Abstract
Humans and cetaceans are exposed to a wide range of contaminants. In this study, we compared the cytotoxic and genotoxic effects of a metal pollutant, hexavalent chromium [Cr(VI)], which has been shown to cause damage in lung cells from both humans and North Atlantic right whales. Our results show that Cr induces increased cell death and chromosome damage in lung cells from both species with increasing intracellular Cr ion levels. Soluble Cr(VI) induced less of a cytotoxic and genotoxic effect on administered dose in right whale (Eubalaena glacialis) cells than in human (Homo sapiens) cells. Whereas, particulate Cr(VI) induced a similar cytotoxic effect but less of a genotoxic effect based on administered dose in right whale cells than in human cells. Differences in chromium ion uptake explained some but not all of the soluble chromate-induced cell death and chromosome damage. Uptake differences of lead ions could explain the differences in particulate chromate-induced toxicity. The data show that both forms of Cr(VI) are less genotoxic to right whale than human lung cells, and that soluble Cr(VI) induces a similar cytotoxic effect in both right whale and human cells, while particulate Cr(VI) is more cytotoxic to right whale lung cells.
Keywords: Chromium, North Atlantic right whale, human, lung cells, cytotoxicity, genotoxicity
Introduction
Hexavalent chromium [(Cr(VI)] industries are still very active in the United States and other parts of the world and as a consequence substantial amounts of Cr-contaminated waste is released into the environment, affecting both the atmosphere and water, including the oceans (ASTDR, 2008). The oceans can be a common sink for pollutants putting aquatic species at risk (Hyxon et al., 2001; Chung, 1986). In the ocean, Cr(VI) is the predominant state for Cr (Geisler and Schmidt, 1992; Pettine and Millero, 1990). Thus, Cr(VI) remains a health concern for both humans and whales.
For humans, Cr(VI) is a well known lung carcinogen (IARC, 1990). Cr(VI) carcinogenicity has been demonstrated in various animal and cell culture studies, and the major mechanism underlying its carcinogenicity are its genotoxic effects of chromosomal aberrations and DNA strand breaks (Holmes et al., 2006; Levi and Venitt, 1986; Wise et al., 2006; Xie et al., 2005).
The clastogenic effects may also cause teratogenic and developmental problems; therefore, Cr(VI) is also a human reproductive concern. Human and rodent studies show that Cr(VI) accumulates in the testes and causes a reduction in testicular weights, seminiferous tubule degeneration, decreased sperm counts and altered reproductive behaviors (Al-Hamood et al., 1998; Bataineh et al., 1997; Chowdhury et al., 1995; Mancuso, 1997; Witmer et al., 1989, 1991). Cr exposure is also a concern for aquatic animals such as whales. We recently reported that free-ranging North Atlantic right whales and sperm whales had very high (means of 7.1 ± 0.8 μg/g and 8.8 μg/g tissue, respectively) Cr skin levels (Wise et al., 2008, 2009). These data show that these whales are accumulating very high levels of Cr typically only seen in occupationally exposed humans. Moreover, the data show that the levels vary by region (Wise et al., 2009) indicating local pollution is a likely source. How the whales are being exposed in uncertain. Cr(VI) can enter the body though dermal, oral and inhalational routes (ATSD, 2008). It is likely that all three routes could occur in whales though dermal and inhalation routes seem the most likely (Wise et al., 2008, 2009). The form of Cr(VI) the whales might be exposed to also is uncertain, but soluble forms are likely delivered by oral and dermal routes (i.e. in the water) while the particulate forms are likely delivered by oral and inhalation routes (i.e. in air and food) (ATSDR, 2009).
The toxic effects of Cr(VI) in whales are poorly understood. We also found that Cr(VI) is genotoxic to right whale lung, skin and testes cells inducing chromosomal aberrations (Wise et al. 2008; Li Chen et al., 2009). The North Atlantic right whale is an endangered species that has been protected from hunting for more than 70 years; however, the population has shown little or no evidence of recovery (Kareiva, 2001). The lack of reproductive success is likely due to a number of factors, both biological and environmental, but it has been suggested that exposure to anthropogenic chemical pollutants may contribute to the lack of recovery of this endangered species population (Knowlton et al., 1994; Kraus et al., 2001; Rolland et al., 2007). It is plausible that if the genotoxic effects of Cr(VI) did occur in vivo for whales that Cr could be a reproductive toxicant for whales as well.
However, the Cr levels observed in right whales are remarkably high and yet Cr does not appear to be inducing any cancer in these species. It is possible that whales with lung tumors may simply be removed from the population by natural means (e.g. predation) and simply not beached or washed ashore to be noticed. On the other hand, comparing the Wise et al. (2008) and Li Chen et al. (2009) results to the published literature, Cr(VI) appears to be less genotoxic to right whale lung cells than human lung cells. These comparisons suggest that whales may have evolved cellular mechanisms to better protect themselves from genotoxic threats, though no direct comparisons between the species have been made. If this were to be true for right whales, it could have important implications for studying the mechanism of Cr(VI) genotoxicity and preventing its effects, as well as implications for the threat that Cr exposure may have for the whales. Accordingly, in this study we directly compared the cytotoxicity and genotoxicity of Cr(VI) in right whale and human lung cells to see if there is a difference in response. Because the form of Cr(VI) the whales might be exposed to is unknown, we considered both particulate and soluble forms.
Materials and Methods
Chemicals and Reagents
Sodium chromate (Na2CrO4), lead chromate (PbCrO4), demecolchicine and potassium chloride (KCl) were purchased from Sigma/Aldrich (St. Louis, MO, USA). Giemsa stain was purchased from Biomedical Specialties Inc. (Santa Monica, CA, USA). Cytoseal 60 slide mounting medium was purchased from VWR (Bridgeport, NJ, USA). Gurr's buffer, trypsin/EDTA, sodium pyruvate, penicillin/streptomycin, and L-Glutamine were purchased from Invitrogen Corporation (Grand Island, NY, USA). Crystal violet, methanol and acetone were purchased from J.T. Baker (Phillipsburg, NJ, USA). Dulbecco's minimal essential medium and Ham's F-12 (DMEM/F-12) 50:50 mixture was purchased from Mediatech Inc. (Herndon, VA, USA). Cosmic calf serum (CCS) was purchased from Hyclone, (Logan, UT, USA). MycoAlert detection kits were purchased from Lonza Rockland, Inc (Rockland, ME, USA). Tissue culture dishes, flasks, and plasticware were purchased from Corning Inc. (Acton, MA, USA).
Cells and Cell Culture
In this study, human lung cells are referred to as WTHBF-6 cells, a cell line of hTERT-expressing human lung clonal cells derived from primary human bronchial fibroblasts (PHBF), exhibiting a diploid karyotype, normal growth parameters and an extended lifespan. North Atlantic right whale lung cells were primary cell lines, isolated from tissue explants obtained from stranded animal biopsies, as described previously (Wise et al., 2008). Both cell lines were cultured in adherent monolayer of cells, being sub-cultured at least once a week. Cells were maintained in Dulbecco's minimal essential medium and Ham's F-12 (DMEM/F-12) 50:50 mixture, supplemented with 15% cosmic calf serum, 2 mM L-glutamine, 100 U/mL penicillin/100 μg/mL streptomycin, and 0.1 mM sodium pyruvate. Human and whale cells were maintained in a 37 °C and 33 °C, respectively, humidified incubator with 5% CO2. Cells were regularly tested for mycoplasma contamination, using the MycoAlert kit. All experiments were conducted on logarithmically growing cells with a doubling time of 24 and 36 h for human and North Atlantic right whale cells, respectively.
Preparation of Chromium Compounds
As a model of soluble Cr(VI), Na2CrO4 (CAS #7775-11-3, ACS reagent minimum 98% purity) (sodium chromate), was administered as a solution in water as previously described (Wise et al., 2002). Briefly, Na2CrO4 was weighed and dissolved in deionized water. Desired treatment concentrations were made from a filtered sample of the stock solution. As a model of particulate Cr(VI), PbCrO4 (CAS# 7758-97-6, ACS reagent minimum 98% purity) (lead chromate), was administered as suspensions of particles as previously described (Wise et al., 2002) to ensure that cells were exposed to intact particles. Briefly, PbCrO4 was weighed, suspended in acetone to sterilize the particles and desired treatment concentrations were made from this stock solution. Treatments ranged from 1-10 μM for sodium chromate and 0.1-5 μg/cm2 for lead chromate.
Cytotoxicity Assay
Cytotoxicity was measured as the impact of the Cr(VI) compound on a cells ability to form colonies in a culture dish (i.e. a colony-forming assay), as described previously (Wise et al., 2008). Briefly, 90,000 cells (100,000 cells for whale) were seeded into each well of a 6-well plate and then the cells were allowed 48 h to resume normal cell cycling followed by a 24 h treatment with lead chromate or sodium chromate. At the end of the exposure time, cells were washed with phosphate-buffered saline (PBS) and then removed from the dish with 0.05% trypsin-0.53 mM EDTA, and centrifuged for 5 min at 400 g and 4°C. Supernatant was aspirated, and cell pellet was resuspended in cell culture media. Cells were counted and seeded into 100 mm tissue culture dish at a density of 1000 cells per dish, four dishes were seeded for each concentration; and let grow for about 2 weeks until colony forming. Dishes were then washed twice with PBS, fixed with 100% methanol and stained with Crystal Violet. Colonies were counted in each dish and compared to the control. Experiments were repeated at least 3 times. All treatment groups were compared to control and expressed as a percentage of the control.
Genotoxicity Assay
Genotoxicity was measured by the ability of Cr(VI) to induce aberrations in metaphase chromosomes as described previously (Wise et al., 2008). Cells were prepared according to Wise et al. (2008). Briefly, cells were seeded into 100 mm tissue culture dishes at a density of 500,000 cells (800,000 cells for whale) per dish and allowed to rest for 48 h. Then, cells were treated with the desired concentrations of lead chromate or sodium chromate for 24 h. 1 h (5 h for whale cells) before the end of chromate treatment; demecolchicine was added to arrest cells in metaphase. At the end of chromate treatment, cells were harvested: culture media was saved to keep all loosened cells, dishes were washed with PBS, removed with 0.05% trypsin-0.53mM EDTA, and centrifuged for 5 min at 400 g and 4 °C. Supernatant was aspirated, and a hypotonic solution of 0.075 M KCl was added for 17 min and then 1 mL of fixative (3:1 methanol:acetic acid) was added; and centrifuged once more. Supernatant was aspirated and 10 mL of fixative was added and allowed to fix for 20 min at room temperature. Cells were centrifuged again and fixative was changed two more times to remove cell debris. At the last fixative change, supernatant was aspirated and cells, suspended in an appropriate amount of fix, were then dropped into a wet clean slide uniformly, stained with Giemsa stain and coverslipped with Cytoseal. Experiments were repeated at least 3 times. Treatment concentrations used were chosen depending on cell's relative survival.
Chromosome Scoring Criteria
Chromosomal structural aberrations were scored in 100 metaphases per each treatment dose, according to standard criteria previously described (Wise et al., 2002). Chromatid and isochromatid gaps and breaks were distinguished by the width of the gap or break compared to the width of the chromatid arm diameter. However, during results analysis, gaps and breaks were pooled together. It has been shown that correlations with treatments are stronger when pooling gaps and breaks together than gaps or breaks alone (Paz-y-Mino et al., 2002). This approach also avoids subjectivity inherent to the different scorers.
Determination of Intracellular Chromium Ion Levels
Cell preparation
Cells were prepared according to Wise et al. (2004), with minor changes. Briefly, cells were seeded into 100 mm tissue culture dishes at a density of 500,000 cells (800,000 cells for whale) per dish and allowed to rest for 48 h. Cells were then treated for 24 h. At the end of the treatment with lead chromate or sodium chromate, 3 mL of culture media was saved for extracellular chromium analysis; cells were washed with PBS and removed from the dishes with 0.05% trypsin-0.53mM EDTA; the number and volume of cells were determined, cells were centrifuged for 5 min at 400 g and 4 °C, supernatant was removed and cells were resuspended in 4 mL of PBS. Process was repeated twice. Cells were resuspended in 1 mL hypotonic solution for 5 min followed by 15 min in 1 mL 2% SDS. Finally the solution was sheered through an 18 gauge needle and filter through a 0.2 μm filter. Samples were stored at −20 °C until metal analysis. Experiments were repeated at least 3 times. The treatment concentrations used were the same as the ones used to test Cr(VI) genotoxicity.
Ion level measurements
Intracellular Cr ion levels were determined using the Inductively Coupled Plasma-Optical Emission Spectrometers (ICP-OES), according to previous published methods (Wise et al., 2004). A VG Axiom MC instrument, equipped with a CETAC U5000AT ultrasonic nebulizer, was used. Samples were introduced to the ultrasonic nebulizer using a peristaltic pump operating at 500-1000 μL/min. Samples of extracellular and intracellular fluids were diluted 50-500 × in 0.16 M aqueous HNO3 prior to analysis. Cr was determined using the 52Cr isotope at a resolving power of 6000 (R=m/Δm); this cleanly separates the 52Cr+ peak from 40Ar12C+ and 35Cl16O1H+ peaks. 51V was used as an internal standard for Cr determination. Pb was determined in the low-resolution mode (R=410) using 209Bi+ as the internal standard. The Cr detection limit in solution was approximately 0.05 micrograms per liter. Cr was analyzed in the high resolution mode (R=6000) which resolves Cr ions from interfering polyatomic ion peaks. Cr levels given by the ICP-OES were then corrected for number and volume of the cells.
Statistics
Statistical analysis was performed as previously described (Wise et al, 2008). Briefly, linear regression analysis was employed to estimate the relationship between administered dose and intracellular concentration for cytotoxicity experiments. Dose-response relationships for cytotoxicity, percent metaphases with damage, and total damage were fit using linear and polynomial regression analysis. For percent of metaphases with damage and total damage, controls were subtracted from each experimental value. Interactions between dose or concentration and species or type of chromium were evaluated by adding product terms to the regression models. Differences between species and between lead versus sodium chromate were assessed using t-tests for linear combinations of parameters obtained from the regression analyses. Ninety-five percent confidence intervals were calculated based on the variance-covariance matrix. All analyses were conducted using SAS (SAS Institute, 2004).
Results
Human lung cells take up more Cr than North Atlantic right whale lung cells
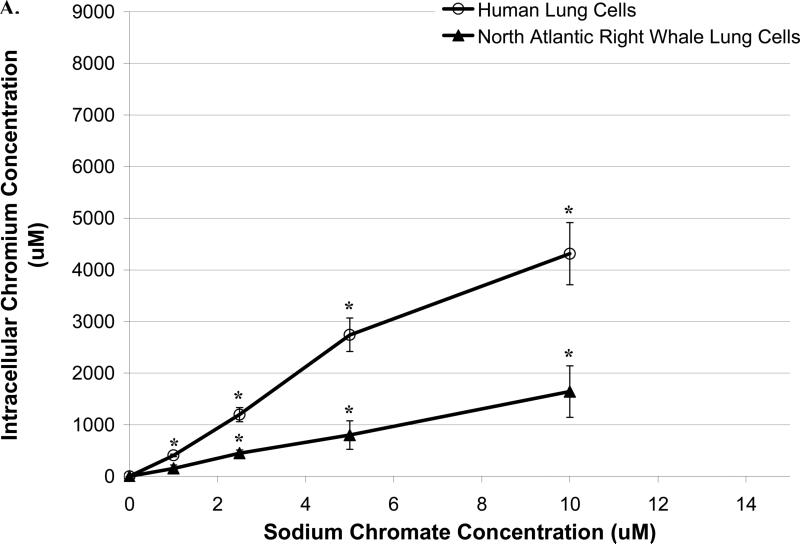
Administered dose does not necessarily reflect the intracellular ion concentration. To account for potential differences in Cr uptake between the two cell lines, the intracellular levels of Cr were determined after a 24 h exposure. Both cell lines took up increasing amounts of Cr ions in a concentration-dependent manner (Figure 1). Specifically, 1, 2.5, 5 and 10 μM sodium chromate produced mean intracellular Cr levels of 407, 1195, 2740 and 4313 μM, respectively, in human lung cells, and 155, 449, 799 and 1641 μM, respectively in right whale lung cells. Significant differences between human and right lung cells were observed at 1, 2.5, 5 and 10 μM (p<0.01) (Figure 1A).
Figure 1. Human Lung Cells Take Up Greater Amounts of Cr(VI) than North Atlantic Right Whale Lung Cells.
This figure shows that exposure to Cr(VI) induces Cr ion uptake in both human and North Atlantic right whale lung cells in a concentration-dependent manner. Data represent the average of 3 independent experiments. Error bars = standard error of the mean. (A) Cr uptake after sodium chromate treatment. (B) Cr uptake after lead chromate treatment.
The pattern was similar for lead chromate with human lung cells again taking up more chromium. Concentrations of 0.1, 0.5, 1 and 5 μg /cm2 lead chromate produced mean intracellular Cr levels of 131, 677, 1095 and 2062 μM, respectively in human lung cells; and in whale lung cells concentrations of 0.5, 1, 2, 4 and 5 μg/cm2 produced mean intracellular Cr levels of 142, 172, 396, 475 and 675 μM, respectively. Significant differences between the two cell lines were observed at concentrations 0.5, 1 and 5 μg/cm2 (p<0.01) (Figure 1B). These data indicate that the human lung cells had greater chromium uptake.
Cr(VI) is cytotoxic to both human and North Atlantic right whale lung cells
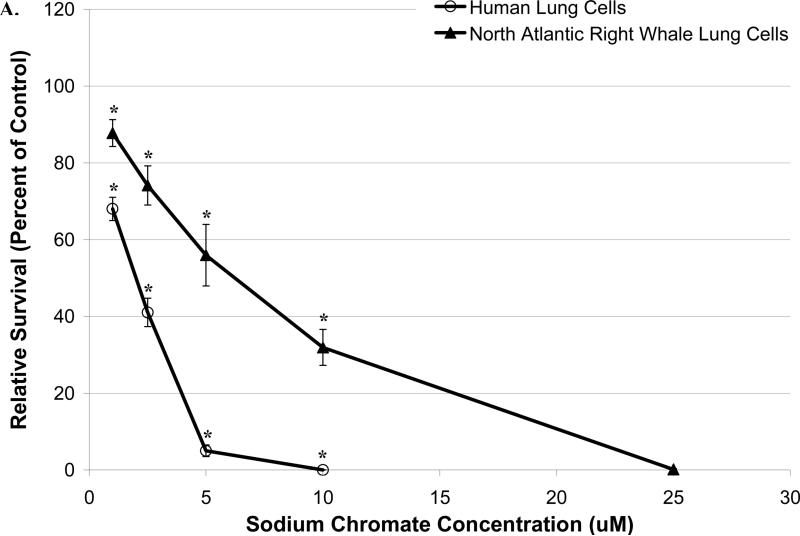
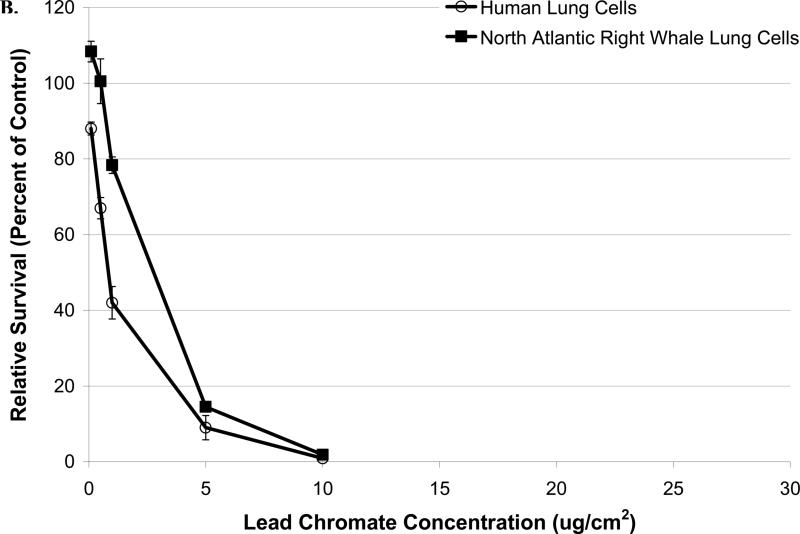
Sodium chromate and lead chromate were cytotoxic to both cell lines in a concentration-dependent manner (Figure 2). In human lung cells, concentrations of 1, 2.5, 5, 10 and 25 μM sodium chromate reduced relative survival to 68, 41, 5, 0 and 0%, respectively; and 88, 74, 56, 32 and 0%, respectively, in whale lung cells. Significant differences between the two cell lines were observed (p<0.01) (Figure 2A). For lead chromate, concentrations of 0.1, 0.5, 1, 5 and 10 μg/cm2 reduced relative survival to 88, 67, 42, 9 and 1%, respectively, in human lung cells, and 108, 100, 78, 14 and 2%, respectively, in whale lung cells. Significant differences between the two cell lines were observed (p<0.01) (Figure 2B).
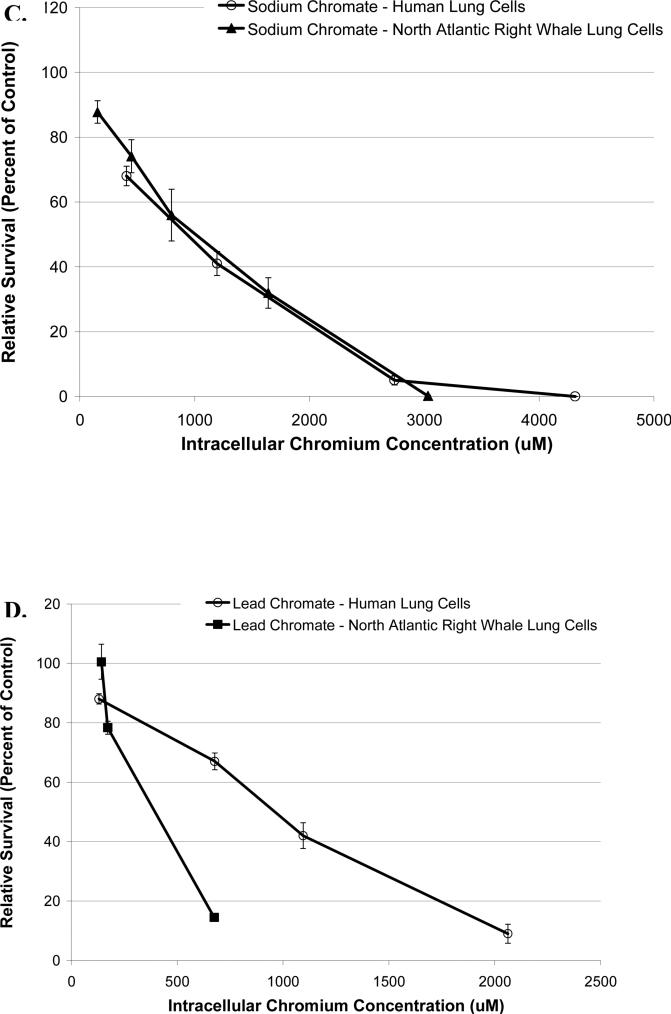
Figure 2. Reduced Cr(VI)-Induced Cytotoxicity in North Atlantic Right Whale Lung Cells Relative to Human Lung Cells Is Due to Differences in Cr Ion Uptake for Soluble Cr(VI), But Not for Particulate Cr(VI).
This figure shows that exposure to Cr(VI) induces cytotoxicity in human and North Atlantic right whale lung cells in a concentration-dependent manner, based on administered dose (p<0.01). Data represent the average of 3 independent experiments. Error bars = standard error of the mean. (A) Sodium chromate cytotoxicity based on administered dose. (B) Lead chromate cytotoxicity based on administered dose. (C) Cytotoxicity for sodium chromate based on intracellular Cr ion level. (D) Cytotoxicity for lead chromate based on intracellular Cr ion level. (E) Cytotoxicity for both sodium and lead chromate based on intracellular Cr ion level for right whale lung cells.
These data indicate that both sodium and lead chromate are more cytotoxic to human lung cells than to whale lung cells, based on administered dose. Correcting for the reduced chromium uptake in the whale cells at these doses, shows that sodium chromate induces similar cytotoxicity in both cell lines. Significant differences between the two cell lines were not observed (p=0.61) (Figure 2C). This indicates that the difference in Cr(VI) potency is due to differences in Cr uptake. However, for lead chromate, correcting for the greater uptake does not account for the differences in potency, the data shows that right whale lung cells were actually more sensitive to intracellular Cr than human lung cells, for example, at an intracellular chromium concentration of 600 μM, the estimated relative survival is 49% for right whale lung cells and 65% for human lung cells (based on the fitted data, 95% confidence interval for differences = 5 to 27%, p<0.01) (Figure 2D). Further inspection of the data shows that for human lung cells, lead chromate and sodium chromate exhibit similar amounts of cytotoxicity, consistent with a published report that the mechanism of lead chromate cytotoxicity is due to the production of the chromate anion (Holmes at al., 2005). However, for right whale lung cells there is significantly more cytotoxicity after lead chromate exposure than for sodium chromate, for example, at an intracellular chromium concentration of 600 μM, the estimated relative survival is 68% after exposure to sodium chromate and 49% after exposure to lead chromate (based on the fitted data, 95% confidence interval for differences = 9 to 28%, p<0.01) (Figure 2E).
Cr(VI) is less genotoxic to both human and North Atlantic right whale lung cells
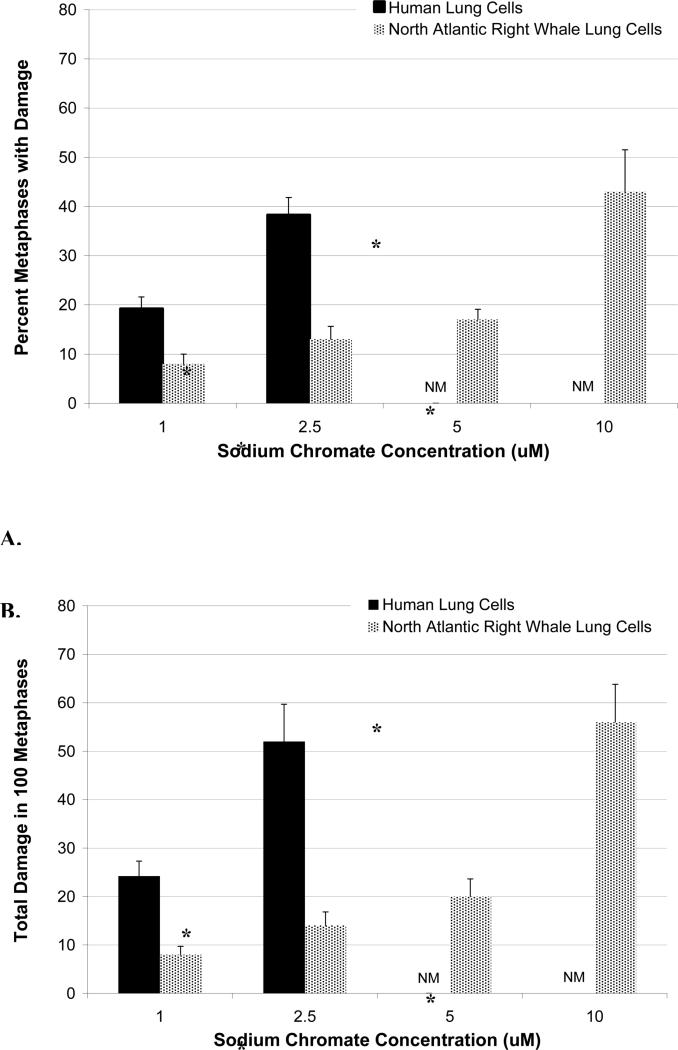
Sodium chromate was genotoxic to both cell lines in a concentration-dependent manner (Figure 3). Significant differences between the two cell lines were observed (p<0.01). First considering the frequency of damage; in human lung cells, concentrations of 1 and 2.5 μM sodium chromate damaged chromosomes in 19 and 38% of metaphases, respectively, while 5 and 10 μM induced cell cycle arrest with no metaphases seen (Figure 3A). In right whale lung cells, sodium chromate was less genotoxic, concentrations of 1, 2.5 and 5 μM sodium chromate damaged chromosomes in 8, 13 and 17% of metaphases, respectively (Figure 3A).
Figure 3. Reduced Soluble Cr(VI)-Induced Genotoxicity in North Atlantic Right Whale Lung Cells Relative to Human Lung Cells Is Not Due to Differences in Cr Ion Uptake.
This figure shows that exposure to sodium chromate induces genotoxicity in both human and right whale lung cells in a concentration-dependent manner. (A) Sodium chromate-induced metaphases with chromosome damage based on administered dose. (B) Sodium chromate-induced total chromosome damage in 100 metaphases based on administered dose. (C) Percent metaphases with damage based on intracellular Cr ion level. (D) Total chromosome damage in 100 metaphases based on intracellular Cr ion level. Data represent the average of 3 independent experiments. Error bars = standard error of the mean. NM – no metaphases observed.
When we considered the effect by the amount of total chromosome damage in 100 metaphases, we found the same trend (Figure 3B). Significant differences between the two cell lines were observed (p<0.01). Concentrations of 1 and 2.5 μM damaged 24 and 52 chromosomes per 100 metaphases in human lung cells, respectively; and in whale lung cells, 1, 2.5 and 5 μM sodium chromate damaged 8, 14 and 20 chromosomes per 100 metaphases, respectively (Figure 3B). The spectrum of sodium chromate-induced chromosome aberrations was not different between the cell lines (data not shown).
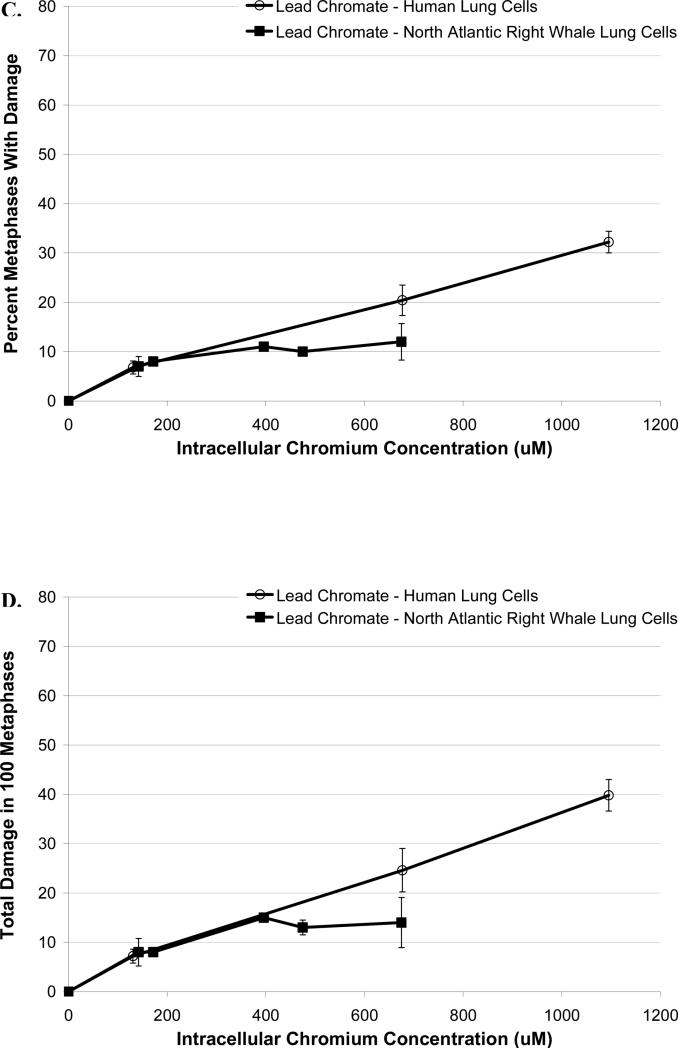
Lead chromate also induced chromosomal damage in a concentration-dependent manner (Figure 4). Significant differences between the two cell lines were observed (p<0.01). Considering the frequency of damage, concentrations of 0.1, 0.5, and 1 μg/cm2 damaged chromosomes in 7, 20 and 32 % of metaphases, respectively, in human lung cells. Cell cycle arrest was induced at 5 μg/cm2 and no metaphases were seen (Figure 4A). In whale lung cells, lead chromate was again less genotoxic, concentrations of 0.5, 1, 2, 4 and 5 μg/cm2 lead chromate damaged chromosomes in 7, 8, 11, 10 and 12% of metaphases, respectively (Figure 4A).
Figure 4. Reduced Particulate Cr(VI)-Induced Genotoxicity in North Atlantic Right Whale Lung Cells Relative to Human Lung Cells May Be Due to Differences in Cr Ion Uptake.
This figure shows that exposure to lead chromate induces genotoxicity in both human and right whale lung cells in a concentration-dependent manner. (A) Lead chromate-induced metaphases with chromosome damage based on administered dose. (B) Lead chromate-induced total chromosome damage in 100 metaphases based on administered dose. (C) Percent metaphases with damage for based on intracellular Cr ion level. (D) Total chromosome damage in 100 metaphases based on intracellular Cr ion level. Data represent the average of 3 independent experiments. Error bars = standard error of the mean. NM – no metaphases observed.
The same trend was found when considering total chromosome damage in 100 metaphases (Figure 4B). Significant differences between the two cell lines were observed (p<0.01).Concentrations of 0.1, 0.5, and 1 μg/cm2 damaged 7, 25 and 40 chromosomes, respectively, in human lung cells; and in whale lung cells, 0.5, 1, 2, 4 and 5 μg/cm2 damaged 8, 8, 15, 13 and 14 chromosomes, respectively (Figure 4B). The spectrum of lead chromate-induced chromosome aberrations was not different between the cell lines (data not shown).
These data indicate that sodium and lead chromate are more genotoxic to human lung cells than to whale lung cells, based on administered dose. Correcting for the lower chromium uptake in the whale lung cells at these doses indicates that the decreased genotoxicity seen in these cells is only partly explained by the uptake difference for both frequency of damage (Figure 3C and 4C) and total chromosome damage (Figure 3D and 4D). We found significant differences when comparing the two cell lines (p>0.01). For example, for sodium chromate, at an intracellular Cr ion level of 600 μM, the difference in frequency of damage is 6% (95% confidence interval = 2 to 9%, p<0.01), and the difference in total chromosome damage is 9 (95% confidence interval = 4 to 15, p<0.01). And for lead chromate, at an intracellular Cr ion level of 600 μM, the difference in frequency of damage is 27% (95% confidence interval = 22 to 32%, p<0.01), and the difference in total chromosome damage is 33 (95% confidence interval = 27 to 40, p<0.01). Because previous studies show that the chemical reduction of intracellular Cr ions are responsible for Cr(VI) genotoxicity in human lung cells (Holmes et al., 2005), these data indicate that additional mechanisms must underlie the decreased genotoxicity seen in the whale lung cells.
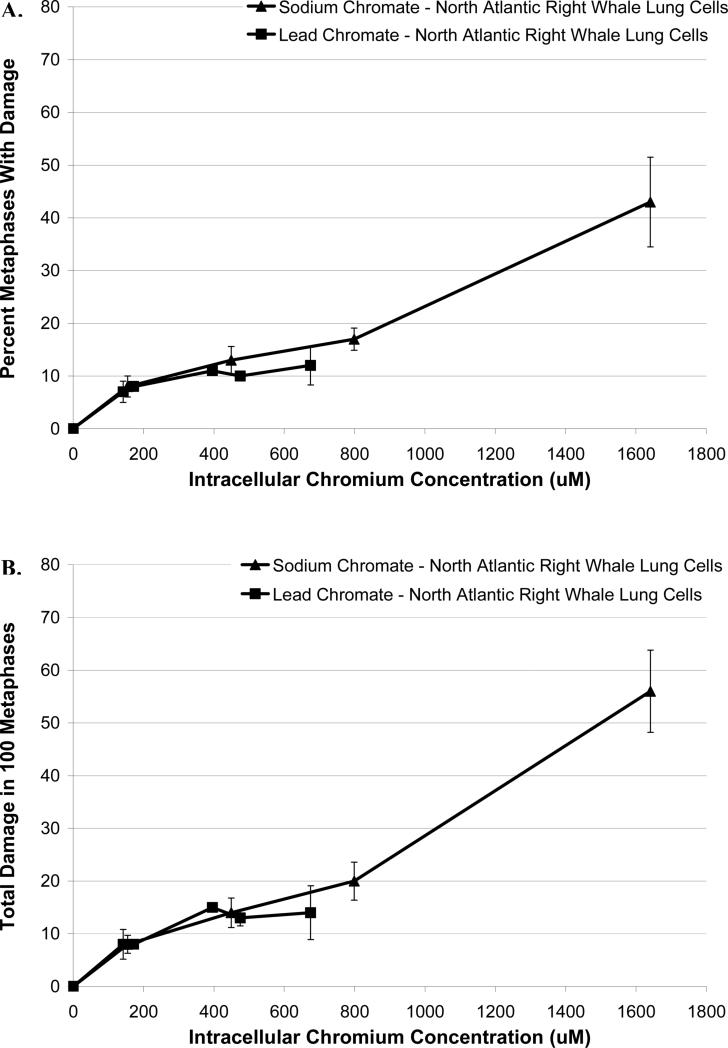
Further inspection of the data also shows that for whale lung cells, lead chromate and sodium chromate exhibit similar amounts of genotoxicity, for both frequency of damage (p=0.10) (Figure 5A) and total chromosome damage (p=0.15) (Figure 5B), consistent with published reports that the mechanism of lead chromate genotoxicity is due to the production of the chromate anion (Wise et al., 2004).
Figure 5. Particulate and Soluble Cr(VI) Induce Similar Amounts of Genotoxicity Based on Intracellular Cr Ion Levels.
This figure shows that exposure to both soluble and particulate Cr(VI) induces genotoxicity with increasing intracellular Cr ion levels in right whale lung cells in a similar manner. (A) Soluble and particulate Cr(VI) induces similar amount of metaphase with chromosome damage in right whale lung cells. (B) Soluble and particulate Cr(VI) induces similar amount of total chromosome damage in 100 metaphases in both human and right whale lung cells. Data represent the average of 3 independent experiments. Error bars = standard error of the mean. ND – not done; NM – no metaphases.
Discussion
Few studies have directly compared the response of human and marine mammals to test whether cetaceans and humans respond similarly to toxicants. This study is the first one to directly compare the cytotoxic and genotoxic effects of a pollutant of concern (chromium) for the marine environment in both human and North Atlantic right whale lung cells. The results of this study indicate that right whale lung cells respond similarly to Cr(VI) as human lung cells, but that there are some different intracellular mechanisms occurring. For example, for soluble Cr(VI), we found that the decreased sensitivity to Cr(VI)-induced cytotoxicity found in whale cells relative to human cells was directly due to differences in Cr ion uptake. However, the decreased sensitivity of right whale cells to Cr(VI)-induced genotoxicity is only partially explained by differences in uptake.
The reasons for the decreased chromosome damage seen in the right whale lung cells at similar intracellular Cr levels are uncertain. The current paradigm for Cr(VI)-induced chromosome damage indicates that the aberrations are produced as a consequence of the repair of other DNA lesions (Wise et al., 2008). Specifically, the mechanism indicates that Cr(VI) ions cross the cell membrane through an anion transport system, by mimicking phosphates and sulfates, and then are reduced inside the cell to Cr(III) via a series of short-lived intermediates. Then Cr(III), and/or one or more of these intermediates, form adducts on the DNA to cause a stalled replication fork, which the cell attempts to repair. During that repair DNA double strand breaks are created. These breaks then go on to form the chromosomal aberrations. Thus, two cellular factors may explain the lower amount of chromosome damage seen in the right whale cells: a more efficient repair of DNA adducts or DNA double strand breaks and a different intracellular metabolism that decreases the amount of the available adducting Cr species. However, more work is needed to distinguish the relative importance of these mechanisms.
A different outcome was observed for particulate chromate. The cytotoxicity of lead chromate did not appear to be different between the two species based on administered dose. However, differential Cr uptake showed that the amount of cytotoxicity was not similar when compared by intracellular Cr ions. This outcome is inconsistent with the results we observed for soluble chromate and suggest that the Pb ion may be playing a role. In addition, when we compared the cytotoxicity of lead chromate and sodium chromate based on intracellular Cr levels in the right whale cells alone, we found that lead chromate was much more toxic. These data suggest that for lead chromate there is more to the cytotoxicity than the effects of the Cr ion alone. Previous studies in human lung cells indicated that Pb ions do not play a role in lead chromate-induced cytotoxicity and genotoxicity because there is limited Pb ion uptake (Holmes et al., 2005). The differences in lead chromate effects seen between human and right whale lung cells could be due to a differential lead uptake.
The comparative genotoxic effects for lead chromate were similar to soluble chromate. Based on administered concentration, there was less chromosome damage in the right whale cells. Correcting for differential uptake showed less chromosome damage in the right whale lung cells. These data are consistent with the results we observed for soluble chromate, where right whale lung cells have a lower amount of chromosome damage than human lung cells, and they also suggest that Pb ions may be playing a role and possibly inhibiting DNA repair. In addition, when we compared the genotoxicity of lead chromate and sodium chromate based on intracellular Cr levels in the right whale cells alone, we found that the two compounds were equipotent. These data suggest that for lead chromate the genotoxicity is due to the Cr ions alone, which is consistent with data from previous studies in human lung cells (Wise et al., 2004).
It is interesting to note that the right whale lung cells take up less Cr(VI) than the human lung cells. The mechanism for this difference is uncertain. Chromate uptake is mediated by anion transport proteins responsible for phosphate and sulfate transport into the cells (Wise et al., 2008). Perhaps the explanation is that the whale cells simply have lower levels of these transport proteins. Alternatively, the whale cells may have evolved a different and less efficient transport system for chromate; perhaps one more specific to sulfate and phosphate. Further research is needed to distinguish between these possibilities.
It is also interesting to note that for sodium chromate, right whale and human cells showed similar cytotoxicity, but whales had less genotoxicity. The reason for this decreased sensitivity is uncertain. The current paradigm for Cr(VI)-induced toxicity was recently reviewed (Wise et al., 2008). It indicates that Cr-DNA adducts cause a stalled replication fork, which lead to DNA double strand breaks. These breaks then become chromosomal aberrations. One possibility, albeit untested, is that whale cells may be more efficient in recognizing this damage and then signaling to the apoptosis machinery more effectively than the human cells leading to more cell death in the presence of less damage. Alternatively, the whale cells may have a different intracellular metabolism that reduces the amount of the adducting Cr species produced.
In summary, our data demonstrate that North Atlantic right whales have evolved better mechanisms to protect them from Cr(VI) genotoxicity. These mechanisms include reducing exposure by taking up less chromium at the start. In addition, once internalized the whale cells have evolved a mechanism to induce cell death at lower levels of genotoxicity, thus removing compromised cells from potentially becoming problematic. They further suggest that whales may be able to tolerate high Cr levels without a carcinogenic outcome because of these protective mechanisms.
Acknowledgements
We would like to thank the New England Aquarium staff for their collection of right whale biopsies, Teri Rowles of National Oceanic and Atmospheric Administration and Margie Peden-Adams of the Medical University of South Carolina for their efforts in the tissue collections of the neonate right whale. We would also like to thank Amie Holmes for assistance with the human chromosome damage scoring and David Kirstein and Christy Gianios, Jr. for administrative and technical support.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Funding
This work was supported by Grant number NA03NMF4720478 from the United States Department of Commerce, National Oceanic and Atmospheric Administration (J.P.W.); National Institute of Environmental Health Sciences Grant number ES10838 (J.P.W.) and the Maine Center for Toxicology and Environmental Health.
References
- Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological profile for Chromium. U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry; Atlanta, GA: 2008. [Google Scholar]
- Al-Hamood MH, Elbetieha A, Bataineh H. Sexual maturation and fertility of male and female mice exposed prenatally and postnatally to trivalent and hexavalent chromium compounds. Reprod. Fertil. Dev. 1998;10:179–183. doi: 10.1071/r97001. [DOI] [PubMed] [Google Scholar]
- Bataineh H, Al-Hamood MH, Elbetieha A, Bani Hani I. Effect of long-term ingestion of chromium compounds on aggression, sex behavior and fertility in adult male rat. Drug Chem. Toxicol. 1997;20:133–149. doi: 10.3109/01480549709003875. [DOI] [PubMed] [Google Scholar]
- Chowdhury AR, Mitra C. Spermatogenic and steroidogenic impairment after chromium treatment in rats. Indian J. Exp. Biol. 1995;33:480–484. [PubMed] [Google Scholar]
- Chung YS. Air pollution detection by satellites: The transport and deposition of air pollutants over oceans. Atmos. Environ. 1986;20:617–630. [Google Scholar]
- Geisler CD, Schmidt D. An overview of chromium in the marine environment. Deut. Hydrogr. Z. 1991;44:185–196. [Google Scholar]
- Hartwig A, Schlepegrell R, Beyersmann D. Indirect mechanism of lead-induced genotoxicity in cultured mammalian cells. Mutat. Res. 1990;241:75–82. doi: 10.1016/0165-1218(90)90110-n. [DOI] [PubMed] [Google Scholar]
- Holmes AL, Wise SS, Xie H, Gordon N, Thompson WD, Wise JP., Sr. Lead ions do not cause human lung cells to escape chromate-induced cytotoxicity. Toxic. Appl. Pharmac. 2005;203:167–176. doi: 10.1016/j.taap.2004.08.006. [DOI] [PubMed] [Google Scholar]
- Holmes AL, Wise SS, Sandwick SJ, Lingle WL, Negron VC, Thompson WD, Wise JP., Sr. Chronic exposure to lead chromate causes centrosome abnormalities and aneuploidy in human lung cells. Cancer Res. 2006;66:4041–4048. doi: 10.1158/0008-5472.CAN-05-3312. [DOI] [PubMed] [Google Scholar]
- Hixon MA, Boersma PD, Hunter ML, Jr., Micheli F, Norse EA, Possingham HP, Snelgrove PVR. Oceans at risk: Research priorities in marine conservation biology. In: Soulé ME, Orians GH, editors. Conservation Biology: Research Priorities for the Next Decade. Island Press; Washington, D.C: 2001. pp. 125–154. [Google Scholar]
- IARC . Monographs on the Evaluation of Carcinogenic Risks to Humans: Chromium, Nickel and Welding. Vol. 49. International Agency for Research on Cancer; Lyons, France: 1990. [PMC free article] [PubMed] [Google Scholar]
- Kareiva P. When one whale matters. Nature. 2001;414:493–494. doi: 10.1038/35107167. [DOI] [PubMed] [Google Scholar]
- Knowlton AR, Kraus SD, Kenney RD. Reproduction in North Atlantic right whales (Eubalaena glacialis). Can. J. Zool. 1994;72:1297–1305. [Google Scholar]
- Kraus SD. Rates and potential causes of mortality in North Atlantic right whale (Eubalaena glacialis). Mar Mammals Sci. 1990;6:278–291. [Google Scholar]
- Kraus SD, Hamilton PK, Kenney RD, Knowlton AR, Slay CK. Reproductive parameters of the North Atlantic right whale. J. Cetacean Res. Manage. 2001;2(special issue):213–236. [Google Scholar]
- Kraus SD, Rolland RM. Right whales in the urban ocean. In: Kraus SD, Rolland RM, editors. The urban whale: North Atlantic right whale at the crossroads. Harvard University Press; Cambridge, Massachusetts: 2007. p. 543. [Google Scholar]
- Levy LS, Venitt S. Carcinogenicity and mutagenicity of chromium compounds: the association between bronchial metaplasia and neoplasia. Carcinogenesis. 1986;7:831–835. doi: 10.1093/carcin/7.5.831. [DOI] [PubMed] [Google Scholar]
- Li Chen T, Wise SS, Kraus S, Shaffiey F, Grau M, Thompson WD, Zheng T, Zhang Y, Romano T, O'Hara T, Wise JP., Sr. Particulate hexavalent chromium is cytotoxic and genotoxic to the North Atlantic right whale (Eubalaena glacialis) lung and skin fibroblasts. Environ. Mol. Mutagen. 2009 doi: 10.1002/em.20471. In Press. [DOI] [PubMed] [Google Scholar]
- Mancuso TF. Chromium as an industrial carcinogen: Part II. Chromium in human tissues. Am. J. Ind. Med. 1997;2:140–7. doi: 10.1002/(sici)1097-0274(19970204)31:2<140::aid-ajim2>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- Paz-y-Miño C, Davalos MV, Sánchez ME, Arevalo M, Leone PE. Should gaps be included in chromosomal aberration analysis? Evidence based on the comet assay. Mutat. Res. 2002;516:57–61. doi: 10.1016/s1383-5718(02)00021-9. [DOI] [PubMed] [Google Scholar]
- Pettine M, Millero FJ. Chromium speciation in seawater: The probable role of hydrogen peroxide. Limnol. Oceanogr. 1990;35:730–736. [Google Scholar]
- Rolland RM, Hamilton PK, Marx MK, Pettis HM, Angell CM, Moore MJ. External perspectives on right whale health. In: Kraus SD, Rolland RM, editors. The urban whale: North Atlantic right whale at the crossroads. Harvard University Press; Cambridge, Massachusetts: 2007. p. 543. [Google Scholar]
- Roy NK, Rossman TG. Mutagenesis and comutagenesis by lead compounds. Mutat. Res. 1992;298:97–103. doi: 10.1016/0165-1218(92)90034-w. [DOI] [PubMed] [Google Scholar]
- SAS Institute . SAS/STAT 9.1 User's Guide. SAS Institute, Inc.; Cary, NC: 2004. p. 5136. [Google Scholar]
- Wise SS, Holmes AL, Ketterer ME, Hartsock WJ, Fomchenko E, Katsifis SP, Thompson WD, Wise JP., Sr Chromium is the proximate clastogenic species for lead chromate-induced clastogenicity in human bronchial cells. Mutat. Res. 2004;560:79–89. doi: 10.1016/j.mrgentox.2004.02.009. [DOI] [PubMed] [Google Scholar]
- Wise SS, Holmes AL, Wise JP., Sr. Particulate and soluble hexavalent chromium are cytotoxic and genotoxic to human lung epithelial cells. Mutat. Res. 2006;610(1-2):2–7. doi: 10.1016/j.mrgentox.2006.06.005. [DOI] [PubMed] [Google Scholar]
- Wise SS, Holmes AL, Wise JP., Sr. Hexavalent chromium-induced DNA damage and repair mechanisms. Rev. Environ. Health. 2008;23:39–57. doi: 10.1515/reveh.2008.23.1.39. [DOI] [PubMed] [Google Scholar]
- Wise JP, Sr., Wise SS, Little JE. The Cytotoxicity and genotoxicity of particulate and soluble hexavalent chromium in human lung cells. Mutat. Res. 2002;517:221–229. doi: 10.1016/s1383-5718(02)00071-2. [DOI] [PubMed] [Google Scholar]
- Wise JP, Sr., Payne R, Wise SS, LaCerte C, Wise J, Gianios C, Jr., Thompson WD, Perkins C, Zheng T, Zhu C, Benedict L, Kerr I. A global assessment of chromium pollution using sperm whales (Physeter macrocephalus) as an indicator species. Chemosphere. 2009;75:1461–1467. doi: 10.1016/j.chemosphere.2009.02.044. [DOI] [PubMed] [Google Scholar]
- Wise JP, Sr., Wise SS, Kraus S, Shaffiey F, Grau M, Li Chen T, Perkins C, Thompson WD, Zheng T, Zhang Y, Romano T, O'Hara T. Hexavalent chromium is cytotoxic and genotoxic to the North Atlantic right whale (Eubalaena glacialis) lung and testes fibroblasts. Mutat. Res. 2008;650:30–38. doi: 10.1016/j.mrgentox.2007.09.007. [DOI] [PubMed] [Google Scholar]
- Witmer CM, Harris R, Shupack SI. Oral bioavailability of chromium from a specific site. Environ. Health Perspect. 1991;92:105–110. doi: 10.1289/ehp.92-1519374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witmer CM, Park H-S, Shupack SI. Mutagenicity and disposition of chromium. Sci. Total Environ. 1989;86:131–148. doi: 10.1016/0048-9697(89)90200-3. [DOI] [PubMed] [Google Scholar]
- Xie H, Wise SS, Holmes AL, Xu B, Wakeman T, Pelsue SC, Singh NP, Wise JP., Sr. Carcinogenic lead chromate induces dna double-strand breaks in human lung cells. Mutat. Res. 2005;586:160–172. doi: 10.1016/j.mrgentox.2005.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]