Abstract
Many studies report an association of cognitive and social experiential factors and related traits with dementia risk. Further, many clinical-pathologic studies find a poor correspondence between levels of neuropathology and the presence of dementia and level of cognitive impairment. The poor correspondence suggests that other factors contribute to the maintenance or loss of cognitive function, with factors associated with the maintenance of function referred to as neural or cognitive reserve. This has led investigators to examine the associations of cognitive and social experiential factors with neuropathology as a first step in disentangling the complex associations between these experiential risk factors, neuropathology, and cognitive impairment. Despite the consistent associations of a range of cognitive and social lifestyle factors with cognitive decline and dementia risk, the extant clinical pathologic data finds only a single factor from one cohort, linguistic ability, related to AD pathology. Other factors, including education, harm avoidance, and emotional neglect, are associated with cerebrovascular disease. Overall, the associations are weak. Some factors, such as education, social networks, and purpose in life modify the relation of neuropathology to cognition. Finally, some factors such as cognitive activity appear to bypass known pathologies altogether suggesting a more direct association with biologic indices that promote person-specific differences in reserve and resilience. Future work will first need to replicate findings across more studies to ensure the veracity of the existing data. Second, effort is need to identify the molecular substrates of neural reserve as potential mediators of the association of lifestyle factors with cognition.
Keywords: Aging, Dementia, Risk Factors, Neuropathology, Neural Reserve, Epidemiology
Introduction
It has long been known that the common neuropathologies associated with dementia, including Alzheimer’s disease (AD), cerebrovascular disease (CVD), and Lewy body disease (LBD) can be found in the brains of persons without obvious dementia [30, 67, 130, 131]. These findings have been replicated and extended in numerous clinical-pathologic studies across a wide range of different cohort studies all with their distinctive strengths and weaknesses. Significant amounts of AD, CVD and LBD can be found in the brains of up to a third of persons without dementia and en even greater proportion of those with mild cognitive impairment (MCI) prior to death [13, 21, 22, 35, 68, 94, 112, 124, 132]. Overall, the discordance between neuropathology and cognitive impairment constitutes prima facie evidence for the role of some type of neural, brain or cognitive reserve [43, 85, 107, 125, 155].
Reserve likely has both structural and functional components. Structural or “neurocentric” components include macrostructure such as head circumference or intracranial volume [16, 51, 114, 115] which reflect brain development. Generally, these studies have replicated a link between smaller cranial size and greater dementia risk, especially in women. There are also microstructure components such as synaptic proteins, neuronal density, and neuronal hypertrophy. These too have been associated with cognition and appear to account in part for neural reserve [3, 35, 59, 61, 62, 100, 151]. The functional component is much more difficult to obtain from post-mortem tissue. Some efforts are being deployed in this direction For example, one group has reported the functional biochemical activation of downstream signaling molecules in postmortem brain tissue to BDNF or insulin stimulation [128, 140]. Another approach is to use psychosocial variables to identify lifestyle factors clustered around complex mental stimulation (e.g., cognitive lifestyle [134]) that predict the rate of cognitive decline and AD risk. Years of formal education was among the first cognitive or social lifestyle factors associated with the prevalence of dementia and thought to be a proxy for some type of neural reserve [159]. The association between education and dementia now has been replicated in many, though not all studies, and has been extended to include incident disease [5, 15, 25, 26, 44, 79, 91, 93, 95, 126, 135, 143, 156]. The reasons for the discrepant findings are numerous. In a quantitative sensitivity analysis of cohort studies that examined education and incident dementia, the overall dementia case-rate for the cohort was highly explanatory – cohorts with higher incidence rates tended to find a protective effect of more education on dementia risk compared to those with low incidence rates [133].
The initial focus on education has since expanded to other related constructs including occupational complexity, linguistic ability, bilingualism, and cognitive, leisure and social engagement, all of which have been linked to dementia, in addition to related personality characteristics [15, 25, 45, 48, 58, 78, 105, 109, 117, 134, 138, 139, 154]. Like education, the association of cognitive and social lifestyle factors with cognitive decline and dementia risk has been interpreted as evidence of reserve. The neuropathologic and neurobiologic mechanisms linking experiential factors with dementia represent a new area of active investigation.
Incorporating Neuropathology into Studies of Lifestyle and Cognition
This review synthesizes the extant data regarding potential neuropathologic bases that underlie the association of cognitive and social lifestyle experiences with cognitive decline and dementia. Nine studies were identified that link one or more cognitive or social lifestyle experiences to neuropathology and cognition (Table 1). To our knowlegde, this represents all of the published data on this specific topic to date.
Table 1.
List of clinical-pathologic studies linking cognitive and social activities to neuropathology and cognition in late life.
| Population-based prospective cohort studies |
| Medical Research Council Cognitive Function and Ageing Study (MRC CFA); England and Wales [21] |
| Cambridge City over-75s Cohort study (CC75); England [47] |
| Vantaa 85+; Finland [92] |
| Epidemiological Clinicopathological Studies in Europe (EClipSE) study (MRC CFA, CC75, and Vantaa 85+) [44] |
| Community-based cohort studies of special populations |
| Rush Memory and Aging Project (MAP); Northeastern Illinois [11] |
| Nun Study; School Sisters of Notre Dame, USA [119] |
| Religious Orders Study (ROS); more than 40 Religious Orders, USA [10] |
| Retrospective population-based study |
| Brazilian Aging Brain Aging Study Group (BABSG); Brazil [53] |
| Clinic-based studies |
| Dementia Study Project (DSP); University of Western Ontario, Canada [57] |
| National AD Coordinating Center (NACC); 27 AD research centers, USA [6] |
Two studies of education are clinic-based. These recruited either cases only (DSP [57]) or cases and controls (NACC [6]) from different sources, i.e., cases evaluated at a specialized dementia center and controls recruited through a variety of recruitment strategies. These studies are subject to the biases of studying risk factors in clinic-based studies using different schemes to identify cases and controls. One is a retrospective population-based autopsy study (BABSG [53]) with all clinical data, including dementia status, obtained after death via structured interview with knowledgeable informants. These studies all suffer from different selection biases and variable autopsy rates with largely specified, if any, denominators (i.e., X autopsies of Y deaths) that allow comparing the characteristics of those who came to autopsy to those who did not [113, 123].
Ideally, the neuropathologic biospecimens necessary to rigorously investigate the associaitions of lifestyle factors across the lifecourse should come from well established, prospective, community-based cohort studies with lifestyle factors determined prior to dementia onset and participants having long follow-up phases to capturing cognitive decline and incident dementia prior to autopsy. However, few such studies exist that meet these criteria. We found three European studies that are truly population-based (MRC, CC75, Vantaa [21, 47, 92]). These lend their findings to greater generalizability which is a major strength. However they tend to have relatively low autopsy rates which is a potential weakness. This is outweighed in large part by the ability to carefully examine and if necessary adjust for differences between those who did and did not come to autopsy. The remaing three are community-based volunteer cohort studies, one of lay persons (MAP [11]) and two restricted to Catholic clergy (Nun Study, ROS [10, 119]). These studies have the advantage of very high clinical follow-up and autopsy rates, thus minimizing biases that can be introduced in studies with occasional autopsies that may not be representative of the overall cohort under study. Thus, community-based studies with high autopsy rates have excellent internal validity. However, they are limited in the inferences that can be drawn to the community because of the inherent selection bias due to their volunteer nature. In particular, they may be enriched for persons with a family history of dementia and thus differ genetically which has the potential to alter the relationship between the lifestyle factors and neuropathology or cognition or both.
All of the community-based studies are subject to all of the potential biases and limitations of causal inferences inherent in even the most well conducted analytic epidemiologic studies. In particular, persons who are more educated, and cognitively and socially engaged differ for from those who are not by many factors leaving the possibility of residual confounding. The exposure factors tend to be studied one by one, while controlling for the others when possible, yet they may not be independent. Only one study to date has made a composite of some of these variables [134]. However, these epidemiologic studies are subject to a few additional limitations and potential biases as a result of incorporating neuropathology. First, we are interested in understanding the neuropathologic basis of the association of several cognitive and social lifestyle experiences to cognitive decline and dementia, i.e., is neuropathology mediating or modifying the association of lifestyle factors with cognition. However, the brain is obtained at the end of the study. Thus, the causal inference that we wish to make results in presenting the data in a temporal order that does not correspond with reality. Thus, the potential for reverse causality must be explored very carefully. Second, as discussed, one needs to pay careful attention to understanding differences between those who did and did not come to autopsy as most studies show important differences that are related to some of the exposure variables of interest. Finally, death itself is both informative censoring and a competing risk for dementia, and is related to some of the lifestyle factors of interest [5, 88, 89]. All of these factors should lead one to be skeptical of findings and to examine the methods of each study in detail in order to arrive at the appropriate conclusion and inference.
It is essential that findings be replicated in multiple studies as is necessary for all data from analytic epidemiologic studies. Unfortunately, the number of studies so far is small and the range of lifestyle factors available in the cohorts is limited. In fact, most data on lifestyle factors come from a single study. (Table 2). The exceptions are education which has been examined in several studies and cognitive activity which has been studied in two.
Table 2.
List of cognitive and social activities examined by cohort.
| Lifestyle Factors | EClipSE | BABSG | Nun Study |
ROS/MAP | DSP | NACC | |||
|---|---|---|---|---|---|---|---|---|---|
| MRC | CC75 | Vantaa | ROS | MAP | |||||
| Education | X | X | X | X | X | X | |||
| Linguistic ability | X | ||||||||
| Late-life Cognitive activity | X | X | |||||||
| Early-life cognitive activity | X | ||||||||
| Social networks | X | ||||||||
| Loneliness | X | ||||||||
| Purpose in life | X | ||||||||
| Harm avoidance | X | ||||||||
| Emotional neglect | X | ||||||||
| Conscientiousness | X | ||||||||
| Cognitive lifestyle composite | X | ||||||||
Education
The Dementia Study Project [32] was the first to examine the relation of education to neuropathology. The sample included 95 patients with dementia evaluated at a tertiary-care dementia clinic that came to autopsy. The authors found associations between education and macroscopic infarctions with a trend for more white matter lesions. They failed to find an association between education and brain weight, neuritic plaques or neurofibrillary tangles, or nigral or limbic Lewy bodies.
The relation of education to neuropathology was also examined in 60 sisters in the Nun Study [84]. The authors failed to find an association between education and the likelihood of meeting pathologic criteria for AD independent of clinical status. They did not report on the association with vascular disease.
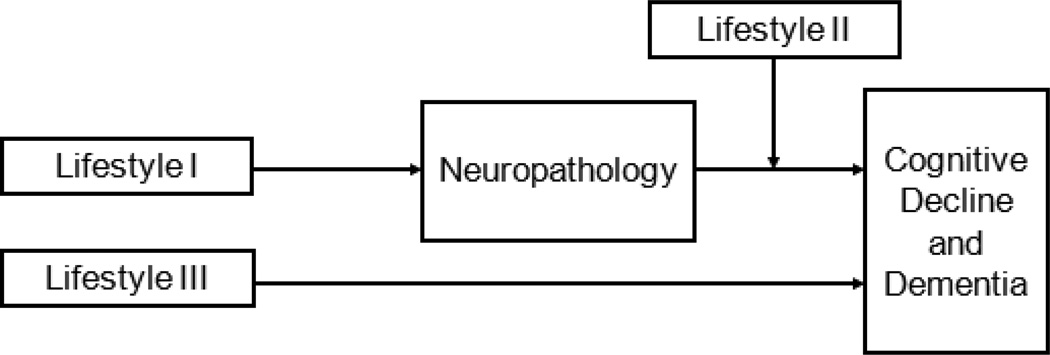
Two papers from the Religious Orders Study examined the relation of education to AD pathology and cognition. The first quantified the density of AD plaque and tangle pathology identified by classic silver staining in 130 autopsied participants [14]. The number of persons with dementia was not provided but the mean minimental status examination (MMSE) was only 23.1 suggesting many were without dementia. Like the prior two studies, the authors failed to find a direct association between education and measures of AD pathology. However, the authors further examined the extent to which education modified the relation of AD pathology to dementia (i.e., Fig. 1, Lifestyle III). Here the authors found strong evidence of effect modification such that more years of formal education reduced the deleterious impact of AD pathology on the odds of dementia. Interestingly, the findings were robust for both neuritic and diffuse plaques but not for neurofibrillary tangles. Similar relationships were seen with a measure of global cognition proximate to death. A subsequent immunohistochemical study in the same cohort quantified amyloid-β load based on image analysis and tau tangle density based on stereological assisted counting for density in 156 participants [12]. Again, the mean MMSE was 23.0. The authors found that education strongly modified the relation of amyloid pathology to cognition but not tau tangle density. The authors hypothesized that the beneficial effect of education was likely early in the disease process when amyloid is more prominently related to cognition [13].
Figure 1.
Schematic representing three potential classes of mechanisms linking cognitive and social lifestyle factors with neuropathology and dementia. Lifestyle I is a direct disease effect on neuropathology such that it is directly associated with more or less pathology. Lifestyle II represents a modulatory effect such that the experiential factor alters the relation of pathology to cognition, i.e., increases or decreases the probability of dementia for any given level of neuropathology. Lifestyle III represents a pathway linking lifestyle to dementia that is unrelated to classic neuropathology.
The relation of education to neuropathology was examined using data from the National AD Coordinating Center (NACC). The first study was restricted to 2,372 persons, 30% without dementia, over age 65 who were evaluated within a year of life and met pathologic criteria for AD [103]. As in the above studies, the authors found that level of eduation was associated with dementia consistent with a main effect of education on dementia status However, in this study limited to persons meeting pathologic criteria for AD, the authors failed to replicate the interaction of education with AD pathology and cognition seen in the Religiouis Orders Study. The same authors conducted a follow-up study restricted to 1563 NACC participants, 18% without dementia, with semi-quantitative measures of AD pathologic burden [102]. Interestingly, they found an interaction of eduation with pathology, but it was in the opposite direction to that reported in the Religious Orders Study. In other words, education increased the likelihood that neuritic plaques were associated dementia. The effect modification was not seen for diffuse plaques or tangles. A third NACC study by a different group of investigators used data from 2,051 participants with MMSE available within 2 years of death (mean MMSE = 11.5) [69]. The findings were similar to the prior study such that edcuation was associated with a much higher MMSE among those with little AD pathology, but not among those with severe AD pathology.
The associations of education, neuropathology, and dementia were also examined using data from the Epidemiological Clinicopathological Studies in Europe (EClipSE) [42]. The authors used data from 872 persons, 44% of whom were dementia free. They did not find a main effect of education on brain weight, neurodegenerative or CVD markers. However, education decreased the odds of dementia among those with low but not high brain weight. By contrast, education reduced the odds of dementia for low but not high Braak stage, similar to the NACC data.
The most recent study used data from the Brazilian Aging Brain Study Group (BABSG) [46]. Unlike the prior studies from North America and Western Europe with relatively high levels of education, this study included persons with extremely low levels of education including many without any formal education. Of 675 persons, 65% had a clinical dementia rating (CDR) scale <1 and 19.3% had no formal education while 60.7% had only 1–4 years of formal schooling. Education modified the relation of burden of CVD on CDR such that among those with more education, CVD was associated with a lower CDR, i.e., reduced odds of dementia. However, education did not modify the relation of AD pathology and Lewy bodies to CDR.
Summary
Overall, the data suggest that education is related to cognition and dementia status irrespective of the presence, amount, or type of pathology. Some studies found that low education was either directly related to CVD or modified the relation of CVD to dementia status. Education was not directly related to AD pathology. However, several studies found that education modified the the relation of AD pathology on dementia and level of cognition. If so, the data sugggest the benefical effect may be early in the disease process. However, with sufficient AD pathology it appears that there is no longer a beneficial effect of education and the effect modification may parodoxically be in the opposite direction of what what would be expected. This crossover effect is similar to that of education with cognitive decline seen clinically, whereby education is associated with a slower rate of decline prior to but a faster rate of decline following dementia onset. Thus, education appears to not only delay the onset of cognitive decline but is also associated with a more rapid progression once decline commences and a greater risk of adverse health outcomes [108, 127]. Recent studies have examined this elegantly with random change point models [54, 157] as well as multistate modeling [76]. The findings illustrate that education is initially associated with a slower rate of cognitive decline, delays the onset of the change point or recovery from an impaired cognitive state, but is then associated with a more rapid decline. The primary explanation for this effect is that more education (and other reserve-related lifestyle activities) may result in the compression of cognitive morbidity in later life [75]. Compression of morbidity results from extending disability free survival and compressing morbidity into fewer years at the end of life [50] While the data from education and other related variables can be viewed in this context, the concept is not universally accepted [29]. The concept of compression of morbidity also may be related to terminal decline where one can show a marked increased rate of cognitive decline in the years prior to death [86, 129, 144]. In fact, there is evidence that education can delay the onset of terminal decline [87]. Finally, the findings suggest that the full range of education levels have a beneficial effect from just a couple of years in BABSG to many years in the Nun Study and the Religious Orders Study. Thus, while education is likely a proxy for many factors related to socioeconomic status that confer a range of health benefits, the beneficial effects do not appear to be present across a wide range of levels of educational attainment.
Linguistic Ability
The relationship of early life linguistic ability to measures of neuropathology was examined in the Nun Study [120]. The authors took advantage of archival autobiographies written by the sisters at the time of their final vows at an average age of 22. They investigated two linguistic measures derived from the essays of 93 sisters from the order in Milwaukee: idea density (i.e., the average number of ideas per 10 words) and grammatical complexity (i.e., sophistication of sentence structure). Both measures were strongly related to MMSE scores in late life. Further, among 14 sisters who came to autopsy, low idea density was associated with AD pathology, especially neurofibrillary tangle density. By contrast, grammatical complexity was not related to neuropathology. A subsequent publication nearly a decade later, with 90 sisters coming to autopsy, confirmed these associations and also demonstrated associations with lower brain weight and more cerebral atrophy [99]. A separate study of 74 sisters failed to find an association between idea density and the severity of atherosclerosis or macroscopic infarctions [84]. This is the only study of which we are aware that found a direct association with measures of AD pathology. Unfortunately, the availability of written essays with uniform instructions is unlikely to be available in any other study. Thus, the findings are likely only to be extended by further work in this unique cohort.
Cognitive Activity
The finding that education buffers the deleterious cognitive effects of AD and possibly CVD pathology raises the possibility that other experiential factors may also have a beneficial effect. One factor is engagement in cognitively stimulating activities. In one study of more than 700 participants in the Rush Memory and Aging Project, the investigators created measures of past and current life participation in cognitively stimulating activities [152]. In this cohort and the Religious Orders Study, an activity was considered cognitive if it included information processing as a central feature, and if physical demands and social requirements were minimal [145]. The measures covered a range of activities that varied by the perceived degree of mental stimulation from puzzles to watching television and listening to the radio. Past life included items about childhood activities (ages 6 and 12), young adulthood (age 18), and middle age (age 40). Current activities were those endorsed at study baseline and annually thereafter. The authors first demonstrated that both past and current activities predicted incident AD, incident MCI, and cognitive decline in separate models, controlling for education and other socioeconomic indices. However, the effect of past activity was attenuated and no longer significant when both past and current activities were examined in a single model. The authors subsequently examined the relation of past and current activities (both at baseline and average over all study years) to amyloid load, tangle density, Braak stage, Lewy bodies and macroscopic infarctions in more than 100 participants and failed to find any associations. In a subsequent paper, the authors modeled the relation of past and current activities to cognitive change over multiple years prior to death controlling for education, amyloid load, tangle density, Lewy bodies and both macro- and microscopic infarctions [149]. In these analyses, both past and current activities were associated with a slower rate of cognitive decline and together accounted for 14% of the variability of cognitive change over several years prior to death suggesting that it is a very important factor.
Another study used latent variable modeling to combine education, cognitive activities and other socioeconomic indices with neuropathologic data from more than 650 persons in the Religious Orders Study and Rush Memory and Aging Project [98]. The authors created a latent neural reserve variable based on the residual term when regressing cognitive function on neuropathologic indices including neuritic and diffuse plaques, tangles, CD, and LBD. They subsequently examined predictors of reserve and found that midlife cognitive activity was the strongest predictor of reserve followed by late life (current) cognitive activity.
Summary
Overall, the data suggest that cognitive activity across the lifespan is related to cognition and dementia status regardless of the presence and amount of pathology. Further, it appears to capture a significant amount of the variability of decline making it worthy of future study.
Social Engagement
Several reports from two cohort studies have examined the relation of various aspects of social engagement and related personality characteristics to measures of neuropathology and dementia status.
The first study used data from 89 participants in the Rush Memory and Aging Project to investigate the potential protective effect of social networks [9]. Social networks were assessed by standard questions regarding the number of children, family and friends with whom the participant feels close to and how often they interacted with them. Impoverished social networks are known to be associated with a wide range of adverse health outcomes. While there was no main effect of social networks on either cognition or amyloid load or tangle density, social networks strongly modified the relation of both amyloid and tangles to cognition proximate to death such that amyloid and tangles had little effect on cognition in the presence of a large network. The finding was robust and persisted after controlling for cognitive, physical, and social activity, depressive symptoms, and number of chronic diseases. It should be noted that the average MMSE proximate to death was 24.0 suggesting that these persons had a relatively high level of cognition. Like education, it’s possible that in the presence of more severe cognitive impairment due to high levels of pathology the effect would be attenuated.
Another report using data from the same cohort study examined the relation of loneliness, a measure of perceived social isolation, to cognitive outcomes and AD pathology [150]. The authors used data from more than 800 participants who completed a loneliness scale and found that loneliness was related to the development of incident AD and to rate of cognitive decline. The finding was robust and persisted after controlling for numerous confounders including social networks, and social, cognitive, and physical activities, disabilty and depressive symptoms. Interestingly, in 135 persons who came to autopsy, loneliness was not related to amyloid load or tangle density, or cerebral infarctions.
A third set of reports from the same cohort examined the relation of purpose in life – the psychological tendency to derive meaning from one’s experiences and a sense of goal directedness to guide behavior – and AD pathology [17]. The authors first demonstrated that purpose in life, assessed in more than 900 participants, was associated with incident AD, incident MCI, and cognitive decline [18]. The finding was robust to controlling for social networks, neuroticism, depressive sysmptoms, and number of comorbidities. Subsequently, they showed that purpose in life modified the association of a global measure of AD pathologic burden and change in cognition over multiple years prior to death [19]. Interestingly, the association was strong for tangle density but not for amyloid load.
A fourth set of analyses from the same cohort examined the relation of harm avoidance, a behavioral trait related to behavior inhibition comprised of four subscales, to dementia risk and neuropathology. In nearly 800 persons followed longitudinally, harm avoidance was associated with risk of AD [146]. The finding was seen in three of the four subscales, anticipatory worry, fear of uncertainty, and fatigability; it was not found with shyness. Harm avaoidance was also associated with risk of MCI and with change in cognitive function across multiple domains. Among more than 100 persons who came to autopsy, it was not associated with measures of AD pathology. In a follow up study with more than 250 autopsies, the authors found associations with both macroscopic and microscopic subcortical infarctions [148].
A final set of analyses using data from more than 200 participants from the same cohort found that emotional neglect, a facet of childhood adversity, was directly associated wih both micro- and macrocopic infarctions, particularly cortical microinfarcts and subcortical macroscopic infarcts [147]. Analyses were robust to controlling for early and late life socioeconomic status, body mass index, physical activity, smoking, diabetes, and blood pressure
A study using data nearly 1000 particpants in the Religious Orders Study examined the association of conscientiousness to cognitive outcomes and neuropathology [153]. Conscientiousness - a component of the 5-factor model of personality, also known as will, work, and dependibitity - is linked to impulse control and goal directedness. Higher consientousness was found to be associated with a reduced risk of incident AD and incident MCI, and to a slower rate of cognitive decline. It had no main effect on pathology among 324 persons with autopsy at the time. However, it had an unanticipated interaction with pathology such that higher conscientiousness increased the impact of tangle density and macroscopic infarctions on cogntive function. There were no associations with amyloid or Lewy bodies.
Summary
Overall, these studies suggest complex relationships between several social experiential factors and related personality constructs to brain pathology and dementia. Some, such as social networks appear to reduce the deleterious effects of AD pathology. However, loneliness or perceived social isolation appears to have an effect irrespective of AD pathology. Harm avoidance, related to behavioral inhibition, and emotional neglect, a facet of childhood adversity, had a direct association with cerebral infarctions. Finally, conscientiousness, a personality component related to goal directedness is associated with a lower incidence of dementia and a slower rate of cognitive decline but increases rather than decreases the relation of AD and infarct pathology on cognition. However, it should be noted that these studies are in need of replication. As there are few cohort studies with neuropathology that include these indices, it is likely that replication will come from neuroimaging studies. Such work is already emerging [24, 33, 66, 77, 110]. Finally, whether these predisposing personality or personal style factors represent reserve-related processes or rather health-related background factors is still a matter of debate and further work.
Cognitive/Social Lifestyle Composites
Investigators with the United Kingdom Medical Research Council Cognitive Function and Ageing Study (MRC-CFAS) created a composite lifestyle variable based on educational attainment, occupational complexity, and social engagement. Individuals were classified as high, medium or low based on sex-specific tertiles. These indices were all previously shown to predict incident dementia in the entire cohort [134]. A total of 329 persons came to autopsy, 153 of whom were without dementia. Pathologic assessments were based on the Consortium to Establish a Registry of Alzheimer’s Disease (CERAD). The lifestyle groups did not differ by semiquantitative indices of AD. However, men with an engaged lifestyle had less cerebrovascular disease, and women had greater brain weight. No group differences were evident in hippocampal neuronal density. In a substudy of Brodmann area 9 in 72 matched pairs of high and low engagement, cognitively active individuals had significantly greater neuronal density and greater cortical thickness. Hence, this study is one of the few to show a potential disease-modifying mechanism in relation to CVD (in men), and possible correlates of compensatory prefrontal changes in both men and women.
Potential Pathways Linking Lifestyle Factors to Neuropathology and Cognition
Overall, there is published data on nine cognitive and social experiential and related factors associated with dementia risk, and their association with measures of common dementia neuropathologies. Conceptually, the data suggest that there are at least three pathways by which measures in the brain, such as neuropathology, may link lifestyle factors to cognition. For discussion, we designate them as lifestyle factors I, II, and III (Fig 1). These lifestyle factors are not mutually exclusive and it’s likely that more than one lifestyle factor will operate through more than one pathway. However, the schematic is useful for helping frame our thinking regarding how we proceed with using our findings to identify potential nodes for intervention and the prevention of clinical disease.
Lifestyle I is a direct disease effect on neuropathology such that it is directly associated with more or less pathology. Remarkably, studies failed to find that many of these factors related to clinical AD risk were also associated with measures of AD pathology. One factor, early life linguistic ability, was associated with a measure of AD pathology, and two factors, harm avoidance and emotional neglect, were associated with cerebrovascular disease, albeit the effects were small suggesting other pathways. One study limited to clinic patients enrolled with dementia found an association of education with infarctions. A second study that created a composite of educational attainment, occupational complexity, and social engagement also found an association with CVD that was limited to men. The lack of an association of many factors with measures of AD neuropathology does not imply that the pathology is not measured well. Studies have reported very robust associations with a range of genetic risk factors and measures of AD pathology, and some have found much greater power with AD pathology relative to clinical measures [31, 8, 118]. It is more like that the other experiential factors do not directly impact the accumulation of AD pathology but affect brain function via other mechanisms.
Lifestyle II represents a modulatory effect such that the experiential factor alters the relation of pathology to cognition, i.e., increases or decreases the probability of dementia for any given level of neuropathology. Four factors that modify the relation of neuropathology to cognition have been identified including education, social networks, purpose in life, and conscientiousness. One study of persons with very low levels of education found an effect modification of education on infarctions, and one other factor, conscientiousness, also modified the relation of infarctions on the odds of dementia. The effect modification provides evidence of person-specific differences in the response of the brain to injury due to AD and CVD. The neurobiologic basis of these plastic response remains to be determined.
Lifestyle III represents a pathway linking lifestyle to dementia that is not directly related to classic neuropathology and does not modify the relation of neuropathologic indices to cognition. Early and late cognitive activity and self-perceived social isolation or loneliness, were in this category. Further, in the study of neuronal density and cortical thickness in matched pairs of high and low engagement participants (based on the composite of educational attainment, occupational complexity, and social engagement), the investigators reported the more engaged group had a higher neuronal density and greater cortical thickness. To our knowledge, this is the only study to date to report an association between a cognitively and socially active lifestyle with a potential structural marker of brain plasticity.
Life Experiences and Animal Models
Animal models are useful for investigating the neurobiology of environmental manipulations at behavioral, neurophysiological, cellular and molecular levels under highly controlled conditions that are not feasible in humans. In rodents, various interventions such as environmentally enriched housing, isolated housing, and chronic variable physical and social stressors (e.g., social defeat paradigms) have been employed to represent, in part, the enhancement or deprivation of constructs related to the cognitive and social activities in humans. The results of these studies provide important clues to the neurobiological basis of these associations which can be used to build testable hypotheses in human clinical-pathologic studies [135]. It is important to note, however, that the housing conditions of experimental animal models do not replicate the nature and complexity of the wild in which these species evolved. Indeed, standard “control” laboratory housing conditions represent what would be severe sensory and social deprivation for humans, with enhanced environments only partly addressing this deprivation.
Environmental enrichment manipulations have included large cages with more animals, running wheels, ramps, various toys and materials for playing and novelties introduced to stimulate exploration. Environmental enrichment has been shown to improve cognitive behavior in juvenile and adult mice, rats and other animals. This probably also holds true for age-related cognitive decline in old rodents, especially when the enrichment is initiated prior to mid-life [49, 122].
Enrichment paradigms have been reported to significantly alter brain chemistry, structure and function. Early studies showed that enrichment improved brain cholinesterase activity [70] and increased cerebral cortex thickness, neuron and glial cell numbers [1, 34]. Subsequent studies have shown that enrichment increases synapses and dendrite complexity [23, 52], neurophysiological synaptic plasticity, cortico-cortical interactions and various molecular mechanisms of synaptic neurotransmission [36, 40, 60, 64, 90], increase neurogenesis and brain neurotrophic factors such as brain-derived neurotrophic factors [63, 104, 137], and alter the brain’s transcriptome [97], proteome [83], and lipidome [106]. A major limitation in interpreting the results of these studies is that it is difficult to distinguish the beneficial effects of physical exercise in an enriched environment from those due to the cognitive or social simulation. Interestingly, differences have been reported in behavioral and pathological effects of exercise when the exercise is voluntary as opposed to forced [158]. Another limitation is that these studies are not akin to randomized trials as they offer the opportunity for animals to engage in these activities based on their own motivation. Despite the use of identical strains and handling there are inter-individual differences among animals in terms of how frequently they take advantage of the opportunities. In this sense they are subject to many of the same limitations of the observational studies in humans described above.
There have also been investigations on how environmental enrichments affect cognitive behaviors as well as the accumulation of amyloid- and tau pathology in transgenic mouse models of AD. Enrichment improves them compared to routinely housed transgenic mice [2, 27, 28, 136]. However, in contrast to clinicalpathological studies in humans which mostly have found no direct associations of education, personality factors, or cognitively stimulating activities on plaque or tangle pathology, most (though not all) of the published AD mouse model studies have reported more amyloid pathology with social isolation and less amyloid accumulation with environmental enrichment [2, 28, 37, 38]. The discrepancy is not necessarily surprising. For example, there is a long history of therapeutics clearing amyloid-β in transgenic mice that do not replicat in humans. Thus, the model most likely has limitations that leads to lack of replication. Alternatively, it is also possible that the transgenic models captured the disease early in the biologic cascade relative to the human studies which often includes persons with moderate to severe dementia. In addition, there may be publication bias with those failing to find an association not inclined to publish a null effect. This, of course, is also an issue for the human studies. Several other cohort studies have pathology and autopsies and have not published on education, which is available in all studies, in addition, perhaps, to one or more additional related variables.
Summary and Future Directions
Over the past two decades, the addition of brain autopsy to prospective cohort studies has provided an opportunity to study the impact of a wide range of experiential and psychological factors on the development of clinically diagnosed dementia and to the common brain pathologies that cause dementia, including AD, CVD, and LBD. Some of these studies start with a population-based sampling scheme (e.g., MRC-CFAS), some are community-based cohorts of lay persons (e.g., Memory and Aging Project), and others are cohorts of special populations (e.g., Religious Orders Study), each of which has its own set of strengths and limitations. Overall, the results of these studies suggest that there is only a small direct relation between these experiential and lifestyle factors and the development of AD pathology despite strong associations with risk of AD dementia. While some direct associations have been reported for cerebrovascular disease, these too are modest in magnitude. Overall, the data suggest that other factors must account for the associations of cognitive and social experiential risk factors with dementia.
Preclinical studies suggest that a number of structural and molecular factors that subserve cognition are related to manipulation of the environment. These include structural changes such as neurogenesis and synaptic density along with functional changes in molecular signaling and neurophysiology. Clinical pathologic studies of humans followed prospectively are also investigating the structural elements that subserve cognition, including neurons, synapses, dendrites, and dendritic spines [3, 7, 55, 59, 61, 62, 65, 100, 111, 151]. Only one study, using a composite measure of educational attainment, occupational complexity, and social engagement, found relation to neuronal density, a key structural element subserving cognitive processes. We suspect that many factors account for lack of association with these indices. Studying these issues in human populations is exceedingly complex due to the many uncontrolled variables related to the multi-dimensional lifestyle factors themselves, the noise in the cognitive measures, the presence of co-morbidities, and peri- and post-mortem factors that might influence the status and integrity of the molecular markers and brain tissue. A further complicating issue is controlling for atrophy when measuring density of structures that are hypothesized to be degenerating and thus “disappearing.” Measures of neuropathology accumulate while the tissue atrophies. This results in magnifying the density, but we can assume it maintains rank order and that the value remains useful for analyses. By contrast, loss of structural elements, e.g., numbers of neurons, synapses, dendrites, and dendritic spines, are decreasing while the tissue atrophies. The result is that the loss of these elements is obscured because the atrophy artificially leads to a higher density. A variety of approaches have been used to overcome this issue with some, such as three-dimensional unbiased counts, but these methods are highly labor intensive and not yet amenable to the high throughput needed for the large numbers that are used in epidemiologic studies. Despite some inherent biases in commonly used high-throughput approaches, a number of researchers have shown that cellular and subcellular structural indices are related to cognition. Further, as noted above, one study also found a relation between a composite lifestyle variable and neuronal density using a combination of large numbers and careful matching [134]. Although not the topic of this review, another study found a relation between dendrites and dendritic spines to a composite measure of depression and anxiety using a scales measure to account for atrophy [121]. Thus, we are optimistic that future efforts in this area will be met with some success. However, additional work will also need to examine other pathologic indices, including those related to response to injury, such as oxidative stress, and inflammation, and blood brain barrier.
Other approaches that are likely to be fruitful in the near future will take advantage of the developments in high throughput genomics and related molecular markers. A recent targeted proteomics study identified a number of factors related to resilience from AD pathology [3]. It will be interesting to see if the indices identified by this study might be related to cognitive and social experiential factors. Future work will likely employ systems biology to interrogate genomic, epigenomics, and transcriptomic datasets under development, in addition to proteomics, to identify novel molecular signals that account for the association of experiential factors with cognitive decline [31]. Epigenetic changes such as DNA methylation, histone marks (e.g., acetylation), and microRNA in particular are of great interest as evidence suggests that behavior can alter epigenomic marks and ultimately transcription. For example, preclinical studies first demonstrated that maternal behavioral influences on her pups’ behavior were associated with reversible epigenetic programming changes in adulthood [141, 142]. In the past several years, epigenetic changes have been reported in humans in relation to suicide, childhood abuse, and other early life trauma [71, 81, 82]. The relation of cognitive and social experiential factors on the epigenome is of great interest and the subject of ongoing studies.
Much work remains to be done to understand the neurobiologic and molecular pathways linking cognitive and social lifestyle factors to dementia. It is hoped that such an understanding will yield novel approaches to the maintenance of cognition in old age. While the majority of preventive measures for common degenerative diseases that cause dementia focus on the prevention or removal of pathology, the body of work summarized here suggests that preventing dementia despite the accumulation of AD and other common pathologies is a viable and potentially powerful approach for the maintenance of cognitive health. The observational studies suggest a substantial fraction of clinically manifest dementia at given ages might be averted through healthier lifestyles, some of which could well be accounted for by compensation and reserve rather than avoidance of pathology. Although the results to date are likely an overestimate due to lack of independence the evidence on terminal decline and reduction of dementia prevalence at death in those with higher education suggests the potential for compression of morbidity including shorter periods of cognitive impairment before death [9]. Recent data from some studies but not others suggest that the prevalence of dementia might be declining already, possibly as a result of secular trends, e.g., more education, lower rates of childhood infectious disease [56, 72, 73, 74, 80, 96, 101, 116]. Such findings, if confirmed and if continued, is a strong reminder that improving lifestyle may have a profound impact on the health and well being of our aging population.
Acknowledgements
This work was supported by NIH grants P30AG10161, R01AG15819, R01AG17917, R01AG39478, MV is supported by a National Health and Medical Research Council of Australia Clinical Career Development Fellowship (#1004156).
References
- 1.Altman J, Das GD. Autoradiographic Examination of the Effects of Enriched Environment on the Rate of Glial Multiplication in the Adult Rat Brain. Nature. 1964;204:1161–1163. doi: 10.1038/2041161a0. [DOI] [PubMed] [Google Scholar]
- 2.Arendash GW, Garcia MF, Costa DA, Cracchiolo JR, Wefes IM, Potter H. Environmental enrichment improves cognition in aged Alzheimer's transgenic mice despite stable beta-amyloid deposition. Neuroreport. 2004;6:1751–1754. doi: 10.1097/01.wnr.0000137183.68847.4e. [DOI] [PubMed] [Google Scholar]
- 3.Arnold SE, Louneva N, Cao K, Wang L-S, Han L-Y, Wolk DA, Negash S, Leurgans SE, Schneider JA, Buchman AS, Wilson RS, Bennett DA. Cellular, synaptic, and biochemical features of resilient cognition in Alzheimer's disease. Neurobiology of Aging. 2013;34:157–168. doi: 10.1016/j.neurobiolaging.2012.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barrett JK, Siannis F, Farewell VT. A semi-competing risks model for data with interval-censoring and informative observation: an application to the MRC cognitive function and ageing study. Stat Med. 2011;30:1–10. doi: 10.1002/sim.4071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Beard CM, Kokmen E, Offord KP, Kurland LT. Lack of association between Alzheimer's disease and education, occupation, marital status, or living arrangement. Neurology. 1992;42:2063–2068. doi: 10.1212/wnl.42.11.2063. [DOI] [PubMed] [Google Scholar]
- 6.Beekly DL, Ramos EM, Lee WW, Deitrich WD, Jacka ME, Wu J, Hubbard JL, Koepsell TD, Morris JC, Kukull WA NIA Alzheimer's Disease Centers. The National Alzheimer's Coordinating Center (NACC) database: the Uniform Data Set. Alzheimer Dis Assoc Disord. 2007;21:249–258. doi: 10.1097/WAD.0b013e318142774e. [DOI] [PubMed] [Google Scholar]
- 7.Beeri MS, Haroutunian V, Schmeidler J, Sano M, Fam P, Kavanaugh A, Barr AM, Honer WG, Katsel P. Synaptic protein deficits are associated with dementia irrespective of extreme old age. Neurobiol Aging. 2012;33:1125.e1–1128.e1. doi: 10.1016/j.neurobiolaging.2011.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bennett DA, De Jager PL, Leurgans SE, Schneider JA. Neuropathologic intermediate phenotypes enhance association to Alzheimer susceptibility alleles. Neurology. 2009;72:1495–1503. doi: 10.1212/WNL.0b013e3181a2e87d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bennett DA, Schneider JA, Arnold SE, Tang Y, Wilson RS. The effect of social networks on the relation between Alzheimer's disease pathology and level of cognitive function in old people: a longitudinal cohort study. The Lancet Neurology. 2006;5:406–412. doi: 10.1016/S1474-4422(06)70417-3. [DOI] [PubMed] [Google Scholar]
- 10.Bennett DA, Schneider JA, Arvanitakis Z, Wilson RS. Overview and findings from the Religious Orders Study. Curr Alzheimer Res. 2012;9:628–645. doi: 10.2174/156720512801322573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bennett DA, Schneider JA, Buchman AS, Barnes LL, Boyle PA, Wilson RS. Overview and findings from the rush Memory and Aging Project. Curr Alzheimer Res. 2012;9:646–663. doi: 10.2174/156720512801322663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bennett DA, Schneider JA, Wilson RS, Bienias JL, Arnold SE. Education modifies the association of amyloid, but not tangles, with cognitive function. Neurology. 2005;65:953–955. doi: 10.1212/01.wnl.0000176286.17192.69. [DOI] [PubMed] [Google Scholar]
- 13.Bennett DA, Wilson RS, Boyle PA, Buchman AS, Schneider JA. Relation of neuropathology to cognition in persons without cognitive impairment. Annals of Neurology. 2012;72:5999–5609. doi: 10.1002/ana.23654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bennett DA, Wilson RS, Schneider JA, Evans DA, Mendes de Leon CF, Arnold SE, Barnes LL, Bienias JL. Education modifies the relation of AD pathology to cognitive function in older persons. Neurology. 2003;60:1909–1915. doi: 10.1212/01.wnl.0000069923.64550.9f. [DOI] [PubMed] [Google Scholar]
- 15.Bonaiuto S, Rocca WA, Lippi A, Giannandrea E, Mele M, Cavarzeran F, Amaducci L. Education and occupation as risk factors for dementia: a population-based case-control study. Neuroepidemiology. 1995;14:101–109. doi: 10.1159/000109785. [DOI] [PubMed] [Google Scholar]
- 16.Borenstein GA, Mortimer JA, Bowen JD, McCormick WC, McCurry SM, Schellenberg GD, Larson EB. Head circumference and incident Alzheimer's disease: modification by apolipoprotein E. Neurology. 2001;57:1453–1460. doi: 10.1212/wnl.57.8.1453. [DOI] [PubMed] [Google Scholar]
- 17.Boyle PA, Barnes LL, Buchman AS, Bennett DA. Purpose in life is associated with mortality among community-dwelling older persons. Psychosomatic Medicine. 2009;71:574–579. doi: 10.1097/PSY.0b013e3181a5a7c0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Boyle PA, Buchman AS, Barnes LL, Bennett DA. Purpose in life is associated with a reduced risk of incident Alzheimer's disease among community-dwelling older persons. Archives of General Psychiatry. 2010;67:304–310. doi: 10.1001/archgenpsychiatry.2009.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boyle PA, Buchman AS, Wilson RS, Yu L, Schneider JA, Bennett DA. Effect of purpose in life on the relation between Alzheimer disease pathologic changes on cognitive function in advanced age. Archives of General Psychiatry. 2012;69:499–504. doi: 10.1001/archgenpsychiatry.2011.1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brayne C, Gao L, Dewey M, Matthews FE Medical Research Council Cognitive Function and Ageing Study Investigators. Dementia before death in ageing societies--the promise of prevention and the reality. PLoS Med. 2006;3:e397. doi: 10.1371/journal.pmed.0030397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Brayne C, McCracken C, Matthews FE. Cohort profile: the Medical Research Council Cognitive Function and Ageing Study (CFAS) Int J Epidemiol. 2006;35:140–1145. doi: 10.1093/ije/dyl199. [DOI] [PubMed] [Google Scholar]
- 22.Brayne C, Richardson K, Matthews FE, Fleming J, Hunter S, Xuereb JH, Paykel E, Mukaetova-Ladinska EB, Huppert FA, O'Sullivan A, Dening T Cambridge City Over-75s Cohort Cc75c Study Neuropathology Collaboration. Neuropathological correlates of dementia in over-80-year-old brain donors from the population-based Cambridge city over-75s cohort (CC75C) study. J Alzheimers Dis. 2009;18:645–658. doi: 10.3233/JAD-2009-1182. [DOI] [PubMed] [Google Scholar]
- 23.Briones TL, Klintsova AY, Greenough WT. Stability of synaptic plasticity in the adult rat visual cortex induced by complex environment exposure". Brain Res. 2004;1018:130–135. doi: 10.1016/j.brainres.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 24.Cacioppo JT, Norris CJ, Decety J, Monteleone G, Nusbaum H. In the eye of the beholder: individual differences in perceived social isolation predict regional brain activation to social stimuli. J Cogn Neurosci. 2009;21:83–92. doi: 10.1162/jocn.2009.21007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Callahan CM, Hall KS, Hui SL, Musick BS, Unverzagt FW, Hendrie HC. Relationship of age, education, and occupation with dementia among a community-based sample of African Americans. Arch Neurol. 1996;53:134–140. doi: 10.1001/archneur.1996.00550020038013. [DOI] [PubMed] [Google Scholar]
- 26.Cobb JL, Wolf PA, Au R, White R, D'Agostino RB. The effect of education on the incidence of dementia and Alzheimer's disease in the Framingham Study. Neurology. 1995;45:1707–1712. doi: 10.1212/wnl.45.9.1707. [DOI] [PubMed] [Google Scholar]
- 27.Costa DA, Cracchiolo JR, Bachstetter AD, Hughes TF, Bales KR, Paul SM, Mervis RF, Arendash GW, Potter H. Enrichment improves cognition in AD mice by amyloid-related and unrelated mechanisms. Neurobiol Aging. 2007;28:831–844. doi: 10.1016/j.neurobiolaging.2006.04.009. [DOI] [PubMed] [Google Scholar]
- 28.Cracchiolo JR, Mori T, Nazian SJ, Tan J, Potter H, Arendash GW. Enhanced cognitive activity--over and above social or physical activity--is required to protect Alzheimer's mice against cognitive impairment, reduce Abeta deposition, and increase synaptic immunoreactivity. Neurobiol Learn Mem. 2007;88:277–294. doi: 10.1016/j.nlm.2007.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Crimmins EM, Beltrán-Sánchez H. Mortality and morbidity trends: is there compression of morbidity? J Gerontol B Psychol Sci Soc Sci. 2011;66:75–86. doi: 10.1093/geronb/gbq088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Crystal H, Dickson D, Fuld P, Masur D, Scott R, Mehler M, Masdeu J, Kawas C, Aronson M, Wolfson L. Clinico-pathologic studies in dementia: nondemented subjects with pathologically confirmed Alzheimer's disease. Neurology. 1988;38:1682–1687. doi: 10.1212/wnl.38.11.1682. [DOI] [PubMed] [Google Scholar]
- 31.De Jager PL, Bennett DA. An Inflection Point in Gene Discovery Efforts for Neurodegenerative Diseases: From Syndromic Diagnoses Toward Endophenotypes and the Epigenome. JAMA Neurol. 2013;70:719–726. doi: 10.1001/jamaneurol.2013.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Del Ser T, Hachinski V, Merskey H, Munoz DG. An autopsy-verified study of the effect of education on degenerative dementia. Brain. 1999;122:2309–2319. doi: 10.1093/brain/122.12.2309. [DOI] [PubMed] [Google Scholar]
- 33.DeYoung CG, Hirsh JB, Shane MS, Papademetris X, Rajeevan N, Gray JR. Testing predictions from personality neuroscience. Brain structure and the big five. Psychol Sci. 2010;1:820–828. doi: 10.1177/0956797610370159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Diamond MC, Krech D, Rosenzweig MR. The Effects of an Enriched Environment on the Histology of the Rat Cerebral Cortex. J. Comp. Neurol. 1964;123:111–120. doi: 10.1002/cne.901230110. [DOI] [PubMed] [Google Scholar]
- 35.Dickson DW, Crystal HA, Bevona C, Honer W, Vincent I, Davies P. Correlations of synaptic and pathological markers with cognition of the elderly. Neurobiol Aging. 1995;16:285–304. doi: 10.1016/0197-4580(95)00013-5. [DOI] [PubMed] [Google Scholar]
- 36.Di Garbo A, Mainardi M, Chillemi S, Maffei L, Caleo M. Environmental enrichment modulates cortico-cortical interactions in the mouse. PLoS One. 2011;6:e25285. doi: 10.1371/journal.pone.0025285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dong H, Csernansky JG. Effects of stress and stress hormones on amyloid-beta protein and plaque deposition. J Alzheimers Dis. 2009;18:459–469. doi: 10.3233/JAD-2009-1152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dong H, Goico B, Martin M, Csernansky CA, Bertchume A, Csernansky JG. Modulation of hippocampal cell proliferation, memory, and amyloid plaque deposition in APPsw (Tg2576) mutant mice by isolation stress. Neuroscience. 2004;127:601–609. doi: 10.1016/j.neuroscience.2004.05.040. [DOI] [PubMed] [Google Scholar]
- 39.Driscoll I, Resnick SM, Troncoso JC, An Y, O'Brien R, Zonderman AB. Impact of Alzheimer's pathology on cognitive trajectories in nondemented elderly. Ann Neurol. 2006;60:688–695. doi: 10.1002/ana.21031. [DOI] [PubMed] [Google Scholar]
- 40.Duffy SN, Craddock KJ, Abel T, Nguyen PV. Environmental enrichment modifies the PKAdependence of hippocampal LTP and improves hippocampus-dependent memory. Learn Mem. 2001;8:26–34. doi: 10.1101/lm.36301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.EClipSE Collaborative Members. Cohort profile: Epidemiological Clinicopathological studies in Europe (EClipSE) J Alzheimers Dis. 2009;18:659–663. doi: 10.3233/JAD-2009-1181. [DOI] [PubMed] [Google Scholar]
- 42.EClipSE Collaborative Members. Brayne C, Ince PG, Keage HA, McKeith IG, Matthews FE, Polvikoski T, Sulkava R. Education, the brain and dementia: neuroprotection or compensation? Brain. 2010;133:2210–2216. doi: 10.1093/brain/awq185. [DOI] [PubMed] [Google Scholar]
- 43.Esiri MM, Chance SA. Cognitive reserve, cortical plasticity and resistance to Alzheimer's disease. Alzheimers Res Ther. 2012;4:7. doi: 10.1186/alzrt105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Evans DA, Hebert LE, Beckett LA, Scherr PA, Albert MS, Chown MJ, Pilgrim DM, Taylor JO. Education and other measures of socioeconomic status and risk of incident Alzheimer disease in a defined population of older persons. Arch Neurol. 1997;54:1399–1405. doi: 10.1001/archneur.1997.00550230066019. [DOI] [PubMed] [Google Scholar]
- 45.Fabrigoule C, Letenneur L, Dartigues JF, Zarrouk M, Commenges D, Barberger-Gateau P. Social and leisure activities and risk of dementia: a prospective longitudinal study. J Am Geriatr Soc. 1995;43:485–490. doi: 10.1111/j.1532-5415.1995.tb06093.x. [DOI] [PubMed] [Google Scholar]
- 46.Farfel JM, Nitrini R, Suemoto CK, Grinberg LT, Ferretti FEL, Leite REP, Menezes PR, Fegni F, Bennett DA, Pasqualucci CA, Jacob-Filho W Brazilian Aging Brain Study Group. Elementary education protects against dementia: a clinicopathological study. Neurology. 2013;81:650–657. [Google Scholar]
- 47.Fleming J, Zhao E, O'Connor DW, Pollitt PA, Brayne C. Cohort profile: the Cambridge City over-75s Cohort (CC75C) Int J Epidemiol. 2007;36:40–46. doi: 10.1093/ije/dyl293. [DOI] [PubMed] [Google Scholar]
- 48.Fratiglioni L, Wang HX, Ericsson K, Maytan M, Winblad B. Influence of social network on occurrence of dementia: a community-based longitudinal study. Lancet. 2000;355:1315–1319. doi: 10.1016/S0140-6736(00)02113-9. [DOI] [PubMed] [Google Scholar]
- 49.Freret T, Billard JM, Schumann-Bard P, Dutar P, Dauphin F, Boulouard M, Bouet V. Rescue of cognitive aging by long-lasting environmental enrichment exposure initiated before median lifespan. Neurobiol. Aging. 2012;33:1005.e1–1010.e1. doi: 10.1016/j.neurobiolaging.2011.09.028. [DOI] [PubMed] [Google Scholar]
- 50.Fries JF, Bruce B, Chakravarty E. Compression of morbidity 1980–2011: a focused review of paradigms and progress. J Aging Res. 2011:261702. doi: 10.4061/2011/261702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Graves AB, Mortimer JA, Larson EB, Wenzlow A, Bowen JD, McCormick WC. Head circumference as a measure of cognitive reserve. Association with severity of impairment in Alzheimer's disease. Br J Psychiatry. 1996;169:86–92. doi: 10.1192/bjp.169.1.86. [DOI] [PubMed] [Google Scholar]
- 52.Greenough WT, Volkmar FR. Pattern of dendritic branching in occipital cortex of rats reared in complex environments". Exp. Neurol. 1973;40:491–504. doi: 10.1016/0014-4886(73)90090-3. [DOI] [PubMed] [Google Scholar]
- 53.Grinberg LT, Ferretti RE, Farfel JM, Leite R, Pasqualucci CA, Rosemberg S, Nitrini R, Saldiva PH, Filho WJ Brazilian Aging Brain Study Group. Brain bank of the Brazilian aging brain study group - a milestone reached and more than 1,600 collected brains. Cell Tissue Bank. 2007;8:151–162. doi: 10.1007/s10561-006-9022-z. [DOI] [PubMed] [Google Scholar]
- 54.Hall CB, Lipton RB, Sliwinski M, Katz MJ, Derby CA, Verghese J. Cognitive activities delay onset of memory decline in persons who develop dementia. Neurology. 2009;73:356–361. doi: 10.1212/WNL.0b013e3181b04ae3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Head E, Corrada MM, Kahle-Wrobleski K, Kim RC, Sarsoza F, Goodus M, Kawas CH. Synaptic proteins, neuropathology and cognitive status in the oldest-old. Neurobiol Aging. 2009;30:1125–1134. doi: 10.1016/j.neurobiolaging.2007.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hebert LE, Bienias JL, Aggarwal NT, Wilson RS, Bennett DA, Shah RC, Evans DA. Change in risk of Alzheimer disease over time. Neurology. 2010;75:786–791. doi: 10.1212/WNL.0b013e3181f0754f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Helmes E, Merskey H, Fox H, Fry RN, Bowler JV, Hachinski VC. Patterns of deterioration in senile dementia of the Alzheimer type. Arch Neurol. 1995;52:306–310. doi: 10.1001/archneur.1995.00540270102026. [DOI] [PubMed] [Google Scholar]
- 58.Holwerda TJ, Deeg DJ, Beekman AT, van Tilburg TG, Stek ML, Jonker C, Schoevers RA. Feelings of loneliness, but not social isolation, predict dementia onset: results from the Amsterdam Study of the Elderly (AMSTEL) J Neurol Neurosurg Psychiatry. 2012 doi: 10.1136/jnnp-2012-302755. [Epub] [DOI] [PubMed] [Google Scholar]
- 59.Honer WG, Barr AM, Sawada K, Thornton AE, Morris MC, Leurgans SE, Schneider JA, Bennett DA. Cognitive reserve, presynaptic proteins and dementia in the elderly. Translational Psychiatry. 2012;2:2114. doi: 10.1038/tp.2012.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Huang FL, Huang KP, Wu J, Boucheron C. Environmental enrichment enhances neurogranin expression and hippocampal learning and memory but fails to rescue the impairments of neurogranin null mutant mice. J Neurosci. 2006;26:6230–6237. doi: 10.1523/JNEUROSCI.1182-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Iacono D, Markesbery WR, Gross M, Pletnikova O, Rudow G, Zandi P, Troncoso JC. The Nun study: clinically silent AD, neuronal hypertrophy, and linguistic skills in early life. Neurology. 2009;73:665–773. doi: 10.1212/WNL.0b013e3181b01077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Iacono D, O'Brien R, Resnick SM, Zonderman AB, Pletnikova O, Rudow G, An Y, West MJ, Crain B, Troncoso JC. Neuronal hypertrophy in asymptomatic Alzheimer disease. J Neuropathol Exp Neurol. 2008;67:578–589. doi: 10.1097/NEN.0b013e3181772794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ickes BR, Pham TM, Sanders LA, Albeck DS, Mohammed AH, Granholm AC. Long-term environmental enrichment leads to regional increases in neurotrophin levels in rat brain. Experimental Neurology. 2000;164:45–52. doi: 10.1006/exnr.2000.7415. [DOI] [PubMed] [Google Scholar]
- 64.Irvine GI, Abraham WC. Enriched environment exposure alters the input-output dynamics of synaptic transmission in area CA1 of freely moving rats. Neurosci Lett. 2005;391:32–37. doi: 10.1016/j.neulet.2005.08.031. [DOI] [PubMed] [Google Scholar]
- 65.Jacobs B, Schall M, Scheibel A. A quantitative dendritic analysis of Wernicke's area in humans. II Gender, hemispheric, and environmental factors. Journal of Comparative Neurology. 1993;327:97–111. doi: 10.1002/cne.903270108. [DOI] [PubMed] [Google Scholar]
- 66.Kanai R, Bahrami B, Duchaine B, Janik A, Banissy MJ, Rees G. Brain structure links loneliness to social perception. Curr Biol. 2012;22:1975–1979. doi: 10.1016/j.cub.2012.08.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Katzman R, Terry R, DeTeresa R, Brown T, Davies P, Fuld P, Renbing X, Peck A. Clinical, pathological, and neurochemical changes in dementia: a subgroup with preserved mental status and numerous neocortical plaques. Ann Neurol. 1998;23:138–144. doi: 10.1002/ana.410230206. [DOI] [PubMed] [Google Scholar]
- 68.Knopman DS, Parisi JE, Salviati A, Floriach-Robert M, Boeve BF, Ivnik RJ, Smith GE, Dickson DW, Johnson KA, Petersen LE, McDonald WC, Braak H, Petersen RC. Neuropathology of cognitively normal elderly. J Neuropathol Exp Neurol. 2003;62:1087–1095. doi: 10.1093/jnen/62.11.1087. [DOI] [PubMed] [Google Scholar]
- 69.Koepsell TD, Kurland BF, Harel O, Johnson EA, Zhou XH, Kukull WA. Education, cognitive function, and severity of neuropathology in Alzheimer disease. Neurology. 2008;70:1732–1739. doi: 10.1212/01.wnl.0000284603.85621.aa. [DOI] [PubMed] [Google Scholar]
- 70.Krech D, Rosenzweig MR, Bennett EL. Effects of environmental complexity and training on brain chemistry. J Comp Physiol Psychol. 1960;53:509–519. doi: 10.1037/h0045402. [DOI] [PubMed] [Google Scholar]
- 71.Labonté B, Suderman M, Maussion G, Navaro L, Yerko V, Mahar I, Bureau A, Mechawar N, Szyf M, Meaney MJ, Turecki G. Genome-wide epigenetic regulation by early-life trauma. Arch Gen Psychiatry. 2012;69:722–731. doi: 10.1001/archgenpsychiatry.2011.2287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Langa KM, Larson EB, Karlawish JH, et al. Trends in the prevalence and mortality of cognitive impairment in the United States: is there evidence of a compression of cognitive morbidity? Alzheimers Dement. 2008;4:134–144. doi: 10.1016/j.jalz.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lobo A, Saz P, Marcos G, et al. the ZARADEMP Workgroup. Prevalence of dementia in a southern European population in two diff erent time periods: the ZARADEMP project. Acta Psychiatr Scand. 2007;116:299–307. doi: 10.1111/j.1600-0447.2007.01006.x. [DOI] [PubMed] [Google Scholar]
- 74.Manton KC, Gu XL, Ukraintseva SV. Declining prevalence of dementia in the U.S. elderly population. Adv Gerontol. 2005;16:30–37. [PubMed] [Google Scholar]
- 75.Marioni RE, Valenzuela MJ, van den Hout A, Brayne C, Matthews FE MRC Cognitive Function and Ageing Study. Active cognitive lifestyle is associated with positive cognitive health transitions and compression of morbidity from age sixty-five. PLoS One. 2012;7:e50940. doi: 10.1371/journal.pone.0050940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Marioni RE, van den Hout A, Valenzuela MJ, Brayne C, Matthews FE MRC Cognitive Function and Ageing Study. Active cognitive lifestyle associates with cognitive recovery and a reduced risk of cognitive decline. J Alzheimers Dis. 2012;28:223–230. doi: 10.3233/JAD-2011-110377. [DOI] [PubMed] [Google Scholar]
- 77.Markett S, Weber B, Voigt G, Montag C, Felten A, Elger C, Reuter M. Intrinsic connectivity networks and personality: the temperament dimension harm avoidance moderates functional connectivity in the resting brain. Neuroscience. 2013;240:98–105. doi: 10.1016/j.neuroscience.2013.02.056. [DOI] [PubMed] [Google Scholar]
- 78.Marquine MJ, Segawa E, Wilson RS, Bennett DA, Barnes LL. The association between cognitive activity and cognitive function in older Hispanics. J Int Neuropsychological Society. 2012;18:1041–1051. doi: 10.1017/S135561771200080X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Mathuranath PS, Cherian PJ, Mathew R, Kumar S, George A, Alexander A, Ranjith N, Sarma PS. Dementia in Kerala, South India: prevalence and influence of age, education and gender. Int J Geriatr Psychiatry. 2010;25:290–297. doi: 10.1002/gps.2338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Matthews FE, Arthur A, Barnes LE, Bond J, Jagger C, Robinson L, Brayne C on behalf of the Medical Research Council Cognitive Function and Ageing Collaboration. A two-decade comparison of prevalence of dementia in individuals aged 65 years and older from three geographical areas of England: results of the Cognitive Function and Ageing Study I and II. Lancet. 2013 Jul 17; doi: 10.1016/S0140-6736(13)61570-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.McGowan PO, Sasaki A, D'Alessio AC, Dymov S, Labonté B, Szyf M, Turecki G, Meaney MJ. Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nat Neurosci. 2009;12:342–348. doi: 10.1038/nn.2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.McGowan PO, Sasaki A, Huang TC, Unterberger A, Suderman M, Ernst C, Meaney MJ, Turecki G, Szyf M. Promoter-wide hypermethylation of the ribosomal RNA gene promoter in the suicide brain. PLoS One. 2008;3:e2085. doi: 10.1371/journal.pone.0002085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.McNair K, Broad J, Riedel G, Davies CH, Cobb SR. Global changes in the hippocampal proteome following exposure to an enriched environment. Neuroscience. 2007;145:413–422. doi: 10.1016/j.neuroscience.2006.12.033. [DOI] [PubMed] [Google Scholar]
- 84.Mortimer JA, Borenstein AR, Gosche KM, Snowdon DA. Very early detection of Alzheimer neuropathology and the role of brain reserve in modifying its clinical expression. J Geriatr Psychiatry Neurol. 2005;18:218–223. doi: 10.1177/0891988705281869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Mortimer JA, Snowdon DA, Markesbery WR. Head circumference, education and risk of dementia: Findings from the Nun Study. J Clin Exp Neuropsychol. 2003;25:671–679. doi: 10.1076/jcen.25.5.671.14584. [DOI] [PubMed] [Google Scholar]
- 86.Muniz-Terrera G, Matthews FE, Stephan B, Brayne C CC75C Collaboration Group. Are terminal decline and its potential indicators detectable in population studies of the oldest old? Int J Geriat r Psychiatry. 2011;26:584–592. doi: 10.1002/gps.2566. [DOI] [PubMed] [Google Scholar]
- 87.Muniz-Terrera G, van den Hout A, Piccinin AM, Matthews FE, Hofer SM. Investigating terminal decline: results from a UK population-based study of aging. Psychol Aging. 2013;28:377–385. doi: 10.1037/a0031000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Murphy TE, Han L, Allore HG, Peduzzi PN, Gill TM, Lin H. Treatment of death in the analysis of longitudinal studies of gerontological outcomes. J Gerontol A Biol Sci Med Sci. 2011;66:109–114. doi: 10.1093/gerona/glq188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Næss O, Hoff DA, Lawlor D, Mortensen LH. Education and adult cause-specific mortality--examining the impact of family factors shared by 871 367 Norwegian siblings. Int J Epidemiol. 2012;41:1683–1691. doi: 10.1093/ije/dys143. [DOI] [PubMed] [Google Scholar]
- 90.Naka F, Narita N, Okado N, Narita M. Modification of AMPA receptor properties following environmental enrichment. Brain Dev. 2005;27:275–278. doi: 10.1016/j.braindev.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 91.Ott A, van Rossum CT, van Harskamp F, van de Mheen H, Hofman A, Breteler MM. Education and the incidence of dementia in a large population-based study: the Rotterdam Study. Neurology. 1999;52:663–666. doi: 10.1212/wnl.52.3.663. [DOI] [PubMed] [Google Scholar]
- 92.Polvikoski T, Sulkava R, Haltia M, Kainulainen K, Vuorio A, Verkkoniemi A, Niinisto L, Halonen P, Kontula K. Apolipoprotein E, dementia, and cortical deposition of betaamyloid protein. N Engl J Med. 1995;333:242–1247. doi: 10.1056/NEJM199511093331902. [DOI] [PubMed] [Google Scholar]
- 93.Prencipe M, Casini AR, Ferretti C, Lattanzio MT, Fiorelli M, Culasso F. Prevalence of dementia in an elderly rural population: effects of age, sex, and education. J Neurol Neurosurg Psychiatry. 1996;60:628–633. doi: 10.1136/jnnp.60.6.628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Price JL, McKeel DW, Buckles VD, Roe CM, Xiong C, Grundman M, Hansen LA, Petersen RC, Parisi JE, Dickson DW, Smith CD, Davis DG, Schmitt FA, Markesbery WR, Kaye J, Kurlan R, Hulette C, Kurland BF, Higdon R, Kukull W, Morris JC. Neuropathology of nondemented aging: presumptive evidence for preclinical Alzheimer disease. Neurobiol Aging. 2009;30:1026–1036. doi: 10.1016/j.neurobiolaging.2009.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Qiu C, Bäckman L, Winblad B, Agüero-Torres H, Fratiglioni L. The influence of education on clinically diagnosed dementia incidence and mortality data from the Kungsholmen Project. Arch Neurol. 2001;58:2034–2039. doi: 10.1001/archneur.58.12.2034. [DOI] [PubMed] [Google Scholar]
- 96.Qiu C, von Strauss E, Backman L, Winblad B, Fratiglioni L. Twenty-year changes in dementia occurrence suggest decreasing incidence in central Stockholm, Sweden. Neurology. 2013;80:1888–1894. doi: 10.1212/WNL.0b013e318292a2f9. [DOI] [PubMed] [Google Scholar]
- 97.Rampon C, Jiang CH, Dong H, Tang YP, Lockhart DJ, Schultz PG, Tsien JZ, Hu Y. Effects of environmental enrichment on gene expression in the brain. Proc Natl Acad Sci U S A. 2000;97:12880–12884. doi: 10.1073/pnas.97.23.12880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Reed BR, Dowling M, Tomaszewski Farias S, Sonnen J, Strauss M, Schneider JA, Bennett DA, Mungas D. Cognitive activities during adulthood are more important than education in building reserve. J Int Neuropsychol Soc. 2011;17:615–624. doi: 10.1017/S1355617711000014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Riley KP, Snowdon DA, Desrosiers MF, Markesbery WR. Early life linguistic ability, late life cognitive function, and neuropathology: findings from the Nun Study. Neurobiol Aging. 2005;26:341–347. doi: 10.1016/j.neurobiolaging.2004.06.019. [DOI] [PubMed] [Google Scholar]
- 100.Riudavets MA, Iacono D, Resnick SM, O'Brien R, Zonderman AB, Martin LJ, Rudow G, Pletnikova O, Troncoso JC. Resistance to Alzheimer's pathology is associated with nuclear hypertrophy in neurons. Neurobiol Aging. 2007;28:1484–1492. doi: 10.1016/j.neurobiolaging.2007.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Rocca WA, Petersen RC, Knopman DS, et al. Trends in the incidence and prevalence of Alzheimer's disease, dementia, and cognitive impairment in the United States. Alzheimers Dement. 2011;7:80–93. doi: 10.1016/j.jalz.2010.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Roe CM, Xiong C, Miller JP, Cairns NJ, Morris JC. Interaction of neuritic plaques and education predicts dementia. Alzheimer Dis Assoc Disord. 2008;22:188–193. doi: 10.1097/WAD.0b013e3181610fff. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Roe CM, Xiong C, Miller JP, Morris JC. Education and Alzheimer disease without dementia: support for the cognitive reserve hypothesis. Neurology. 2007;68:223–228. doi: 10.1212/01.wnl.0000251303.50459.8a. [DOI] [PubMed] [Google Scholar]
- 104.Rossi C, Angelucci A, Costantin L, Braschi C, Mazzantini M, Babbini F, et al. Brain-derived neurotrophic factor (BDNF) is required for the enhancement of hippocampal neurogenesis following environmental enrichment. European Journal of Neuroscience. 2006;24:1850–1856. doi: 10.1111/j.1460-9568.2006.05059.x. [DOI] [PubMed] [Google Scholar]
- 105.Saczynski JS, Pfeifer LA, Masaki K, Korf ES, Laurin D, White L, Launer LJ. The effect of social engagement on incident dementia: the Honolulu-Asia Aging Study. Am J Epidemiol. 2006;63:433–440. doi: 10.1093/aje/kwj061. [DOI] [PubMed] [Google Scholar]
- 106.Sato Y, Bernier F, Suzuki I, Nakagawa M, Oda Y. Comparative lipidomics of mouse brain exposed to enriched environment. J Lipid Res. 2013;54:2687–2696. doi: 10.1194/jlr.M038075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Satz P, Morgenstern H, Miller EN, Selnes OA, McArthur JC, Cohen BA, Wesch J, Becker JT, Jacobson L, D'Elia LF, et al. Low education as a possible risk factor for cognitive abnormalities in HIV-1: findings from the multicenter AIDS Cohort Study (MACS) J Acquir Immune Defic Syndr. 1993;6:503–511. [PubMed] [Google Scholar]
- 108.carmeas N, Albert SM, Manly JJ, Stern Y. Education and rates of cognitive decline in incident Alzheimer's disease. J Neurol Neurosurg Psychiatry. 2006;77:308–316. doi: 10.1136/jnnp.2005.072306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Scarmeas N, Levy G, Tang MX, Manly J, Stern Y. Influence of leisure activity on the incidence of Alzheimer's disease. Neurology. 2001;57:2236–2242. doi: 10.1212/wnl.57.12.2236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Scarmeas N, Zarahn E, Anderson KE, Habeck CG, Hilton J, Flynn J, Marder KS, Bell KL, Sackeim HA, Van Heertum RL, Moeller JR, Stern Y. Association of life activities with cerebral blood flow in Alzheimer disease: implications for the cognitive reserve hypothesis. Arch Neurol. 2003;60:359–365. doi: 10.1001/archneur.60.3.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Scheibel A, Paul A, Fried I, Forsythe A, Tomiyasu U, Wechsler A. A quantitative study of dendrite complexity in selected areas of the human cortex. Brain and Cognition. 12:85–101. doi: 10.1016/0278-2626(90)90006-a. (19990) [DOI] [PubMed] [Google Scholar]
- 112.Schmitt FA, Davis DG, Wekstein DR, Smith CD, Ashford JW, Markesbery WR. "Preclinical" AD revisited: neuropathology of cognitively normal older adults. Neurology. 2000;55:370–376. doi: 10.1212/wnl.55.3.370. [DOI] [PubMed] [Google Scholar]
- 113.Schneider JA, Aggarwal NT, Barnes LL, Boyle PA, Bennett DA. The neuropathology of older persons with and without dementia from community vs. clinic cohorts. Journal of Alzheimer's Disease. 2009;18:691–701. doi: 10.3233/JAD-2009-1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Schofield PW, Logroscino G, Andrews HF, Albert S, Stern Y. An association between head circumference and Alzheimer's disease in a population-based study of aging and dementia. Neurology. 1997;49:30–37. doi: 10.1212/wnl.49.1.30. [DOI] [PubMed] [Google Scholar]
- 115.Schofield P, Mosesson R, Stern Y, Mayeux R. The age at onset of Alzheimer's disease and an intracranial area measurement: a relationship. Archives of Neurology. 1995;52:95–98. doi: 10.1001/archneur.1995.00540250103019. [DOI] [PubMed] [Google Scholar]
- 116.Schrijvers EM, Verhaaren BF, Koudstaal PJ, Hofman A, Ikram MA, Breteler MM. Is dementia incidence declining? Trends in dementia incidence since 1990 in the Rotterdam Study. Neurology. 2012;78:1456–1463. doi: 10.1212/WNL.0b013e3182553be6. [DOI] [PubMed] [Google Scholar]
- 117.Schweizer TA, Ware J, Fischer CE, Craik FI, Bialystok E. Bilingualism as a contributor to cognitive reserve: evidence from brain atrophy in Alzheimer's disease. Cortex. 2012;48:991–996. doi: 10.1016/j.cortex.2011.04.009. [DOI] [PubMed] [Google Scholar]
- 118.Shulman JM, Chen K, Keenan BT, Chibnik LB, Fleisher A, Thiyyagura P, Roontiva A, McCabe C, Patsopoulos NA, Corneveaux J, Yu L, Huentelman MJ, Evans DA, Schneider JA, Reiman E, De Jager PL, Bennett DA. Genetic susceptibility for Alzheimer's disease neuritic plaque pathology. JAMA Neurology. 2013;70:1150–1157. doi: 10.1001/jamaneurol.2013.2815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Snowdon DA. Nun Study. Healthy aging and dementia: findings from the Nun Study. Ann Intern Med. 2003;139:450–454. doi: 10.7326/0003-4819-139-5_part_2-200309021-00014. [DOI] [PubMed] [Google Scholar]
- 120.Snowdon DA, Kemper SJ, Mortimer JA, Greiner LH, Wekstein DR, Markesbery WR. Linguistic ability in early life and cognitive function and Alzheimer's disease in late life. Findings from the Nun Study. JAMA. 1996;275:528–532. [PubMed] [Google Scholar]
- 121.Soetanto A, Wilson RS, Talbot K, Un A, Schneider JA, Sobiesk M, Kelly JF, Leurgans SE, Bennett DA, Arnold SE. Association of anxiety and depression with microtubule-associated protein 2-and synaptopodin-immunolabeled dendrite and spine densities in hippocampal CA3 of older humans. Archives of General Psychiatry. 2010;67:448–457. doi: 10.1001/archgenpsychiatry.2010.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Soffie M, Hahn K, Terao E, Eclancher Behavioural and glial changes in old rats following environmental enrichment. Behav. Brain Res. 1999;101:37–49. doi: 10.1016/s0166-4328(98)00139-9. [DOI] [PubMed] [Google Scholar]
- 123.Sonnen JA, Larson EB, Crane PK, Haneuse S, Li G, Schellenberg GD, Craft S, Leverenz JB, Montine TJ. Pathological correlates of dementia in a longitudinal, population-based sample of aging. Ann Neurol. 2007;62:406–413. doi: 10.1002/ana.21208. [DOI] [PubMed] [Google Scholar]
- 124.Sonnen JA, Santa Cruz K, Hemmy LS, et al. Ecology of the aging human brain. Arch Neurol. 2011;68:1049–1056. doi: 10.1001/archneurol.2011.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Stern Y. Cognitive reserve in ageing and Alzheimer's disease. Lancet Neurol. 2012;11:1006–1012. doi: 10.1016/S1474-4422(12)70191-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Stern Y, Gurland B, Tatemichi TK, Tang MX, Wilder D, Mayeux R. Influence of education and occupation on the incidence of Alzheimer's disease. JAMA. 1994;271:1004–1010. [PubMed] [Google Scholar]
- 127.Stern Y, Tang MX, Denaro J, Mayeux R. Increased risk of mortality in Alzheimer's disease patients with more advanced educational and occupational attainment. Ann Neurol. 1995;37:590–595. doi: 10.1002/ana.410370508. [DOI] [PubMed] [Google Scholar]
- 128.Talbot K, Wang HY, Kazi H, Han LY, Bakshi KP, Stucky A, Fuino RL, Kawaguchi KR, Samoyedny AJ, Wilson RS, Arvanitakis Z, Schneider JA, Wolf BA, Bennett DA, Trojanowski JQ, Arnold SE. Brain insulin resistance demonstrated in Alzheimer's disease without diabetes is associated with dysregulated IRS-1, IFG-1, and cognitive decline. Journal of Clinical Investigation. 2013;122:1316–1338. doi: 10.1172/JCI59903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Thorvaldsson V, Hofer SM, Berg S, Skoog I, Sacuiu S, Johansson B. Onset of terminal decline in cognitive abilities in individuals without dementia. Neurology. 2008;71:882–887. doi: 10.1212/01.wnl.0000312379.02302.ba. [DOI] [PubMed] [Google Scholar]
- 130.Tomlinson BE, Blessed G, Roth M. Observations on the brains of non-demented old people. J Neurol Sci. 1968;7:331–356. doi: 10.1016/0022-510x(68)90154-8. [DOI] [PubMed] [Google Scholar]
- 131.Tomlinson BE, Blessed G, Roth M. Observations on the brains of demented old people. J Neurol Sci. 1970;11:205–242. doi: 10.1016/0022-510x(70)90063-8. [DOI] [PubMed] [Google Scholar]
- 132.Troncoso JC, Martin LJ, Dal Forno G, Kawas CH. Neuropathology in controls and demented subjects from the Baltimore Longitudinal Study of Aging. Neurobiol Aging. 1996;17:365–371. doi: 10.1016/0197-4580(96)00028-0. [DOI] [PubMed] [Google Scholar]
- 133.Valenzuela MJ. Brain reserve and the prevention of dementia. Curr Opin Psychiatry. 2008;21:296–302. doi: 10.1097/YCO.0b013e3282f97b1f. [DOI] [PubMed] [Google Scholar]
- 134.Valenzuela M, Brayne C, Sachdev P, Wilcock G, Matthews F Medical Research Council Cognitive Function and Ageing Study. Cognitive lifestyle and long-term risk of dementia and survival after diagnosis in a multicenter population-based cohort. Am J Epidemiol. 2011;173:1004–1012. doi: 10.1093/aje/kwq476. [DOI] [PubMed] [Google Scholar]
- 135.Valenzuela MJ, Breakspear M, Sachdev P. Complex mental activity and the aging brain: molecular, cellular and cortical network mechanisms. Brain Res Rev. 2007;56:198–213. doi: 10.1016/j.brainresrev.2007.07.007. [DOI] [PubMed] [Google Scholar]
- 136.Valero J, España J, Parra-Damas A, Martín E, Rodríguez-Álvarez J, Saura CA. Short-term environmental enrichment rescues adult neurogenesis and memory deficits in APP(Sw,Ind) transgenic mice. PLoS One. 2011;9:e16832. doi: 10.1371/journal.pone.0016832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Van Praag H, Kempermann G, Gage FH. Neural consequences of environmental enrichment. Nature Reviews Neuroscience. 2000;1:191–198. doi: 10.1038/35044558. [DOI] [PubMed] [Google Scholar]
- 138.Verghese J, Lipton RB, Katz MJ, Hall CB, Derby CA, Kuslansky G, Ambrose AF, Sliwinski M, Buschke H. Leisure activities and the risk of dementia in the elderly. N Engl J Med. 2003;48:2508–2516. doi: 10.1056/NEJMoa022252. [DOI] [PubMed] [Google Scholar]
- 139.Wang HX, Karp A, Winblad B, Fratiglioni L. Late-life engagement in social and leisure activities is associated with a decreased risk of dementia: a longitudinal study from the Kungsholmen project. Am J Epidemiol. 2002;155:1081–1087. doi: 10.1093/aje/155.12.1081. [DOI] [PubMed] [Google Scholar]
- 140.Wang HY, Stucky A, Hahn CG, WEilson RS, Bennett DA, Arnold SE. BDNF-TrkB signaling in late life cognitive decline and Alzheimer's disease. Translational Neuroscience. 2011;2:91–100. [Google Scholar]
- 141.Weaver IC, Cervoni N, Champagne FA, D'Alessio AC, Sharma S, Seckl JR, Dymov S, Szyf M, Meaney MJ. Epigenetic programming by maternal behavior. Nat Neurosci. 2004;7:847–854. doi: 10.1038/nn1276. [DOI] [PubMed] [Google Scholar]
- 142.Weaver IC, Meaney MJ, Szyf M. Maternal care effects on the hippocampal transcriptome and anxiety-mediated behaviors in the offspring that are reversible in adulthood. Proc Natl Acad Sci U S A. 2006;103:3480–3485. doi: 10.1073/pnas.0507526103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.White L, Katzman R, Losonczy K, Salive M, Wallace R, Berkman L, Taylor J, Fillenbaum G, Havlik R. Association of education with incidence of cognitive impairment in three established populations for epidemiologic studies of the elderly. J Clin Epidemiol. 1994;47:363–374. doi: 10.1016/0895-4356(94)90157-0. [DOI] [PubMed] [Google Scholar]
- 144.Wilson RS, Beck TL, Bienias JL, Bennett DA. Terminal cognitive decline: accelerated loss of cognition in the last years of life. Psychosom Med. 2007;69:131–137. doi: 10.1097/PSY.0b013e31803130ae. [DOI] [PubMed] [Google Scholar]
- 145.Wilson RS, Bennett DA, Beckett LA, Morris MC, Gilley DW, Bienias J, Scherr PA, Evans DA. Cognitive activity in older persons from a geographically defined population. Journal of Gerontology: Psychological Sciences. 1999;54:P155–P160. doi: 10.1093/geronb/54b.3.p155. [DOI] [PubMed] [Google Scholar]
- 146.Wilson RS, Boyle PA, Buchman AS, Arnold SE, Bennett DA. Harm avoidance and risk of Alzheimer's disease. Psychosomatic Medicine. 2011;73:690–696. doi: 10.1097/PSY.0b013e3182302ale. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Wilson RS, Boyle PA, Levine SR, Yu L, Anagnos SE, Buchman AS, Schneider JA, Bennett DA. Emotional neglect in childhood and cerebral infarction in old age. Neurology. 2012;79:1534–1539. doi: 10.1212/WNL.0b013e31826e25bd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Wilson RS, Boyle PA, Levine SR, Yu L, Hoganson GM, Buchman AS, Schneider JA, Bennett DA. Harm avoidance and cerebral infarcts. Neuropsychology. doi: 10.1037/neu0000022. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wilson RS, Boyle PA, Yu L, Barnes LL, Schneider JA, Bennett DA. Life span cognitive activity, neuropathologic burden, and cognitive aging. Neurology. 2013;81:314–321. doi: 10.1212/WNL.0b013e31829c5e8a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Wilson RS, Krueger KR, Arnold SE, Schneider JA, Kelly JF, Barnes LL, Tang Y, Bennett DA. Loneliness and risk of Alzheimer's disease. Archives of General Psychiatry. 2007;64:234–240. doi: 10.1001/archpsyc.64.2.234. [DOI] [PubMed] [Google Scholar]
- 151.Wilson RS, Nag S, Boyle PA, Hizel LP, Yu L, Buchman AS, Schneider JA, Bennett DA. Neural reserve, neuronal density in the locus coeruleous and cognitive decline. Neurology. 2013;80:1202–1208. doi: 10.1212/WNL.0b013e3182897103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wilson RS, Scherr PA, Schneider JA, Tang Y, Bennett DA. The relation of cognitive activity to risk of developing Alzheimer's disease. Neurology. 2007;69:1911–1920. doi: 10.1212/01.wnl.0000271087.67782.cb. [DOI] [PubMed] [Google Scholar]
- 153.Wilson RS, Schneider JA, Arnold SE, Bienias JL, Bennett DA. Conscientiousness and the incidence of Alzheimer's disease and mild cognitive impairment. Arch Gen Psychiatry. 2007;64:1204–1212. doi: 10.1001/archpsyc.64.10.1204. [DOI] [PubMed] [Google Scholar]
- 154.Wilson RS, Segawa E, Boyle PA, Bennett DA. Influence of late life cognitive activity on cognitive health. Neurology. 2012;78:1123–1129. doi: 10.1212/WNL.0b013e31824f8c03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Wilson RS, Yu L, Trojanowski JQT, Chen EY, Bennett DA, Schneider JA. TDP-43 pathology, cognitive decline, and dementia in old age. JAMA Neurology. 2013 doi: 10.1001/jamaneurol.2013.3961. Epub. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Yip AG, Brayne C, Matthews FE MRC Cognitive Function and Ageing Study. Risk factors for incident dementia in England and Wales: The Medical Research Council Cognitive Function and Ageing Study. A population-based nested case-control study. Age Ageing. 2006;35:154–160. doi: 10.1093/ageing/afj030. [DOI] [PubMed] [Google Scholar]
- 157.Yu L, Boyle PA, Wilson RS, Segawa E, Leurgans SE, De Jager PL, Bennett DA. A random change point model for cognitive decline in Alzheimer's disease and mild cognitive impairment. Neuroepidemiology. 2012;39:73–83. doi: 10.1159/000339365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Yuede CM, Zimmerman SD, Dong H, Kling MJ, Bero AW, Holtzman DM, Timson BF, Csernansky JG. Effects of voluntary and forced exercise on plaque deposition, hippocampal volume, and behavior in the Tg2576 mouse model of Alzheimer's disease. Neurobiol Dis. 2009;35:426–432. doi: 10.1016/j.nbd.2009.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhang MY, Katzman R, Salmon D, Jin H, Cai GJ, Wang ZY, Qu GY, Grant I, Yu E, Levy P, et al. The prevalence of dementia and Alzheimer's disease in Shanghai, China: impact of age gender, and education. Ann Neurol. 1990;27:428–437. doi: 10.1002/ana.410270412. [DOI] [PubMed] [Google Scholar]