Summary
Myelofibrosis (MF) patients can present with a wide spectrum of disease characteristics. We analysed the consistency of ruxolitinib efficacy across patient subgroups in the COntrolled MyeloFibrosis Study With ORal JAK Inhibitor Treatment (COMFORT-I,) a double-blind trial, where patients with intermediate-2 or high-risk MF were randomized to twice-daily oral ruxolitinib (n = 155) or placebo (n = 154). Subgroups analysed included MF subtype (primary, post-polycythaemia vera, post-essential thrombocythaemia), age (≤65, > 65 years), International Prognostic Scoring System risk group, baseline Eastern Cooperative Oncology Group performance status (0, 1, ≥2), JAK2 V617F mutation (positive, negative), baseline haemoglobin level (≥100, <100 g/l), baseline platelet count (100–200 × 109/l, >200 × 109/l), baseline palpable spleen size (≤10, >10 cm), and baseline quartile of spleen volume and Total Symptom Score (TSS; Q1 = lowest, Q4 = highest). Mean percentage change from baseline to week 24 in spleen volume and TSS were calculated for ruxolitinib and placebo in each subgroup. Overall survival was estimated by Kaplan–Meier method according to original randomization group. In ruxolitinib-treated patients, reductions in spleen volume and TSS and evidence of improved survival relative to placebo across subgroups were consistent with those seen in the COMFORT-I population, confirming that ruxolitinib is an effective therapy for the spectrum of MF patients studied in COMFORT-I.
Keywords: Myelofibrosis, ruxolitinib, subgroups, spleen volume, symptoms
Myelofibrosis (MF) is a rare and life-threatening myeloproliferative neoplasm that can arise de novo [primary MF (PMF)] or evolve from polycythaemia vera (PV), i.e. post-polycythaemia vera MF (PPV-MF), or essential thrombocythaemia (ET), i.e. post-ET MF (PET-MF) (Barosi et al, 2008; Vardiman et al, 2009; Tefferi, 2011). Dysregulated Janus kinase/signal transducers and activators of transcription (JAK-STAT) signalling, resulting from gain- or loss-of-function mutations and/or high circulating levels of inflammatory cytokines, plays a key role in the pathogenesis of MF (Vainchenker et al, 2011). The JAK2 V617F mutation is present in approximately 50–60% of patients with PMF or ET and in over 95% of patients with PV (Nguyen & Gotlib, 2012). Dysregulation of the JAK-STAT signalling pathway in MF is additionally related to mutations in genes such as JAK2 exon 12, MPL exon 10, SH2B3, members of the Casitas B-cell lymphoma family and post-translational modifications of suppressor of cytokine signalling proteins (Vainchenker et al, 2011). Proinflammatory cytokines that have been implicated in MF are known to signal through JAK1 and JAK2 (Vainchenker et al, 2008) and symptoms of MF have been linked to elevated levels of these cytokines (Verstovsek, 2009; Tefferi et al, 2011). An association between elevated cytokines and decreased survival has also been reported (Tefferi et al, 2011).
A broad spectrum of disease characteristics exists within the MF patient population (Cervantes et al, 1997, 2009). Patients often experience constitutional symptoms (e.g. fever, night sweats, weight loss) and splenomegaly, which may cause disability and have a profound impact on quality of life (Mesa et al, 2007); however, other MF-related symptoms, such as fatigue (84%), itching (50%) and bone pain (47%), are also common and burdensome to patients (Mesa et al, 2007).
Patients with MF have shortened survival, and those with advanced MF have a poor prognosis (Barbui et al, 2011). The major prognostic scoring systems used to categorize risk in patients with MF are the International Prognostic Scoring System (IPSS) (Cervantes et al, 2009), the Dynamic International Prognostic Scoring System (DIPSS) (Passamonti et al, 2010) and DIPSS Plus (Gangat et al, 2011). Risk factors for shorter survival in both the IPSS and DIPSS are age >65 years, presence of constitutional symptoms, haemoglobin <100 g/l, white blood cell count >25 × 109/l and peripheral blood blasts ≥1% (Cervantes et al, 2009; Passamonti et al, 2010). The DIPSS Plus adds platelet count <100 × 109/l, red blood cell transfusion status and unfavourable karyotype to these prognostic criteria (Gangat et al, 2011). Depending on risk factors/category, median survival can range from 11·2 years (low risk) to 2·2 years (high risk) according to the IPSS (Cervantes et al, 2009), with wider ranges for survival across risk groups for the DIPSS (Passamonti et al, 2010) and DIPSS Plus (Gangat et al, 2011).
Although factors that influence disease burden and survival have been published, there is little information regarding how these factors influence treatment efficacy. In the randomized, double-blind, placebo-controlled COMFORT (COntrolled MyeloFibrosis Study With ORal JAK Inhibitor Treatment)-I trial, ruxolitinib, a JAK1/JAK2 inhibitor, significantly reduced spleen volume and improved MF-related symptoms, and was associated with a survival advantage compared with placebo (Verstovsek et al, 2012). The current analysis evaluated the consistency of ruxolitinib efficacy across MF patient subgroups in the COMFORT-I trial.
Methods
Patients and study design
The COMFORT-I study design was previously published (Verstovsek et al, 2012). The primary inclusion criteria were age 18 years or older; diagnosis of PMF, PPV-MF or PET-MF based on 2008 World Health Organization criteria (Tefferi & Vardiman, 2008); life expectancy 6 months or longer; IPSS score (Cervantes et al, 2009) of 2 (intermediate-2 risk) or ≥3 (high risk); Eastern Cooperative Oncology Group (ECOG) performance status ≤3 (Oken et al, 1982); peripheral blood blasts <10%; absolute peripheral blood CD34 + cell count >20 × 106/l; platelet count ≥100 × 109/l; and palpable splenomegaly (≥5 cm below left costal margin). An institutional review board at each site approved the protocol. The study was conducted in accordance with Good Clinical Practice guidelines per the International Conference on Harmonization. All patients provided written informed consent. The study is registered at ClinicalTrials.gov (NCT00952289).
Patients were randomized 1:1 to placebo or ruxolitinib at an oral dose of 15 mg or 20 mg twice daily depending on baseline platelet count (100–200 × 109/l or >200 × 109/l respectively). The dose was optimized for efficacy and safety during treatment. The primary analysis data cut-off occurred when half of the patients remaining in the study completed the week 36 visit and all had either completed the week 24 evaluation or discontinued treatment. All patients receiving placebo were eligible for crossover to ruxolitinib after the primary analysis data cut-off. Early unblinding and crossover from placebo to ruxolitinib was permitted prior to week 24 for a ≥25% increase in spleen volume from baseline accompanied by worsening early satiety with weight loss or worsening splenic pain (demonstrated by increased requirement for narcotics). The primary endpoint was the proportion of patients achieving ≥35% reduction from baseline in spleen volume at week 24. Secondary endpoints included the duration of maintenance of spleen volume reduction, the proportion of patients with ≥50% reduction in Total Symptom Score (TSS) from baseline to week 24 using the modified Myelofibrosis Symptom Assessment Form v2.0 electronic diary, the change in TSS from baseline to week 24, and overall survival. The MF symptoms assessed were night sweats, itching (pruritus), abdominal discomfort, pain under the ribs on the left side, feeling of fullness (early satiety), muscle/bone pain and inactivity on a scale of 0 (absent) to 10 (worst imaginable). The TSS was the average of the daily sum of individual symptom scores over a 7-day period for baseline and over a 28-day period for week 24, except for the inactivity score, which was analysed separately (Verstovsek et al, 2012).
Statistical analyses
Patient subgroups were based on the following: MF subtype (PMF, PPV-MF, PET-MF), age (≤65, >65 years), IPSS risk group, baseline ECOG performance status (0, 1, ≥2), presence/absence of JAK2 V617F mutation, baseline haemoglobin (≥100, <100 g/l), baseline platelet count (100–200 × 109/l, >200 × 109/l) and baseline palpable spleen size (≤10, >10 cm). Additional subgroups included baseline quartile of palpable spleen size and baseline quartile of TSS (Q1 = lowest and Q4 = highest). Mean percentage change from baseline in spleen volume and TSS were calculated for each subgroup. Possible subgroup by treatment interaction was evaluated by using analysis of covariance method with baseline, sex, age group, myelofibrosis type, previous hydroxycarbamide use, JAK2 V617F mutation status, subgroup, treatment and subgroup by treatment interaction as the model effects.
Overall survival was estimated by Kaplan–Meier method according to original randomization group, regardless of crossover to ruxolitinib, for the intention-to-treat population (N = 309). The analysis was carried out for each subgroup separately. The COMFORT-I study was designed to follow patients even after they discontinued study treatment (Verstovsek et al, 2012). The survival analysis in this evaluation included 4 additional months of follow-up beyond the primary analysis data cut-off, corresponding to the time of a pre-specified safety update. Hazard ratios (HRs) for ruxolitinib versus placebo with 95% confidence intervals (CIs) were estimated using the Cox proportional hazard regression method.
The trial was not designed or powered to detect differences in efficacy between treatment arms within a subgroup. Subgroup analyses were intended only to assess the uniformity of treatment effect found in the overall patient population (Second International Study of Infarct Survival (ISIS-2) Collaborative Group, 1988; Cuzick, 2005).
Results
A total of 309 patients were randomized, 155 to ruxolitinib (median age, 66 years) and 154 to placebo (median age, 70 years). As previously reported, the study arms were balanced in terms of demographics and baseline disease characteristics (Verstovsek et al, 2012). Among the 154 patients in the placebo arm, 111 crossed over to ruxolitinib, 40 before study unblinding and 71 after study unblinding. The median time to crossover was 41·1 weeks.
Changes in spleen volume and TSS across subgroups
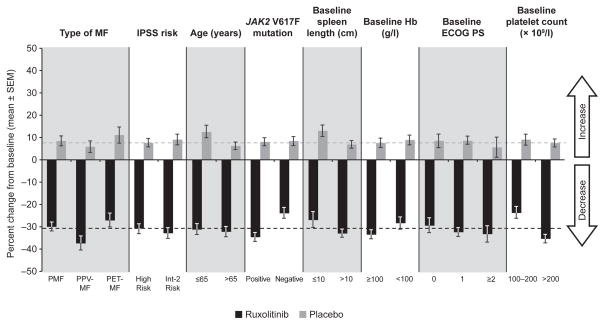
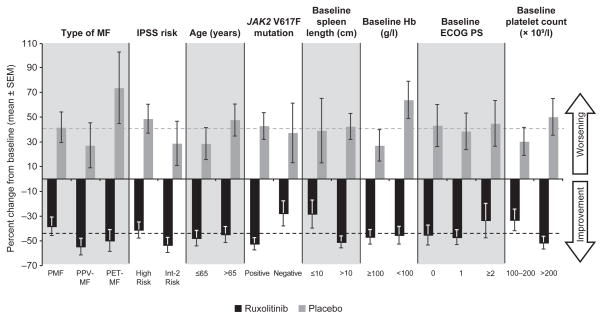
Ruxolitinib demonstrated a benefit over placebo with respect to both spleen volume (Fig 1) and TSS (Fig 2) across all subgroups evaluated. The mean percentage change from baseline to week 24 in spleen volume (Fig 1) and TSS (Fig 2) consistently improved (i.e. decreased) in patients receiving ruxolitinib treatment and worsened in patients receiving placebo across all subgroups evaluated. Mean percentage changes from baseline to week 24 in ruxolitinib-treated patients in each subgroup were similar to mean percentage changes in spleen volume (−31·6%) and TSS (−46·1%) in ruxolitinib-treated patients in the overall study population (Verstovsek et al, 2012).
Fig. 1.
Percentage change in spleen volume from baseline to week 24 by patient subgroup. Dashed lines represent the mean percentage change from baseline for overall treatment group (Verstovsek et al, 2012). ECOG PS, Eastern Cooperative Oncology Group performance status; Hb, haemoglobin; Int-2, Intermediate-2; IPSS, International Prognostic Scoring System; MF, myelofibrosis; PET, post-essential thrombocythaemia; PMF, primary myelofibrosis; PPV, post-polycythaemia vera; SEM, standard error of the mean.
Fig. 2.
Total Symptom Score percentage change from baseline to week 24 by patient subgroup. Dashed lines represent the mean percentage change from baseline for overall treatment group (Verstovsek et al, 2012). ECOG PS, Eastern Cooperative Oncology Group performance status; Hb, haemoglobin; Int-2, Intermediate-2; IPSS, International Prognostic Scoring System; MF, myelofibrosis; PET, post-essential thrombocythaemia; PMF, primary myelofibrosis; PPV, post-polycythaemia vera; SEM, standard error of the mean.
Although there was no subgroup that did not benefit from ruxolitinib therapy with respect to spleen volume reductions and TSS improvements, interaction tests were performed to further assess if baseline characteristics affect treatment outcome. For the mean percentage change from baseline to week 24 in spleen volume, subgroup by treatment interaction P-values for age, JAK2 V617F mutation status and baseline platelet count were all <0·10. The results for JAK2 V617F mutation status (P-value = 0·07) and baseline platelet count (P-value = 0·06) were driven predominantly by differences in the magnitude of spleen volume reductions between ruxolitinib- treated patients, with similar levels of spleen growth relative to baseline noted in placebo-treated patients. In contrast, the result for age (P-value = 0·02) was driven by differences in the magnitude of spleen volume increases between patients receiving placebo, with similar reductions in spleen volume relative to baseline noted in ruxolitinib-treated patients regardless of age. For the mean percentage change from baseline to week 24 in TSS, the subgroup by treatment interaction for baseline haemoglobin (P-value = 0·07) was primarily driven by differences in the magnitude of TSS worsening between patients receiving placebo. Patients receiving placebo with baseline haemoglobin <100 g/l had greater worsening of their TSS than those with haemoglobin ≥100 g/l, while similar degrees of improvement in TSS relative to baseline were observed in ruxolitinib-treated patients regardless of baseline haemoglobin. Regardless of the quantitative differences observed in these subgroups, ruxolitinib-treated patients consistently experienced reductions in spleen volume and improvements in TSS with therapy while patients receiving placebo experienced worsening on these measures. For the remaining subgroup by treatment interaction tests, all P-values were ≥0·10.
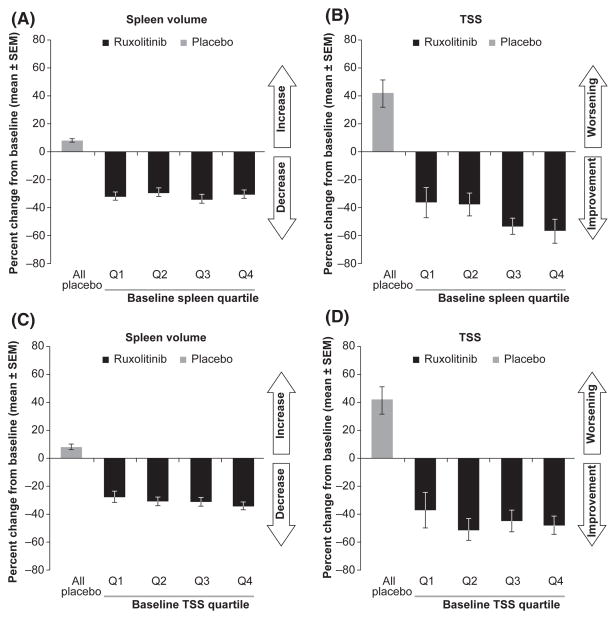
Patients treated with ruxolitinib experienced reductions in spleen volume and improvements in TSS regardless of spleen volume or symptom severity at baseline. Analysis by baseline spleen volume quartile showed mean reductions in spleen volume that ranged from −29.2% to −33.9% (Fig 3A) and mean changes in TSS that ranged from −36.2% to −56.7% (Fig 3B). Analysis by baseline TSS quartile showed mean reductions in spleen volume ranging from −28·3% to −34·8% (Fig 3C). Mean changes in TSS ranged from −37·2% to −51·4% (Fig 3D). In contrast, mean changes in spleen volume and TSS in patients receiving placebo (all quartiles combined) were +8·2% and +41·8% respectively.
Fig. 3.
Change in spleen volume and TSS (baseline to week 24) by (A, B) baseline spleen volume quartile and (C, D) baseline TSS. Patients with missing baseline values for spleen volume or TSS were not included in the analyses for panels B or C, respectively. Q, quartile; SEM, standard error of the mean; TSS, Total Symptom Score.
Survival analysis
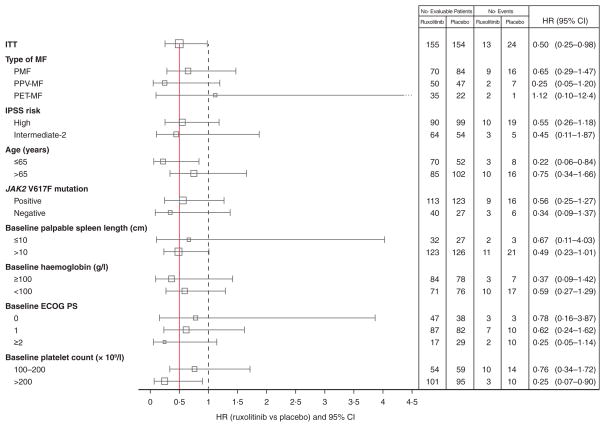
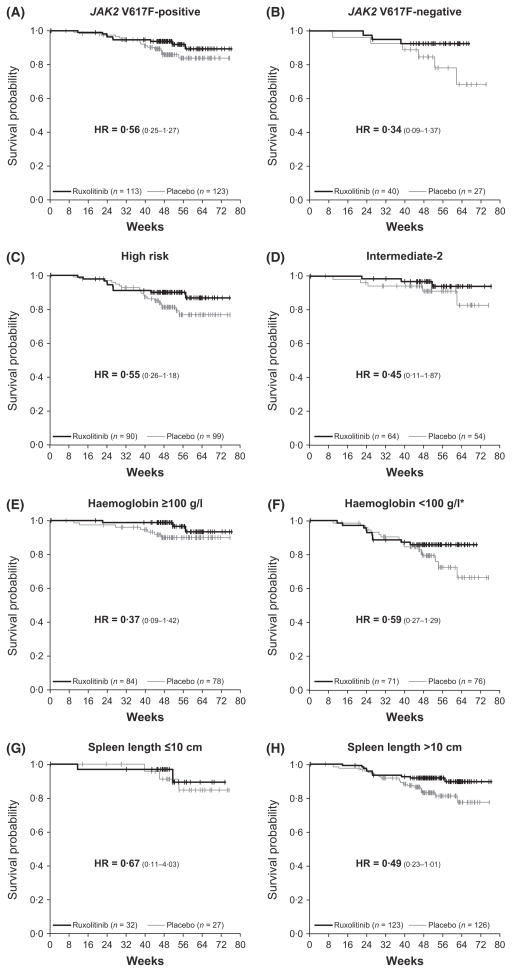
Overall, there were 13 deaths in the ruxolitinib group and 24 in the placebo group during the follow-up period (median follow-up, 51 weeks), representing a HR of 0·50 (95% CI: 0·25–0·98) (P = 0·04) (Verstovsek et al, 2012). In the subgroup analyses of overall survival (Fig 4 and 5), HRs consistently favoured ruxolitinib over placebo (range, 0·22–0·67), with the exception of the PET-MF subgroup (1·12); this probably was the result of the low frequency of events (three in total) in this small subgroup. Among patients in the placebo group, those with low haemoglobin had a particularly poor prognosis (Fig 5F), whereas patients with low haemoglobin who received ruxolitinib appeared to achieve benefits (HR = 0·59) consistent with those observed in other ruxolitinib-treated patients.
Fig. 4.
Forest plot of survival by patient subgroup. Red line represents the hazard ratio (HR) of the ITT population and the dashed line represents an HR of 1. The squares represent the HR and sample size for each subgroup, where the area of the square is proportional to the subgroup sample size. CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; IPSS, International Prognostic Scoring System; ITT, intention-to-treat; PET-MF, post-essential thrombocythaemia myelofibrosis; PMF, primary myelofibrosis; PPV-MF, post-polycythaemia vera myelofibrosis.
Fig. 5.
Overall survival by (A, B) JAK2 V617F mutation status, (C, D) baseline IPSS risk category, (E, F) baseline haemoglobin level and (G, H) baseline palpable spleen length. *Patients who received ≥1 unit of RBC transfusions within 12 weeks before baseline were assigned to the haemoglobin <100-g/l subgroup. HR, hazard ratio; IPSS, International Prognostic Scoring System; RBC, red blood cell.
Discussion
Heterogeneity in patient disease characteristics has been observed in the MF population (Tefferi, 2000; Mesa et al, 2007; Cervantes et al, 2009). Thus, a ‘typical’ MF patient is difficult to characterize. In this analysis, ruxolitinib was shown to be effective in reducing spleen volume and improving MF-related symptoms regardless of MF subtype, age group, IPSS risk group, baseline ECOG performance status, presence/absence of JAK2 V617F mutation, baseline haemoglobin, baseline platelet count, baseline palpable spleen size, baseline spleen volume quartile or baseline TSS quartile. In contrast, spleen volume and symptoms worsened across all evaluated subgroups of patients receiving placebo. These data indicate that, among the population studied in COMFORT-I, there was no subgroup that did not benefit from ruxolitinib therapy and there was no subgroup that did not worsen with placebo.
The COMFORT-I study was designed to follow patients for survival, even after they discontinued study treatment. The current evaluation of survival is an intention-to-treat analysis, so patients were grouped according to the original randomization, regardless of crossover. At the time of the updated survival analyses (4 additional months of follow-up beyond the primary analysis data cut-off; median follow-up duration of 51 weeks), all but two patients had crossed over from the placebo arm to ruxolitinib. Although subgroup comparisons were not powered to show statistically significant differences, the observed prolongation of survival in favour of ruxolitinib relative to placebo was seen across subgroups and was generally consistent with that observed in the total COMFORT-I study population (Verstovsek et al, 2012).
Baseline patient characteristics, such as age >65 years, presence of constitutional symptoms and haemoglobin <100 g/l, are prognostic for shorter survival (Cervantes et al, 2009; Passamonti et al, 2010). In this study, ruxolitinib was associated with a survival advantage over placebo in intermediate-2 and high-risk patients based on IPSS, as well as in subgroups defined by factors included in IPSS risk stratification (e.g. age, haemoglobin). Notably, patients in the placebo arm who had low haemoglobin (<100 g/l) had a particularly poor prognosis, and in patients with low haemoglobin, ruxolitinib treatment was associated with evidence of improved survival. The mechanism underlying the prolonged survival relative to placebo associated with ruxolitinib treatment in COMFORT-I is not clear and is probably multifactorial. However, we believe that reductions in spleen volume and resolution of MF symptoms may have contributed to the observed prolongation of survival in this study. Among 517 deaths in PMF patients described by Cervantes et al (2009), 86 (17%) and 50 (10%) were because of progression to acute myeloid leukaemia and progression of PMF respectively. Thrombosis and cardiovascular complications, infection, bleeding (not related to acute transformation) and portal hypertension accounted for 92 deaths (18%). The prolonged survival associated with ruxolitinib relative to placebo in this study may in part be the result of a modulatory or secondary effect related to those causes of death not attributable to disease progression. For example, resolution of splenomegaly may reduce the risk of portal hypertension in patients with MF.
Although the IPSS (Cervantes et al, 2009) and DIPSS (Passamonti et al, 2010) are tools used for risk stratification of MF patients (and also entry in the COMFORT-I study and ongoing clinical trials), a great degree of patient heterogeneity may exist within IPSS/DIPSS risk categories. For example, symptom assessment within these instruments is limited to the presence/absence of constitutional symptoms. Both exclude potentially significant symptoms that result from splenomegaly, as well as bothersome symptoms such as pruritus, abdominal pain/discomfort, early satiety and bone pain. These symptoms, although not ‘constitutional,’ may be associated with advanced disease and cause substantial disability and/or adversely affect quality of life. Thus, it is important to assess the effects of therapy on aspects of the disease that are not captured by the current prognostic scoring systems. The COMFORT-I study assessed the effects of ruxolitinib on splenomegaly and TSS (a composite score representing six MF-related symptoms, including constitutional and other symptoms). In this present analysis, ruxolitinib improved both spleen volume and TSS compared with placebo, regardless of the degree of splenomegaly or symptom burden at baseline.
As with all subgroup analyses, there are limitations to the present analyses. The purpose of subgroup analyses is to explore the overall heterogeneity of treatment effect across subgroups relative to the overall treatment effect observed in the trial. Small sample sizes within individual subgroups and a large number of different comparisons preclude firm conclusions regarding the absence or presence of benefit limited to any particular subgroup (Second International Study of Infarct Survival (ISIS-2) Collaborative Group, 1988; Cuzick, 2005). However, the results support the treatment effects of ruxolitinib across subgroups that are similar to those observed in the overall COMFORT-I population.
In conclusion, results from these subgroup analyses confirm that there was little heterogeneity of treatment benefit in COMFORT-I, and that ruxolitinib is an effective therapy for the spectrum of MF patients enrolled in this study.
Acknowledgments
The COMFORT-I study was supported by Incyte Corporation. We thank Monica Nicosia, PhD, from the Curry Rockefeller Group, LLC, and Melinda Ramsey, PhD, of Melinda Ramsey, LLC for providing medical writing assistance (funded by Incyte Corporation) with earlier drafts of the manuscript. S.V., R.A.M. and H.A.K. contributed to concept design, data acquisition, data analysis and data interpretation. R.S.L. and V.S. contributed to concept design and data interpretation. J.G., V.G., J.F.D., R.P., J.V.C., R.M.L., E.F.W., R.T.S., C.M. and E.H. contributed to data acquisition and data interpretation. M.D., J.H.H., M.T., M.O.A and A.R. contributed to data acquisition. W.S. performed the statistical analyses. K.V. contributed to concept design. S.E.-V. contributed to data interpretation. All authors assisted with manuscript development or revised it critically for intellectual content as well as approved the final draft of the manuscript.
Footnotes
Disclosure and competing interests statement
S.V. reports receiving grant support through his institution from Incyte Corporation, Exelixis, Celgene, NS Pharma, Infinity Pharmaceuticals, SBIO, Lilly Oncology, AstraZeneca, Geron, Bristol-Myers Squibb, YM BioSciences, Gilead and Roche; RAM., receiving research funding from Incyte Corporation, Lilly, Sanofi, NS Pharma and YM Bioscience; JG., receiving Consultancy, Honoraria and Support for travel to meeting for the study or other purposes from Incyte Corporation; R.S.L., is an employee of Incyte Corporation and owns stock in Incyte Corporation; V.G., receiving grant support through his institution from Incyte Corporation and Novartis, consulting fees from Incyte Corporation and Novartis, and lecture fees from Novartis; J.F.P. has no relationships to disclose; J.V.C., receiving consulting fees from Incyte Corporation; M.D., receiving consulting fees from Bristol-Myers Squibb, Novartis and Ariad, and grant support through his institution from Bristol-Myers Squibb; C.M., receiving grant support through her institution, consulting fees, and lecture fees from Novartis, and payments for development of educational presentations from Incyte Corporation and Novartis; R.T.S., receiving grant support through his institution from Incyte Corporation and lecture fees from Incyte Corporation, and holding stock in Incyte Corporation both individually and through his institution; M.T., receiving membership on an entity’s Board of Directors or advisory committees from Novartis, BMS, Sanofi, Teva and Pfizer and research funding from Novartis, BMS, Ariad, and Sanofi and speakers bureau for Novartis; E.F.W., receiving consultancy and honoraria from Incyte Corporation; J.H.H. has no relationships to disclose; M.O.A., receiving research funding from Incyte Corporation; E.H., has no relationships to disclose; R.M.L., receiving grant support through his institution from Incyte Corporation; R.P., receiving consulting fees paid through his institution from Incyte Corporation; A.R., has no relationships to disclose; K.V., W.S. and V.S. are employees of Incyte Corporation and own stock in Incyte Corporation; S.E.-V. is a former employee of Incyte Corporation and owns stock in Incyte Corporation. R.M.K., receiving grant support through his institution from Incyte Corporation.
References
- Barbui T, Barosi G, Birgegard G, Cervantes F, Finazzi G, Griesshammer M, Harrison C, Hasselbalch HC, Hehlmann R, Hoffman R, Kiladjian JJ, Kroger N, Mesa R, McMullin MF, Pardanani A, Passamonti F, Vannucchi AM, Reiter A, Silver RT, Verstovsek S, Tefferi A European Leukemia Net. Philadelphia-negative classical myeloproliferative neoplasms: critical concepts and management recommendations from European LeukemiaNet. Journal of Clinical Oncology. 2011;29:761–770. doi: 10.1200/JCO.2010.31.8436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barosi G, Mesa RA, Thiele J, Cervantes F, Campbell PJ, Verstovsek S, Dupriez B, Levine RL, Passamonti F, Gotlib J, Reilly JT, Vannucchi AM, Hanson CA, Solberg LA, Orazi A, Tefferi A International Working Group for Myelofibrosis Research Treatment (IWG-MR) Proposed criteria for the diagnosis of post-polycythemia vera and post-essential thrombocythemia myelofibrosis: a consensus statement from the International Working Group for Myelofibrosis Research and Treatment. Leukemia. 2008;22:437–438. doi: 10.1038/sj.leu.2404914. [DOI] [PubMed] [Google Scholar]
- Cervantes F, Pereira A, Esteve J, Rafel M, Cobo F, Rozman C, Montserrat E. Identification of ‘short-lived’ and ‘long-lived’ patients at presentation of idiopathic myelofibrosis. British Journal of Haematology. 1997;97:635–640. doi: 10.1046/j.1365-2141.1997.782714.x. [DOI] [PubMed] [Google Scholar]
- Cervantes F, Dupriez B, Pereira A, Passamonti F, Reilly JT, Morra E, Vannucchi AM, Mesa RA, Demory JL, Barosi G, Rumi E, Tefferi A. New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood. 2009;113:2895–2901. doi: 10.1182/blood-2008-07-170449. [DOI] [PubMed] [Google Scholar]
- Cuzick J. Forest plots and the interpretation of subgroups. Lancet. 2005;365:1308. doi: 10.1016/S0140-6736(05)61026-4. [DOI] [PubMed] [Google Scholar]
- Gangat N, Caramazza D, Vaidya R, George G, Begna K, Schwager S, Van Dyke D, Hanson C, Wu W, Pardanani A, Cervantes F, Passamonti F, Tefferi A. DIPSS plus: a refined Dynamic International Prognostic Scoring System for primary myelofibrosis that incorporates prognostic information from karyotype, platelet count, and transfusion status. Journal of Clinical Oncology. 2011;29:392–397. doi: 10.1200/JCO.2010.32.2446. [DOI] [PubMed] [Google Scholar]
- Mesa RA, Niblack J, Wadleigh M, Verstovsek S, Camoriano J, Barnes S, Tan AD, Atherton PJ, Sloan JA, Tefferi A. The burden of fatigue and quality of life in myeloproliferative disorders (MPDs): an international Internet-based survey of 1179 MPD patients. Cancer. 2007;109:68–76. doi: 10.1002/cncr.22365. [DOI] [PubMed] [Google Scholar]
- Nguyen H, Gotlib J. Insights into the molecular genetics of myeloproliferative neoplasms. American Society of Clinical Oncology (ASCO) Education Book. 2012:411–418. doi: 10.14694/EdBook_AM.2012.32.85. Available at: http://www.asco.org/ASCOv2/Home/Education%20&%20Training/Educational%20Book/PDF%20Files/2012/zds00112000411.PDF. [DOI] [PubMed]
- Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP. Toxicity and response criteria of the Eastern Cooperative Oncology Group. American Journal of Clinical Oncology. 1982;5:649–655. [PubMed] [Google Scholar]
- Passamonti F, Cervantes F, Vannucchi AM, Morra E, Rumi E, Pereira A, Guglielmelli P, Pungolino E, Caramella M, Maffioli M, Pascutto C, Lazzarino M, Cazzola M, Tefferi A. A dynamic prognostic model to predict survival in primary myelofibrosis: a study by the IWG-MRT (International Working Group for Myeloproliferative Neoplasms Research and Treatment) Blood. 2010;115:1703–1708. doi: 10.1182/blood-2009-09-245837. [DOI] [PubMed] [Google Scholar]
- Second International Study of Infarct Survival (ISIS-2) Collaborative Group. Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2. Lancet. 1988;2:349–360. [PubMed] [Google Scholar]
- Tefferi A. Myelofibrosis with myeloid metaplasia. New England Journal of Medicine. 2000;342:1255–1265. doi: 10.1056/NEJM200004273421706. [DOI] [PubMed] [Google Scholar]
- Tefferi A. How I treat myelofibrosis. Blood. 2011;117:3494–3504. doi: 10.1182/blood-2010-11-315614. [DOI] [PubMed] [Google Scholar]
- Tefferi A, Vardiman JW. Classification and diagnosis of myeloproliferative neoplasms: the 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia. 2008;22:14–22. doi: 10.1038/sj.leu.2404955. [DOI] [PubMed] [Google Scholar]
- Tefferi A, Vaidya R, Caramazza D, Finke C, Lasho T, Pardanani A. Circulating interleukin (IL)-8, IL-2R, IL-12, and IL-15 levels are independently prognostic in primary myelofibrosis: a comprehensive cytokine profiling study. Journal of Clinical Oncology. 2011;29:1356–1363. doi: 10.1200/JCO.2010.32.9490. [DOI] [PubMed] [Google Scholar]
- Vainchenker W, Dusa A, Constantinescu SN. JAKs in pathology: role of Janus kinases in hematopoietic malignancies and immunodeficiencies. Seminars in Cell and Developmental Biology. 2008;19:385–393. doi: 10.1016/j.semcdb.2008.07.002. [DOI] [PubMed] [Google Scholar]
- Vainchenker W, Delhommeau F, Constantinescu SN, Bernard OA. New mutations and pathogenesis of myeloproliferative neoplasms. Blood. 2011;118:1723–1735. doi: 10.1182/blood-2011-02-292102. [DOI] [PubMed] [Google Scholar]
- Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A, Harris NL, Le Beau MM, Hellstrom-Lindberg E, Tefferi A, Bloomfield CD. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114:937–951. doi: 10.1182/blood-2009-03-209262. [DOI] [PubMed] [Google Scholar]
- Verstovsek S. Therapeutic potential of JAK2 inhibitors. Hematology/the Education Program of the American Society of Hematology. 2009:636–642. doi: 10.1182/asheducation-2009.1.636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verstovsek S, Mesa RA, Gotlib J, Levy RS, Gupta V, DiPersio JF, Catalano JV, Deininger M, Miller C, Silver RT, Talpaz M, Winton EF, Harvey JH, Jr, Arcasoy MO, Hexner E, Lyons RM, Paquette R, Raza A, Vaddi K, Erickson-Viitanen S, Koumenis IL, Sun W, Sandor V, Kantarjian HM. A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis. New England Journal of Medicine. 2012;366:799–807. doi: 10.1056/NEJMoa1110557. [DOI] [PMC free article] [PubMed] [Google Scholar]