Abstract
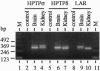
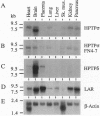
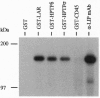
The transmembrane protein-tyrosine-phosphatases (PTPases) LAR, PTP delta, and PTP sigma each contain two intracellular PTPase domains and an extracellular region consisting of Ig-like and fibronectin type III-like domains. We describe the cloning and characterization of human PTP sigma (HPTP sigma) and compare the structure, alternative splicing, tissue distribution, and PTPase activity of LAR, HPTP delta, and HPTP sigma, as well their ability to associate with the intracellular coiled-coil LAR-interacting protein LIP.1. Overall, these three PTPases are structurally very similar, sharing 64% amino acid identity. Multiple isoforms of LAR, HPTP delta, and HPTP sigma appear to be generated by tissue-specific alternative splicing of up to four mini-exon segments that encode peptides of 4-16 aa located in both the extracellular and intracellular regions. Alternative usage of these peptides varies depending on the tissue mRNA analyzed. Short isoforms of both HPTP sigma and HPTP delta were also detected that contain only four of the eight fibronectin type III-like domains. Northern blot analysis indicates that LAR and HPTP sigma are broadly distributed whereas HPTP delta expression is largely restricted to brain, as is the short HPTP sigma isoform containing only four fibronectin type III-like domains. LAR, HPTP delta, and HPTP sigma exhibit similar in vitro PTPase activities and all three interact with LIP.1, which has been postulated to recruit LAR to focal adhesions. Thus, these closely related PTPases may perform similar functions in various tissues.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brady-Kalnay S. M., Flint A. J., Tonks N. K. Homophilic binding of PTP mu, a receptor-type protein tyrosine phosphatase, can mediate cell-cell aggregation. J Cell Biol. 1993 Aug;122(4):961–972. doi: 10.1083/jcb.122.4.961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Clark E. A., Brugge J. S. Integrins and signal transduction pathways: the road taken. Science. 1995 Apr 14;268(5208):233–239. doi: 10.1126/science.7716514. [DOI] [PubMed] [Google Scholar]
- Doherty P., Moolenaar C. E., Ashton S. V., Michalides R. J., Walsh F. S. The VASE exon downregulates the neurite growth-promoting activity of NCAM 140. Nature. 1992 Apr 30;356(6372):791–793. doi: 10.1038/356791a0. [DOI] [PubMed] [Google Scholar]
- Edelman G. M., Crossin K. L. Cell adhesion molecules: implications for a molecular histology. Annu Rev Biochem. 1991;60:155–190. doi: 10.1146/annurev.bi.60.070191.001103. [DOI] [PubMed] [Google Scholar]
- Fischer E. H., Charbonneau H., Tonks N. K. Protein tyrosine phosphatases: a diverse family of intracellular and transmembrane enzymes. Science. 1991 Jul 26;253(5018):401–406. doi: 10.1126/science.1650499. [DOI] [PubMed] [Google Scholar]
- Gebbink M. F., Zondag G. C., Wubbolts R. W., Beijersbergen R. L., van Etten I., Moolenaar W. H. Cell-cell adhesion mediated by a receptor-like protein tyrosine phosphatase. J Biol Chem. 1993 Aug 5;268(22):16101–16104. [PubMed] [Google Scholar]
- Gebbink M. F., van Etten I., Hateboer G., Suijkerbuijk R., Beijersbergen R. L., Geurts van Kessel A., Moolenaar W. H. Cloning, expression and chromosomal localization of a new putative receptor-like protein tyrosine phosphatase. FEBS Lett. 1991 Sep 23;290(1-2):123–130. doi: 10.1016/0014-5793(91)81241-y. [DOI] [PubMed] [Google Scholar]
- Grumet M., Mauro V., Burgoon M. P., Edelman G. M., Cunningham B. A. Structure of a new nervous system glycoprotein, Nr-CAM, and its relationship to subgroups of neural cell adhesion molecules. J Cell Biol. 1991 Jun;113(6):1399–1412. doi: 10.1083/jcb.113.6.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gyuris J., Golemis E., Chertkov H., Brent R. Cdi1, a human G1 and S phase protein phosphatase that associates with Cdk2. Cell. 1993 Nov 19;75(4):791–803. doi: 10.1016/0092-8674(93)90498-f. [DOI] [PubMed] [Google Scholar]
- Hunter T. Protein kinases and phosphatases: the yin and yang of protein phosphorylation and signaling. Cell. 1995 Jan 27;80(2):225–236. doi: 10.1016/0092-8674(95)90405-0. [DOI] [PubMed] [Google Scholar]
- Jiang Y. P., Wang H., D'Eustachio P., Musacchio J. M., Schlessinger J., Sap J. Cloning and characterization of R-PTP-kappa, a new member of the receptor protein tyrosine phosphatase family with a proteolytically cleaved cellular adhesion molecule-like extracellular region. Mol Cell Biol. 1993 May;13(5):2942–2951. doi: 10.1128/mcb.13.5.2942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krueger N. X., Streuli M., Saito H. Structural diversity and evolution of human receptor-like protein tyrosine phosphatases. EMBO J. 1990 Oct;9(10):3241–3252. doi: 10.1002/j.1460-2075.1990.tb07523.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizuno K., Hasegawa K., Katagiri T., Ogimoto M., Ichikawa T., Yakura H. MPTP delta, a putative murine homolog of HPTP delta, is expressed in specialized regions of the brain and in the B-cell lineage. Mol Cell Biol. 1993 Sep;13(9):5513–5523. doi: 10.1128/mcb.13.9.5513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Grady P., Krueger N. X., Streuli M., Saito H. Genomic organization of the human LAR protein tyrosine phosphatase gene and alternative splicing in the extracellular fibronectin type-III domains. J Biol Chem. 1994 Oct 7;269(40):25193–25199. [PubMed] [Google Scholar]
- Ogata M., Sawada M., Kosugi A., Hamaoka T. Developmentally regulated expression of a murine receptor-type protein tyrosine phosphatase in the thymus. J Immunol. 1994 Nov 15;153(10):4478–4487. [PubMed] [Google Scholar]
- Pan M. G., Rim C., Lu K. P., Florio T., Stork P. J. Cloning and expression of two structurally distinct receptor-linked protein-tyrosine phosphatases generated by RNA processing from a single gene. J Biol Chem. 1993 Sep 15;268(26):19284–19291. [PubMed] [Google Scholar]
- Pulido R., Krueger N. X., Serra-Pagès C., Saito H., Streuli M. Molecular characterization of the human transmembrane protein-tyrosine phosphatase delta. Evidence for tissue-specific expression of alternative human transmembrane protein-tyrosine phosphatase delta isoforms. J Biol Chem. 1995 Mar 24;270(12):6722–6728. doi: 10.1074/jbc.270.12.6722. [DOI] [PubMed] [Google Scholar]
- Saito H., Streuli M. Molecular characterization of protein tyrosine phosphatases. Cell Growth Differ. 1991 Jan;2(1):59–65. [PubMed] [Google Scholar]
- Sap J., Jiang Y. P., Friedlander D., Grumet M., Schlessinger J. Receptor tyrosine phosphatase R-PTP-kappa mediates homophilic binding. Mol Cell Biol. 1994 Jan;14(1):1–9. doi: 10.1128/mcb.14.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlessinger J., Ullrich A. Growth factor signaling by receptor tyrosine kinases. Neuron. 1992 Sep;9(3):383–391. doi: 10.1016/0896-6273(92)90177-f. [DOI] [PubMed] [Google Scholar]
- Serra-Pagès C., Kedersha N. L., Fazikas L., Medley Q., Debant A., Streuli M. The LAR transmembrane protein tyrosine phosphatase and a coiled-coil LAR-interacting protein co-localize at focal adhesions. EMBO J. 1995 Jun 15;14(12):2827–2838. doi: 10.1002/j.1460-2075.1995.tb07282.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stitt T. N., Conn G., Gore M., Lai C., Bruno J., Radziejewski C., Mattsson K., Fisher J., Gies D. R., Jones P. F. The anticoagulation factor protein S and its relative, Gas6, are ligands for the Tyro 3/Axl family of receptor tyrosine kinases. Cell. 1995 Feb 24;80(4):661–670. doi: 10.1016/0092-8674(95)90520-0. [DOI] [PubMed] [Google Scholar]
- Streuli M., Hall L. R., Saga Y., Schlossman S. F., Saito H. Differential usage of three exons generates at least five different mRNAs encoding human leukocyte common antigens. J Exp Med. 1987 Nov 1;166(5):1548–1566. doi: 10.1084/jem.166.5.1548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streuli M., Krueger N. X., Ariniello P. D., Tang M., Munro J. M., Blattler W. A., Adler D. A., Disteche C. M., Saito H. Expression of the receptor-linked protein tyrosine phosphatase LAR: proteolytic cleavage and shedding of the CAM-like extracellular region. EMBO J. 1992 Mar;11(3):897–907. doi: 10.1002/j.1460-2075.1992.tb05128.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streuli M., Krueger N. X., Hall L. R., Schlossman S. F., Saito H. A new member of the immunoglobulin superfamily that has a cytoplasmic region homologous to the leukocyte common antigen. J Exp Med. 1988 Nov 1;168(5):1523–1530. doi: 10.1084/jem.168.5.1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streuli M., Krueger N. X., Thai T., Tang M., Saito H. Distinct functional roles of the two intracellular phosphatase like domains of the receptor-linked protein tyrosine phosphatases LCA and LAR. EMBO J. 1990 Aug;9(8):2399–2407. doi: 10.1002/j.1460-2075.1990.tb07415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trowbridge I. S., Thomas M. L. CD45: an emerging role as a protein tyrosine phosphatase required for lymphocyte activation and development. Annu Rev Immunol. 1994;12:85–116. doi: 10.1146/annurev.iy.12.040194.000505. [DOI] [PubMed] [Google Scholar]
- Volkmer H., Hassel B., Wolff J. M., Frank R., Rathjen F. G. Structure of the axonal surface recognition molecule neurofascin and its relationship to a neural subgroup of the immunoglobulin superfamily. J Cell Biol. 1992 Jul;118(1):149–161. doi: 10.1083/jcb.118.1.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner J., Boerboom D., Tremblay M. L. Molecular cloning and tissue-specific RNA processing of a murine receptor-type protein tyrosine phosphatase. Eur J Biochem. 1994 Dec 15;226(3):773–782. doi: 10.1111/j.1432-1033.1994.00773.x. [DOI] [PubMed] [Google Scholar]
- Walton K. M., Martell K. J., Kwak S. P., Dixon J. E., Largent B. L. A novel receptor-type protein tyrosine phosphatase is expressed during neurogenesis in the olfactory neuroepithelium. Neuron. 1993 Aug;11(2):387–400. doi: 10.1016/0896-6273(93)90193-u. [DOI] [PubMed] [Google Scholar]
- Williams A. F., Barclay A. N. The immunoglobulin superfamily--domains for cell surface recognition. Annu Rev Immunol. 1988;6:381–405. doi: 10.1146/annurev.iy.06.040188.002121. [DOI] [PubMed] [Google Scholar]
- Yan H., Grossman A., Wang H., D'Eustachio P., Mossie K., Musacchio J. M., Silvennoinen O., Schlessinger J. A novel receptor tyrosine phosphatase-sigma that is highly expressed in the nervous system. J Biol Chem. 1993 Nov 25;268(33):24880–24886. [PubMed] [Google Scholar]
- Yu Q., Lenardo T., Weinberg R. A. The N-terminal and C-terminal domains of a receptor tyrosine phosphatase are associated by non-covalent linkage. Oncogene. 1992 Jun;7(6):1051–1057. [PubMed] [Google Scholar]
- Zhang J. S., Longo F. M. LAR tyrosine phosphatase receptor: alternative splicing is preferential to the nervous system, coordinated with cell growth and generates novel isoforms containing extensive CAG repeats. J Cell Biol. 1995 Feb;128(3):415–431. doi: 10.1083/jcb.128.3.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W. R., Hashimoto N., Ahmad F., Ding W., Goldstein B. J. Molecular cloning and expression of a unique receptor-like protein-tyrosine-phosphatase in the leucocyte-common-antigen-related phosphate family. Biochem J. 1994 Aug 15;302(Pt 1):39–47. doi: 10.1042/bj3020039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]