Abstract
Aims
Diabetes increases the risk of tuberculosis and the prevalence of diabetes is rising in tuberculosis-endemic regions such as sub-Saharan Africa. Resource-appropriate strategies for tuberculosis case finding among African adults with diabetes are needed. The aims of this study were to determine prevalence of tuberculosis and evaluate one screening strategy among adult Tanzanians with diabetes.
Methods
In this prospective cohort study, we evaluated a “cough-triggered” strategy for tuberculosis case finding among adults with diabetes at our zonal hospital in Tanzania. All adults with diabetes and cough underwent further tuberculosis symptom assessment and those with productive cough had sputum collected for microscopy and M. tuberculosis culture.
Results
Between September 2011 and March 2012, 700 adults with diabetes attended our hospital. A total of 693 were enrolled, 121/693 (17.5%) had cough and 32/693 (4.6%) had at least 2 of the classic symptoms of tuberculosis. Of note, 87/121 (71.9%) of patients with cough could not produce sputum spontaneously. Nine patients were diagnosed with tuberculosis for a prevalence of 1299/100,000 (1.3%), 7-fold greater than the national average.
Conclusions
Tuberculosis is common among Tanzanian adults with diabetes but tuberculosis case finding is challenging due to the high prevalence of non-productive cough. This low-cost, ‘cough-triggered’ tuberculosis case-finding strategy may serve as a reasonable first step for improving tuberculosis screening among adults with diabetes in sub-Saharan Africa.
INTRODUCTION
The prevalence of diabetes mellitus is increasing worldwide, particularly in developing regions such as sub-Saharan Africa where tuberculosis prevalence is also high [1–3]. An estimated 70% of all persons with diabetes currently reside in low and middle-income countries where 95% of all persons with tuberculosis also live [2]. As diabetes becomes increasingly prevalent in developing regions, these 2 epidemics will continue to converge [4].
Diabetes increases the relative risk for tuberculosis by an average of 3-fold [5–7], with larger effect estimates in regions of higher tuberculosis prevalence [7]. In studies from India and the USA/Mexico border, 15%–25% of tuberculosis was related to diabetes [5,8]. The prevalence of tuberculosis among patients with diabetes varies widely depending on region, population and tuberculosis case-finding strategy [9], and very few studies have been done among adults with diabetes in Africa [9–11].
Tanzania is one of the world’s 22 high-burden countries for tuberculosis with a national prevalence of 177/100,000 and >60% of new cases occurring in the HIV-uninfected [12,13]. According to the International Diabetes Foundation, the prevalence of diabetes is 3.2% among Tanzanian adults with higher prevalences in urban areas [14], but this is likely an underestimate. A recent study from our city in Tanzania, for example, showed that the prevalence of diabetes mellitus among adults with tuberculosis was 16.7% versus 9.4% among uninfected adults (p<0.001) [12].
For this reason, the World Health Organization (WHO) has recently urged further research to determine the best strategy for tuberculosis screening among adults with diabetes, particularly in resource-poor settings [2,15]. WHO guidelines state that, at a minimum, people with diabetes should be screened for cough at the time of diagnosis and during regular check-ups [15], but in our experience, this is often not done in busy diabetic clinics in Tanzania. In this prospective cohort study, we assessed a simple tuberculosis screening protocol among adults with diabetes presenting to our zonal hospital in Tanzania [16,17]. We hypothesized that the prevalence of tuberculosis would be >3-fold more than the national prevalence.
PATIENTS AND METHODS
Study area
The study was conducted at Bugando Medical Centre (BMC) in Mwanza, Tanzania between September 2011 and March 2012. BMC is the referral hospital for Tanzania’s Lake Zone, with 850 inpatient beds and ~2400 patients receiving care annually at the diabetes clinic. The prevalence of tuberculosis in our region has been estimated at 80/100,000, lower than the national prevalence [18]. The BMC diabetes clinic provides primary care for persons with diabetes who live in the city of Mwanza. In Tanzania, most diabetes care is provided in hospital clinics since these are the only health facilities that have a reliable supply of insulin and common oral hypoglycemic agents. In our clinic, the diagnosis of diabetes is made in patients who have symptoms of diabetes and fasting blood glucose ≥7 mmol/liter and/or random blood glucose ≥11.1 mmol/liter.
Study design and population
This was a prospective cohort study for which all adults with diabetes (age ≥18 years) seen at BMC were eligible. Adult outpatients and inpatients with diabetes were asked whether they currently had a cough. Patients reporting cough of any duration completed a structured questionnaire and standardized physical examination to elicit other symptoms/signs of tuberculosis. Cough of any duration triggered further tuberculosis screening since research in Tanzanian HIV-infected adults has shown that cough duration was not helpful in discriminating between patients with and without tuberculosis [16]. All patients with productive cough underwent sputum microscopy for acid-fast bacilli (AFB), sputum culture for Mycobacterium tuberculosis, and chest radiograph (CXR). Patients were followed until the diagnosis of tuberculosis was either confirmed or excluded. A required sample size of 384 was calculated using the Leslie Kish formula for prevalence in a cross sectional study assuming a prevalence of tuberculosis of 0.5% (approximately 4× the national tuberculosis prevalence) and allowing a standard error of 0.05%. For the sake of assessing the longer-term feasibility of this new screening strategy, we decided to enroll all patients seen in the diabetes clinic over the 6-month enrollment period.
Investigations
In patients with productive cough, two sputum samples were collected (spot and morning) and examined under fluorescence microscopy for AFB. Culture for Mycobacterium tuberculosis was performed using Lowenstein-Jensen solid culture medium after sodium hydroxide digestion, decontamination and concentration. CXRs were obtained in postero-anterior view and interpreted with a standard grading sheet. Two physicians independently interpreted each CXR and the hospital’s radiologist resolved discordant results. HIV counseling and voluntary testing was conducted in all patients with productive cough according to the Tanzanian National HIV Guidelines [19].
Case definition
Our primary case definition for tuberculosis is based upon the American Thoracic Society definition [17,20–22]. Patients must meet 2 of the 3 following criteria: i) symptoms of tuberculosis (cough, fever, night sweats, etc); ii) either visible AFB or growth of M. tuberculosis from sputum; iii) CXR independently interpreted as highly suggestive of tuberculosis. For patients with sputum-negative tuberculosis, we also required clinical response to anti-tuberculosis treatment within the first 3 months of treatment. We also classified tuberculosis cases according to the WHO definitions as bacteriologically confirmed tuberculosis or clinically diagnosed tuberculosis [23]. For clinically diagnosed tuberculosis we required symptoms and CXR findings consistent with tuberculosis + response to anti-tuberculosis treatment.
Data analysis
The primary outcome was tuberculosis diagnosis. Data were double entered, verified and cleaned using Microsoft Excel 2007, and analysis was done using STATA version 10 (College Station, Texas). Categorical variables were reported as proportions (%). Continuous data were described as means (standard deviation) or medians [interquartile range] depending on the distribution of data.
Ethical considerations
All participants provided written informed consent either by signature or fingerprint. Ethical approval for the study was obtained from BMC and Weill Cornell Medical College. The decision to treat for tuberculosis was made by hospital clinicians according to Tanzanian national guidelines, and anti-tuberculosis treatment was provided as directly-observed therapy according to the same guidelines [24].
RESULTS
Enrollment
A total of 700 adult patients with diabetes attended BMC from September 2011-March 2012 and 693 (99%) agreed to participate in the study. Of these, 121 (17.5%) reported cough and underwent further evaluation for tuberculosis. See Fig. 1.
Figure 1.
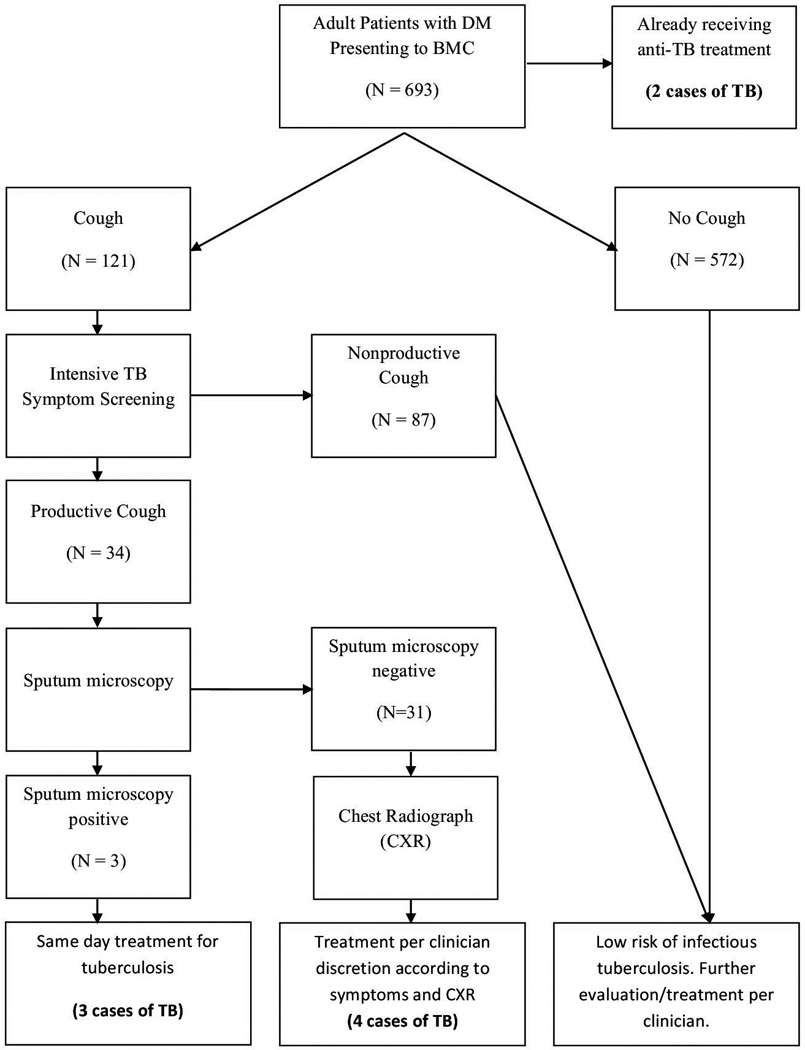
Case-finding strategy for identification of patients with tuberculosis (TB) among adults with diabetes mellitus (DM) presenting to Bugando Medical Centre (BMC), a zonal hospital in Tanzania.
Study population
Among the 121 patients with diabetes and cough, the median age was 60 years [50–66], and 62/121 (51.2%) were women. The large majority had Type 2 diabetes (109/121, 90.0%) and the remaining patients had Type 1 diabetes.
Cough had persisted for >2 weeks in 91/121 (75.2%), and 87/121 (71.9%) reported a dry cough without sputum production. Among these, 47/87 (54.0%) were using ACE-inhibitors. Other symptoms of tuberculosis were also common. See Table 1 (center column) for details. Sputum samples for microscopy and culture were sent for all 34 patients who had productive cough. Of these 34 patients, 26 (76.5%) agreed to HIV testing, 2 (5.9%) were positive and both of these were referred to the HIV clinic for further management.
Table 1.
Demographic and clinical characteristics of 121 consecutive adults with diabetes mellitus presenting to BMC with cough (center column) and among the 7 of these adults who were newly diagnosed with tuberculosis during the screening period (right column). All results are reported as proportion (%) or median [interquartile range].
| Characteristic | All Adults with Diabetes and Cough (N=121) |
Adults with Newly Diagnosed Tuberculosis (N=7) |
|---|---|---|
| Female | 62 (51.2%) | 2 (28.6%) |
| Age (years) | 60 [50–66] | 45 [40–60] |
| Outpatient | 96 (79.3%) | 1 (14.3%) |
| BMI (kg/m2) | 26.3 [23.1–30.9] | 23.3 [20.8–26.0] |
| Poorly-controlled diabetes* | 56 (46.3%) | 5 (71.4%) |
| Type 2 Diabetes | 109 (90.0%) | 7 (100%) |
| Duration of diabetes (months) | 54 [9–96] | 36 [1–96] |
| Use of ACE inhibitors | 61 (50.4%) | 1 (14.3%) |
| Prior tuberculosis | 9 (7.4%) | 1 (14.3%) |
| Duration of cough (weeks) | 4.3 [2.0–12.9] | 4.3 [3.0–21.4] |
| Coughing for >2 weeks | 91 (75.2%) | 7 (100%) |
| Non-productive (dry) cough | 87 (71.9%) | 0 (0%) |
| Hemoptysis | 1 (0.8%) | 1 (14.3%) |
| Fever | 22 (18.2%) | 3 (42.9%) |
| Duration of fever (days) | 7 [7–14] | 30 [14–60] |
| Reported fever > 2 weeks | 9 (7.4%) | 3 (42.9%) |
| Drenching night sweats | 16 (13.2%) | 2 (28.6%) |
| Duration of night sweats (days) | 14 [6–30] | 52 [14–90] |
| Weight loss >5kg | 21 (17.4%) | 5 (71.4%) |
| Number of classic tuberculosis symptoms† | ||
| No symptom | 18 (14.9%) | 0 (0%) |
| One symptom | 71 (58.7%) | 0 (0%) |
| Two or more symptoms | 32 (26.4%) | 7 (100%) |
Poorly controlled diabetes is defined as fasting blood glucose > 7 mmol/L or a random blood glucose > 12 mmol/L at the time of enrollment.
Classic tuberculosis symptoms: cough > 2 weeks, fever, weight loss >5kg and drenching night sweats.
Chest radiographs
CXRs were obtained in 25/34 (73.5%) of patients with productive cough. The remaining 9 patients presented at times when the chest radiograph machine was not functional. Twenty of 25 (80.0%) films were abnormal, and 14/25 (56.0%) had multiple abnormal findings. Radiographic abnormalities included unilateral effusion (8/20,40.0%), lower lobe opacity (7/20,35.0%), cavitations (6/20,30.0%), upper lobe opacity (6/20,30.0%), pulmonary edema (6/20,30.0%), hilar adenopathy (2/20,10.0%) and emphysema (1/20,5.0%). Eight out of the 25 (32.0%) films were poor quality due to obesity and poor penetration.
Tuberculosis diagnosis
A total of 9/693 (1.3%) adults with diabetes had tuberculosis, giving a point prevalence of 1299/100,000 persons, 7.1-fold greater than the national prevalence and 16.3-fold greater than the regional prevalence. Of these, according to the WHO definitions, 5/9 had bacteriologically confirmed tuberculosis and 4/9 had clinically diagnosed tuberculosis with both symptoms and CXR that were consistent with tuberculosis and response to anti-tuberculosis treatment.
Among the 7 adults with newly diagnosed tuberculosis during the screening period, 2/7 (28.6%) were women, the median age was 45 [40–60] and all had Type 2 diabetes. One of these patients was HIV-infected. Three of these adults had smear positive sputum and the same 3 adults had positive sputum cultures for Mycobacterium tuberculosis. See Table 1 (right column) for details.
The chest radiograph was abnormal in 6/7 (85.7%) with all 6 having multiple abnormal findings. The most common findings on chest radiograph were lower lobe opacification in 5/7 (71.4%), upper lobe opacification in 4/7 (57.1%), cavitation in 4/7 (57.1%), and unilateral pleural effusion in 3/7 (42.9%).
DISCUSSION
In our study in Tanzania, the prevalence of tuberculosis in adults with diabetes was 1299/100,000, >7-fold greater than the national prevalence of 177/100,000 [13]. To the best of our knowledge, the only other study of tuberculosis screening among sub-Saharan Africans with diabetes revealed a prevalence of 488/100,000 in Ethiopia [25]. Studies in African children with Type I diabetes revealed even higher prevalences [10,11,26]. Together, these data demonstrate an alarmingly high prevalence of tuberculosis among adults with diabetes in sub-Saharan Africa where the number of people with diabetes is projected to increase from 7 million in 2000 to 18 million in 2030, a regional increase of 161% that is second only to the Middle East [3].
A recent WHO committee urged that “prospective observational cohort studies should be conducted” to determine “whether, when and how to screen for tuberculosis in patients with diabetes [2].” Inspired by that report, we designed this study to assess a simple, “cough-triggered” screening algorithm that we have previously used successfully in HIV-infected adults [17]. This low-cost strategy yielded the diagnosis of 7 new tuberculosis cases over 6 months and may represent a reasonable first step for tuberculosis screening among adults diabetes in resource-limited settings. Future studies should evaluate the addition of screening for other tuberculosis symptoms such as fevers, weight loss and night sweats as is already recommended in HIV-infected adults [27].
Among our African adults with diabetes and cough, nearly 5% had at least 2 of the classic tuberculosis symptoms. Any of these symptoms alone may be attributable to diabetes itself, medications used for diabetes (particularly ACE-inhibitors) or other complications of diabetes (including pneumonia, other infection, heart failure or autonomic dysfunction). The presence of these symptoms together, though, raises the possibility of undiagnosed pulmonary tuberculosis in our population in patients who either could not produce sputum or were smear-negative. In one recent study from India, more than half of adults with diabetes and tuberculosis either could not produce sputum or were smear-negative [8], and smear microscopy is known to have low sensitivity in HIV-prevalent countries such as Tanzania [28]. Clear guidelines should be developed for when empiric anti-tuberculosis treatment should be considered in adults with diabetes and tuberculosis symptoms, similar to the guidelines already available for HIV [27,28].
Our study also highlights several challenges of tuberculosis case finding among adults with diabetes in Africa. The high rates of non-productive cough (15% of all adults) made diagnosis difficult in a setting where clinicians rely on sputum microscopy to confirm tuberculosis. Sputum induction is a logical next step [29], but operational studies are needed assess the feasibility of sputum induction in busy diabetes clinics in Africa. Additionally, our hospital administration felt that CXR could not be performed on all adults with diabetes because its cost (≈10 USD) would not be sustainable. Tuberculosis screening services, including CXR, should be offered cost-free in Africa for patients with diabetes as they are for patients with HIV. In addition, given the high prevalence of tuberculosis among adults with diabetes in Africa, diabetic clinics must develop infection control plans that includes both administrative and environmental control measures to reduce nosocomial tuberculosis transmission [15].
One limitation for this study is the potential for bias leading to under diagnosis of tuberculosis. Among patients without productive cough, no tests were available to confirm the diagnosis of tuberculosis. Another limitation of this study was that HIV counseling and testing was not considered feasible for all patients due to the large number of patients and limited private space in our diabetes clinic. Facilities and systems for HIV testing among adults with diabetes in Africa need to be improved.
In conclusion, the prevalence of tuberculosis among adults with diabetes attending our hospital in Tanzania was 7-fold higher than that reported in the general population. Many additional patients had classic symptoms of tuberculosis but were not started on anti-tuberculosis therapy because of concern that these symptoms may be due to diabetes medications or complications of diabetes. These symptomatic patients may have had tuberculosis but did not meet our studies case definition; thus the actual prevalence of tuberculosis among our adults with diabetes may be even higher than that reported in this study. This study illustrates the importance and challenges of screening for tuberculosis among adults with diabetes in Africa and provides one possible strategy for resource-limited settings.
Novelty Statement.
Data regarding tuberculosis among adults with diabetes in sub-Saharan Africa are few and the World Health Organization has advocated research regarding tuberculosis screening strategies in this population, suggesting screening for cough as a first step.
In this prospective tuberculosis screening study among adults with diabetes in Tanzania, the incidence of tuberculosis was 7-fold greater than the general adult population.
We also assessed the strengths and weaknesses of a cough-triggered tuberculosis screening strategy that may be appropriate for resource-limited settings.
These data illustrate the importance of screening for tuberculosis among adults with diabetes in Africa and provide one possible low-cost strategy.
ACKNOWLEDGEMENTS
The authors acknowledge the administrative support and guidance of Prof. Charles Majinge, Director General of BMC.
FUNDING:
No funding.
Footnotes
CONFLICTS OF INTEREST:
The authors have no relevant conflicts of interest to disclose.
REFERENCES
- 1.Hall V, Thomsen RW, Henriksen O, Lohse N. Diabetes in Sub Saharan Africa 1999–2011: epidemiology and public health implications. A systematic review. BMC Pub Health. 2011;11(1):564. doi: 10.1186/1471-2458-11-564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Harries AD, Murray MB, Jeon CY, Ottmani S-E, Lonnroth K, Barreto ML, et al. Defining the research agenda to reduce the joint burden of disease from diabetes mellitus and tuberculosis. Trop Med Int Health. 2010;15(6):659–663. doi: 10.1111/j.1365-3156.2010.02523.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27(5):1047–1053. doi: 10.2337/diacare.27.5.1047. [DOI] [PubMed] [Google Scholar]
- 4.Dooley KE, Chaisson RE. Tuberculosis and diabetes mellitus: convergence of two epidemics. Lancet Infect Dis. 2009;9(12):737–746. doi: 10.1016/S1473-3099(09)70282-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pablos-Méndez A, Blustein J, Knirsch C. The role of diabetes mellitus in the higher prevalence of tuberculosis among Hispanics. Am J Pub Health. 1997;87(4):574–579. doi: 10.2105/ajph.87.4.574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pérez A, Brown HS, Restrepo BI. Association between tuberculosis and diabetes in the Mexican border and non-border regions of Texas. Am J Trop Med Hyg. 2006;74(4):604–611. [PMC free article] [PubMed] [Google Scholar]
- 7.Jeon CY, Murray MB. Diabetes mellitus increases the risk of active tuberculosis: a systematic review of 13 observational studies. PLoS Med. 2008;5(7):e152. doi: 10.1371/journal.pmed.0050152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.India Diabetes Mellitus Tuberculosis Study Group. Screening of patients with diabetes mellitus for tuberculosis in India. Trop Med Int Health. 2013;18(5):646–654. doi: 10.1111/tmi.12083. [DOI] [PubMed] [Google Scholar]
- 9.Jeon CY, Harries AD, Baker Ma, Hart JE, Kapur A, Lönnroth K, et al. Bi-directional screening for tuberculosis and diabetes: a systematic review. Trop Med Int Health. 2010;15(11):1300–1314. doi: 10.1111/j.1365-3156.2010.02632.x. [DOI] [PubMed] [Google Scholar]
- 10.Gill GV, Huddle KR, Krige LP. Intensive health screening of young black diabetics. S Afr Med J. 1984;65(20):815–816. [PubMed] [Google Scholar]
- 11.Webb E, Hesseling A, Schaaf H, Gie R, Lombard C, Spitaels A, et al. High prevalence of Mycobacterium tuberculosis infection and disease in children and adolescents with type 1 diabetes mellitus. Int J Tuberc Lung Dis. 2009;13(7):868–874. [PubMed] [Google Scholar]
- 12.Faurholt-Jepsen D, Range N, PrayGod G, Jeremiah K, Faurholt-Jepsen M, Aabye MG, et al. Diabetes Is a Risk Factor for Pulmonary Tuberculosis: A Case-Control Study from Mwanza, Tanzania. PLoS ONE. 2011;6(8):e24215. doi: 10.1371/journal.pone.0024215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.World Health Organization. Tanzania Tuberculosis Profile - 2012 [online] [Accessed 25 January 2013]; Available from: www.who.int/tb/data.
- 14.International Diabetes Federation. Diabetes Atlas - 2012 [online] [Accessed 11 July 2013]; Available from: http://www.idf.org/diabetesatlas. [Google Scholar]
- 15.World Health Organization. Collaborative Framework for Care and Control of Tuberculosis and Diabetes. Geneva, Switzerland: 2011. [Accessed 11 July 2013]. Available from: www.who.int/diabetes/publications/tb_diabetes2011/en. [PubMed] [Google Scholar]
- 16.Ngadaya ES, Mfinanga GS, Wandwalo ER, Morkve O. Detection of pulmonary tuberculosis among patients with cough attending outpatient departments in Dar Es Salaam, Tanzania: does duration of cough matter? BMC Health Serv Res. 2009;9:112. doi: 10.1186/1472-6963-9-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Burgess AL, Fitzgerald DW, Severe P, Joseph P, Noel E, Rastogi N, et al. Integration of tuberculosis screening at an HIV voluntary counselling and testing centre in Haiti. AIDS. 2001;15(14):1875–1879. doi: 10.1097/00002030-200109280-00018. [DOI] [PubMed] [Google Scholar]
- 18.National Tuberculosis and Leprosy Control Program. Regional Prevalences [internet]. [Accessed 6 June 2013]; Available from: http://ntlp.go.tz/index.php?option=com_content&view=article&id=69&Itemid=125. [Google Scholar]
- 19.Tanzanian National Aids Control Program. National Guidelines for the Management of HIV/AIDS. 3rd edition. Dar es Salaam, Tanzania: 2009. [Accessed 11 July 2013]. Available from: http://www.who.int/hiv/pub/guidelines/tanzania_art.pdf. [Google Scholar]
- 20.Blumberg H, Burman W, Chaisson RE, Daley CL, Etkind SC, Friedman LN, et al. American Thoracic Society, Centers for Disease Control and Prevention and Infectious Diseases Society of America: treatment of tuberculosis. Am J Resp Crit Care Med. 2003;167(4):603–662. doi: 10.1164/rccm.167.4.603. [DOI] [PubMed] [Google Scholar]
- 21.Kidenya BR, Kabangila R, Peck RN, Mshana SE, Webster LE, Koenig SP, et al. Early and efficient detection of mycobacterium tuberculosis in sputum by microscopic observation of broth cultures. PLoS One. 2013;8(2):4–8. doi: 10.1371/journal.pone.0057527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peck R, Luhanga A, Kalluvya S, Todd J, Lugoba S, Fitzgerald D, et al. Predictors of tuberculosis in first 6 months after initiation of antiretroviral therapy: a case-control study. Int J Tuberc Lung Dis. 2012;16:1047–1051. doi: 10.5588/ijtld.11.0772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.World Health Organization. Definitions and reporting framework for tuberculosis – 2013 revision. Geneva, Switzerland: 2013. [Accessed 11 July 2013]. Available from: http://apps.who.int/iris/bitstream/10665/79199/1/9789241505345_eng.pdf. [Google Scholar]
- 24.Tanzania Ministry of Health. Manual of the national tuberculosis and leprosy program in Tanzania. Dar es Salaam, Tanzania: 2006. [Accessed 20 March 2013]. Available from: http://www.who.int/hiv/pub/guidelines/tanzania_tb.pdf. [Google Scholar]
- 25.Lester F. Tuberculosis in Ethiopian diabetics. Ethiop Med J. 1984;22(3):129–133. [PubMed] [Google Scholar]
- 26.Swai AB, McLarty DG, Mugusi F. Tuberculosis in diabetic patients in Tanzania. Trop Doct. 1990;20(4):147–150. doi: 10.1177/004947559002000402. [DOI] [PubMed] [Google Scholar]
- 27.World Health Organization. Guidelines for intensified tuberculosis case-finding and isoniazid preventive therapy for people living with HIV in resource-constrained settings. Geneva, Switzerland: 2011. [Accessed 11 July 2013]. Available from: http://whqlibdoc.who.int/publications/2011/9789241500708_eng.pdf. [Google Scholar]
- 28.World Health Organization. Improving the diagnosis and treatment of smear-negative pulmonary and extrapulmonary tuberculosis among adults and adolescents. Recommendations for HIV-prevalent and resource-constrained settings. Geneva, Switzerland: 2007. [Accessed 11 July 2013]. Available from: http://www.who.int/tb/publications/2006/tbhiv_recommendations.pdf. [Google Scholar]
- 29.Brown M, Varia H, Bassett P, Davidson RN, Wall R, Pasvol G. Prospective study of sputum induction, gastric washing, and bronchoalveolar lavage for the diagnosis of pulmonary tuberculosis in patients who are unable to expectorate. Clin Infect Dis. 2007;44(11):1415–1420. doi: 10.1086/516782. [DOI] [PubMed] [Google Scholar]


