Abstract
We report a rare case of progressive parkinsonism and cognitive dysfunction due to dural arteriovenous fistula (DAVF). A 69-year-old man, with a history of hypertension and diabetes, was admitted to our hospital because of parkinsonism and dementia. Susceptibility-weighted imaging (SWI) revealed a thrombus in the superior sagittal sinus (SSS) and marked dilation of the medullary vein suggestive of the presence of comorbid DAVF. A single-photon emission CT (SPECT) showed widespread hypoperfusion in the bilateral frontal lobes. Selective cerebral angiography revealed a DAVF in SSS. These symptoms were significantly ameliorated following transvenous embolisation of the venous sinus at the shunting point. Reversible parkinsonism and dementia after embolisation was correlated with decreased dilation of medullary vein on SWI and improved cerebral blood flow on SPECT in the frontal lobes. Differentiation of parkinsonian and dementia symptoms due to DAVF from those associated with neurodegenerative disease is of great importance because DAVF-associated deficits may be reversed by endovascular therapy.
Background
The clinical manifestations of dural arteriovenous fistula (DAVF) depend on the anatomical location and on the venous drainage pattern, and may include headache, tinnitus, proptosis, visual disturbances, seizures and the neurological deficits associated with intracranial haemorrhage. The clinical and imaging features of DAVF are non-specific and diagnosis is often delayed or incorrect. However, early diagnosis and prompt treatment of DAVF is very important because DAVF with cortical venous reflux as in this case carry a high risk for intracranial haemorrhage, non-hemorrhagic neurological deficit or death.1
We report a rare case of progressive parkinsonism and cognitive dysfunction due to DAVF.
Case presentation
A 69-year-old man developed a tremor that ocurred while writing and mild memory deficits about 2 years before admission. About 6 months before admission, he began to walk with a shuffle with shortened strides. Owing to this festinating gait, he was unable to walk long distances and fell easily. These symptoms were slowly progressive. Four months before admission, levodopa was prescribed by the neurology department of a local hospital for the parkinsonian syndrome but did not produce clinical improvement at doses up to 300 mg/day. Two months before admission, he retired from his job as a corporate executive officer because he was no longer able to solve problems that required quick decisions. He was also unable to sit squarely because of postural instability. By 1 month before admission, assisted care was needed for eating and bathing. Three weeks before admission, he was referred to the neurology department of our hospital from the local hospital. He had a history of hypertension and diabetes but no history of head trauma, meningitis or skull surgery. Family history of Parkinson's disease was absent. At admission, he was taking levodopa (300 mg/day), an antidiabetic agent and antihypertension agent.
Investigations
His vital signs were stable. He had no headache, and a machinery murmur characteristic of an arteriovenous fistula was not heard from his head. On neurological examination, he was alert and showed no signs of aphasia, apraxia or agnosia. However, he demonstrated attention disturbance and deficits in executive function. He also exhibited motor impersistence, including ocular vacillation. His Mini-Mental State Examination (MMSE) score on admission was 26 and frontal assessment battery (FAB) score was 6, suggesting frontal lobe hypofunction. There were no abnormal findings at fundoscopy. No tinnitus, hearing loss or other signs of cranial nerve palsy were detected. He had mild rigidity of the upper limbs bilaterally with bradykinesia and postural tremor in the right upper limb with an intentional component on targeting; resting tremor was absent. The postural tremor had moderate amplitude with a frequency of 5−6 Hz. His gait consisted of short strides with anteversion and decreased arm swing, and he tended to lose balance easily. No ataxia or sensory disturbances were detected. His deep tendon reflexes were normal and no pathological reflexes were observed.
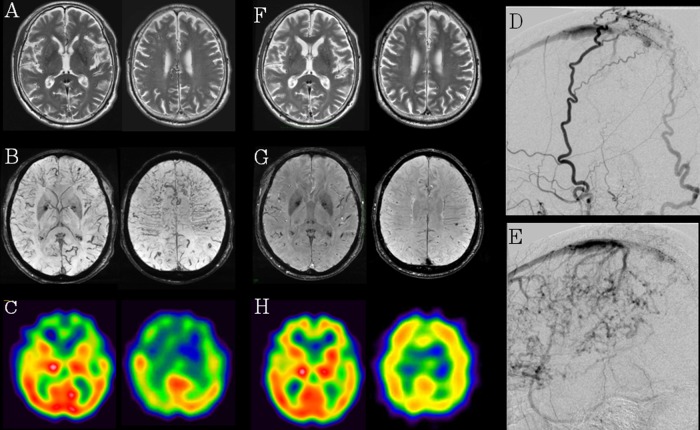
Results of routine haematological and biochemical tests were normal. T2-weighted MRI revealed the slight degree of dilation of the medullary vein in the bilateral frontal and parietal lobes (figure 1A). No hyperintense lesions on T2-weighted or diffusion-weighted MRI were detected. Susceptibility-weighted imaging (SWI) revealed a thrombus in the superior sagittal sinus (SSS) and marked dilation of the medullary vein in the bilateral frontal and parietal lobes (figure 1B). MR venography revealed extreme distension of the collateral vein (data not shown), whereas single-photon emission CT (SPECT) with N-isopropyl-p-123I-iodoamphetamine (123IMP-SPECT) of the brain showed widespread hypoperfusion in the frontal lobes bilaterally at rest (figure 1C). We confirmed the absence of thrombotic diathesis. Marked dilation of the medullary vein suggested the presence of comorbid DAVF.
Figure 1.
(A–C) (Pre-embolisation therapy): T2-weighted MRI revealed the slight degree of dilation of the medullary vein in the bilateral frontal and parietal lobes (A). Susceptibility-weighted imaging (SWI) showed marked dilation of the medullary vein in the bilateral frontal and parietal lobes (B). IMP-SPECT of the brain showed hypoperfusion in the frontal lobes bilaterally at rest (C). (D and E) (Selective cerebral angiography): cerebral angiography revealed dural arteriovenous fistula in superior sagittal sinus with cortical venous reflux (D) fed by the superficial temporal artery and occipital artery (E). (F–H) (postembolisation therapy): T2-weighted image (F) and SWI (G) revealed decreased dilation of the medullary vein in the bilateral frontal and parietal lobes and IMP-SPECT showed improved regional cerebral blood flow in the frontal lobes bilaterally (H).
Treatment
Selective cerebral angiography revealed a DAVF (Borden type II) in SSS supplied by the superficial temporal artery and occipital artery invading through the skull (figure 1D). The latter part of SSS was occluded and there was shunting at the anterior end of the occlusion. In addition, there was retrograde flow into the cortical veins in the frontal and parietal lobes downstream of the occlusion (figure 1E). Venous reflux to the brain surface was eliminated by transvenous embolisation using platinum coils of the venous sinus at the shunting point. Anticoagulant therapy was not performed in consideration of the risk of intracranial haemorrhage.
Outcome and follow-up
This treatment resulted in a marked clinical improvement, particularly of the patient's gait. Timed Up and Go Test results improved from 16 s on admission to 8 s on postoperative day 5. His MMSE score improved from 26 on admission to 27 on postoperative day 2, and the FAB score increased dramatically from 6 on admission to 13 on postoperative day 2. Levodopa could be tapered on postoperative day 1 and discontinued on postoperative day 7. A T2-weighted image and SWI revealed decreased dilation of the medullary vein in the bilateral frontal and parietal lobes (figure 1F,G). IMP-SPECT showed improved regional cerebral blood flow in the frontal lobes bilaterally (figure 1H).
Discussion
DAVF accounts for approximately 10−15% of all intracranial arteriovenous malformations.2 The clinical manifestations depend on the anatomical location and on the venous drainage pattern. To the best of our knowledge, only 13 reports have described individual patients with DAVF who have parkinsonism with or without cognitive dysfunction (table 1).3–11 Ten of these patients were male and had a mean age of 63 years (range 40−81 years). The duration from initial presentation to diagnosis of DAVF ranged from 1 week to 3 years.
Table 1.
Neurological presentation and imaging findings of DAVF presented with progressive dementia and parkinsonism
| Case number (reference number) | Age/sex | Neurological presentation |
Time from onset to treatment | Hyperintense lesions on T2-weighted image | Hypoperfusion lesions on SPECT | Outcome | |||
|---|---|---|---|---|---|---|---|---|---|
| Tremor | Rigidity | Gait disturbance | Dementia | ||||||
| 1 (3) | 81/M | – | + | + | + | 6 months | Bilateral cerebral white matter | Bilateral cortex | Improved |
| 2 (4) | 55/M | − | − | + | + | 8 months | Bilateral deep and subcortical white matter | Left thalamus | Improved |
| 3 (4) | 78/M | − | + | + | + | 9 months | NL | Bilateral cortex | Improved |
| 4 (4) | 69/F | − | + | – | + | Several years | Bilateral cerebral white matter | NE | Unimproved |
| 5 (5) | 44/M | − | − | + | − | 1 week | NL | Bilateral cortex | Improved |
| 6 (6) | 60/F | Bilateral action tremor |
+ | + | + | 18 months | Bilateral subcortical white matter | Bilateral basal ganglia and left frontotemporal lobes | Improved |
| 7 (7) | 75/M | Bilateral postural tremor | + | + | + | 14 weeks | NL | Frontal lobes and basal ganglia | Improved |
| 8 (8) | 65/M | − | + | + | + | 11 months | Basal ganglia and deep white matter | Left frontal lobe and basal ganglia | Improved |
| 9 (9) | 79/M | Bilateral postural tremor | + | + | − | 2 years | NL | NE | Normalised |
| 10 (10) | 54/M | Bilateral postural tremor | + | + | + | 3 years | Bilateral thalamus and globus pallidus | NE | Only minimal change |
| 11 (10) | 40/M | – | + | – | + | 3 months | NE | NE | Unchanged |
| 12 (11) | 52/F | − | − | + | + | 3 months | Basal ganglia and deep white matter | Bilateral cortex and basal ganglia | Improved |
| 13 (present case) | 69/M | Right hand postural tremor | + | + | + | 2 years | NL | Frontal lobes and basal ganglia | Improved |
NE, not examined; NL, normal; SPECT, single-photon emission CT.
Eleven of 13 past case reports showed cognitive manifestation, of which seven showed cognitive manifestations suggestive of frontal lobe dysfunction, including decreased attention span, executive dysfunction and decreased speech output. The other four cases showed cognitive manifestation such as impaired calculation; disorientation in time, place and person; and an abnormal result on the clock drawing test.
The pathophysiology of parkinsonism due to DAVF is still poorly understood. It has been reported that parkinsonism due to DAVF may stem from two distinct pathogenic processes. One possible pathogenic mechanism for DAVF-associated parkinsonism is thought to be hypoperfusion in the frontal lobes due to a venous hypertension caused by either increased blood flow through the draining veins or an obstruction to drainage.6 12 Another possible pathogenic mechanism is thought to be basal ganglia dysfunction due to impaired drainage of the deep internal veins.6 Cerebral hypoperfusion was observed in the bilateral frontal lobes but not in the basal ganglia in our case. Reversible parkinsonism after embolisation was correlated with perfusion status, suggesting that hypoperfusion in the frontal lobes was responsible for the pathogenesis of the parkinsonism in our case. Following decreased dilation of the medullary vein in the bilateral frontal lobes on SWI after embolisation, IMP-SPECT showed improved regional cerebral blood flow in the frontal lobes bilaterally, suggesting that hypoperfusion in the frontal lobes may be caused by retrograde reflux into the medullary vein in our case.
It has been reported that DAVF with cortical venous drainage (Borden types II and III) carry a high risk for intracranial haemorrhage.13 In these lesions, annual risk of intracranial haemorrhage of 8.1% have been reported.14 Therefore, we considered this patient has a high major bleeding risk. Anticoagulant therapy was not performed in consideration of the risk of intracranial haemorrhage.
SWI is an MRI technique that maximises sensitivity to magnetic susceptibility effects.15 16 DAVF may be less conspicuous on conventional MRI sequences. Owing to the ability of SWI to detect and delineate intravascular deoxygenated blood and venous structures, prominence of venous vasculature due to the prolonged passage of time of intracranial blood, venous engorgement and possible functional obstruction caused by DAVF can be identified by SWI.17 To the best of our knowledge, this is an important report showing reversible dilation of medullary vein on SWI in cases of DAVF presenting with progressive parkinsonism and cognitive dysfunction. The use of SWI may be useful in early diagnosis of DAVF by detecting the dilation of the medullary vein accompanied with DAVF as in our case.
We report a rare case of progressive parkinsonism and cognitive dysfunction due to DAVF. Differentiation of parkinsonian and dementia symptoms due to DAVF from those associated with neurodegenerative disease is of great importance because DAVF-associated deficits may be reversed by endovascular therapy.
Learning points.
The clinical manifestations of dural arteriovenous fistula (DAVF) depend on the anatomical location and on the venous drainage pattern, and may include headache, tinnitus, proptosis, visual disturbances, seizures and the neurological deficits associated with intracranial haemorrhage.
Early diagnosis and prompt treatment of DAVF is very important because DAVF with cortical venous reflux as in this case carry a high risk for intracranial haemorrhage, non-hemorrhagic neurological deficit or death.
While DAVF presenting with signs of dementia and progressive parkinsonism is a rare condition, DAVF should be considered and eliminated as a potential cause of progressive parkinsonism or dementia because DAVF-associated deficits may be reversed by endovascular therapy.
The use of susceptibility-weighted imaging may be useful for earlier diagnosis of DAVF presenting with dementia and parkinsonism.
Acknowledgments
The authors thank Kazuhide Ochi and Takeshi Nakamura, Department of Neurology, Hiroshima University Hospital for advising as to diagnosis and treatment. They thank Hiroki Ueno, Department of Neurology, Hiroshima University Hospital for advising as to imaging findings. The authors also thank Takahito Okazaki, Shigeyuki Sakamoto, Yoshihiro Kiura and Kaoru Kurisu, Department of Neurosurgery, Hiroshima University Hospital for treating this case by endovascular therapy.
Footnotes
Contributors: HF and YN were involved in assessment of the neurological finding. NH and MM were involved in providing advise for diagnosis and treatment of the patient. HF wrote the first draft of the manuscript. All authors contributed to drafting of the article or revising it critically for important intellectual content and gave final approval of the version to be published.
Competing interests: None.
Patient consent: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Awad IA, Little JR, Akarawi WP, et al. Intracranial dural arteriovenous malformations: factors predisposing to an aggressive neurological course. J Neurosurg 1990;72:839–50 [DOI] [PubMed] [Google Scholar]
- 2.Newton TH, Cronqvist S. Involvement of dural arteries in intracranial arteriovenous malformations. Radiology 1969;93:1071–8 [DOI] [PubMed] [Google Scholar]
- 3.Okuizumi K, Watanabe K, Yamazaki M, et al. A case of dural arteriovenous malformation associated with progressive dementia showing marked improvement with endovascular treatment. Rinsho Shinkeigaku 1998;38:112–17 [PubMed] [Google Scholar]
- 4.Matsuda S, Waragai M, Shinotoh H, et al. Intracranial dural arteriovenous fistula (DAVF) presenting progressive dementia and parkinsonism. J Neurol Sci 1999;165:43–7 [DOI] [PubMed] [Google Scholar]
- 5.Hamada Y, Yamakawa Y, Fukui M. A case of dural arteriovenous fistula in the anterior cranial fossa presenting frozen gait. Jpn J Neurosurg 2003;12:798–802 [Google Scholar]
- 6.Lee PH, Lee JS, Shin DH, et al. Eur parkinsonism as an initial manifestation of dural arteriovenous fistula. J Neurol 2005;12:403–6 [DOI] [PubMed] [Google Scholar]
- 7.Kajitani M, Yagura H, Kawahara M, et al. Treatable fluctuating Parkinsonism and dementia in a patient with a dural arteriovenous fistula. Mov Disord 2007;22:437–9 [DOI] [PubMed] [Google Scholar]
- 8.Miura S, Noda K, Shiramizu N, et al. Parkinsonism and ataxia associated with an intracranial dural arteriovenous fistula presenting with hyperintense basal ganglia in T1-weighted MRI. J Clin Neurosci 2009;16:341–3 [DOI] [PubMed] [Google Scholar]
- 9.Nogueira RG, Baccin CE, Rabinov JD, et al. Reversible parkinsonism after treatment of dural arteriovenous fistula. J Neuroimaging 2009;19:183–4 [DOI] [PubMed] [Google Scholar]
- 10.Netravathi M, Pal PK, Bharath RD, et al. Intracranial dural arteriovenous fistula presenting as parkinsonism and cognitive dysfunction. J Clin Neurosci 2011;18:138–40 [DOI] [PubMed] [Google Scholar]
- 11.Hattori T, Takeuchi T, Kabeya R, et al. Transverse-sigmoid sinus dural arteriovenous fistula presenting with parkinsonism. Neurol Med Chir 2013;53:224–7 [DOI] [PubMed] [Google Scholar]
- 12.Cognard C, Casasco A, Toevi M, et al. Dural arteriovenous fistulas as a cause of intracranial hypertension due to impairment of cranial venous outflow. J Neurol Neurosurg Psychiatry 1998;65:308–16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gandhi D, Chen J, Pearl M, et al. Intracranial dural arteriovenous fistulas: classification, imaging findings, and treatment. Am J Neuroradiol 2012;33:1007–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van Dijk JM, terBrugge KG, Willinsky RA, et al. Clinical course of cranial dural arteriovenous fistulas with long-term persistent cortical venous reflux. Stroke 2002;33:1233–6 [DOI] [PubMed] [Google Scholar]
- 15.Reichenbach JR, Venkatesan R, Schillinger DJ, et al. Small vessels in the human brain: MR venography with deoxyhemoglobin as an intrinsic contrast agent. Radiology 1997;204:272–7 [DOI] [PubMed] [Google Scholar]
- 16.Reichenbach JR, Jonetz-Mentzel L, Fitzek C, et al. High-resolution blood oxygen-level dependent MR venography (HRBV): a new technique. Neuroradiology 2001;43:364–9 [DOI] [PubMed] [Google Scholar]
- 17.Tsui YK, Tsai FY, Hasso AN, et al. Susceptibility-weighted imaging for differential diagnosis of cerebral vascular pathology: a pictorial review. J Neurol Sci 2009;287:7–16 [DOI] [PubMed] [Google Scholar]