amnesia associated with clinical use of benzodiazepines. As a result, the clinical efficacy of benzodiazepines is restricted to generalized anxiety disorders, social phobia, and panic disorders,3 while the antidepressant classes of drugs, including selective serotonin reuptake inhibitors, tricyclic antidepressants, and serotonin noradrenaline reuptake inhibitors, have a slow onset of action (4–6 weeks) and their own side-effect profiles.3 Moreover, several clinical studies have shown that patients with generalized anxiety disorder who do not achieve remission are resistant to first-line medications, such as selective serotonin reuptake inhibitors and serotonin noradrenaline reuptake inhibitors. Therefore, there is a pressing need to use hydroxyzine, a first-generation antihistamine, as an adjunctive treatment.4 Consequently, there is an ongoing need to discover new therapeutic targets for the development of novel, more effective, and safer drugs with anxiolytic-like and antidepressant-like activities.
Since their discovery, there has been increasing evidence supporting a role for central histamine H3 receptors (H3Rs) in various brain functions, including cognition, emotion, stress, and feeding.5 Moreover, the most recent advances in preclinical and clinical trials using H3R antagonists have shown distinct pharmacologic actions, indicating their importance for diverse central nervous system-related therapeutic applications, such as depression, schizophrenia, sleep-wake disorders, dementia, and epilepsy.6,7
Central histamine plays an important role in anxiety and depression. There have been numerous studies indicating a functional relationship between anxiety and histaminergic neurotransmission in classical animal models. The H1R antagonist chlorpheniramine improved anxiety in the rat elevated plus maze test (EPM) and the open field test (OFT).8 It has also been reported that anxiety-like behavior is decreased in the EPM test for mice lacking H1Rs.9
The possible involvement of H3R function in depression has been described previously.10,11 Lamberti et al found that the highly selective H1R agonist 2-(3-trifluoromethylphenyl) histamine, the better known H1R agonist 2-thiazolylethylamine, and the standard H3R antagonist/inverse agonist thioperamide had antidepressant-like activity in the mouse forced swim test (FST).12 Moreover, it has been reported very recently that the newly developed non-imidazole H3R antagonist, 3,5-dimethyl-isoxazole-4-carboxylic acid [2-methyl-4-((2S,3′S)-2-methyl-[1,3′]bipyrrolidinyl-1′-yl)phenyl] amide, was active in the FST, suggesting the potential therapeutic utility of H3R antagonists/inverse agonists as antidepressive agents.13
Taken together, these findings support the concept that the histaminergic system enhances an anxiogenic-like response mainly via activation of H1Rs and inhibition of H3Rs. However, there is still quite limited information available regarding the psychopharmacologic profiles of H3R antagonists/inverse agonists, in particular their therapeutic value for anxiety and depression disorders.14 The H3R subtype, as a presynaptic autoreceptor, was found to suppress the synthesis and release of histamine in the central nervous system.15,16 It was also shown to behave as a presynaptic heteroreceptor, modulating the release of many other important neurotransmitters, such as dopamine, noradrenaline, acetylcholine, gamma aminobutyric acid, and serotonin.16–19
Therefore, in this study the effects of the novel highly potent and selective non-imidazole H3R antagonist, ST-1283 [3-(5-methyl-4-(4-(3-(piperidin-1-yl)propoxy)phenyl)-4H-1,2,4-triazol-3-yl)pyridine], with high in vitro human H3R affinity in the subnanomolar concentration range and a pKi value of 9.6220 was investigated for its effect on anxiety-related and depression-related behaviors in adult male C57BL/6 mice.
Materials and methods
Animals
Adult male C57BL/6 mice (aged 14 weeks and weighing 24–30 g) bred in the local central animal facility of the College of Medicine and Health Sciences (United Arab emirates University) were used in all the experiments. The animals were group-housed (five per cage) in a temperature-controlled vivarium (approximately 22°C), on a 12–12-hour light–dark cycle, with lights on at 6 am. The mice were acclimated to our testing facility for 7 days before any experimental procedure. Bedding was produced locally and autoclaved before use, and the mice had free access to tap water and a standard rodent chow diet (except as specified later) obtained from the National Feed and Flour Production and Marketing Company LLC (Abu Dhabi, United Arab Emirates). All procedures were approved by the College of Medicine and Health Sciences Animal Research Care and Use Ethics Committee (approvals A15-11 and A28-12). All efforts were made to minimize suffering and the number of animals used.
Drugs
The H3R antagonist 3-(5-methyl-4-(4-(3-(piperidin-1-yl) propoxy)phenyl)-4H-1,2,4-triazol-3-yl) pyridine (ST-1283, 5 mg/kg and 7.5 mg/kg) and the H3R agonist R-α-methylhistamine (RAMH, 10 mg/kg) were synthesized by the Institut für Pharmazeutische Chemie, Goethe University Frankfurt am Main, Germany, and were validated in a previous study.20 Diazepam 1 mg/kg manufactured by Gulf Pharmaceutical Industries (Ras Al Khaimah, United Arab Emirates) was obtained from Dr Essam Emam (Department of Medicine, Tawam Hospital, Al Ain, United Arab Emirates) and fluoxetine (Prozac®, 10 mg/kg) was obtained from Eli Lilly (Indianapolis, IN, USA). Both diazepam and fluoxetine were used as reference compounds. All drugs were diluted in isotonic saline and injected intraperitoneally at a volume of 10 mL/kg adjusted to body weight 30 minutes before each behavioral experiment. The experimental groups were as follows: vehicle (n=9), ST-1283 5 mg/kg (n=10), ST-1283 7.5 mg/kg (n=7), diazepam (n=8), and fluoxetine (n=8).
Behavioral studies
These experiments provide the first behavioral assessment of the ST-1283-injected mice in anxiety-like and depression-like behaviors in mice.
Elevated plus maze test
The EPM test was performed as previously described.21–24 Briefly, a four wooden-armed apparatus was elevated 40 cm above the ground and consisted of two opposite open arms (40 cm × 6 cm) and two opposite closed arms of the same size with high black-painted walls. The arms were connected by a central square (6 cm × 6 cm). An animal was placed in the center of the maze facing an open arm. Testing took place between 9 am and 1 pm in an order randomized for drug treatment. The maze was kept light with a 60 W bulb placed at a height of approximately one meter above the maze. The amount of time spent with head and forepaws on the open arms and closed arms of the maze as well as the number of entries into each arm was manually scored for 5 minutes. The maze was thoroughly cleaned with a tissue dampened with 70% (volume/volume; v/v) alcohol to remove the odor after each mouse was tested. The total number of entries into the closed arms is usually used as an index of locomotor activity in the test.21–24
Open field test
The OFT was performed as previously described for C57BL/6 mice.21–24 Briefly, the open field was a 32 cm × 32 cm white plexiglass square arena surrounded by a 20 cm high wall and divided into 64 equal squares by black lines. The 16 central squares are regarded as the “center” of the field. A 60 W light bulb was positioned approximately one meter above the arena. Mice were transferred one hour beforehand to the testing room and placed in the center of the field. The following parameters were manually scored: the number of lines crossed (defined as at least three paws in a square) and time spent in the center of the arena during a 10-minute test. Less time spent in the central area is usually taken as a measure of a higher level of anxiety and vice versa. After each test, the arena was sprayed with 70% ethanol and wiped thoroughly to remove the residual odor.
Marble burying test
We followed the test as previously described21,23 for analysis of the effects of ST-1283 on the marble burying test (MBT). Briefly, this test was performed using a white plexiglass cage with approximately 5 cm deep sawdust bedding lightly pressed to give a flat surface. Twenty glass marbles were evenly spaced over the bedding. The mice were placed in the center of the marble-containing cage and the total duration of digging bouts was manually recorded for each animal in a 10-minute test. The mouse was then returned to its home cage, and the number of buried marbles was scored. A marble was considered partially buried if at least 70% of its surface was covered by bedding.21,23 Marbles that were no longer visible were considered completely buried.
Tail suspension test
This method, reported originally by Steru et al,25 was followed with slight modifications.23 In brief, each mouse was suspended on the edge of a rod 50 cm above a table top using adhesive Scotch tape placed approximately 1 cm from the tip of the tail. Tail climbing was prevented by passing the mouse’s tail through a small plastic cylinder prior to suspension, as described by Can et al.26 The duration of immobility was manually scored for a 6-minute observation period. Mice were considered immobile only when they hung down passively and were completely motionless. The parameter recorded was the number of seconds spent immobile.
Novelty suppressed feeding test
The novelty suppressed feeding (NSF) test was performed as described previously.23 Briefly, 24 hours before testing, the mice were food-deprived, and only water was available. At the time of testing, each mouse was placed in one corner of a clear plastic box and allowed to explore for a maximum of 15 minutes. The test box (32 cm × 32 cm × 15 cm) was filled with 2 cm of fresh autoclaved bedding and three preweighed food pellets were placed on a circular white filter paper in the center of the arena. The time taken to bite a food pellet was manually scored. Immediately after an eating event, the mouse was placed back to its home cage and allowed to feed freely for 5 minutes. The amount of food consumption in the home cage was measured. For this test, latency until eating food was measured as well as amount of food consumed.
Forced swim test
The FST test was performed according to the method originally described by Porsolt et al.27 Mice were individually placed into glass cylinders containing 15 cm of water at ~25°C. The mice were left in the cylinders for 6 minutes and immobility was analyzed. The mice were then removed from the container and left to dry in a heated enclosure before being returned to their home cages. The mice were judged to be immobile when they ceased struggling and remained floating motionless in the water (without any vertical or horizontal movements), making only the movements necessary to keep their heads above the water level as described previously.23
Statistical analysis
IBM® SPSS Statistics® version 20 software (IBM Middle East, Dubai, United Arab Emirates) was used for all statistical comparisons. Mean values and standard errors were calculated for each group. Dependent variables for each behavioral model were analyzed using one-way analysis of variance, with “dose” as a between-subject factor. When relevant, post hoc analyses were performed by Student’s t-tests with Bonferroni corrections for multiple comparisons. P<0.05 denotes a statistically significant difference.
Results
C57BL/6 mice exhibited anxiolytic activity following ST-1283 administration
Elevated plus maze test
Figure 1 shows a dose-response curve for the effects of acute administration of ST-1283 (0, 5, or 7.5 mg/kg) on the anxiety indices (percentage of time spent in open arms, number of entries into open arms, and percentage entries into open arms) and locomotor activity (number of entries into closed arm) of mice exposed to the EPM. One-way analysis of variance showed that ST-1283 dose-dependently increased the percentage of time spent exploring the open arms of the maze during a 5-minute session (F(2,23) =3.959, P=0.033, Figure 1A). Relative to the vehicle condition, only the 7.5 mg/kg dose of ST-1283 produced a significant increase in the percentage of time spent on the open arms (P=0.038 and P=0.180 for each dose, respectively). As a positive control, and compared with vehicle, diazepam 1 mg/kg induced a significant increase in the percentage of time spent exploring the open arms (F(1,15) =26.220, P<0.001). Analyses of data characterizing the number of entries into the open arms of the maze (F(2,23) =5.270, P=0.013) yielded essentially the same results. As shown in Figure 1B, only the highest dose of ST-1283 (7.5 mg/kg) was significantly different from that obtained with vehicle pretreatment (P=0.011). Diazepam 1 mg/kg also increased the number of entries into the open arms (F(1,15) =31.424, P<0.001). Similarly, pretreatment with ST-1283 altered the percentage of entries into the open arms (F(2,23) =4.894, P=0.017). Post hoc evaluation revealed that, compared with vehicle, the mice injected with the higher dose of ST-1283 (7.5 mg/kg) displayed a higher percentage of entries into the open arms (P=0.015). In contrast, no significant difference was found between vehicle- and ST-1283 (5 mg/kg)-treated mice (P=0.221, Figure 1C). As a positive control, diazepam 1 mg/kg induced a significant increase in the percentage of entries into the open arms (F(1,15) =24.436, P<0.001). Interestingly, no significant changes were found in the number of closed arm entries following ST-1283 injection (F(2,23) =0.044, P=0.957, Figure 1D), nor following diazepam injection (F(1,15) =0.302, P=0.590), indicating that locomotor activity per se was not affected following ST-1283 injection relative to that obtained with saline pretreatment. Thus, the observed behavioral changes were not accompanied by any significant alterations in the distance traveled during this period.
Figure 1.
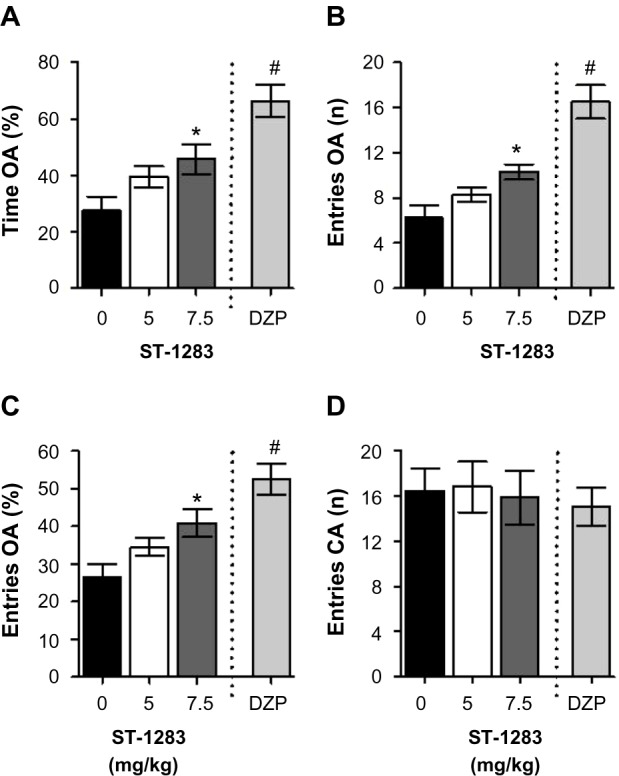
Effects of acute ST-1283 pretreatment on exploratory behavior on the ele vated plus maze test. ST-1283 dose-dependently increased the percentage of time spent on the open arms of the elevated plus maze (A), increased the number of entries into the open arms (B) and the percentage of entries into the open arms (C). Pretreatment with the H3R antagonist did not affect the number of closed arm entries (D).
Notes: *Denotes significant differences between drug doses and saline controls (P<0.05); #denotes significant differences between diazepam and saline controls (P<0.001). Vehicle n=9; ST-1283 5 mg/kg n=10; ST-1283 7.5 mg/kg n=7, diazepam n=8. Data are shown as the mean ± standard error of the mean.
Abbreviations: DZP, diazepam; OA, open arms.
Open field test
We used the OFT to further corroborate anxiety-like behavior and simultaneously rule out possible intrinsic impairment of spontaneous locomotor activity. Locomotor activity was measured by the number of line crossings in the arena. One-way analysis of variance showed that, compared with vehicle, both ST-1283 and diazepam had no effect on total line crossings (F(2,23) =0.247, P=0.783) and (F(1,15) =0.055, P=0.818), respectively (Figure 2A). However, significant treatment differences were found with regard to time spent in the central area (F(2,23) =4.309, P=0.026). In fact, mice injected with the ST-1283 (7.5 mg/kg) spent more time in the central area (P=0.026 compared to vehicle). However, at the lower dose, ST-1283 had no effect on time spent in the central area (P=0.198 compared to vehicle) (Figure 2B). As a positive control, diazepam 1 mg/kg also increased the amount of time spent in the center of the arena (F(1,15) =51.575, P<0.001).
Figure 2.
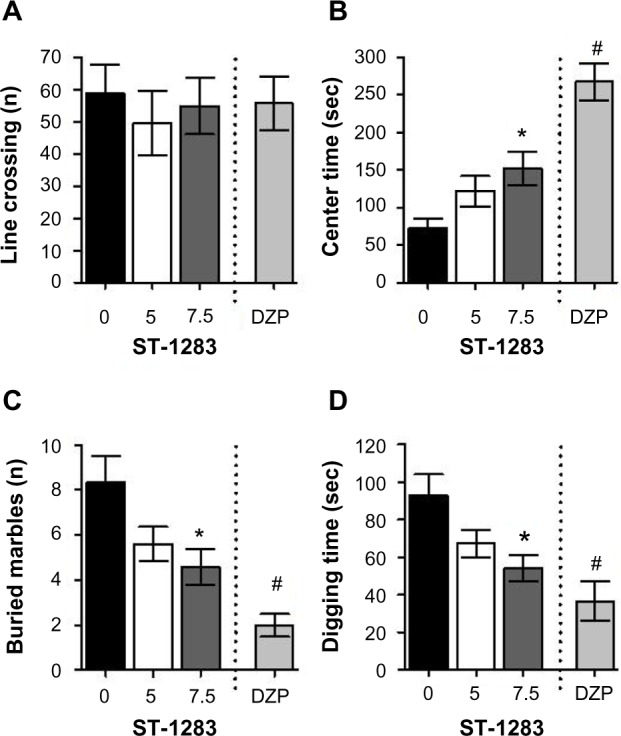
Effects of acute ST-1283 pretreatment on anxiety-like behavior in the open field test and marbles burying test. In the open field test, ST-1283 had no effect on total line crossing (A) but dose-dependently increased the time spent in the center of the arena (B). In the marbles burying test, acute ST-1283 decreased the number of buried marbles (C) and the time spent digging (D).
Notes: *Denotes significant differences between drug doses and saline controls (P<0.05); #denotes significant differences between diazepam and saline controls (P<0.001). Vehicle n=9; ST-1283 5 mg/kg n=10; ST-1283 7.5 mg/kg n=7, DZP n=8. Data are shown as the mean ± standard error of the mean.
Abbreviation: DZP, diazepam.
Marbles burying test
We then explored differences in anxiety-like and obsessive– compulsive-like behavior in vehicle-treated and ST-1283-treated mice using the digging test and the MBT. In parallel with our findings in the OFT and EPM tests, ST-1283-treated mice showed a significant decrease in the number of buried marbles compared with vehicle-injected mice (F(2,23) =4.114, P=0.030). Post hoc evaluations revealed that mice injected with the higher (7.5 mg/kg) dose showed an approximately 1.8-fold decrease in the number of buried marbles (P=0.039 and P=0.127 for vehicle versus ST-1283 5 mg/kg, Figure 2C). Diazepam 1 mg/kg also decreased the number of buried marbles (F(1,15) =23.166, P<0.001). In addition, analysis of digging duration showed a main effect of treatment (F(2,23) =4.517, P=0.022). In fact, mice injected with the ST-1283 (7.5 mg/kg) spent more time digging (P=0.025 compared to vehicle). However, at the lower dose, ST-1283 had no effect on time spent digging (P=0.141 compared to vehicle) (Figure 2D). Further, compared with vehicle, the positive diazepam control showed a decrease in time spent digging (F(1,15) =12.912, P=0.003).
ST-1283 injection reduced depression-like behavior in C57BL/6 mice
Because anxiety and depression are often comorbid, we next analyzed the effects of H3R blockade with ST-1283 using three measures of depression, ie, the tail suspension test (TST), NSF, and FST.
Tail suspension test
In the TST, acute treatment with either ST-1283 (F(2,23) =3.846, P=0.036) or fluoxetine 10 mg/kg intraperitoneally (F(1,15) =17.020, P=0.001) elicited a significant change in immobility time compared with vehicle. As shown in Figure 3A, there was a significant effect of ST-1283 on immobility time at the high dose of 7.5 mg/kg (P=0.042 versus vehicle). In contrast, low-dose peripheral administration of ST-1283 5 mg/kg did not significantly alter immobility time in the mouse TST model (P=0.182 versus vehicle).
Figure 3.
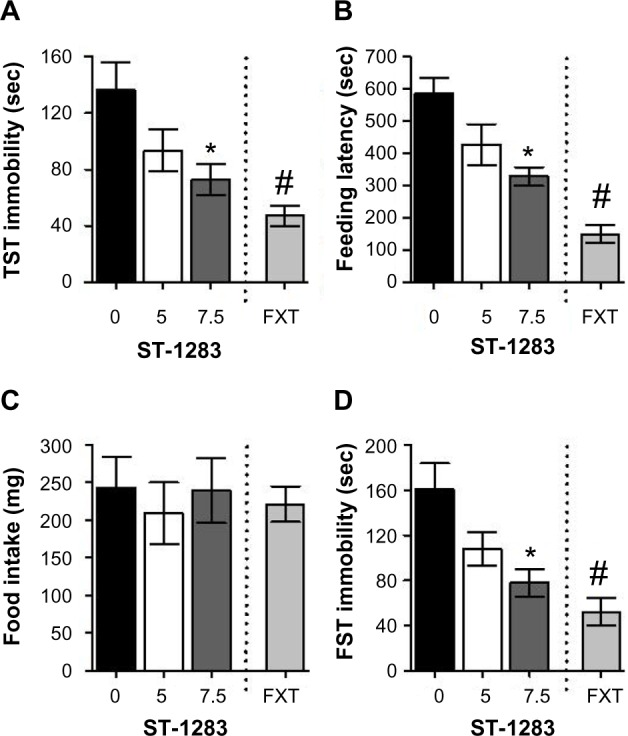
Effects of acute ST-1283 pretreatment on depression-like behavior in the TST, the novelty suppressed feeding test, and the forced swim test. Acute ST-1283 decreased immobility time in the TST (A) and decreased the feeding latency in the novelty suppressed feeding test (B). However, ST-1283 did not affect eating in the home cage (C). Similarly, ST-1283 dose-dependently decreased the immobility time in the forced swim test (D).
Notes: *Denotes significant differences between drug doses and saline controls (P<0.05); #denotes significant differences between fluoxetine and saline controls (P<0.001). Vehicle n=9; ST-1283 5 mg/kg n=10; ST-1283 7.5 mg/kg n=7, fluoxetine n=8. Data are shown as the mean ± standard error of the mean.
Abbreviations: TST, tail suspension test; FXT, fluoxetine; FST, forced swim test.
Novelty suppressed feeding
We next tested the groups of mice using the NSF test. Acute treatment with ST-1283 significantly improved the NSF test results by reducing feeding latency (F(2,23) =5.530, P=0.011). As shown in Figure 3B, at the higher dose, ST-1283-treated mice showed significantly shorter feeding latencies than control mice (P=0.011). However, at the 5 mg/kg dose, ST-1283-injected animals did not show significantly reduced feeding latency compared with controls (P=0.115). This was not due to increased appetite because food consumption in home cage was not significantly changed following acute admin istration of ST-1283 (F(2,23) =0.217, P=0.807, Figure 3C). As the positive control, treatment with fluoxetine led to an overall decrease in time to first eating event (F(1,15) =58.013, P<0.001, Figure 3B) in the NDF test, but did not affect food intake in the home cage (F(1,15) =0.208, P=0.655, Figure 3C).
Forced swim test
Similar to the antidepressant-like activity reported in the TST and the NSF, acute treatment with ST-1283 significantly affected immobility time in the mouse FST (F(2,23) =4.750, P=0.019). As shown in Figure 3D, only high-dose administration of ST-1283 decreased immobility times in the FST (P=0.019). However, no change in immobility time was observed when mice were injected with ST-1283 5 mg/kg (P=0.153 versus vehicle). As the positive control, fluoxetine 10 mg/kg induced a significant decrease in immobility (F(1,15) =15.808, P=0.001).
RAMH blocked ST-1283-attenuated anxiety-like behavior in mice
In this set of experiments we tested whether the anxiolytic and antidepressant effects induced by ST-128 can be abrogated by pretreatment with the H3R-selective agonist RAMH injected at a dose of 10 mg/kg 15 minutes before administration of ST-1283 7.5 mg/kg.
Elevated plus maze
One-way analysis of variance indicated that there were significant differences between the groups with regard to the percentage of time spent by the mice in the open arm compartments of the maze (F(2,18) =5.507, P=0.014). Figure 4A shows that, compared with vehicle, treatment with ST-1283, as expected, increased the percentage of time spent by mice in the open arms of the EPM apparatus (anxiolytic-like effect; P=0.023). However, acute administration of RAMH at the 10 mg/kg dose counteracted the anxiolytic-like effect of ST-1283 (P=0.032 versus ST-1283 and P=1.000 versus vehicle). When the number of entries in the open arms was measured, there were significant differences between the groups (F(2,18) =7.355, P=0.005). As shown in Figure 4B, post hoc evaluations revealed that ST-1283 increased the number of entries into the open arms (P=0.007 versus vehicle). However, preinjection with RAMH reversed the anxiolytic-like effect of ST-1283 (P=0.014) and lowered the number of open arm entries. Similarly, one-way analysis of variance showed a significant effect of drug treatment on the percentage of entries into the open arms in the EPM test (F(2,18) =5.540, P=0.013). Post hoc comparisons revealed that ST-1283 7.5 mg/kg significantly increased the percentage of entries into the open arms compared with the vehicle-treated group (P=0.023), and treatment with RAMH abrogated the ST-1283-induced anxiolytic effect (P=0.031, Figure 4C). As shown in Figure 4D, measures of general activity (closed arm entries) did not differ significantly between any of the groups (F(2,18) =1.142, P=0.341).
Figure 4.
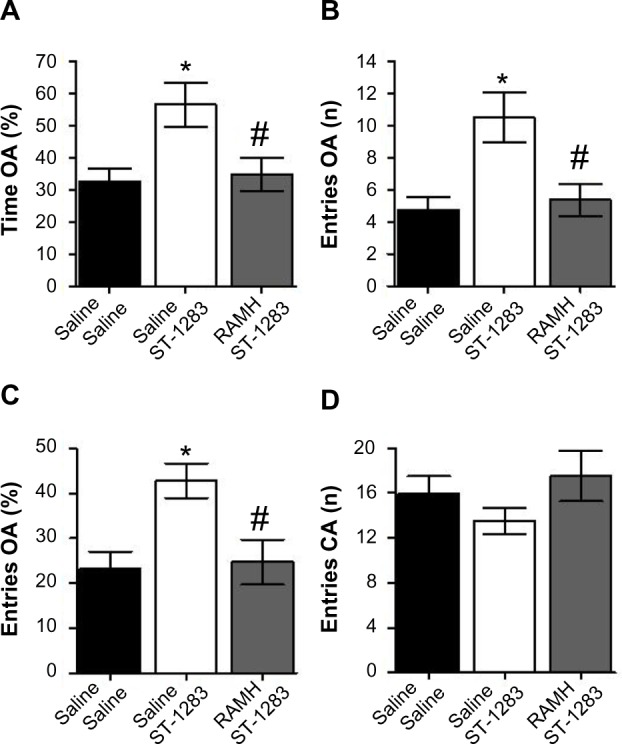
Effects of acute RAMH pretreatment on ST-1283-induced anxiolytic effects in the elevated plus maze test. RAMH pretreatment abrogated the effects of ST-1283 on the percentage of time spent in the open arms (A), the number of entries into the open arms (B), and the percentage of entries into the open arms (C). Pretreatment with the H3R agonist RAMH did not affect the number of closed arm entries (D).
Notes: *Denotes significant differences between ST-1283 and saline controls (P<0.05); #denotes significant differences between RAMH and ST-1283 (P<0.05). Vehicle n=7; ST-1283 7.5 mg/kg n=6, RAMH n=8. Data are shown as the mean ± standard error of the mean.
Abbreviations: CA, closed arm; OA, open arm; RAMH, R-α-methylhistamine.
Open field test
In addition to EPM, the OFT was used to examine anxiety-like behavior and locomotion. As seen in Figure 5A, there were no significant effects of acute exposure to ST-1283 and RAMH on overall locomotor activity (F(2,18) =0.015, P=0.985). In contrast, one-way analysis of variance revealed that there was a main effect of treatment on time spent in the center of the arena (F(2,18) =5.095, P=0.018). Post hoc evaluations demonstrated that, as expected, mice treated with ST-1283 spent significantly more time in the center of the arena than those treated with vehicle (P=0.032). Interestingly, this effect was inhibited when RAMH was injected before ST-1283 (P=0.038, Figure 5B).
Figure 5.
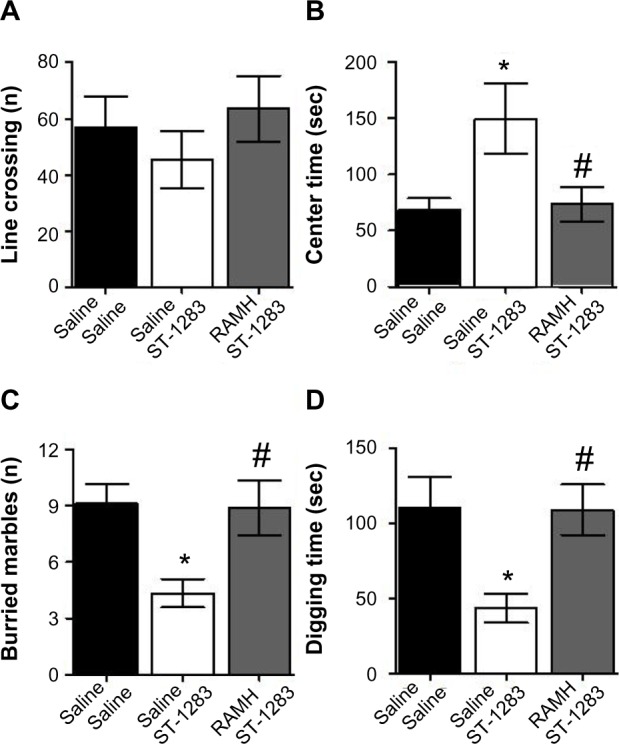
Effects of acute RAMH pretreatment on ST-1283-induced anxiolytic effects in the open field test and marbles burying test. In the open field test, RAMH pretreatment had no effect on total line crossing (A) but abrogated the effects of ST-1283 on the time spent in the center of the arena (B). In the marbles burying test, acute RAMH pretreatment blocked the effects of ST-1283 on the number of buried marbles (C) and time spent digging (D).
Notes: *Denotes significant differences between ST-1283 and saline controls (P<0.05); #denotes significant differences between RAMH and ST-1283 (P<0.05). Vehicle n=7; ST-1283 7.5 mg/kg n=6, RAMH n=8. Data are shown as the mean ± standard error of the mean.
Abbreviation: RAMH, R-α-methylhistamine.
Marbles burying test
The results of the MBT are shown in Figure 5C and D. One-way analysis of variance revealed that there was a significant treatment effect with regard to the number of marbles buried (F(2,18) =4.758, P=0.022, Figure 5C). Bonferroni post hoc comparison testing showed that the response of the control mice was significantly different from the mice that received ST-1283 (P=0.039). However, RAMH-injected mice buried significantly more marbles than the ST-1283-treated animals (P=0.046). To assess whether impaired marble burying is associated with digging behavior, we measured the time spent digging. As seen in Figure 5D, one-way analysis of variance showed that time spent digging was affected by drug treatment (F(2,18) =4.592, P=0.024). Post hoc comparison showed that mice injected with ST-1283 spent significantly less time digging than controls (P=0.048). However, acute injection of RAMH reversed the effect of ST-1283 (P=0.045).
RAMH blocked ST-1283-attenuated depression-like behavior in mice
Tail suspension test
To examine further the role of H3R in the behavioral effects of ST-1283, we investigated whether RAMH could block the effects of ST-1283. The immobility time in the TST for animals treated with ST-1283 with or without RAMH is shown in Figure 6A. One-way analysis of variance showed that there was a main effect of treatment (F(2,18) =5.502, P=0.014). Post hoc analysis indicated a significant decrease in immobility time elicited by administration of ST-1283 7.5 mg/kg (P=0.021). In contrast, pretreatment with RAMH significantly increased the amount of immobility time in the TST as compared with the group treated with ST-1283 alone (P=0.036).
Figure 6.
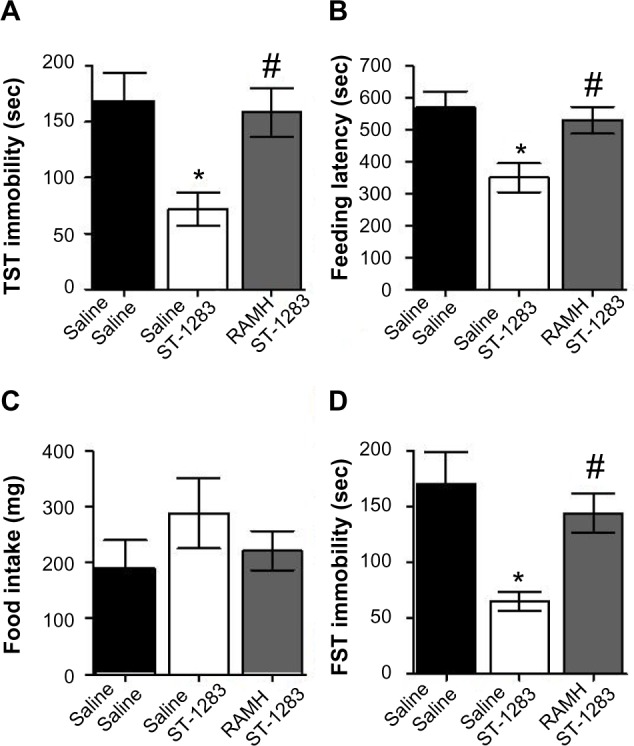
Effects of acute RAMH pretreatment on ST-1283-induced antidepressant effects in the TST, feeding latency and food intake in the NSF, and FST.
Notes: *Denotes significant differences between ST-1283 and saline controls (P<0.05); #denotes significant differences between RAMH and ST-1283 (P<0.05). Vehicle n=7; ST-1283 7.5 mg/kg n=6, RAMH n=8. Data are shown as the mean ± standard error of the mean.
Abbreviations: RAMH, R-α-methylhistamine; FST, forced swim test; NSF, novelty suppressed feeding; TST, tail suspension test.
Novelty suppressed feeding
The effects of acute ST-1283 with and without RAMH treatment were tested in C57BL/6 mice using the NSF test. One-way analysis of variance revealed a significant effect of drug treatment (F(2,18) =5.841, P=0.011) on feeding latency (Figure 6B). Bonferroni post hoc evaluation indicated that, compared with vehicle, acute intraperitoneal administration of ST-1283 7.5 mg/kg reduced the latency to feed in the NSF test (P=0.014). These effects of ST-1283 were completely blocked by RAMH (P=0.041), with a similar magnitude of effect to that seen in the vehicle group (P=1.000). Home cage feeding was assessed for each mouse by returning it to its familiar environment immediately after the NSF test and measuring the amount of food consumed over a period of 5 minutes. As depicted in Figure 6C, acute administration of ST-1283 alone or with RAMH had no effect on home food consumption (F(2,18) =0.985, P=0.393).
Forced swim test
The findings of the FST are shown in Figure 6D. One-way analysis of variance revealed that the drug treatments had a significant effect on time spent immobile (F(2,18) =6.261, P=0.009). Post hoc comparison showed that, as expected, ST-1283 7.5 mg/kg significantly decreased immobility time (P=0.009 versus vehicle). These effects of ST-1283 were completely blocked by RAMH (P=0.049), with no significant difference in behavior found between the RAMH and vehicle groups (P=1.000).
Discussion
An extensive in vivo pharmacologic and behavioral evaluation was performed and clearly indicated the efficacy profile of the H3R antagonist ST-1283 in animal models of anxiety and depression. To the authors’ knowledge, these studies provide the first evidence that an H3 antagonist belonging to the non-imidazole class has anxiolytic-like and antidepressant-like effects. In consideration of anxiolytic-like effects, our major finding was that acute blockade of H3Rs altered anxiety-like behavior in the EPM mouse model. At a dose of 7.5 mg/kg, ST-1283 significantly increased the percentage of time spent in the open arms of the maze. The number and percentage of open arm entries were also significantly increased in response to treatment with ST-1283. Typically, in studies based on EPM, either the number of open arm entries or the percentage of time spent or both have been taken into consideration.28–30 When both parameters were included, the anxiolytic-like action of ST-1283 was only evident at a dose of 7.5 mg/kg and was significantly higher when compared with the 5 mg/kg dose. Interestingly, our results showed that both ST-1283 7.5 mg/kg and diazepam 1 mg/kg did not change the number of entries into the closed arms, thereby excluding the possibility of artifactual changes in general behavior or activity following pretreatment with ST-1283 as compared with saline pretreatment. The suggestion that the changes are associated with anxiolytic-like effects is consistent with previous experimental results indicating that H3−/− knockout mice show fewer anxiety-like effects in the EPM model.31 However, our results are in conflict with those of a recent study by Mohsen et al who reported that the specific H3R antagonist JNJ-10181457 (10 mg/kg) was anxiogenic when C57BL/6 mice were tested in the elevated zero maze.32 The discrepancy between these studies regarding the effect of H3R antagonists could be explained by differences in procedure (EPM versus elevated zero maze) and mouse age (14 weeks versus 7 weeks). It should be also emphasized that Mohsen et al used a repeated exposure approach whereby mice were first habituated to the maze on day 1 and tested in the presence of the H3R antagonist JNJ-10181457 24 hours later.32 However, in our study, the mice were naïve to the EPM and were only tested once in the presence of the drug. Consequently, care should be taken with regard to such factors in future studies. It is clear that more research is required to determine further the reasons for the different observations with H3R antagonists regarding anxiety-like behaviors. Therefore, local stereotaxic injection of viral vectors to modulate H3R function in discrete brain regions is currently under consideration to test the impact of the H3R on emotion-related behavior.
Moreover, by testing in the EPM it is important to ensure that there are no differences in overall activity levels in the experimental groups, since reduced activity could otherwise wrongfully be interpreted as increased measures of anxiety.29 Therefore, we investigated ST-1283 further in the mouse OFT model, and our results show that neither ST-1283 (5 mg/kg and 7.5 mg/kg) nor the reference anxiolytic drug diazepam (1 mg/kg) altered the total amount of line crossings when compared with vehicle. However, significant treatment differences were found for time spent in the center of the arena, with mice injected with the higher dose (7.5 mg/kg) but not the lower dose (5 mg/kg) showing higher scores than those injected with vehicle. Therefore, the results obtained clearly validate the anxiolytic-like behavior and at the same time exclude possible intrinsic impairment in spontaneous locomotor activity following treatment with ST-1283.
To examine further the possible effect of acute H3R blockade on anxiety-like behavior in the mouse, ST-1283 was tested in the MBT. Interestingly, the results for the MBT were consistent with those for the EPM and OFT, since only pretreatment with ST-1283 7.5 mg/kg significantly decreased the number of buried marbles and significantly shortened the digging duration, demonstrating the anxiolytic-like effects of ST-1283.
It is well known that both anxiety disorders and depressive disorders are highly prevalent and frequently comorbid conditions. Therefore, the antidepressant-like effect of pretreatment with ST-1283 (5 mg/kg and 7.5 mg/kg, intraperitoneally) was tested in the mouse TST, NSF, and FST models. Our results showed that acute H3R blockade significantly reduced immobility time and feeding latency when compared with vehicle. These results further support the role of H3Rs in neurobehavioral disorders and also corroborate the main conclusion of the current study, ie, that H3R blockade is associated with an antidepressant-like effect in mice. Importantly, the NSF test results showed that food intake was not altered following acute treatment with ST-1283 (5 mg/kg and 7.5 mg/kg) or with the reference drug fluoxetine (10 mg/kg, intraperitoneally). Further, the antidepressant-like activity observed for ST-1283 in the TST and NSF were confirmed by the mouse FST, and immobility time was significantly decreased when the mice were injected with 7.5 mg/kg. Interestingly, our results are in agreement with a previous study in which the H3R antagonist thioperamide (10 mg/kg, intraperitoneally) showed antidepressant-like effects in the mouse FST model.33 Moreover, consistent with the antidepressant-like effect of ST-1283 observed in our study, the histamine-N-methyltransferase inhibitor, metoprine (2, 7, and 20 mg/kg, subcutaneously) and the histamine precursor L-histidine (500 and 1,000 mg/kg subcutaneously) also showed an antidepressant-like effect in an earlier study by Lamberti at al using the FST.12
The mouse FST is normally used to assess potential antidepressant compounds based on the abilities of clinically effective antidepressants to reduce the immobility that animals typically display after active and unsuccessful attempts when exposed to inevitable stressors.27 The FST is sufficiently specific, given that it discriminates antidepressants from neuroleptics and anxiolytics.3,7 However, ligands enhancing locomotor activity may give rise to a false-positive effect in this test.34,35 Therefore, the number of entries into the closed arms in the EPM test and the number of line crossings in the OFT were used as indicators of locomotor activity. Our results showed that ST-1283 at a dose of 7.5 mg/kg did not alter locomotor activity in mice. Thus, the anxiolytic and antidepressant actions observed for ST-1283 seem unlikely to be due to an increase in locomotor activity of the mice.
Interestingly, ST-1283 at a dose of 7.5 mg/kg exhibited both anxiolytic-like and antidepressant-like actions. However, the question remains as to the mechanism by which ST-1283 exerts these effects. At least part of the mechanism behind the ST-1283-induced anxiolytic and antidepressant effects appears to be associated with the inhibitory effect of ST-1283 on H3Rs and subsequently enhanced synthesis and release of histamine. Pretreatment with 10 mg/kg of the H3R agonist RAMH abrogated the anxiolytic-like effects observed for ST-1283 in the EPM, OFT, and MBT, without affecting the general activity of the treated mice. Moreover, the antidepressant-like behaviors observed for ST-1283 in the mouse TST, NSF, and FST were also blocked following administration of RAMH 10 mg/kg. Importantly, previous studies have shown that RAMH and immepip dihydrobromide, both H3R agonists, do not affect spontaneous locomotor activity, even at doses much higher than those in the pharmacologically effective range, suggesting that these H3 agonists do not induce hypolocomotion, which may result from suppression of histaminergic neurotransmission.36,37 Moreover, previous studies have shown that anxiolytic drugs such as diazepam and buspirone (a serotonin 5-HT1A agonist) significantly inhibit brain histamine turnover in rodents.38,39 Zolantidine, a central nervous system-penetrating H2R antagonist, was also found to amplify the anxiogenic-like effect induced by thioperamide, an imidazole-substituted standard H3R antagonist/inverse agonist, in the mouse light/dark box test.40 These findings further corroborate the association of histaminergic neurotransmission with antianxiety-like and antidepressant-like behaviors. Further, the H3R subtype acts as a presynaptic autoreceptor and heteroreceptor primarily in the central nervous system, controlling the synthesis and release of histamine and modifying the release of several other neurotransmitters, eg, dopamine, serotonin, gamma aminobutyric acid, noradrenaline, and acetylcholine.6,7 Therefore, further investigations are needed to clarify whether other monaminergic neurotransmitters are involved in the effects observed with ST-1283. Further, species differences in affinity for H3R between humans and mice have to be considered when evaluating the preliminary results obtained for ST-1283.41 Moreover, anxiety-related behaviors were recently found to be mainly mediated via H2Rs, highlighting the potential importance of investigating the affinity of ST-1283 for H2Rs in humans and mice to further corroborate the results obtained in the current study.
Conclusion
The H3R antagonist ST-1283 appears to be an antidepressant compound with anxiolytic properties at the same dose level. The results of this study could lead to a search for a new group of antidepressant and anxiolytic compounds. However, the exact mechanisms by which H3R antagonists exert their action in animal models of anxiety and depression await full elucidation. Nonetheless, considerable experimental effort is still required to fully understand the mechanisms of the histaminergic interaction in anxiety and depression and to assess the potential utility of H3R antagonists/inverse agonists as therapeutic possibilities in the treatment of anxiety-like and depression-like disorders.
Acknowledgments
This work was supported by grants from the United Arab Emirates University (to AB and BS) and by the EU COST Actions (BM0806, BM1007, CM1103, and CM1207), Hesse LOEWE Schwerpunkte Fh-TMP, OSF and NEFF, the Else KrönerStiftung, TRIP, and the Deutsches Konsortium für Translationale Krebsforschung DKTK (to HS). The funders played no part in the study design, or in the collection, analysis, or interpretation of the data, writing of the report, or the decision to submit the paper for publication. The authors would like to thank Dr Essam Emam (Department of Medicine, Tawam Hospital, Al Ain, United Arab Emirates) for providing the diazepam used in this study, Mohamed Elwasila and Mohamed Shafiullah for their technical assistance, and Dr Mahmoud Hag Ali from the Central Animal Facility for his advice regarding veterinary care. The authors would also like to acknowledge Professor Keith Bagnall for his critical and careful proofreading.
Author contributions
AB and BS were responsible for the study concept and design. AB contributed to the acquisition and analysis of the animal data. HS, BS, JSS, and MW were responsible for the generation, synthesis, and in vitro pharmacologic characterization of ST-1283. AB and BS drafted the manuscript. HS critically revised the manuscript. All authors critically reviewed the content of the manuscript and approved the final version for publication.
Disclosure
The authors have no financial interests that might be perceived to influence the results or the discussion reported in this article.
References
- 1.Kent JM, Coplan JD, Gorman JM. Clinical utility of the selective serotonin reuptake inhibitors in the spectrum of anxiety. Biol Psychiatry. 1998;44(9):812–824. doi: 10.1016/s0006-3223(98)00210-8. [DOI] [PubMed] [Google Scholar]
- 2.McLeod DR, Hoehn-Saric R, Zimmerli WD, De Souza EB, Oliver LK. Treatment effects of alprazolam and imipramine: physiological versus subjective changes in patients with generalized anxiety disorder. Biol Psychiatry. 1990;28(10):849–861. doi: 10.1016/0006-3223(90)90567-l. [DOI] [PubMed] [Google Scholar]
- 3.Borsini F, Podhorna J, Marazziti D. Do animal models of anxiety predict anxiolytic-like effects of antidepressants? Psychopharmacology (Berl) 2002;163(2):121–141. doi: 10.1007/s00213-002-1155-6. [DOI] [PubMed] [Google Scholar]
- 4.Zahreddine N, Richa S. Non-antidepressant treatment of generalized anxiety disorder. Curr Clin Pharmacol. 2013 Feb 4; doi: 10.2174/15748847113089990058. [Epub ahead of print.] [DOI] [PubMed] [Google Scholar]
- 5.Leurs R, Bakker RA, Timmerman H, de Esch IJ. The histamine H3 receptor: from gene cloning to H3 receptor drugs. Nat Rev Drug Discov. 2005;4(2):107–120. doi: 10.1038/nrd1631. [DOI] [PubMed] [Google Scholar]
- 6.Witkin JM, Nelson DL. Selective histamine H3 receptor antagonists for treatment of cognitive deficiencies and other disorders of the central nervous system. Pharmacol Ther. 2004;103(1):1–20. doi: 10.1016/j.pharmthera.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 7.Bhowmik M, Khanam R, Vohora D. Histamine H3 receptor antagonists in relation to epilepsy and neurodegeneration: a systemic consideration of recent progress and perspectives. Br J Pharmacol. 2012;167(7):1398–1414. doi: 10.1111/j.1476-5381.2012.02093.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hasenohrl RU, Weth K, Huston JP. Intraventricular infusion of the histamine H(1) receptor antagonist chlorpheniramine improves maze performance and has anxiolytic-like effects in aged hybrid Fischer 344xBrown Norway rats. Exp Brain Res. 1999;128(4):435–440. doi: 10.1007/s002210050866. [DOI] [PubMed] [Google Scholar]
- 9.Yanai K, Son LZ, Endou M, et al. Behavioural characterization and amounts of brain monoamines and their metabolites in mice lacking histamine H1 receptors. Neuroscience. 1998;87(2):479–487. doi: 10.1016/s0306-4522(98)00167-5. [DOI] [PubMed] [Google Scholar]
- 10.Ghi P, Ferretti C, Blengio M. Effects of different types of stress on histamine-H3 receptors in the rat cortex. Brain Res. 1995;690(1):104–107. doi: 10.1016/0006-8993(95)00542-x. [DOI] [PubMed] [Google Scholar]
- 11.Ghi P, Ferretti C, Blengio M, Portaleone P. Stress-induced changes in histaminergic system: effects of diazepam and amitriptyline. Pharmacol Biochem Behav. 1995;51(1):65–68. doi: 10.1016/0091-3057(94)00359-q. [DOI] [PubMed] [Google Scholar]
- 12.Lamberti C, Ipponi A, Bartolini A, Schunack W, Malmberg-Aiello P. Antidepressant-like effects of endogenous histamine and of two histamine H1 receptor agonists in the mouse forced swim test. Br J Pharmacol. 1998;123(7):1331–1336. doi: 10.1038/sj.bjp.0701740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gao Z, Hurst WJ, Czechtizky W, et al. Identification and profiling of 3,5-dimethyl-isoxazole-4-carboxylic acid [2-methyl-4-((2S,3′S)-2-methyl-[1,3′]bipyrrolidinyl-1′-yl)phenyl] amide as histamine H(3) receptor antagonist for the treatment of depression. Bioorg Med Chem Lett. 2013;23(23):6269–6273. doi: 10.1016/j.bmcl.2013.09.081. [DOI] [PubMed] [Google Scholar]
- 14.Raber J. Histamine receptor-mediated signaling during development and brain function in adulthood. Cell Mol Life Sci. 2007;64(6):735–741. doi: 10.1007/s00018-007-6442-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Arrang JM, Garbarg M, Schwartz JC. Autoregulation of histamine release in brain by presynaptic H3-receptors. Neuroscience. 1985;15(2):553–562. doi: 10.1016/0306-4522(85)90233-7. [DOI] [PubMed] [Google Scholar]
- 16.Arrang JM, Garbarg M, Schwartz JC. Autoinhibition of histamine synthesis mediated by presynaptic H3-receptors. Neuroscience. 1987;23(1):149–157. doi: 10.1016/0306-4522(87)90279-x. [DOI] [PubMed] [Google Scholar]
- 17.Arrang JM, Garbarg M, Schwartz JC. Auto-inhibition of brain histamine release mediated by a novel class (H3) of histamine receptor. Nature. 1983;302(5911):832–837. doi: 10.1038/302832a0. [DOI] [PubMed] [Google Scholar]
- 18.Lovenberg TW, Roland BL, Wilson SJ, et al. Cloning and functional expression of the human histamine H3 receptor. Mol Pharmacol. 1999;55(6):1101–1107. [PubMed] [Google Scholar]
- 19.Schlicker E, Betz R, Gothert M. Histamine H3 receptor-mediated inhibition of serotonin release in the rat brain cortex. Naunyn Schmiedebergs Arch Pharmacol. 1988;337(5):588–590. doi: 10.1007/BF00182737. [DOI] [PubMed] [Google Scholar]
- 20.Sadek B, Schwed JS, Subramanian D, et al. Non-imidazole histamine H3 receptor ligands incorporating antiepileptic moieties. Eur J Med Chem. 2014;77:269–279. doi: 10.1016/j.ejmech.2014.03.014. [DOI] [PubMed] [Google Scholar]
- 21.Bahi A. Individual differences in elevated plus-maze exploration predicted higher ethanol consumption and preference in outbred mice. Pharmacol Biochem Behav. 2013;105:83–88. doi: 10.1016/j.pbb.2013.01.023. [DOI] [PubMed] [Google Scholar]
- 22.Bahi A. Increased anxiety, voluntary alcohol consumption and ethanol-induced place preference in mice following chronic psychosocial stress. Stress. 2013;16(4):441–451. doi: 10.3109/10253890.2012.754419. [DOI] [PubMed] [Google Scholar]
- 23.Bahi A, Dreyer JL. Hippocampus-specific deletion of tissue plasminogen activator “tPA” in adult mice impairs depression- and anxiety-like behaviors. Eur Neuropsychopharmacol. 2012;22(9):672–682. doi: 10.1016/j.euroneuro.2012.01.008. [DOI] [PubMed] [Google Scholar]
- 24.Bahi A, Dreyer JL. Chronic psychosocial stress causes delayed extinction and exacerbates reinstatement of ethanol-induced conditioned place preference in mice. Psychopharmacology (Berl) 2013;231(2):367–377. doi: 10.1007/s00213-013-3243-1. [DOI] [PubMed] [Google Scholar]
- 25.Steru L, Chermat R, Thierry B, Simon P. The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology (Berl) 1985;85(3):367–370. doi: 10.1007/BF00428203. [DOI] [PubMed] [Google Scholar]
- 26.Can A, Dao DT, Terrillion CE, Piantadosi SC, Bhat S, Gould TD. The tail suspension test. J Vis Exp. 2012;(59):e3769. doi: 10.3791/3769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Porsolt RD, Bertin A, Jalfre M. Behavioral despair in mice: a primary screening test for antidepressants. Arch Int Pharmacodyn Ther. 1977;229(2):327–336. [PubMed] [Google Scholar]
- 28.Johnston AL, File SE. Sex differences in animal tests of anxiety. Physiol Behav. 1991;49(2):245–250. doi: 10.1016/0031-9384(91)90039-q. [DOI] [PubMed] [Google Scholar]
- 29.Pellow S, Chopin P, File SE, Briley M. Validation of open:closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Methods. 1985;14(3):149–167. doi: 10.1016/0165-0270(85)90031-7. [DOI] [PubMed] [Google Scholar]
- 30.Dunn RW, Corbett R, Fielding S. Effects of 5-HT1A receptor agonists and NMDA receptor antagonists in the social interaction test and the elevated plus maze. Eur J Pharmacol. 1989;169(1):1–10. doi: 10.1016/0014-2999(89)90811-x. [DOI] [PubMed] [Google Scholar]
- 31.Rizk A, Curley J, Robertson J, Raber J. Anxiety and cognition in histamine H3 receptor−/− mice. Eur J Neurosci. 2004;19(7):1992–1996. doi: 10.1111/j.1460-9568.2004.03251.x. [DOI] [PubMed] [Google Scholar]
- 32.Mohsen A, Yoshikawa T, Miura Y, et al. Mechanism of the histamine H receptor-mediated increase in exploratory locomotor activity and anxiety-like behaviours in mice. Neuropharmacology. 2014;81C:188–194. doi: 10.1016/j.neuropharm.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 33.Perez-Garcia C, Morales L, Cano MV, Sancho I, Alguacil LF. Effects of histamine H3 receptor ligands in experimental models of anxiety and depression. Psychopharmacology (Berl) 1999;142(2):215–220. doi: 10.1007/s002130050882. [DOI] [PubMed] [Google Scholar]
- 34.Borsini F, Meli A. Is the forced swimming test a suitable model for revealing antidepressant activity? Psychopharmacology (Berl) 1988;94(2):147–160. doi: 10.1007/BF00176837. [DOI] [PubMed] [Google Scholar]
- 35.Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977;266(5604):730–732. doi: 10.1038/266730a0. [DOI] [PubMed] [Google Scholar]
- 36.Haas H, Panula P. The role of histamine and the tuberomamillary nucleus in the nervous system. Nat Rev Neurosci. 2003;4(2):121–130. doi: 10.1038/nrn1034. [DOI] [PubMed] [Google Scholar]
- 37.Yokoyama F, Yamauchi M, Oyama M, et al. Anxiolytic-like profiles of histamine H3 receptor agonists in animal models of anxiety: a comparative study with antidepressants and benzodiazepine anxiolytic. Psychopharmacology (Berl) 2009;205(2):177–187. doi: 10.1007/s00213-009-1528-1. [DOI] [PubMed] [Google Scholar]
- 38.Oishi R, Itoh Y, Saeki K. Inhibition of histamine turnover by 8-OH-DPAT, buspirone and 5-hydroxytryptophan in the mouse and rat brain. Naunyn Schmiedebergs Arch Pharmacol. 1992;345(5):495–499. doi: 10.1007/BF00168939. [DOI] [PubMed] [Google Scholar]
- 39.Oishi R, Nishibori M, Itoh Y, Saeki K. Diazepam-induced decrease in histamine turnover in mouse brain. Eur J Pharmacol. 1986;124(3):337–342. doi: 10.1016/0014-2999(86)90236-0. [DOI] [PubMed] [Google Scholar]
- 40.Imaizumi M, Onodera K. The behavioral and biochemical effects of thioperamide, a histamine H3-receptor antagonist, in a light/dark test measuring anxiety in mice. Life Sci. 1993;53(22):1675–1683. doi: 10.1016/0024-3205(93)90204-g. [DOI] [PubMed] [Google Scholar]
- 41.Strasser A, Wittmann HJ, Buschauer A, Schneider EH, Seifert R. Species-dependent activities of G-protein-coupled receptor ligands: lessons from histamine receptor orthologs. Trends Pharmacol Sci. 2013;34(1):13–32. doi: 10.1016/j.tips.2012.10.004. [DOI] [PubMed] [Google Scholar]


