Abstract
Racial disparities in clinic attendance may contribute to racial disparities in plasma human immunodeficiency virus type 1 (HIV-1) RNA levels among HIV-positive patients in care. Data from 946 African American and 535 Caucasian patients receiving HIV care at the University of North Carolina Center for AIDS Research HIV clinic between January 1, 1999, and August 1, 2012, were used to estimate the association between African American race and HIV virological suppression (i.e., undetectable HIV-1 RNA) when racial disparities in clinic attendance were lessened. Clinic attendance was measured as the proportion of scheduled clinic appointments attended (i.e., visit adherence) or the proportion of six 4-month intervals with at least 1 attended scheduled clinic appointment (i.e., visit constancy). In analyses accounting for patient characteristics, the risk ratio for achieving suppression when comparing African Americans with Caucasians was 0.91 (95% confidence interval: 0.85, 0.98). Lessening disparities in adherence or constancy lowered disparities in virological suppression by up to 44.4% and 11.1%, respectively. Interventions that lessen disparities in adherence may be more effective in eliminating disparities in suppression than interventions that lessen disparities in constancy. Given that gaps in care were limited to be no more than 2 years for both attendance measures, the impact of lessening disparities in adherence may be overstated.
Keywords: clinic visits, cohort studies, health status disparities, human immunodeficiency virus, viral load
Human immunodeficiency virus (HIV)–positive African Americans in care are more likely than Caucasians to miss scheduled clinic appointments (1–4) and less likely than Caucasians to suppress plasma human immunodeficiency virus type 1 (HIV-1) RNA levels while on antiretroviral therapy (ART) (4, 5). Contextual and behavioral factors likely contribute to less frequent scheduled clinic attendance among African Americans (4). Missing scheduled clinic appointments may inhibit the receipt and timely modification of, as well as adherence to, ART among African Americans. Using data from the University of Alabama at Birmingham (UAB) 1917 Clinic Cohort, Mugavero et al. (4) demonstrated that the proportion of scheduled appointments that were missed partially explained observed racial disparities in virological response (i.e., plasma HIV-1 RNA level).
Mugavero et al. (6), however, later noted that, as a measure of clinic attendance and in turn engagement in care, the proportion of scheduled visits that were missed is limited among patients who have gaps in care and therefore do not have scheduled appointments to miss. Visit constancy, which reflects how regularly a patient receives care, is an alternative measure of engagement for etiological research that better captures clinic attendance among those with gaps in care. Visit constancy has also been shown to be associated with achieving an undetectable HIV-1 RNA level (i.e., virological suppression) (7).
Even in the absence of gaps in care, prior work (4) may underestimate the extent to which the association between race and virological response remains when disparities in clinic attendance are lessened or induce selection bias in estimating this extent by conditioning on common causes of clinic attendance and virological response that are likely influenced by race. Selection bias could be induced in the presence of an unmeasured factor that determines the measured common causes and virological response. Such underestimation or bias can be minimized with the use of inverse probability weighting (8, 9). Here, we use inverse probability weighted models, as well as data from 946 African American and 535 Caucasian patients in the University of North Carolina (UNC) Center for AIDS Research (CFAR) HIV clinical cohort to estimate the association between African American race and virological suppression when racial disparities in scheduled appointment adherence and visit constancy are lessened. To be consistent with the causal mediation analysis literature, we henceforth refer to this association as the direct effect or the controlled direct effect (8, 10, 11).
METHODS
Study population
The UNC CFAR HIV clinical cohort (henceforth, the cohort) collects sociodemographic, psychosocial, and clinical information from all HIV-positive patients who provided written informed consent in English or Spanish and received care at the UNC HIV clinic. All study forms were approved by the UNC institutional review board. Additional details concerning this clinic cohort are provided elsewhere (12).
As shown in Figure 1, data were ascertained on 3,275 enrolled patients with a clinic appointment (i.e., scheduled or walk-in) in the UNC HIV clinic during the study period, January 1, 1999, through August 1, 2012. The first clinic visit was defined as the first attended appointment (i.e., scheduled or walk-in) that occurred within the study period. Among these 3,275 patients, 43 were excluded because they did not have a first clinic visit, and another 114 were excluded because they did not have an “attended” or “no show” scheduled clinic appointment in the UNC HIV clinic within 2 years of their first clinic visit dates. Of the remaining 3,118 patients, 314 were excluded because they were not African American or Caucasian or their race was unknown. Among the remaining 2,804 patients, the following were also excluded: 118 patients (4%) because of the absence of information on 1 or more characteristics at the first clinic visit; 177 patients (7%) because of reaching August 1, 2012, prior to 2 years after their first clinic visits; 168 patients (6%) because of dying prior to 2 years after their first clinic visits; and 860 patients (37%) because of the absence of any RNA assessments in the window of 2–3 years after their first clinic visits. As described below, measured differences between the 1,481 included patients (53% of the 2,804) and 860 of the excluded patients (31% of the 2,804) were accounted for by using inverse probability weights (13). Inverse probability weights were not used to account for differences between those included and the other 463 excluded patients (16% of the 2,804) because of the absence of information necessary to estimate inverse probability weights or a small percent excluded.
Figure 1.
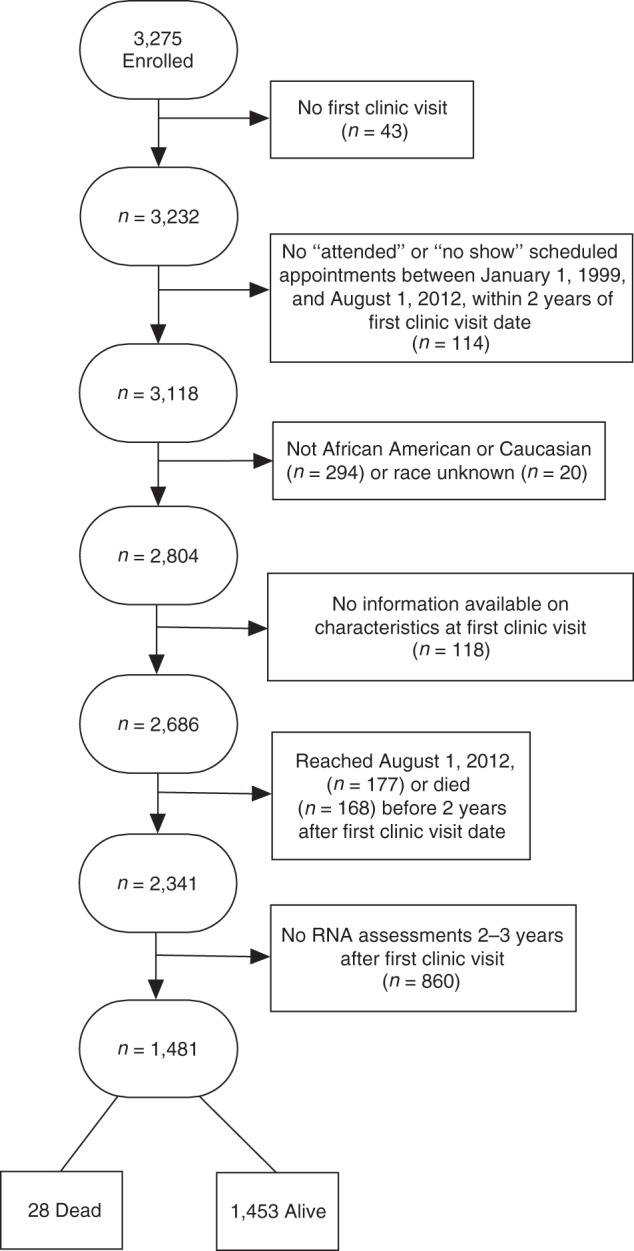
Exclusion criteria applied to identify the 1,481 human immunodeficiency virus (HIV)–infected patients who were included in the analysis sample, University of North Carolina Center for AIDS Research HIV clinical cohort, 1999–2012. The numbers of patients who died and remained alive among the 1,481 included patients during the 1,456 person-years of follow-up that occurred during the window of 2–3 years after their first clinic visit are also included.
Theoretical framework
Figure 2 shows a causal diagram for the influence of African American race on virological suppression in the 2–3 years after the first clinic visit as mediated by clinic attendance in the first 2 years after the first clinic visit among HIV-positive patients in the cohort. In our conceptual model, and described in detail by Jones (14), race is considered to be a socially constructed classification that measures a combination of factors including genes, social class, and culture. African American race is thought to have direct and indirect influences on virological suppression in the 2–3 years after the first clinic visit. In this analysis, the indirect influence (i.e., indirect effect) is hypothesized to operate through clinic attendance in the first 2 years after the first clinic visit, whereas the direct influence (i.e., controlled direct effect) may operate through patient characteristics such as age, disease stage, mental health, alcohol and drug abuse, education, ART use/adherence, and health insurance status at the first and subsequent clinic visits. Note that our “direct” and “indirect” labeling is based on clinic attendance being the mediating pathway of interest. The Figure 2 causal diagram was informed by prior literature (1–4, 12, 15–22).
Figure 2.
Causal diagram for the influence of African American race on virological suppression, University of North Carolina Center for AIDS Research HIV clinical cohort, 1999–2012. Boxes represent conditioning.
Assessment of race, attendance, and other patient characteristics
Data on race were obtained from patient medical records. In terms of the mediating pathway of interest, the 2 measures of clinic attendance used in this analysis were the proportion of scheduled clinic appointments attended by a given patient (i.e., visit adherence) and the proportion of six 4-month time intervals in which a given patient attended at least 1 scheduled clinic appointment (i.e., visit constancy). Six 4-month time intervals for the constancy measure were selected to be consistent with prior work (7). Both measures of clinic attendance had 4 levels where cut points were defined on the basis of quartiles. However, given the distribution of the attendance data, 25% of patients could not always be included in a given attendance category. Quartiles were used, rather than continuous attendance measures, to facilitate the construction of stable inverse probability weights as described below.
Appointments included all scheduled clinic appointments with primary HIV health care providers that occurred in the 2 years after the first clinic visit between January 1, 1999, and August 1, 2012. As done previously (4, 7, 23), only scheduled appointments were used to assess constancy. In addition, we excluded from analysis scheduled appointments that were canceled or rescheduled by the patient or provider, as well as scheduled urgent care or subspecialty appointments (e.g., dermatology). Walk-in visits were also excluded from the definition of a scheduled appointment. A 2-year assessment window for clinic attendance was selected to be consistent with prior work (4) and to allow for gaps in care (i.e., no attended scheduled appointment for at least 1 year) when examining whether inference changed on the basis of the measure of clinic attendance. Information on patient characteristics at the first clinic visit, such as date of birth, calendar date, sex, CD4 cell count, clinical acquired immune deficiency syndrome diagnoses, health insurance status, ART use history, mental illness diagnoses, and history of drug or alcohol abuse was also obtained from patient medical records.
Ascertainment of virological suppression
The primary outcome of interest in this analysis was a repeated measure and corresponded to achieving virological suppression at a given HIV-1 RNA assessment 2–3 years after the first clinic visit. The 2- to 3-year interval was selected to ensure appropriate temporal ordering between race, attendance in the first 2 years, and virological suppression while allowing for the estimation of more recent effects of attendance in the first 2 years on virological suppression. To harmonize HIV-1 RNA data across time, we set all RNA values of less than 400 copies/mL to 200 copies/mL and regarded them as undetectable. Virological suppression was defined as achieving undetectable plasma HIV RNA (i.e., ≤200 copies/mL) at a given RNA level assessment. Patients were not required to have previously initiated ART for a given RNA level assessment to be included in the analysis.
Among the 1,481 study participants, patients were censored at the minimum of their death dates (if applicable), August 1, 2012, or 3 years after their first clinic visits. To assess the impact on inference given the abovementioned limitations of visit adherence among patients with gaps in care, we did not censor follow-up during the first 2 years after the first clinic visit on the basis of the pattern of scheduled appointment attendance in this analysis.
Statistical analysis
Characteristics of included and excluded patients with necessary information measured were examined and compared using Pearson's χ2 and Wilcoxon-Mann-Whitney tests. Unadjusted and inverse probability weighted modified Poisson regression models (8, 9, 24) were used to estimate risk ratios for the total and direct effects of African American race on virological suppression. The total effect was defined as the influence of African American race on virological suppression mediated through all causal pathways shown in Figure 2. The controlled direct effect was defined as the association between African American race and virological suppression when racial disparities in visit adherence or constancy were lessened.
Weighted analyses accounted for sources of confounding and selection bias related to age, calendar date, sex, CD4 cell count, acquired immune deficiency syndrome diagnosis, health insurance, time on ART, history of mental illness, and history of drug or alcohol abuse at the first clinic visit, as well as calendar date of birth, and time since last virological success. Pattern of attendance in the 2–3 years after the first clinic visit was also used in the weight estimation. “Pattern of attendance” refers to the frequency with which a given patient was hospitalized or attended scheduled clinic appointments, walk-in clinic visits, or scheduled urgent care or subspecialty appointments (e.g., dermatology). Given the few deaths (i.e., 28), any bias related to censoring due to death was assumed to be negligible. The proportion of the racial disparity in virological suppression that could be eliminated by lessening disparities in attendance on the excess relative risk scale (i.e., proportion eliminated) was calculated on the basis of the estimated total and direct effect(s) (25). The proportion eliminated was set to 0 for all corresponding direct effects that were further from or on the opposite side of the null compared with the total effect.
Adjusted models estimating the total and direct effect(s), as well as the proportion eliminated, are provided for comparison. Details regarding specification and estimation of the unadjusted, adjusted, and weighted models are provided in Web Appendix 1 available at http://aje.oxfordjournals.org/. All analyses were conducted using SAS, version 9.3, software (SAS Institute, Inc., Cary, North Carolina).
RESULTS
The 1,481 patients in the UNC CFAR HIV clinical cohort who were included in the analysis were followed for 1,456 person-years during the window of 2–3 years after their first clinic visits. By the end of follow-up during the window of 2–3 years after their first clinic visits, 28 patients died, and 1,453 patients were alive. Table 1 shows the characteristics of the 1,481 included patients along with characteristics of those patients who were excluded from the analysis because of the absence of RNA measurements 2–3 years after the first clinic visit.
Table 1.
Characteristics of HIV-Infected Patients of African American or Caucasian Race, University of North Carolina Center for AIDS Research HIV Clinical Cohort, 1999–2012
| Characteristica | Included Patients (n = 1,481) |
Excludedb Patients (n = 860) |
P Valuec | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Median | 25th Percentile, 75th Percentile | No. | % | Median | 25th Percentile, 75th Percentile | No. | % | ||
| Calendar dated | 2001 | 1999, 2005 | 2003 | 2000, 2007 | <0.01 | ||||
| Calendar date of birthd | 1962 | 1956, 1969 | 1964 | 1958, 1972 | <0.01 | ||||
| African American race | 946 | 64 | 592 | 69 | 0.01 | ||||
| Age, years | 40 | 33, 46 | 38 | 32, 45 | 0.01 | ||||
| Male sex | 1,017 | 69 | 597 | 69 | 0.71 | ||||
| Prior clinical AIDS diagnosis | 344 | 23 | 173 | 20 | 0.08 | ||||
| CD4 cell count, cells/μL | 333 | 129, 539 | 376 | 187, 578 | <0.01 | ||||
| Time on antiretroviral therapy, years | 1 | 0, 4 | 1 | 0, 5 | <0.01 | ||||
| Health insurance | |||||||||
| Private | 437 | 29 | 173 | 20 | |||||
| Publice | 562 | 38 | 336 | 39 | <0.01 | ||||
| Uninsured | 482 | 33 | 351 | 41 | |||||
| History of mental illness | 424 | 29 | 287 | 33 | 0.02 | ||||
| History of alcohol or drug abuse | 399 | 27 | 333 | 39 | <0.01 | ||||
| No. of scheduled appointments within 2 years after first clinic visit | 8 | 6, 11 | 5 | 3, 8 | <0.01 | ||||
| Proportion of scheduled appointments attended within 2 years after first clinic visit | |||||||||
| 0–0.732 | 372 | 25 | 438 | 51 | |||||
| 0.733–0.874 | 336 | 23 | 153 | 18 | <.01 | ||||
| 0.875–0.999 | 242 | 16 | 45 | 5 | |||||
| 1 | 531 | 36 | 224 | 26 | |||||
| Proportion of six 4-month intervals within 2 years after first clinic visit with attended scheduled appointment | |||||||||
| 0–0.500 | 483 | 33 | 600 | 70 | |||||
| 0.667 | 316 | 21 | 126 | 15 | <.01 | ||||
| 0.833 | 391 | 26 | 82 | 9 | |||||
| 1 | 291 | 20 | 52 | 6 | |||||
| No. of RNA measurements taken 2–3 years after first clinic visit | 3 | 2, 4 | 0 | ||||||
| Proportion of HIV-1 RNA measurements taken 2–3 years after first clinic visit achieving virological suppression | 1 | 0, 1 | |||||||
Abbreviations: AIDS, acquired immune deficiency syndrome; HIV, human immunodeficiency virus; HIV-1, human immunodeficiency virus type 1.
a At first clinic visit unless noted otherwise.
b No RNA measurements taken 2–3 years after first clinic visit.
c Based on Wilcoxon-Mann-Whitney or Pearson's χ2 test.
d Expressed as year only.
e Medicaid, Medicare, or other US public insurance (e.g., AIDS Drug Assistance Program, Veterans Administration, Department of Defense for prisoners).
Sixty-four percent of included patients were African American, 69% were male, and 23% had previously received a clinical acquired immune deficiency syndrome diagnosis at the time of their first clinic visits. The median (25th percentile, 75th percentile) age and CD4 count at the first clinic visit were 40 (33, 46) years and 333 (129, 539) cells/μL, respectively. During the 2 years after their first clinic visits, 36% of patients attended all of their scheduled appointments, and 20% of patients had at least 1 attended scheduled appointment during all of the six 4-month intervals. Sixty-two percent of HIV-1 RNA measurements taken 2–3 years after the first clinic visit revealed suppressed RNA levels.
Compared with the 860 patients who were excluded from the analysis because of the absence of RNA measurements taken 2–3 years after the first clinic visits, patients who were included were born earlier, less likely to be African American, and tended to have earlier first clinic visit calendar dates. They were older and had more advanced HIV disease but shorter times on ART; they were also less likely to be uninsured and to have a history of mental illness or alcohol/drug abuse at the first clinic visit. Given that excluded patients were likely to be less engaged in care, included patients, as expected, also tended to have more scheduled appointments, as well as greater visit adherence and constancy in the first 2 years after the first clinic visit.
Table 2 provides the unadjusted and weighted total effect of African American race on virological suppression. Compared with Caucasians, African Americans had an unadjusted risk ratio for achieving virological suppression of 0.86 (95% confidence interval (CI): 0.79, 0.92). The corresponding adjusted and weighted risk ratios were 0.92 (95% CI: 0.86, 0.98) and 0.91 (95% CI: 0.85, 0.98), respectively.
Table 2.
Total Effect of Race on Virological Suppression Among 946 African American Patients Compared With 535 Caucasian Patients, University of North Carolina Center for AIDS Research HIV Clinical Cohort, 1999–2012
| Race | HIV-1 RNA Measurements Taken 2–3 Years After First Clinic Visit Where RNA Level was Suppressed |
HIV-1 RNA Measurements Taken 2–3 Years After First Clinic Visit |
Unadjusted |
Weighted |
||||
|---|---|---|---|---|---|---|---|---|
| No. Observed | No. Weighted | No. Observed | No. Weighted | Risk Ratio | 95% CI | Risk Ratioa | 95% CI | |
| African American | 1,383 | 1,382 | 2,378 | 2,385 | 0.86 | 0.79, 0.92 | 0.91 | 0.85, 0.98 |
| Caucasian | 1,003 | 938 | 1,477 | 1,410 | 1.00 | 1.00 | ||
| Total | 2,386 | 2,320 | 3,855 | 3,795 | ||||
Abbreviations: AIDS, acquired immune deficiency syndrome; CI, confidence interval; HIV, human immunodeficiency virus; HIV-1, human immunodeficiency virus type 1.
a Accounts for age, calendar date, sex, CD4 cell count, an AIDS diagnosis, health insurance, time on antiretroviral therapy, history of mental illness, and history of drug and alcohol abuse at the first clinic visit, as well as calendar year of birth, visit adherence and visit constancy in the first 2 years after the first clinic visit, and time since last virological success.
Table 3 shows the unadjusted and weighted effects of African American race on virological suppression when racial disparities in visit adherence were lessened. Compared with Caucasians, African Americans had unadjusted risk ratios for achieving virological suppression ranging from 0.81 (95% CI: 0.69, 0.95) to 1.13 (95% CI: 0.86, 1.48). The corresponding adjusted and weighted risk ratios ranged from 0.84 (95% CI: 0.74, 0.96) to 1.05 (95% CI: 0.84, 1.32) and 0.87 (95% CI: 0.76, 0.99) to 1.12 (95% CI: 0.88, 1.44), respectively. When interaction terms between African American race and adherence were omitted from the unadjusted, adjusted, and weighted models, the risk ratios for the controlled direct effect of African American race on virological suppression were 0.91 (95% CI: 0.84, 0.98), 0.94 (95% CI: 0.88, 1.00) and 0.97 (95% CI: 0.91, 1.04), respectively.
Table 3.
Controlled Direct Effects of African American Race on Virological Suppression When Racial Disparities in Visit Adherence are Lessened Between 946 African American and 535 Caucasian Patients, University of North Carolina Center for AIDS Research HIV Clinical Cohort, 1999–2012
| Proportion of Scheduled Appointments Attended Within 2 Years After First Clinic Visit by Race | HIV-1 RNA Measurements Taken 2–3 Years After First Clinic Visit Where RNA Level was Suppressed |
HIV-1 RNA Measurements Taken 2–3 Years After First Clinic Visit |
Unadjusted |
Weighted |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No. Observed | No. Weighted | No. Observed | No. Weighted | Risk Ratio | 95% CI | PE, % | Risk Ratioa | 95% CI | PE, % | |
| 0–0.732 | ||||||||||
| African American | 305 | 454 | 637 | 897 | 1.13 | 0.86, 1.48 | 0 | 1.12 | 0.88, 1.44 | 0 |
| Caucasian | 80 | 109 | 189 | 261 | 1.00 | Referent | 1.00 | Referent | ||
| 0.733–0.874 | ||||||||||
| African American | 309 | 297 | 562 | 512 | 0.81 | 0.69, 0.95 | 0 | 0.87 | 0.76, 0.99 | 0 |
| Caucasian | 196 | 195 | 288 | 275 | 1.00 | Referent | 1.00 | Referent | ||
| 0.875–0.999 | ||||||||||
| African American | 225 | 178 | 384 | 287 | 0.90 | 0.75, 1.08 | 28.57 | 1.02 | 0.87, 1.19 | 0 |
| Caucasian | 183 | 137 | 282 | 212 | 1.00 | Referent | 1.00 | Referent | ||
| 1 | ||||||||||
| African American | 544 | 485 | 795 | 704 | 0.90 | 0.82, 1.00 | 28.57 | 0.95 | 0.87, 1.03 | 44.44 |
| Caucasian | 544 | 473 | 718 | 637 | 1.00 | Referent | 1.00 | Referent | ||
| 0–1b | ||||||||||
| African American | 1,383 | 1,413 | 2,378 | 2,400 | 0.91 | 0.84, 0.98 | 35.71 | 0.97 | 0.91, 1.04 | 66.67 |
| Caucasian | 1,003 | 915 | 1,477 | 1,384 | 1.00 | Referent | 1.00 | Referent | ||
| Total | 2,386 | 2,328 | 3,855 | 3,784 | ||||||
Abbreviations: AIDS, acquired immune deficiency syndrome; CI, confidence interval; HIV, human immunodeficiency virus; HIV-1, human immunodeficiency virus type 1; PE, proportion eliminated.
a Accounts for age, calendar date, sex, CD4 cell count, an AIDS diagnosis, health insurance, time on antiretroviral therapy, history of mental illness, and history of drug and alcohol abuse at the first clinic visit, as well as calendar year of birth, visit adherence and visit constancy in the first 2 years after the first clinic visit, and time since last virological success.
b Interaction terms between African American race and visit adherence omitted.
Table 4 shows the unadjusted and weighted effects of African American race on virological suppression when racial disparities in visit constancy were lessened. Compared with Caucasians, African Americans had unadjusted risk ratios for achieving virological suppression ranging from 0.81 (95% CI: 0.69, 0.95) to 0.89 (95% CI: 0.77, 1.03). The corresponding adjusted and weighted risk ratios ranged from 0.89 (95% CI: 0.78, 1.00) to 0.95 (95% CI: 0.85, 1.05) and 0.89 (95% CI: 0.77, 1.03) to 0.93 (95% CI: 0.81, 1.07), respectively. When interaction terms between African American race and constancy were omitted from the unadjusted, adjusted, and weighted models, the risk ratios for the controlled direct effect of African American race on virological suppression were 0.85 (95% CI: 0.79, 0.92), 0.91 (95% CI: 0.86, 0.97), and 0.92 (95% CI: 0.85, 0.98), respectively.
Table 4.
Controlled Direct Effects of African American Race on Virological Suppression When Racial Disparities in Visit Constancy are Lessened Between 946 African American and 535 Caucasian Patients, University of North Carolina Center for AIDS Research HIV Clinical Cohort, 1999–2012
| Proportion of Six 4-month Intervals Within 2 Years After First Clinic Visit With Attended Scheduled Appointment by Race | HIV-1 RNA Measurements Taken 2–3 Years After First Clinic Visit Where RNA Level was Suppressed |
HIV-1 RNA Measurements Taken 2–3 Years After First Clinic Visit |
Unadjusted |
Weighted |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No. Observed | No. Weighted | No. Observed | No. Weighted | Risk Ratio | 95% CI | PE, % | Risk Ratioa | 95% CI | PE, % | |
| 0–0.500 | ||||||||||
| African American | 426 | 681 | 747 | 1,224 | 0.89 | 0.77, 1.03 | 21.43 | 0.93 | 0.81, 1.07 | 22.22 |
| Caucasian | 235 | 336 | 367 | 558 | 1.00 | Referent | 1.00 | Referent | ||
| 0.677 | ||||||||||
| African American | 235 | 236 | 432 | 419 | 0.82 | 0.69, 0.98 | 0 | 0.91 | 0.80, 1.03 | 0 |
| Caucasian | 245 | 236 | 371 | 351 | 1.00 | Referent | 1.00 | Referent | ||
| 0.833 | ||||||||||
| African American | 433 | 365 | 684 | 570 | 0.88 | 0.77, 1.01 | 14.29 | 0.92 | 0.82, 1.02 | 11.11 |
| Caucasian | 280 | 258 | 390 | 348 | 1.00 | Referent | 1.00 | Referent | ||
| 1 | ||||||||||
| African American | 289 | 174 | 515 | 292 | 0.81 | 0.69, 0.95 | 0 | 0.89 | 0.77, 1.03 | 0 |
| Caucasian | 243 | 145 | 349 | 203 | 1.00 | Referent | 1.00 | Referent | ||
| 0–1b | ||||||||||
| African American | 1,383 | 1,457 | 2,378 | 2,505 | 0.85 | 0.79, 0.92 | 0 | 0.92 | 0.85, 0.98 | 11.11 |
| Caucasian | 1,003 | 975 | 1,477 | 1,460 | 1.00 | Referent | 1.00 | Referent | ||
| Total | 2,386 | 2,432 | 3,855 | 3,965 | ||||||
Abbreviations: AIDS, acquired immune deficiency syndrome; CI, confidence interval; HIV, human immunodeficiency virus; HIV-1, human immunodeficiency virus type 1; PE, proportion eliminated.
a Accounts for age, calendar date, sex, CD4 cell count, an AIDS diagnosis, health insurance, time on antiretroviral therapy, history of mental illness, and history of drug and alcohol abuse at the first clinic visit, as well as calendar year of birth, visit adherence and visit constancy in the first 2 years after the first clinic visit, and time since last virological success.
b Interaction terms between African American race and visit constancy omitted.
Significant associations between African American race and adherence, as well as constancy, were observed when estimating the adherence and constancy weights. Adherence was also significantly associated with suppression in the weighted models that did and did not allow for an interaction between African American race and adherence. Constancy was not significantly associated with suppression (results included in Web Appendix 2, Web Tables 1–4).
DISCUSSION
Among 1,481 HIV-positive patients in the UNC CFAR HIV clinical cohort followed for 1,456 person-years, weighted analyses showed that African Americans were less likely than Caucasians to suppress their HIV-1 RNA. Assuming the presence of multiplicative interaction between African American race and visit adherence (coefficients for interaction terms: 0.17 (P = 0.21), −0.09 (P = 0.26), and 0.07 (P = 0.42)), assigning African American and Caucasians to be 100% adherent lessened racial disparities in virological suppression by 44.4%. Assuming the absence of multiplicative interaction between African American race and visit constancy (coefficients for interaction terms: 0.04 (P = 0.67), 0.02 (P = 0.86), and 0.03 (P = 0.78)), lessening disparities in constancy reduced racial disparities in virological suppression by 11.1%.
These weighted results suggest that interventions that lessen disparities in adherence may be more effective in eliminating disparities in virological suppression than interventions that lessen disparities in constancy. However, given that gaps in care were limited to be no longer than 2 years, the impact of lessening disparities in adherence may be overstated. In addition, some estimates summarizing the effect of intervention on disparities in adherence or constancy were imprecise, likely because of the greater amount of information that is required to estimate direct effects.
Similar to the results of this study, prior work (4) in the UAB 1917 Clinic Cohort indicated that lessening disparities in adherence would reduce disparities in virological response. However, the UAB study assumed the absence of a multiplicative interaction between African American race and adherence based on lack of statistical significance of the corresponding interaction term. When a multiplicative interaction between African American race and adherence was assumed to be absent in our patient population, 66.7% of racial disparities in virological suppression were removed when disparities in adherence were lessened. Therefore, ignoring a potential interaction between race and adherence because of lack of statistical significance in this study led to an overestimation of the impact of lessening disparities in adherence.
Furthermore, the prior UAB 1917 Clinic Cohort study adjusted for common causes of adherence and virological response that are also likely influenced by race and that may have induced selection bias or resulted in the removal of indirect effects. Such selection bias or removal of indirect effects was minimized in our weighted analysis (8, 9). Performing an equivalent adjusted analysis yielded values for the proportion eliminated (results not shown) that were lower in some cases and higher in others compared with the weighted results.
The fact that visit constancy was not significantly associated with suppression in our weighted analysis likely contributed to the lack of a substantial observed reduction in disparities in virological suppression with a reduction in disparities in constancy. Prior work (7) based on combined data from 6 academic HIV clinics in the United States found a statistically significant association between constancy and virological suppression, in which patients with greater constancy were more likely to have suppressed HIV-1 RNA levels. The discrepancy between this prior work and our present results may be due to differences in study population characteristics, in which CD4 cell counts at study entry were higher, visit adherence was lower, and 4-month visit constancy was higher in the combined study. In addition, virological suppression was determined on the basis of a single RNA measurement at the end of the study period rather than on multiple RNA measurements as done in the present study.
There are limitations to the present research. The sample sizes within levels of race and visit adherence (or visit constancy) resulted in imprecise estimates in some cases and increased the amount of random error. Because of changes in the standard of care at the UNC HIV clinic, patients who were virologically suppressed may have been seen at the clinic less frequently in later study years (i.e., after 2011), which might bias results pertaining to the visit constancy measure of clinic attendance. Although inverse probability weighting was used to correct for measured sources of confounding and selection bias (13, 26, 27), unmeasured potential sources of these biases related to factors such as educational level and ART adherence may remain. Although methods are available to assess the sensitivity of estimates for controlled direct effects to unmeasured sources of confounding bias of the mediator-outcome relationship, such methods are not applicable in the settings explored here, in which the exposure determines common causes of the mediator and outcome (28). Bias can also occur in the presence of measurement error, model misspecification, nonpositivity, or lack of consistency (29).
Despite these limitations, there are several noteworthy strengths of this study. The use of prospective cohort data combined with lagging helped ensure the appropriate temporal ordering. In prior work (4), the measure of association used was the odds ratio. When the outcome is not rare, as in this and the prior study, the odds ratio is prone to overestimate the risk ratio, which is the effect measure of interest, and the odds ratio also suffers from noncollapsibility (30, 31). Here, we directly estimated risk ratios using modified Poisson regression (24). We also used inverse probability weighting to correct for potential sources of selection and confounding bias, which helped to avoid selection bias and the removal of indirect effects (8, 9). Lastly, correct model specification was facilitated by including indicator variables for categories and linear and quadratic terms or restricted quadratic splines for continuous predictors.
In summary, our study found that among 1,481 HIV-positive patients in the UNC CFAR HIV clinical cohort, African Americans were less likely than Caucasians to have suppressed HIV-1 RNA levels. The lessening of disparities in visit adherence or constancy lowered disparities in virological suppression by up to 44.4% and 11.1%, respectively. These results suggest that interventions that lessen disparities in visit adherence may be more effective in eliminating disparities in virological suppression than interventions that lessen disparities in visit constancy. However, given that gaps in care were limited to be no longer than 2 years in this study, the impact of lessening disparities in adherence may be overstated. In addition, some estimates were imprecise. Future studies should replicate our analysis in other, larger HIV clinic cohorts while allowing for interactions between race and attendance and using inverse probability weighting to correct for potential sources of selection and confounding bias. Future analyses should also ascertain suppression at multiple RNA assessments.
Supplementary Material
ACKNOWLEDGMENTS
Author affiliations: Department of Epidemiology, Center for Population Health and Clinical Epidemiology, Brown University School of Public Health, Providence, Rhode Island (Chanelle J. Howe, Beth Elston); Department of Epidemiology, University of North Carolina Gillings School of Global Public Health, Chapel Hill, North Carolina (Sonia Napravnik, Stephen R. Cole, Adaora A. Adimora); Division of Infectious Diseases, Department of Medicine, University of North Carolina School of Medicine, Chapel Hill, North Carolina (Sonia Napravnik, Adaora A. Adimora, Joseph J. Eron, Jr); Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Canada (Jay S. Kaufman); and Division of Infectious Diseases, Department of Medicine, University of Alabama at Birmingham, Birmingham, Alabama (Michael J. Mugavero).
This work was supported by the National Institutes of Health (grants K22-AI-095021, K24-HD-059358, and P30 AI50410).
We thank Sam Stinnette for assistance with the University of North Carolina Center for AIDS Research HIV clinical cohort data, Dr. Tyler VanderWeele for expert advice, and the University of North Carolina Center for AIDS Research HIV clinical cohort study staff.
Conflicts of interest: none declared.
REFERENCES
- 1.Kissinger P, Cohen D, Brandon W, et al. Compliance with public sector HIV medical care. J Natl Med Assoc. 1995;87(1):19–24. [PMC free article] [PubMed] [Google Scholar]
- 2.Catz SL, McClure JB, Jones GN, et al. Predictors of outpatient medical appointment attendance among persons with HIV. AIDS Care. 1999;11(3):361–373. doi: 10.1080/09540129947983. [DOI] [PubMed] [Google Scholar]
- 3.Israelski D, Gore-Felton C, Power R, et al. Sociodemographic characteristics associated with medical appointment adherence among HIV-seropositive patients seeking treatment in a county outpatient facility. Prev Med. 2001;33(5):470–475. doi: 10.1006/pmed.2001.0917. [DOI] [PubMed] [Google Scholar]
- 4.Mugavero MJ, Lin HY, Allison JJ, et al. Racial disparities in HIV virologic failure: Do missed visits matter? J Acquir Immune Defic Syndr. 2009;50(1):100–108. doi: 10.1097/QAI.0b013e31818d5c37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gulick RM, Ribaudo HJ, Shikuma CM, et al. Three- vs four-drug antiretroviral regimens for the initial treatment of HIV-1 infection: a randomized controlled trial. JAMA. 2006;296(7):769–781. doi: 10.1001/jama.296.7.769. [DOI] [PubMed] [Google Scholar]
- 6.Mugavero MJ, Davila JA, Nevin CR, et al. From access to engagement: measuring retention in outpatient HIV clinical care. AIDS Patient Care STDS. 2010;24(10):607–613. doi: 10.1089/apc.2010.0086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mugavero MJ, Westfall AO, Zinski A, et al. Measuring retention in HIV care: the elusive gold standard. J Acquir Immune Defic Syndr. 2012;61(5):574–580. doi: 10.1097/QAI.0b013e318273762f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.VanderWeele TJ. Marginal structural models for the estimation of direct and indirect effects. Epidemiology. 2009;20(1):18–26. doi: 10.1097/EDE.0b013e31818f69ce. [DOI] [PubMed] [Google Scholar]
- 9.Robins JM, Hernán MA, Brumback B. Marginal structural models and causal inference in epidemiology. Epidemiology. 2000;11(5):550–560. doi: 10.1097/00001648-200009000-00011. [DOI] [PubMed] [Google Scholar]
- 10.Hafeman DM, Schwartz S. Opening the Black Box: a motivation for the assessment of mediation. Int J Epidemiol. 2009;38(3):838–845. doi: 10.1093/ije/dyn372. [DOI] [PubMed] [Google Scholar]
- 11.Valeri L, Vanderweele TJ. Mediation analysis allowing for exposure-mediator interactions and causal interpretation: theoretical assumptions and implementation with SAS and SPSS macros. Psychol Methods. 2013;18(2):137–150. doi: 10.1037/a0031034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Howe CJ, Cole SR, Napravnik S, et al. Enrollment, retention, and visit attendance in the University of North Carolina Center for AIDS Research HIV clinical cohort, 2001–2007. AIDS Res Hum Retroviruses. 2010;26(8):875–881. doi: 10.1089/aid.2009.0282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hernán MA, Hernández-Díaz S, Robins JM. A structural approach to selection bias. Epidemiology. 2004;15(5):615–625. doi: 10.1097/01.ede.0000135174.63482.43. [DOI] [PubMed] [Google Scholar]
- 14.Jones CP. Invited commentary: “race,” racism, and the practice of epidemiology. Am J Epidemiol. 2001;154(4):299–304. doi: 10.1093/aje/154.4.299. [DOI] [PubMed] [Google Scholar]
- 15.Andersen RM. Revisiting the behavioral model and access to medical care: Does it matter? J Health Soc Behav. 1995;36(1):1–10. [PubMed] [Google Scholar]
- 16.Valdez H, Lederman MM, Woolley I, et al. Human immunodeficiency virus 1 protease inhibitors in clinical practice: predictors of virological outcome. Arch Intern Med. 1999;159(15):1771–1776. doi: 10.1001/archinte.159.15.1771. [DOI] [PubMed] [Google Scholar]
- 17.Lucas GM, Chaisson RE, Moore RD. Highly active antiretroviral therapy in a large urban clinic: risk factors for virologic failure and adverse drug reactions. Ann Intern Med. 1999;131(2):81–87. doi: 10.7326/0003-4819-131-2-199907200-00002. [DOI] [PubMed] [Google Scholar]
- 18.Messeri PA, Abramson DM, Aidala African American, et al. The impact of ancillary HIV services on engagement in medical care in New York City. AIDS Care. 2002;14(suppl 1):S15–S29. doi: 10.1080/09540120220149948. [DOI] [PubMed] [Google Scholar]
- 19.Rastegar DA, Fingerhood MI, Jasinski DR. Highly active antiretroviral therapy outcomes in a primary care clinic. AIDS Care. 2003;15(2):231–237. doi: 10.1080/0954012031000068371. [DOI] [PubMed] [Google Scholar]
- 20.Cargill VA, Stone VE. HIV/AIDS: a minority health issue. Med Clin North Am. 2005;89(4):895–912. doi: 10.1016/j.mcna.2005.03.005. [DOI] [PubMed] [Google Scholar]
- 21.Robbins GK, Daniels B, Zheng H, et al. Predictors of antiretroviral treatment failure in an urban HIV clinic. J Acquir Immune Defic Syndr. 2007;44(1):30–37. doi: 10.1097/01.qai.0000248351.10383.b7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Robbins GK, Johnson KL, Chang Y, et al. Predicting virologic failure in an HIV clinic. Clin Infect Dis. 2010;50(5):779–786. doi: 10.1086/650537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mugavero MJ, Lin HY, Willig JH, et al. Missed visits and mortality among patients establishing initial outpatient HIV treatment. Clin Infect Dis. 2009;48(2):248–256. doi: 10.1086/595705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zou GY, Donner A. Extension of the modified Poisson regression model to prospective studies with correlated binary data. Stat Methods Med Res. 2013;22(6):661–670. doi: 10.1177/0962280211427759. [DOI] [PubMed] [Google Scholar]
- 25.Suzuki E, Evans D, Chaix B, et al. On the “proportion eliminated” for risk differences versus excess relative risks. Epidemiology. 2014;25(2):309–10. doi: 10.1097/EDE.0000000000000060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hernán MA, Hernández-Díaz S, Werler MM, et al. Causal knowledge as a prerequisite for confounding evaluation: an application to birth defects epidemiology. Am J Epidemiol. 2002;155(2):176–184. doi: 10.1093/aje/155.2.176. [DOI] [PubMed] [Google Scholar]
- 27.Hernán MA, McAdams M, McGrath N, et al. Observation plans in longitudinal studies with time-varying treatments. Stat Methods Med Res. 2009;18(1):27–52. doi: 10.1177/0962280208092345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.VanderWeele TJ. Bias formulas for sensitivity analysis for direct and indirect effects. Epidemiology. 2010;21(4):540–551. doi: 10.1097/EDE.0b013e3181df191c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cole SR, Hernán MA. Constructing inverse probability weights for marginal structural models. Am J Epidemiol. 2008;168(6):656–664. doi: 10.1093/aje/kwn164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miettinen OS, Cook EF. Confounding: essence and detection. Am J Epidemiol. 1981;114(4):593–603. doi: 10.1093/oxfordjournals.aje.a113225. [DOI] [PubMed] [Google Scholar]
- 31.Greenland S. Absence of confounding does not correspond to collapsibility of the rate ratio or rate difference. Epidemiology. 1996;7(5):498–501. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.



