Abstract
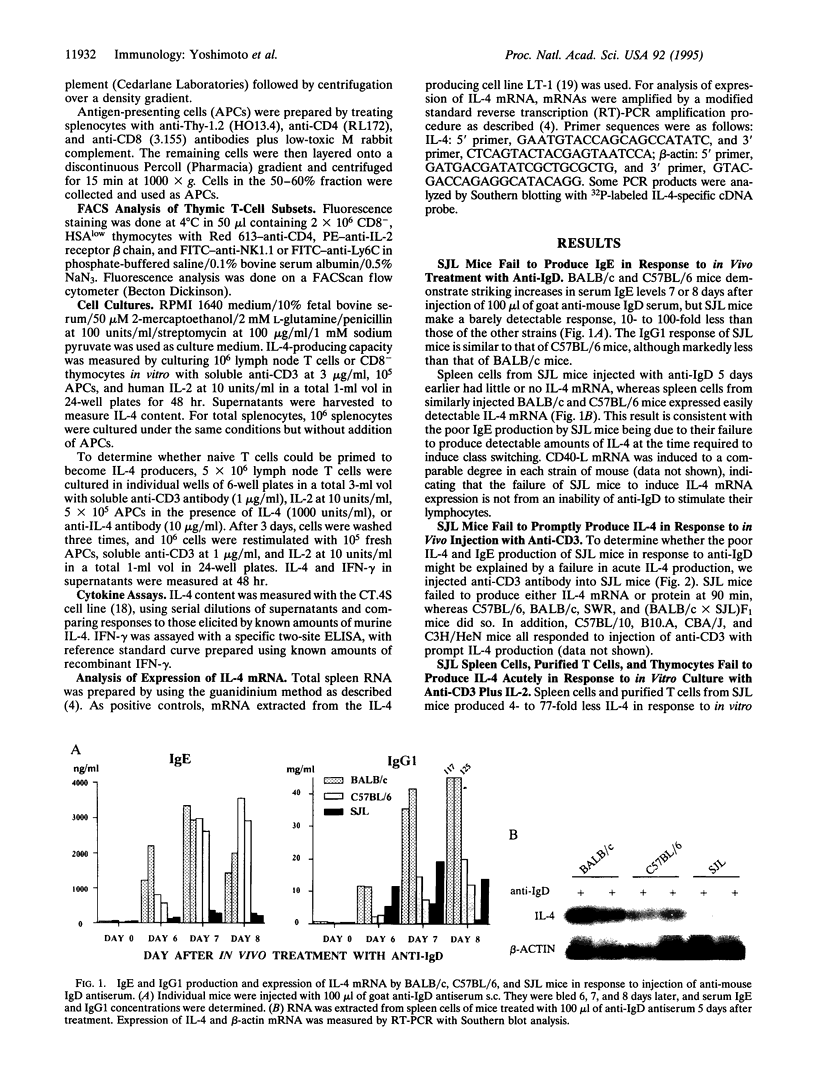
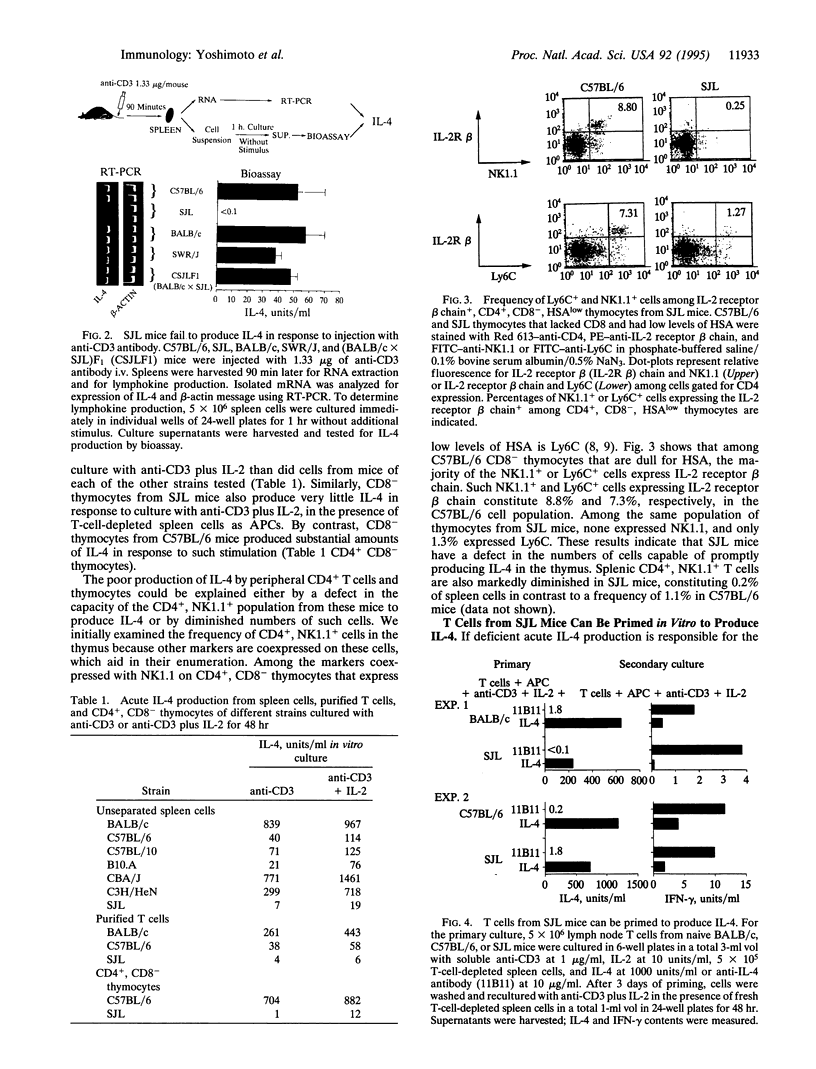
SJL mice produce little or no IgE in response to polyclonal stimulation with anti-IgD antibody and fail to express interleukin 4 (IL-4) mRNA in the spleen 5 days after injection of anti-IgD, in contrast to other mouse strains that produce substantial amounts of IgE and IL-4. Because IL-4 is critical in IgE production, the possibility that SJL mice are poor IgE producers because their naive T cells fail to differentiate into IL-4 producers must be seriously considered. IL-4 itself is the principal factor determining that naive T cells develop into IL-4 producers. A major source of IL-4 for such differentiation is a population of CD1-specific CD4+ T cells that express NK1.1. These cells produce IL-4 within 90 min of anti-CD3 injection. T cells from SJL mice fail to produce IL-4 in response to injection of anti-CD3. Similarly, SJL T cells and CD4+ thymocytes do not produce IL-4 in response to acute in vitro stimulation. SJL T cells show a marked deficiency in CD4+ cells that express the surface receptors associated with the NK1.1+ T-cell phenotype. This result indicates that the SJL defect in IgE and IL-4 production is associated with, and may be due to, the absence of the CD4+, NK1.1+ T-cell population.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arase H., Arase N., Nakagawa K., Good R. A., Onoé K. NK1.1+ CD4+ CD8- thymocytes with specific lymphokine secretion. Eur J Immunol. 1993 Jan;23(1):307–310. doi: 10.1002/eji.1830230151. [DOI] [PubMed] [Google Scholar]
- Behlke M. A., Henkel T. J., Anderson S. J., Lan N. C., Hood L., Braciale V. L., Braciale T. J., Loh D. Y. Expression of a murine polyclonal T cell receptor marker correlates with the use of specific members of the V beta 8 gene segment subfamily. J Exp Med. 1987 Jan 1;165(1):257–262. doi: 10.1084/jem.165.1.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bendelac A., Killeen N., Littman D. R., Schwartz R. H. A subset of CD4+ thymocytes selected by MHC class I molecules. Science. 1994 Mar 25;263(5154):1774–1778. doi: 10.1126/science.7907820. [DOI] [PubMed] [Google Scholar]
- Bendelac A., Lantz O., Quimby M. E., Yewdell J. W., Bennink J. R., Brutkiewicz R. R. CD1 recognition by mouse NK1+ T lymphocytes. Science. 1995 May 12;268(5212):863–865. doi: 10.1126/science.7538697. [DOI] [PubMed] [Google Scholar]
- Bendelac A., Matzinger P., Seder R. A., Paul W. E., Schwartz R. H. Activation events during thymic selection. J Exp Med. 1992 Mar 1;175(3):731–742. doi: 10.1084/jem.175.3.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bendelac A., Schwartz R. H. CD4+ and CD8+ T cells acquire specific lymphokine secretion potentials during thymic maturation. Nature. 1991 Sep 5;353(6339):68–71. doi: 10.1038/353068a0. [DOI] [PubMed] [Google Scholar]
- Bradbury A., Calabi F., Milstein C. Expression of CD1 in the mouse thymus. Eur J Immunol. 1990 Aug;20(8):1831–1836. doi: 10.1002/eji.1830200830. [DOI] [PubMed] [Google Scholar]
- Coffman R. L., Carty J. A T cell activity that enhances polyclonal IgE production and its inhibition by interferon-gamma. J Immunol. 1986 Feb 1;136(3):949–954. [PubMed] [Google Scholar]
- Coffman R. L., Ohara J., Bond M. W., Carty J., Zlotnik A., Paul W. E. B cell stimulatory factor-1 enhances the IgE response of lipopolysaccharide-activated B cells. J Immunol. 1986 Jun 15;136(12):4538–4541. [PubMed] [Google Scholar]
- Epstein R., Sham G., Womack J., Yagüe J., Palmer E., Cohn M. The cytotoxic T cell response to the male-specific histocompatibility antigen (H-Y) is controlled by two dominant immune response genes, one in the MHC, the other in the Tar alpha-locus. J Exp Med. 1986 Apr 1;163(4):759–773. doi: 10.1084/jem.163.4.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelman F. D., Katona I. M., Urban J. F., Jr, Snapper C. M., Ohara J., Paul W. E. Suppression of in vivo polyclonal IgE responses by monoclonal antibody to the lymphokine B-cell stimulatory factor 1. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9675–9678. doi: 10.1073/pnas.83.24.9675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayakawa K., Lin B. T., Hardy R. R. Murine thymic CD4+ T cell subsets: a subset (Thy0) that secretes diverse cytokines and overexpresses the V beta 8 T cell receptor gene family. J Exp Med. 1992 Jul 1;176(1):269–274. doi: 10.1084/jem.176.1.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu-Li J., Ohara J., Watson C., Tsang W., Paul W. E. Derivation of a T cell line that is highly responsive to IL-4 and IL-2 (CT.4R) and of an IL-2 hyporesponsive mutant of that line (CT.4S). J Immunol. 1989 Feb 1;142(3):800–807. [PubMed] [Google Scholar]
- Kaminsky S. G., Nakamura I., Cudkowicz G. Genetic control of the natural killer cell activity in SJL and other strains of mice. J Immunol. 1985 Jul;135(1):665–671. [PubMed] [Google Scholar]
- Kanagawa O. Antibody-mediated activation of T cell clones as a method for screening hybridomas producing antibodies to the T cell receptor. J Immunol Methods. 1988 Jun 13;110(2):169–178. doi: 10.1016/0022-1759(88)90100-7. [DOI] [PubMed] [Google Scholar]
- Lantz O., Bendelac A. An invariant T cell receptor alpha chain is used by a unique subset of major histocompatibility complex class I-specific CD4+ and CD4-8- T cells in mice and humans. J Exp Med. 1994 Sep 1;180(3):1097–1106. doi: 10.1084/jem.180.3.1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Gros G., Ben-Sasson S. Z., Seder R., Finkelman F. D., Paul W. E. Generation of interleukin 4 (IL-4)-producing cells in vivo and in vitro: IL-2 and IL-4 are required for in vitro generation of IL-4-producing cells. J Exp Med. 1990 Sep 1;172(3):921–929. doi: 10.1084/jem.172.3.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lebrun P., Spiegelberg H. L. Concomitant immunoglobulin E and immunoglobulin G1 formation in Nippostrongylus brasiliensis-infected mice. J Immunol. 1987 Sep 1;139(5):1459–1465. [PubMed] [Google Scholar]
- Levine S., Sowinski R. Experimental allergic encephalomyelitis in inbred and outbred mice. J Immunol. 1973 Jan;110(1):139–143. [PubMed] [Google Scholar]
- Okudaira H., Mori A., Akiyoshi K., Yamamoto K., Suko M., Watanabe N., Ito M., Takahashi K., Juji T. Interleukin-4 gene expression and IgE responsiveness. Int Arch Allergy Appl Immunol. 1991;94(1-4):184–186. doi: 10.1159/000235358. [DOI] [PubMed] [Google Scholar]
- Paul W. E., Seder R. A. Lymphocyte responses and cytokines. Cell. 1994 Jan 28;76(2):241–251. doi: 10.1016/0092-8674(94)90332-8. [DOI] [PubMed] [Google Scholar]
- Racke M. K., Bonomo A., Scott D. E., Cannella B., Levine A., Raine C. S., Shevach E. M., Röcken M. Cytokine-induced immune deviation as a therapy for inflammatory autoimmune disease. J Exp Med. 1994 Nov 1;180(5):1961–1966. doi: 10.1084/jem.180.5.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snapper C. M., Paul W. E. Interferon-gamma and B cell stimulatory factor-1 reciprocally regulate Ig isotype production. Science. 1987 May 22;236(4804):944–947. doi: 10.1126/science.3107127. [DOI] [PubMed] [Google Scholar]
- Subba Rao P. V., McCartney-Francis N. L., Metcalfe D. D. An avidin--biotin microELISA for rapid measurement of total and allergen-specific human IgE. J Immunol Methods. 1983 Feb 25;57(1-3):71–85. doi: 10.1016/0022-1759(83)90066-2. [DOI] [PubMed] [Google Scholar]
- Swain S. L., Weinberg A. D., English M., Huston G. IL-4 directs the development of Th2-like helper effectors. J Immunol. 1990 Dec 1;145(11):3796–3806. [PubMed] [Google Scholar]
- Tepper R. I., Coffman R. L., Leder P. An eosinophil-dependent mechanism for the antitumor effect of interleukin-4. Science. 1992 Jul 24;257(5069):548–551. doi: 10.1126/science.1636093. [DOI] [PubMed] [Google Scholar]
- Watanabe N., Kojima S., Ovary Z. Suppression of IgE antibody production in SJL mice. I. Nonspecific suppressor T cells. J Exp Med. 1976 Apr 1;143(4):833–845. doi: 10.1084/jem.143.4.833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimoto T., Paul W. E. CD4pos, NK1.1pos T cells promptly produce interleukin 4 in response to in vivo challenge with anti-CD3. J Exp Med. 1994 Apr 1;179(4):1285–1295. doi: 10.1084/jem.179.4.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]