Abstract
The sperm acrosome reaction (AR), an essential exocytosis step in mammalian fertilization, is mediated by a species-specific interaction of sperm surface molecules with glycans on the egg. Previous studies indicate that a subset of terminal carbohydrates on the mouse egg zona pellucida (ZP) trigger the AR by cross-linking or aggregating receptors on the sperm membrane. However, the exact role of those carbohydrates in AR has not been identified and the mechanism underlying the AR still needs further investigation. To study this process, a series of glycopolymers was synthesized. The glycopolymers are composed of a multivalent scaffold (norbornene), a functional ligand (previously identified ZP terminal monosaccharides), and a linker connecting the ligand and the scaffold. The polymers were tested for their ability to initiate AR and through which signaling pathways AR induction occurred. Our data demonstrate that mannose, fucose, and β-N-acetylglucosamine 10-mers and 100-mers initiate AR in a dose-dependent manner, and the 100-mers are more potent on a per monomer basis than the 10-mers. Although nearly equipotent in inducing the AR at the optimal concentrations, their AR activation kinetics are not identical. Similar to mouse ZP3, all 100-mer-activated AR are sensitive to guanine-binding regulatory proteins (G-proteins), tyrosine kinase, protein kinase A, protein kinase C, and Ca2+-related antagonists. Thus, the chemotypes of synthetic glycopolymers imitate the physiologic AR-activation agents and provide evidence that occupation of one of at least three different receptor binding sites is sufficient to initiate the AR.
Mammalian fertilization involves a series of sophisticated steps. It starts with sperm–egg binding, sperm penetration of the egg outer membrane, the zona pellucida (ZP), and finally sperm–egg fusion to form an embryo.1 In the mouse, the ZP consists of three glycoproteins (ZP1, ZP2, and ZP3) that form a cross-linked extracellular matrix surrounding the egg. Each glycoprotein contains glycans covalently linked to asparagine (N-linked) and/or serine and threonine (O-linked).2 Previous studies indicated that the adhesion of sperm to the egg is mainly mediated by a specific interaction of sperm surface molecules with the glycan moieties on the ZP.3−5 Glycosidase digestion and mass spectrometric analyses have indicated that several mouse ZP3 terminal monosaccharide residues, including β-N-acetylgalactosamine (GalNAc),6 β-N-acetylglucosamine (GlcNAc),7 mannose,8 galactose,9 and sialyl,10 may be critical for sperm binding to the ZP. Although a fucosylated glycan was identified on human ZP11 and the addition of a fucose residue to the GlcNAc and galactose trisaccharides enhances mouse sperm–egg binding affinity,12 the participation of fucose in mouse gamete binding is not definitive. The complex heterogeneity of zona glycosides makes definition of the exact function of these sugars a challenge.
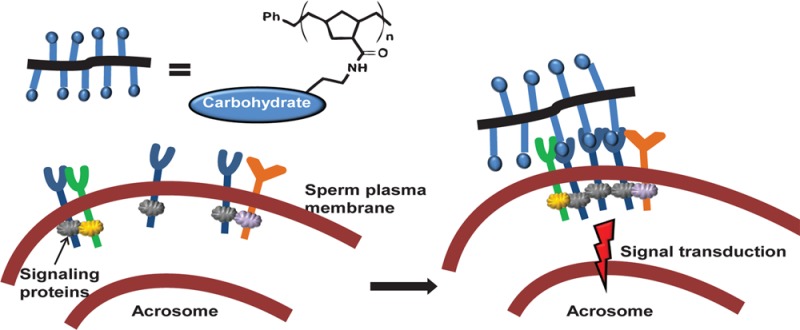
To penetrate the thick ZP layer, sperm need to undergo a cellular exocytosis known as the acrosome exocytosis or acrosome reaction (AR). Structurally, the acrosome is composed of a membrane vesicle filled with soluble components and acrosomal matrix proteins.2 When the AR occurs (Figure 1), the sperm plasma membrane fuses with the outer acrosomal membrane; the soluble contents in the vesicle, e.g., hydrolases and proteases, are sequentially released.13 The acrosome reaction plays an essential role in fertilization, and only acrosome-reacted sperm can participate in the following fertilization steps that lead to sperm–egg fusion.14
Figure 1.
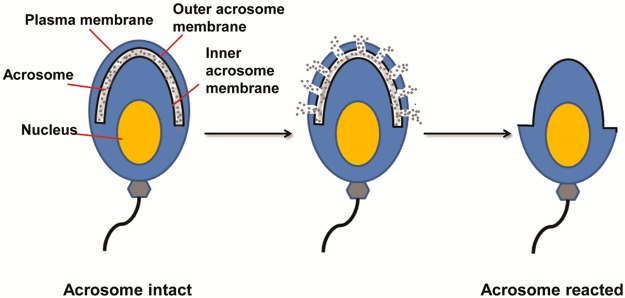
Overview of mouse sperm acrosome reaction.
A number of physiological and nonphysiological agonists can activate the AR. Though controversial, ZP3 was widely accepted as the primary AR activator. Multiple sperm–carbohydrate binding events occur to stimulate the AR by cross-linking or aggregating receptors on the sperm plasma membrane.15,16 This hypothesis is based on inhibition and activation studies with deglycosylated ZP and fragmented ZP.
To elucidate the role of individual carbohydrates in initiating the AR, activity studies of neoglycoconjugates have been undertaken. Loeser et al. (17) examined the properties of several neoglycoproteins that are bovine serum albumin (BSA) conjugates with an average of 8 copies of the glycan of interest per protein molecule. They concluded that mannose-BSA, GlcNAc-BSA, and GalNAc-BSA could initiate the AR while glucose-BSA and galactose-BSA had no effect. Moreover, unconjugated sugars failed to block neoglycoprotein-induced AR, suggesting that a scaffold to display the sugars is necessary. Later, Hanna et al.18 found that Lewis X (Galβ4[Fucα3]GlcNAc) and Lewis A (Galβ3[Fucα4]GlcNAc) when conjugated to BSA could initiate the AR, and LewisX-BSA was more potent than LewisA-BSA. This finding indicated that fucose may be another glycan ligand involved in AR initiation in addition to the three monosaccharides identified by Loeser et al.17 However, Hanna et al.18 did not observe AR activation with GlcNAc-BSA, in contrast to the results of Loeser et al.17 Interpretation is difficult as little is known about the spatial display of the saccharides on the BSA. Moreover, fucosyl bioconjugates were not directly studied.
Recent experiments have further challenged the proposed roles of some glycan residues in the AR and the concept of ZP3 as the AR stimulus. Oocyte-specific deletion of both the β-1,3-galactosyl transferase (T-syn) and the mannoside acetylglucosaminyltransferase 1 (Mgat1) genes generated mouse eggs lacking core-1-derived O-glycans and with modified N-glycans lacking terminal Gal and GlcNAc residues.19 These mice, in which O-linked or N-linked galactose and GlcNAc were genetically deleted from the ZP are still fertile, suggesting these two monosaccharides are genetically unessential.19 However, the knockout mice display considerably lower fecundity compared to wild-type mice, and the remaining glycans on the ZP, for example, mannose or GalNAc, may still contribute to fertilization. A model through which sperm induce acrosome exocytosis by mechanosensory signal transduction without requiring sperm binding to the ZP was also reported,20 but it is difficult to imagine how mechanical signals can be transmitted without molecular binding interactions between sperm and the egg surface. It has also been suggested that most fertilizing sperm begin acrosomal exocytosis before binding to the ZP through molecular interactions between sperm and the female reproductive tract.21,22 It appears that there are multiple interactions that lead to the AR, most likely involving carbohydrates. To what extent signaling is initiated through a single type of receptor–ligand interaction versus multiple types of interactions is poorly understood.
Although the signaling pathways initiating the AR are not completely understood, considerable progress in understanding downstream events has been made.23,24 At minimum, the AR is triggered by a cascade of signal events including activation of G-proteins, protein phosphorylation, and elevation of intracellular calcium and pH levels.25−27 However, the identity of the cell surface receptor or receptors that initiates these signals is unknown, despite the identification of several candidates.26
Controversy about the mechanism of the AR calls for a modified strategy to elucidate the molecular players in this complicated process. We hypothesized that synthetic glycopolymers will provide further insights into the molecular complexity of sperm AR activation.28 The length, substitution, and ligand density of glycopolymers can be easily controlled. Thus, we applied neoglycopolymers prepared by ring-opening metathesis polymerization (ROMP) to investigation of the AR. We further examined the glycopolymer-activated AR in the absence and presence of established pharmacological agents known to prevent the AR by blocking specific signaling pathways. Our data demonstrate that synthetic mannose, fucose, and GlcNAc polymers activate mouse acrosome reaction through convergent signaling pathways.
Results and Discussion
Design and Preparation of Glycopolymers
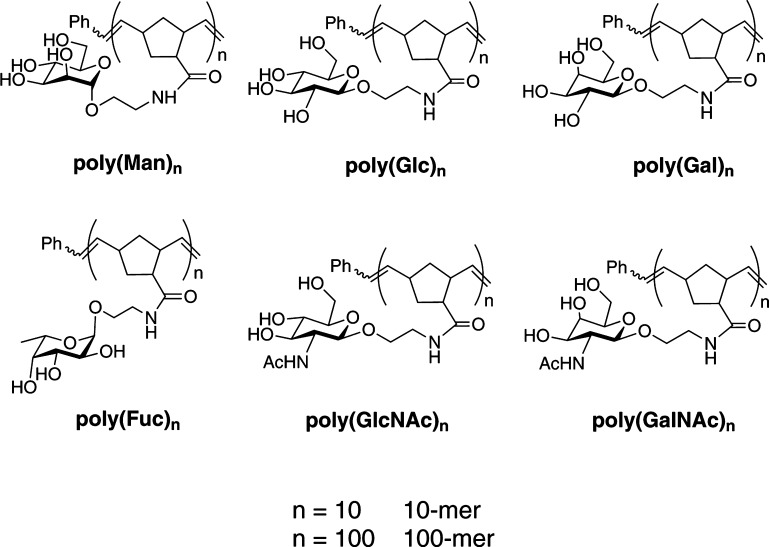
Synthetic polymeric probes provide a versatile strategy to investigate ligand–receptor interactions.29,30 Their design, which is simple and flexible, requires a multivalent scaffold, a minimal ligand, and a spacer to link the ligand to the scaffold. All of the ZP monosaccharides proposed to be involved in the AR, i.e., mannose, fucose, GlcNAc, and GalNAc, were chosen as ligands. Although Loeser et al.17 demonstrated that galactose-BSA and glucose-BSA did not induce AR and glucose is not present on the ZP,31 the properties of norbornene-derived galactose and glucose polymers were still tested to confirm these results. Previously utilized linkers are 3 or 14 atoms long; however, their structures are proprietary. In this work, we designed a simple ethyl amide linker to connect the norbornene-derived backbone and the monosaccharide ligand. Norbornene (NB) served as the scaffold in our work due to its high rigidity and widespread adoption. The desired polymers were generated by ROMP that affords polymers with defined lengths and narrow molecular mass distributions.32,33 Moreover, the density of ligands on norbornyl ROMP polymers favors clustering receptors and activating signaling transduction.34 Hence, a series of homogeneous glycopolymers with two different average lengths (10-mer and 100-mer) (Figure 2) were synthesized (Supplementary Schemes 1–7) and tested to determine the better scaffold length for activating the AR with monosaccharide ligands.
Figure 2.
Structures of glycopolymer probes.
Effect of Homoglycopolymers on Acrosome Reaction
We examined the effect of homoglycopolymers on the sperm AR by sperm immunofluorescence assay (Supplementary Figure 1). In the assay, sperm were capacitated by treatment with 0.3% bovine serum albumin (BSA), which is known to facilitate sperm capacitation by altering fatty acids and/or cholesterol on the sperm plasma membrane. Calcium ionophore A23187, a known sperm AR stimulus, was used as a positive control instead of ZP, because the role of ZP in AR activation is controversial and the calcium concentration has been demonstrated to be essential to activate the AR. Since all polymer samples were prepared in phosphate buffered saline (PBS), sperm treated with PBS was used as a negative control. Sperm samples were incubated with controls and glycopolymers at varying concentrations for 30 min and then stained with rhodamine-labeled peanut agglutinin (PNA). PNA, which has been identified to bind both the outer acrosomal membrane and certain components of the acrosomal matrix, is widely used to discriminate between acrosome-intact and acrosome-reacted sperm.35
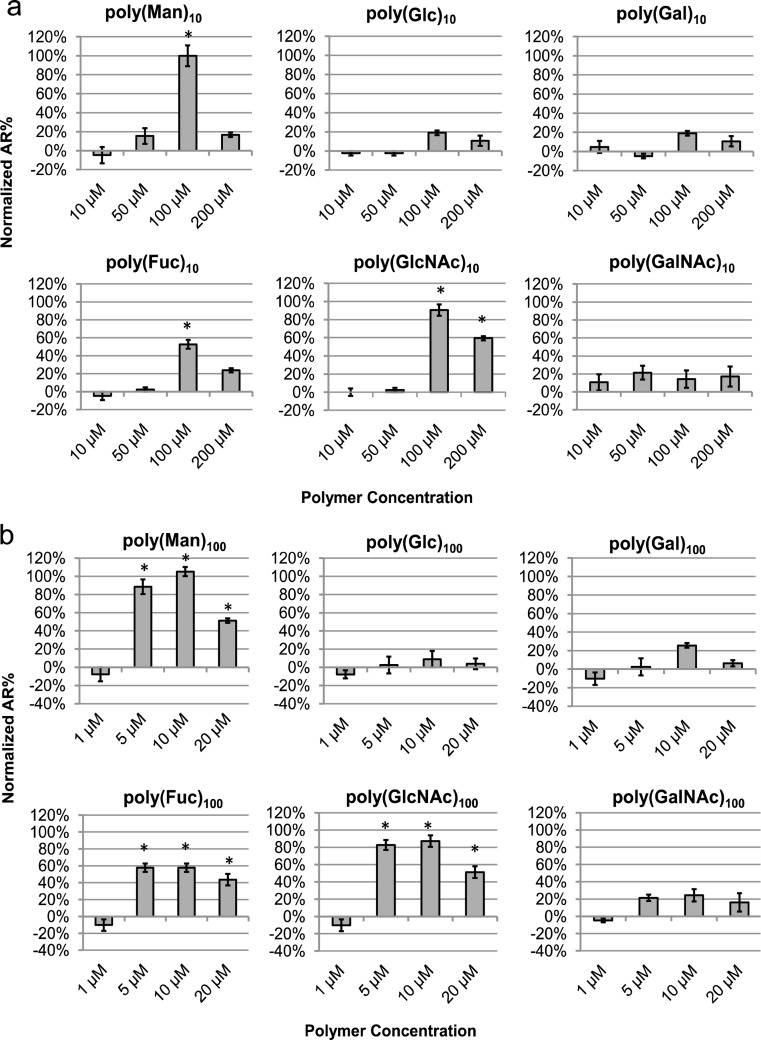
A significantly greater number of sperm undergo the AR when treated with 100 μM poly(Man)10, poly(Fuc)10, or poly(GlcNAc)10 than with the other three glycopolymers (Figure 3a). The activation of the AR by these three glycopolymers is steeply dose-dependent; at a 2-fold lower concentration, the AR is not activated. Sperm samples treated with a 2-fold higher concentration (200 μM) were AR activated with a lower efficiency or no efficacy. These data suggest that at high polymer concentrations, multivalent binding and thus the clustering effect are not favored.34 Neither poly(Glc)10, poly(Gal)10, nor poly(GalNAc)10 triggered the AR at concentrations of 100 or 200 μM. Induction of the AR by poly(Fuc)10 is not as effective as with poly(Man)10 or poly(GlcNAc)10. Similarly, dose-dependent AR initiation was observed when sperm were incubated with poly(Man)100, poly(Fuc)100, and poly(GlcNAc)100, but not poly(Glc)100, poly(Gal)100, and poly(GalNAc)100 (Figure 3b). There is no statistically significant difference between the efficacy of mannose polymers and GlcNAc polymers. Again, the AR initiation efficacy of poly(Fuc)100 is lower than for poly(Man)100 and poly(GlcNAc)100, consistent with the lowered efficacy of poly(Fuc)10. Taken together, these results indicate that galactose and GalNAc may play a role in sperm–egg binding but not in activation of the AR. Mannose, GlcNAc, and fucose function as sperm AR activators, but fucose is less efficient at AR activation. The concentrations reported here are polymer concentrations. If the bulk concentration of glycan ligand utilized is considered in comparing the efficacies of 10-mers versus 100-mers, we observe that the 100-mers are more potent. Sperm did not undergo the AR when incubated with the 10-mers at 500 μM ligand concentration (50 μM polymer) (Figure 3a), whereas 500 μM poly(Man)100, poly(Fuc)100, and poly(GlcNAc)100 (5 μM polymer) (Figure 3b) successfully initiated the AR. The higher potency of the 100-mers further confirms that the polymers stimulate the AR through a multivalent interaction with sperm.
Figure 3.
Activation of sperm acrosome reaction by homoglycopolymers. Capacitated sperm were incubated with glycopolymers at different concentrations (shown as polymer concentration). (a) 10-mers. (b) 100-mers. The average acrosome reaction percentages (AR%) of glycopolymer-treated sperm were normalized using [AR%(glycopolymers) – AR%(negative control)]/[AR%(positive control) – AR%(negative control)]. The average AR% for the positive control, A23187-treated (5 μM) sperm, was 24% and for the negative control, PBS-treated sperm, was 10%. Data represent mean ± SEM of at least three independent experiments. * p < 0.05 when compared to the negative control.
Effect of Pairs of 100-mers on AR
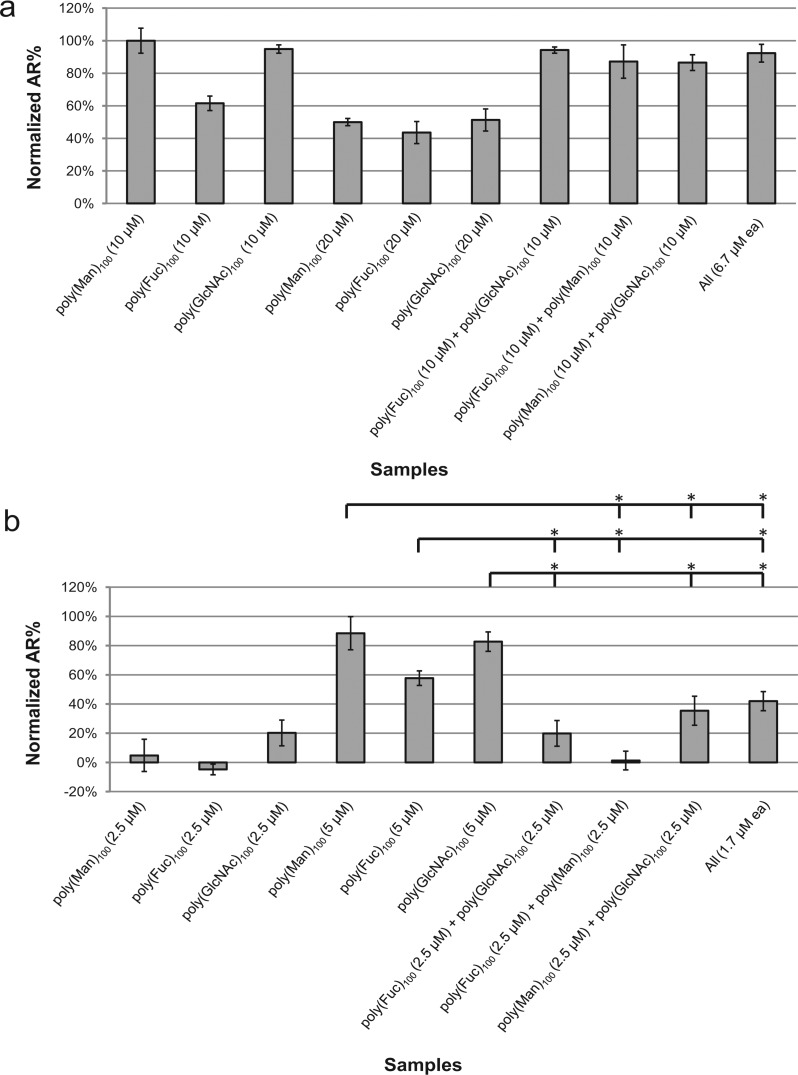
Next, we paired the active 100-mers poly(Man)100, poly(Fuc)100, and poly(GlcNAc)100 at their optimal (10 μM) and at much lower concentrations (2.5 μM) to examine the effect of inducing the AR simultaneously with two different ligands. As glucose has not been identified on ZP and poly(Glc)100 had no AR activation ability, poly(Glc)100 was used as a negative control. We saw no further increase in the amount of sperm AR comparing the polymer pairs and single glycopolymers at their optimal concentrations; the efficacy remained at 100% of the positive control (Figure 4a). Mixtures of three glycopolymers at their optimal concentrations were also tested, but no significant differences in AR percentage between a pair and a mixture of three were observed.
Figure 4.
Comparison of mixed 100-mers and the corresponding single 100-mers. (a) 100-mers paired at 10 μM each. (b) 100-mers paired at 2.5 μM each. The concentration shown in the chart is polymer concentration. The average AR% of glycopolymer-treated sperm were normalized using [AR%(glycopolymers) – AR%(negative control)]/[AR%(positive control) – AR%(negative control)]. The average AR% for the positive control, A23187-treated (5 μM) sperm, was 24% and for the negative control, poly(Glc)100-treated (10 μM) sperm, was 11%. Data represent mean ± SEM of at least three independent experiments. * p < 0.05 when compared to the corresponding single 100-mers.
Although poly(GlcNAc)100 and poly(Man)100 had similar dose-dependent AR activating patterns (Figure 3b), poly(GlcNAc)100 was more effective than poly(Man)100 at 2.5 μM (Figure 4b). Poly(GlcNAc)100 and poly(Man)100 paired at 2.5 μM each showed a slight enhancement in AR activation compared to poly(GlcNAc)100 at 2.5 μM, yet the mixture did not activate AR to the same level as 5 μM of a poly(GlcNAc)100 or poly(Man)100 (Figure 4b). This result suggests that the two carbohydrate ligands bind to different receptors on the sperm.
Sperm samples treated with the other two combinations of activating polymer (2.5 μM each) showed efficacies equal to treatment with a single polymer at 2.5 μM. In addition, the pairwise mixtures (2.5 μM each) were less effective activators of AR than a single polymer at 5 μM, which is equal to the total concentration of polymer in the paired mixture. The data suggest that there is a concentration threshold for glycopolymer-receptor binding and signal transduction and that the three sugars act independently to activate the AR.
We also used dynamic light scattering to investigate whether polymer aggregation, which could interfere with sperm activation, had occurred. No aggregation was observed (data not shown). These results together suggest that maximal sperm AR is achieved upon treatment with a single homopolymer at its optimal concentration (Figure 4a) and that the carbohydrate ligands bind to different receptors on the sperm (Figure 4b).
Kinetics of AR Induced by 100-mers
We further studied the effects of the three active 100-mers on the AR as they were more potent than the 10-mers. The time courses for the three 100-mers were monitored in parallel for 45 min (Figure 5). Data for longer incubation periods were not included due to high AR in the negative control and reduced sperm viability.
Figure 5.
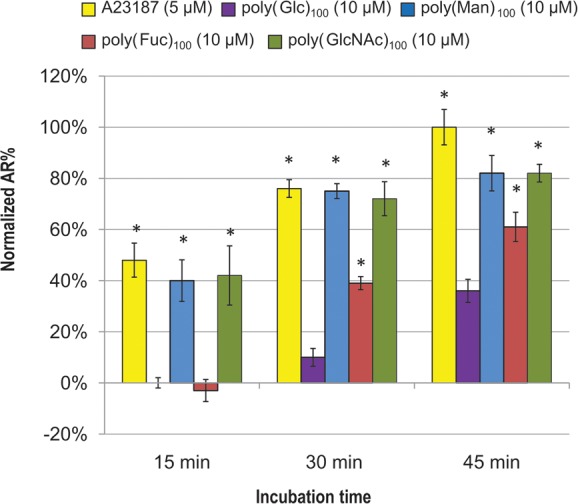
Poly(Fuc)100, poly(Man)100, and poly(GlcNAc)100 have different AR activation rates. The concentration shown in the chart is polymer concentration. The average AR% of glycopolymer-treated sperm were normalized using [AR%(glycopolymers) – AR%(negative control)]/[AR%(positive control) – AR%(negative control)]. The average AR% for the positive control, A23187-treated (5 μM) sperm at 45 min, was 33% and for the negative control, poly(Glc)100-treated (10 μM) sperm at 15 min, was 9%. Data represent mean ± SEM of at least three independent experiments. * p < 0.05 when compared to the AR% of poly(Glc)100 at each time point.
At the three time points, the extent of AR in the positive control and the poly(Man)100- and poly(GlcNAc)100-treated samples is the same. However, poly(Fuc)100 induced lower levels of the AR than poly(Man)100 and poly(GlcNAc)100 at 30 or 45 min, and there was no initiation after 15 min. These data indicate that the induction of the AR by poly(Man)100 and poly(GlcNAc)100 is more rapid than by poly(Fuc)100, which is in agreement with the previous observation that poly(Man)100 and poly(GlcNAc)100 are more effective than poly(Fuc)100. The reason for the slower AR activation kinetics of poly(Fuc)100 compared to the other two 100-mers is unclear. One possibility is that differences in glycopolymer conformations arising from substitution with different monosaccharides affect efficacy of signal transduction. Note that the AR in the negative control increased sharply at 45 min due to spontaneous AR. We selected 30 min for all of our end point assays because the level of spontaneous AR was much lower.
Signaling Pathway of Glycopolymers-Induced AR
We examined which signaling transduction events are activated by AR-active glycopolymers using well characterized inhibitors for protein kinase A (PKA), protein kinase C (PKC), protein tyrosine kinase (PTK), G-protein, T-type/L-type Ca2+ channels, and extracellular Ca2+. These signaling molecules and channels have been detected in both mouse and human sperm and have been suggested to play an important role in both the mouse and the human ZP-induced acrosome reaction.36,37 The inhibitors do not have absolute specificity; they block AR activation nonselectively at high concentrations. The doses of the inhibitors were carefully chosen in order to not affect sperm viability and motility. Conversely, inhibitors do not completely abolish the spontaneous AR.
All seven inhibitors significantly suppressed poly(Man)100-, poly(GlcNAc)100-, and poly(Fuc)100-activated AR with at least 60% inhibition (Figure 6). As the three glycopolymers all activate AR, it is not surprising to find that they share signaling pathways in common. These results demonstrate that poly(Man)100, poly(GlcNAc)100, and poly(Fuc)100 activate the AR though convergent signaling pathways. Sperm treated with inhibitors alone also show a low AR percentage, consistent with a low level of spontaneous AR that is independent of these pathways.
Figure 6.
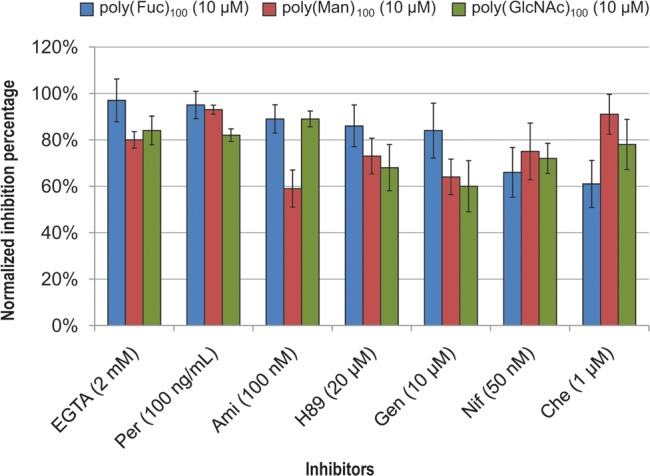
Signaling pathways of AR activation by the three effective glycopolymers are similar. EGTA: ethylene glycol tetraacetic acid, extracellular Ca2+ inhibitor. Per: pertussis toxin, G-protein inhibitor. Ami: amiloride hydrochloride, T-type Ca2+ channel inhibitor. H89: protein kinase A inhibitor. Gen: genistein, protein tyrosine kinase inhibitor. Nif: nifedipine, L-type Ca2+ channel inhibitor. Che: chelerylthrine, protein kinase C inhibitor. The average inhibition percentage of inhibitor and glycopolymer treated sperm were normalized using [AR%(positive control) – AR%(glycopolymers)] /[AR%(positive control) – AR%(negative control)]. The average AR% for the positive control, A23187-treated (5 μM) sperm, was 24%, and for the negative control, inhibitor-treated sperm, was 9–13%. Data represent mean ± SEM of three independent experiments.
The precise sperm AR signaling pathways are not completely elucidated, though several tentative signaling pathway mechanisms of ZP-initiated AR have been proposed.26,38,39 In these mechanisms, ZP is thought to bind to at least two receptors on the sperm membrane. One is a G-protein coupled receptor, which is reported to regulate adenylyl cyclase and activate PKA, phospholipase C (PLC) β1, and H+ efflux. Upon activation, PKA phosphorylates and further triggers downstream factors. The other is a PTK receptor, which is suggested to trigger a sperm Na+/H+ exchanger promoting cell alkalinization, membrane depolarization, and T-type and L-type calcium channels activation on the sperm plasma membrane. The calcium channels play vital roles in elevating intracellular Ca2+ and pH preceding the AR. G-protein and PTK can also activate PKC, which mediates calcium entry into the sperm cytosol from intracellular stores. These signaling factors all lead to an increase in cytosolic calcium, resulting in the fusion of sperm plasma membrane and the outer acrosomal membrane and eventually the AR.
All of the above-described signaling factors are involved in activation of the AR by poly(Man)100, poly(GlcNAc)100, and poly(Fuc)100. Moreover, these homopolymers activate through the same pathways as mouse ZP (Table 1). Our data strongly support that glycopolymer-activated AR is analogous to ZP-activated AR and that these glycopolymers are suitable mimics of the ZP and/or other physiologic ligands for activating sperm AR. Our work emphasizes the high redundancy of carbohydrate ligands that can be used to activate the AR. In contrast to conditional genetic knockouts for which no AR phenotypes were observed, the use of glycopolymers has enabled the identification of which terminal carbohydrates are important for the AR. The glycopolymer chemotypes observed in this work suggest that at least three different sperm receptor binding sites can be utilized to initiate the AR in mouse. After receptor activation by glycopolymer, signaling converges onto the same pathways intracellularly.
Table 1. Comparison of Signaling Pathways Initiated by Different Activators.
| signaling molecule |
|||||||
|---|---|---|---|---|---|---|---|
| AR activator | extracellular Ca2+ | T-type Ca2+ | L-type Ca2+ | PKC | PKA | PTK | G protein |
| BSA-mannose/GalNAc/GlcNAca | –b | – | +c | – | – | – | – |
| BSA-Lewis Xd | + | + | NAe | NA | NA | NA | – |
| BSA-Lewis Af | + | + | NA | NA | NA | NA | – |
| Mouses ZPg | + | + | + | + | + | + | + |
| poly(Man)100 | + | + | + | + | + | + | + |
| poly(GlcNAc)100 | + | + | + | + | + | + | + |
| poly(Fuc)100 | + | + | + | + | + | + | + |
The receptors activated by poly(Man)100, poly(GlcNAc)100, and poly(Fuc)100 have not been definitively identified. None of the large number of egg binding receptors proposed and characterized has been demonstrated to be essential.40−42 Previous results also suggested a high level of redundancy, but whether multiple egg binding receptors acted individually or as a multiprotein complex was unclear. Our results favor the single ligand–receptor interaction model and provide further evidence that induction of the sperm acrosome reaction proceeds through duplicative sperm–egg interactions.
Conclusion
A multivalent display of mannose, GlcNAc, or fucose triggers the sperm acrosome reaction in a concentration-dependent manner, and high valency polymers (100 ligands) are more effective than low valency polymers (10 ligands). Although fucose showed lower AR activation potency and the kinetics of fucose-induced AR are distinct from those of mannose or GlcNAc-induced AR, all of the glycopolymers rely on G-proteins, protein kinase C, protein kinase A, extracellular Ca2+, L- and T-type Ca2+ channels, and a protein tyrosine kinase. The three single ligand–receptor interactions all induce the acrosome reaction and are functionally equivalent but are redundant. Thus, the chemotypes of ROMP-derived glycopolymers mimic the biological function of physiologic AR-activation agents and provide evidence that occupation of one of at least three different receptor binding sites is sufficient to initiate the AR.
Methods
General Methods and Materials
All experiments performed with mice were in accordance with the National Institute of Health and United States Department of Agriculture guidelines, and the specific procedures performed were approved by the Stony Brook University IACUC (protocol 0616). Chemicals for assay buffers were purchased from Sigma-Aldrich, Fisher Scientific, and VWR.
Sperm Treatment
Sperm were isolated from the cauda epididymis of two 10- to 12-week-old ICR male breeders (Taconic) in M16 medium (6 mL) supplemented with 0.3% BSA (w/v). The sperm suspension was then gently pipetted into a polypropylene culture tube (12 mm × 75 mm) and incubated at 37 °C for 30 min under 5% CO2 (v/v) in air. Once the incubation was complete, the sperm motility was examined by phase-contrast microscopy (20×/0.5 air). Only samples of capacitated sperm displaying >80% motility were used in subsequent experiments. The concentration of sperm was assessed by hemocytometer. Aliquots (20 μL) containing about 5 × 105 capacitated sperm were transferred to microcentrifuge tubes and incubated with glycopolymers, negative, and positive controls at 37 °C under 5% CO2 for the specified time (15, 30, or 45 min). The positive control used in our assay is the well-studied Ca2+ transporting ionophore A23187 (Sigma-Aldrich). After incubation, the sperm were pelleted by centrifugation at 500g for 6 min. The supernatant was removed, and the pelleted sperm were washed once with 40 μL of PBS and fixed with 40 μL of 70% ethanol. After fixing at 4 °C for 30 min, the sperm were pelleted and washed twice with PBS. The final pellet was resuspended in 40 μL of DDI water. Aliquots (10 μL) of each sample were transferred to coverslips and air-dried.
Assessment of Sperm Acrosome Reaction
Ten microliters of rhodamine-labeled peanut agglutinin (PNA) (Vector laboratories) at a concentration of 20 μg mL–1 was incubated with fixed sperm (from which polymers had been removed by extensive washing) on coverslips for 10 min at rt. After washing with 2 mL of DDI water (two × 10 min), the coverslips were mounted on SuperFrost Plus microscope slides (Fisher Scientific) over a drop (6 μL) of mounting medium Vectashield (Vector laboratories) and sealed with nail polish, and the acrosomal status was assessed by inverse fluorescence microscopy (Zeiss). Sperm that displayed continuous red fluorescence along their acrosomal arcs were scored as acrosome-intact; those that displayed no red or punctuate fluorescence were scored as acrosome-reacted. The slides were coded and counted blindly; all experiments were conducted at least three times. Each time, three independent replicates of each test group were analyzed, and 200 sperm from each replicate were counted.
Signaling Pathway Inhibition Assay
Inhibitor stock solutions were prepared by dissolving the reagents either in distilled water (pertussis toxin) or in DMSO. Aliquots of the stock solutions were mixed with the capacitated sperm solution to achieve the desired concentration and preincubated for 5 min before treatment with polymers or A23187. The concentrations of inhibitors were chosen on the basis of references36,37 and toxicity tests. The acrosomal status of sperm was evaluated as described above.
Statistical Analysis
Comparisons of the average values for the control and experimental groups were carried out by a paired two-tailed t-test to determine statistically significant differences (p < 0.05). The results are presented as mean ± SEM.
Acknowledgments
We thank Brookhaven Instruments (Brookhaven, NY) for the dynamic light scattering software. Funding was providing by the National Institutes of Health (R01GM097971/R01HD38519 to N.S.S.)
Glossary
Abbreviations
- AR
acrosome reaction
- ZP
zona pellucida
- ZP3
zona pelucida protein 3
- NB
norbornene
- GalNAc
β-N-acetylgalactosamine
- GlcNAc
β-N-acetylglucosamine
- Man
mannose
- Glc
glucose
- Gal
galactose
- Fuc
fucose
- BSA
bovine serum albumin
- PKA
protein kinase A
- PKC
protein kinase C
- PTK
protein tyrosine kinase
Supporting Information Available
Schemes of all syntheses and experimental procedures. This material is available free of charge via the Internet at http://pubs.acs.org.
The authors declare no competing financial interest.
Funding Statement
National Institutes of Health, United States
Supplementary Material
References
- Wassarman P. M.; Jovine L.; Litscher E. S. (2001) A profile of fertilization in mammals. Nat. Cell Biol. 3, 59–64. [DOI] [PubMed] [Google Scholar]
- Wassarman P. M. (1999) Mammalian fertilization: Molecular aspects of gamete adhesion, exocytosis, and fusion. Cell 96, 175–183. [DOI] [PubMed] [Google Scholar]
- Benoff S. (1997) Carbohydrates and fertilization: An overview. Mol. Hum. Reprod. 3, 599–637. [DOI] [PubMed] [Google Scholar]
- Clark G. F.; Dell A. (2006) Molecular models for murine sperm-egg binding. J. Biol. Chem. 281, 13853–13856. [DOI] [PubMed] [Google Scholar]
- Bedford J. M (1977) Sperm/egg interaction: The specificity of human spermatozoa. Anat. Rec. 188, 477–487. [DOI] [PubMed] [Google Scholar]
- Miller D. J.; Gong X.; Shur D. B. (1993) Sperm require β-n-acetylglucosaminidase to penetrate through the egg zona pellucida. Development 118, 1279–1289. [DOI] [PubMed] [Google Scholar]
- Nagdas S. K.; Araki Y.; Chayko C. A.; Orgebin-Crist M. C.; Tulsiani D. R. (1994) O-linked trisaccharide and N-linked poly-N-acetyllactosaminyl glycans are present on mouse ZP2 and ZP3. Biol. Reprod. 51, 262–272. [DOI] [PubMed] [Google Scholar]
- Cornwell G. A.; Tulsiani D. R. P. (1991) Inhibition of the mouse sperm surface alpha-D-mannosidase inhibits sperm-egg binding in vitro. Biol. Reprod. 44, 913–921. [DOI] [PubMed] [Google Scholar]
- Dell A.; Chalabi S.; Easton R. L.; Haslam S. M.; Sutton-Smith M.; Patankar M. S.; Lattanzio F.; Panico M.; Morris H. R.; Clark G. F. (2003) Murine and human zona pellucida 3 derived from mouse eggs express identical O-glycans. Proc. Natl. Acad. Sci. U.S.A. 100, 15631–15636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambert H. (1984) Role of sperm-surface glycoproteins in gamete recognition in two mouse species. J. Reprod. Fertil. 70, 281–284. [DOI] [PubMed] [Google Scholar]
- Pang P.-C.; Chiu P. C. N.; Lee C.-L.; Chang L.-Y.; Panico M.; Morris H. R.; Haslam S. M.; Khoo K.-H.; Clark G. F.; Yeung W. S. B.; Dell A. (2011) Human sperm binding is mediated by the sialyl-LewisX oligosaccharide on the zona pellucida. Science 333, 1761–1764. [DOI] [PubMed] [Google Scholar]
- Johnston D. S.; Wright W. W.; Shaper J. H. (1998) Murine sperm-zona binding, a fucosyl residue is required for a high affinity sperm-binding ligand. J. Biol. Chem. 273, 1888–1895. [DOI] [PubMed] [Google Scholar]
- Kim K.-S.; Gerton G. L. (2003) Differential release of soluble and matrix components: Evidence for intermediate states of secretion during spontaneous acrosomal exocytosis in mouse sperm. Dev. Biol. 264, 141–152. [DOI] [PubMed] [Google Scholar]
- Wassarman P. M.; Jovine L.; Qi H.; Williams Z.; Darie C.; Litscher E. S. (2004) Recent aspects of mammalian fertilization research. Mol. Cell. Endocrinol. 234, 95–103. [DOI] [PubMed] [Google Scholar]
- Tulsiani D. R. P. (2000) Carbohydrates mediate sperm-ovum adhesion and triggering of the acrosome reaction. Asian J. Androl. 2, 87–97. [PubMed] [Google Scholar]
- Wassarman P. M. (2005) Contribution of mouse egg zona pellucida glycoproteins to gamete recognition during fertilization. J. Cell. Physiol. 204, 388–391. [DOI] [PubMed] [Google Scholar]
- Loeser C. R.; Tulsiani D. R. P. (1999) The role of carbohydrates in the induction of the acrosome reaction in mouse spermatozoa. Biol. Reprod. 60, 94–101. [DOI] [PubMed] [Google Scholar]
- Hanna W. F.; Kerr C. L.; Shaper J. H.; Wright W. W. (2004) Lewis X-containing neoglycoproteins mimic the intrinsic ability of zona pellucida glycoprotein ZP3 to induce the acrosome reaction in capacitated mouse sperm. Biol. Reprod. 71, 778–789. [DOI] [PubMed] [Google Scholar]
- Williams S. A.; Xia L.; Cummings R. D.; McEver R. P.; Stanley P. (2007) Fertilization in mouse does not require terminal galactose or N-acetylglucosamine on the zona pellucida glycans. J. Cell Sci. 120, 1341–1349. [DOI] [PubMed] [Google Scholar]
- Baibakov B.; Gauthier L.; Talbot P.; Rankin T. L.; Dean J. (2007) Sperm binding to the zona pellucida is not sufficient to induce acrosome exocytosis. Development 134, 933–943. [DOI] [PubMed] [Google Scholar]
- Jin M.; Fujiwara E.; Kakiuchi Y.; Okabe M.; Satouh Y.; Baba S. A.; Chiba K.; Hirohashi N. (2011) Most fertilizing mouse spermatozoa begin their acrosome reaction before contact with the zona pellucida during in vitro fertilization. Proc. Natl. Acad. Sci. U.S.A. 108, 4892–4896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagimachi R. (2011) Mammalian sperm acrosome reaction: Where does it begin before fertilization. Biol. Reprod. 85, 4–5. [DOI] [PubMed] [Google Scholar]
- Avella M. A.; Dean J. (2011) Fertilization with acrosome-reacted mouse sperm: Implications for the site of exocytosis. Proc. Natl. Acad. Sci. U.S.A. 108, 19843–19844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buffone M. G.; Ijiri T. W.; Cao W.; Merdiushev T.; Agajanian H. K.; Gerton G. L. (2012) Heads or tails? Structural events and molecular mechanisms that promote mammalian sperm acrosomal exocytosis and motility. Mol. Reprod. Dev. 79, 4–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tulsiani D. R. P.; Abou-Haila A.; Loeser C. R.; Pereira B. M. J. (1998) The biological and functional significance of the sperm acrosome and acrosomal enzymes in mammalian fertilization. Exp. Cell Res. 240, 151–164. [DOI] [PubMed] [Google Scholar]
- Breitbart H.; Spungin B. (1997) The biochemistry of the acrosome reaction. Mol. Hum. Reprod. 3, 195–202. [DOI] [PubMed] [Google Scholar]
- Ickowicz D.; Finkelstein M.; Breitbart H. (2012) Mechanism of sperm capacitation and the acrosome reaction: Role of protein kinases. Asian J. Androl. 14, 816–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becer C. R. (2012) The glycopolymer code: Synthesis of glycopolymers and multivalent carbohydrate–lectin interactions. Macromol. Rapid Commun. 33, 742–752. [DOI] [PubMed] [Google Scholar]
- Baessler K. A.; Lee Y. J.; Sampson N. S. (2009) Beta1 integrin is an adhesion protein for sperm binding to eggs. ACS Chem. Biol. 4, 357–366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney A. H.; Puffer E. B.; Pontrello J. K.; Yang Z. Q.; Kiessling L. L. (2009) Sialylated multivalent antigens engage CD22 in trans and inhibit B cell activation. Proc. Natl. Acad. Sci. U.S.A. 106, 2500–2505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Easton R. L.; Patankar M. S.; Lattanzio F. A.; Leaven T. H.; Morris H. R.; Clark G. F.; Dell A. (2000) Structural analysis of murine zona pellucida glycans. Evidence for the expression of core 2-type O-glycans and the Sd(a) antigen. J. Biol. Chem. 275, 7731–7742. [DOI] [PubMed] [Google Scholar]
- Lee Y.; Sampson N. S. (2006) Romping the cellular landscape: Linear scaffolds for molecular recognition. Curr. Opin. Struct. Biol. 16, 544–550. [DOI] [PubMed] [Google Scholar]
- Bielawski C. W.; Grubbs R. H. (2007) Living ring-opening metathesis polymerization. Prog. Polym. Sci. 32, 1–29. [Google Scholar]
- Gestwicki J. E.; Cairo C. W.; Strong L. E.; Oetjen K. A.; Kiessling L. L. (2002) Influencing receptor-ligand binding mechanisms with multivalent ligand architecture. J. Am. Chem. Soc. 124, 14922–14933. [DOI] [PubMed] [Google Scholar]
- Love J. A.; Morgan J. P.; Trnka T. M.; Grubbs R. H. (2002) A practical and highly active ruthenium-based catalyst that effects the cross metathesis of acrylonitrile. Angew. Chem., Int. Ed. 41, 4035–4037. [DOI] [PubMed] [Google Scholar]
- Chiu P. C. N.; Wong B. S. T.; Chung M.-K.; Lam K. K. W.; Pang R. T. K.; Lee K.-F.; Sumitro S. B.; Gupta S. K.; Yeung W. S. B. (2008) Effects of native human zona pellucida glycoproteins 3 and 4 on acrosome reaction and zona pellucida binding of human spermatozoa. Biol. Reprod. 79, 869–877. [DOI] [PubMed] [Google Scholar]
- Loeser C. R.; Lynch C.; Tulsiani D. R. P. (1999) Characterization of the pharmacological-sensitivity profile of neoglycoprotein induced acrosome reaction in mouse spermatozoa. Biol. Reprod. 61, 629–634. [DOI] [PubMed] [Google Scholar]
- Gupta S. K.; Bhandari B. (2011) Acrosome reaction: Relevance of zona pellucida glycoproteins. Asian J. Androl. 13, 97–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tulsiani D. R. P. (2012) Mechanisms of mammalian sperm-egg interaction leading to fertilization, Gynecol. Obstet. 2, doi: 10.4172/2161-0932.1000e4107. [DOI] [Google Scholar]
- Lu Q.; Shur B. D. (1997) Sperm from beta 1,4-galactosyltransferase-null mice are refractory to ZP3-induced acrosome reactions and penetrate the zona pellucida poorly. Development 124, 4121–4131. [DOI] [PubMed] [Google Scholar]
- Tsai J.-Y.; Silver L. M. (1996) Sperm-egg binding protein or proto-oncogene?. Science 271, 1432–1434. [DOI] [PubMed] [Google Scholar]
- Muro Y.; Buffone M. G.; Okabe M.; Gerton G. L. (2012) Function of the acrosomal matrix: Zona pellucida 3 receptor (ZP3R/sp56) is not essential for mouse fertilization. Biol. Reprod. 86, 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.