Abstract
Introduction
Bisulfite treatment of DNA introduces methylation-dependent sequence changes through selective chemical conversion of nonmethylated cytosine to uracil and serves as pretreatment step for the majority of DNA methylation analysis methods.
Methods
We have evaluated the conversion performance of five of the most commonly used bisulfite treatment kits [MethylDetector (Active Motif), Epitect+ (Qiagen), Zymo Methylation, Zymo Gold and Zymo Lightning (all from Zymo Research)] by pyrosequencing four different regions with variable methylation levels, including: a repetitive element (ALUSX), a gene with low levels of methylation (IL6ST), an imprinted gene expected to be approximately 50% methylated (IGF2), and a fully methylated gene (ST3GAL2). In addition, we have studied the influence of duration (3 vs. 16 h) and type (fixed temperature vs. cycling program) of incubation protocol on the conversion efficiency of each evaluated kit.
Results
All kits produced similar conversion rates of ALUSX, IGF2 and ST3GAL2, while the conversion of the low methylated IL6ST gene was variable between kits. The Zymo kits were highly consistent in their performance even when different protocols of incubation were applied, generating full conversion at the low methylated gene IL6; this was not true for the MethylDetector and Epitect+ kits. However, long-cycling incubation could produce similar conversion rates for the same locus in combination with Active Motif and Qiagen kits.
Conclusions
The selection of a long-cycling protocol during conversion permits standardization of protocols, improving the reproducibility of methylation estimates across laboratories for gene-specific, genome-wide and bisulfite-based sequencing analyses.
Keywords: DNA methylation, Bisulfite treatment, Pyrosequencing Abstract
Introduction
DNA methylation is an important molecular mechanism regulating several biological processes, such as genomic imprinting, X-inactivation, and tissue-specific gene expression. Since its original identification as an epigenetic regulator, numerous assays have been developed to study cytosine methylation, including methylation-sensitive restriction enzyme mapping, deamination of unmethylated cytosines with sodium bisulfite, and enrichment with targeting antibodies [1, 2]. In the last 10 years, profiling and fingerprinting the human methylome has become increasingly focused on developing methodologies that enable single base pair resolution due to the observed functional consequences of both regional and single CpG site methylation changes [3-6].
Most of these methods rely on bisulfite conversion of genomic DNA. Introduced in 1970 by Hayatsu and colleagues [7, 8], bisulfite treatment opened new possibilities in the field of the DNA methylation study by providing a means to quantitatively assess site-specific methylation [9], Bisulfite treatment deaminates cytosine to uracil and leaves 5-methylcytosine unchanged [9,10], enabling estimation of the average methylation level as the proportion of cytosines in a given DNA sequence. However, a number of problems can arise during bisulfite treatment of genomic DNA that lead to incorrect conversion of the unmethylated cytosines. Failed conversion occurs when an unmethylated cytosine is not deaminated, resulting in false methylation detection [11], This can inflate estimations of methylation densities, and can typically be reduced both by increasing the duration of bisulfite treatment [12] or the number of denaturation steps used during conversion [13, 14], Inappropriate conversion occurs instead when a methylated cytosine is deaminated to thymine, the reasons for which remain unknown [11,12,15].
Many DNA bisulfite treatment kits are now available on the market. However, each kit proposes different protocols and conditions that may lead to different efficiency and accuracy of DNA conversion. The main differences reside in the duration (from 1 to 16 h) and type (fixed vs. cycling protocol) of incubation during the desulfonation step of the reaction. We evaluated the bisulfite conversion performance of five different commercially available kits by pyrosequencing a set of genes with variable expected DNA methylation status. In addition, we studied the influence of duration and type of incubation protocol on the conversion efficiency of each evaluated kit.
Materials and Methods
Genomic DNA was extracted from whole blood of two anonymous healthy Caucasian male donors (individuals A and B, aged between 40–50 years) by using the Gentra Puregene Blood Kit (Qiagen) according to the manufacturer’s protocol. DNA bisulfite treatment was performed using the MethylDetector (Active Motif), Epitect+ (Qiagen), Zymo Methylation, Zymo Gold, and the Zymo Lightning (all from Zymo Research) kits according to their respective manufacturer’s instructions. In addition, three different incubation protocols (‘cycling 16 h’, ‘cycling 8 h’ and ‘fixed 3 h’) were used to test the performance of the MethylDetector, Epitect+ and Zymo Gold kits. The evaluated kits and protocols are summarized in table 1. Each incubation protocol was tested in duplicate, and for each sample of bisulfite-treated DNA, two PCR replicates were performed. All bisulfite conversions were undertaken on the same thermo-cycler. After bisulfite treatment, all samples were immediately stored at −20°C and simultaneously analyzed by pyrosequencing in a second phase of the study.
Table 1.
Alternative incubation protocols tested to evaluate the performance ofthe bisulfite treatment kits
| Name of the protocol | Incubation steps |
|---|---|
| Cycling 16 h | 16 cycles of 95° C for 30 s and 50° C for 1 h, hold at 4° C |
| Cycling 8 h | 95°C for 5 s, 60° C for 30 s, 8 cycles of 95° C for 6 s and 60° C for 30 s, hold at 4° C |
| Fixed 3 h | 98°C for l0 s and 64° C for 2 h 40 min, hold at 4° C |
All five kits included in this study [Epitect+ (Qiagen], MethylDetector (Active Motif), Zymo Gold, Zymo Methylation and Zymo Lightning (all three from Zymo Research]] were tested according to the protocol specified by the respective manufacturer for each kit. In addition, the Epitect+, MethylDetector, Zymo Gold and Zymo Methylation were tested using the protocols herein listed to evaluate the influence of type and duration ofDNA incubation ontheir performance.
Four different regions were pyrosequenced to assess the bisulfite conversion of DNA methylation: ALUSX (a repetitive element), IL6ST (low methylated gene), IGF2 (imprinted gene with expected 50% methylation), and ST3GAL2 (fully methylated gene), IL6ST and ST3GAL2 were selected from an existing Illumina Infinium 450K study performed on blood buffy coat genomic DNA from a sample of healthy individuals.ALUSX [16] and IGF2 [17, 18] pyrosequencing assays have been described previously. New methylation assays were designed for IL6ST and ST3GAL2 using the PyroMark Assay Design Software 2.0 (www.qiagen.com). The locations of the amplicons are described in table 2. The IL6ST and ST3GAL2 assays were validated with a DNA methylation scale (from 0 to 100%), which was created from whole-genome amplified DNA (hypomethylated DNA), and DNA treated with CpG methyl-transferase M.SssI (hypermethylated DNA; New England Biolabs, Ipswich, Mass., USA), The methylation scales were tested in duplicate for each assay as described [18, 19]. Each assay included a bisulfite conversion check to verify full conversion of the DNA. Methylation levels for all CpG sites were assessed using the Pyromark Q24 pyrosequencer (Qiagen). The reported methylation levels are the average of two distinct bisulfite treatments performed for each of the protocol and kit combination. Replicates had an SD below 5%.
Table 2.
Location and characteristics of pyrosequencing amplicons for ALUSX, IL6ST, IGF2 and ST3GAL2 used inthe study
| Name of assay |
Chromo- some |
Start | End | No. of CpGs analyzed |
Forward | Reverse | Sequencing | Biotiny- lated |
Annealing temperature |
|---|---|---|---|---|---|---|---|---|---|
| ALUSX | _ | _ | _ | 6 | TTTTTATTAAAAA- | CCCAAACTAAAA- | AATAACTAAAA- | Forward | 50° C |
| TATAAAAATT | TACAATAA | TTACAAAC | |||||||
| IGF2 | 11 | 2169369 | 2169402 | 3 | TGAGGATGGGTTTTT- | TCCTCGATCCACCC- | GGGGTGGA- | Reverse | 56° C |
| GTTTGGTAT | AAAATAATAT | GGGTGTA | |||||||
| IL6ST | 5 | 55290797 | 55290852 | 9 | TGAAGGAGTAGGGA- | ACCTCT GCGGAG | TGAAGGAGTAGGG- | Reverse | 54° C |
| TTTGAAGC | AAG GAT CT | ATTTGAAGC | |||||||
| ST3GAL2 | 16 | 70423257 | 70432300 | 5 | GGTTTTTTTGTTGGT- | CTTACCCCAAAACC- | GGGTTTTGG- | Reverse | 55°C |
| GTTTATTATGT | TCTCCTTACTAAA | ATGGGA |
Nucleotide position according to the February 2009 human reference sequence (GRCh37/hgl9) produced by the International Human Genome Sequencing Consortium.
Results
All analyses were performed using blood samples from two healthy male individuals. The patterns of methylation variation induced by the kit protocol were similar between the individuals. For this reason, methylation levels are only displayed for one individual, with the findings for the second reported in the online supplementary information (for all online suppl. material, see www.karger.com/doi/10.1159/000358882).
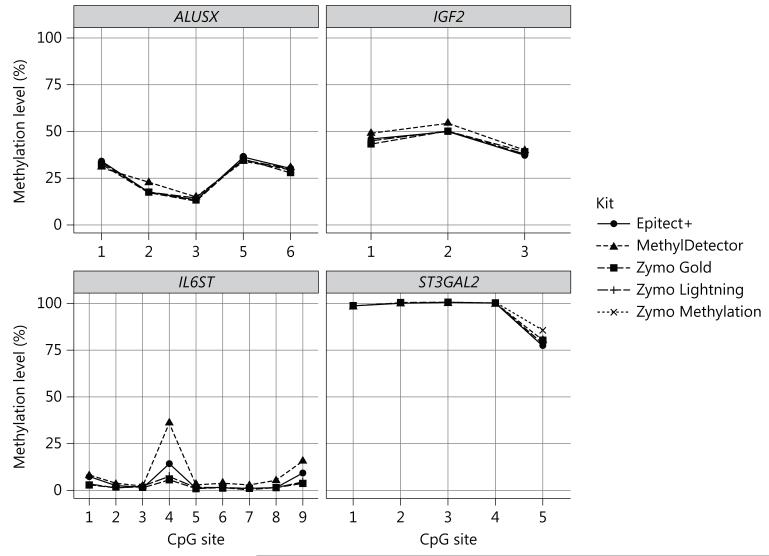
We first evaluated the bisulfite conversion performance of MethylDetector, Epitect+, Zymo Methylation and Zymo Gold by processing the samples according to the manufacturer’s instructions. While the methylation levels of ALUSX, IGF2 and ST3GAL2 were relatively constant across the kits, the methylation level of the hypomethylated IL6ST was more variable at certain CpG sites (CpG site 4 and 9, fig. 1), The methylation levels estimated using the Zymo Methylation and Gold kits were comparable, while MethylDetector and Epitect+ produced apparent incomplete conversion at IL6ST CpG4 (fig. 1), We also tested the conversion efficiency of Zymo Lightning for the same regions and compared it with the performance of both Zymo Methylation and Zymo Gold. Zymo Lightning is a recently released fast bisulfite treatment option. According to the manufacturers’ protocol, efficient conversion of cytosines can be achieved after an incubation protocol of 1 h and 8 min, dramatically reducing the time of the bisulfite-treated DNA sample preparation. The methylation level of each gene was similar across the three Zymo kits (fig. 1).
Fig. 1.
Performance of Active Motif, Epitect+, Zymo Methylation, Zymo Gold and Zymo Lightning kits according to the respective manufacturer’s protocols (individual A). ALUSX, IGF2, IL6ST and ST3GAL2 methylation (%) estimates assessed on human whole-blood DNA from individual A using the Epitect+, MethylDetector, Zymo Gold, Zymo Lightning and Zymo Methylation kits according to the manufacturer’s protocols. Data are reported as mean of two different bisulfite treatments after which two technical replicates have been performed (SD across bisulfite and technical replicates was <5% for each of the CpG sites analyzed).
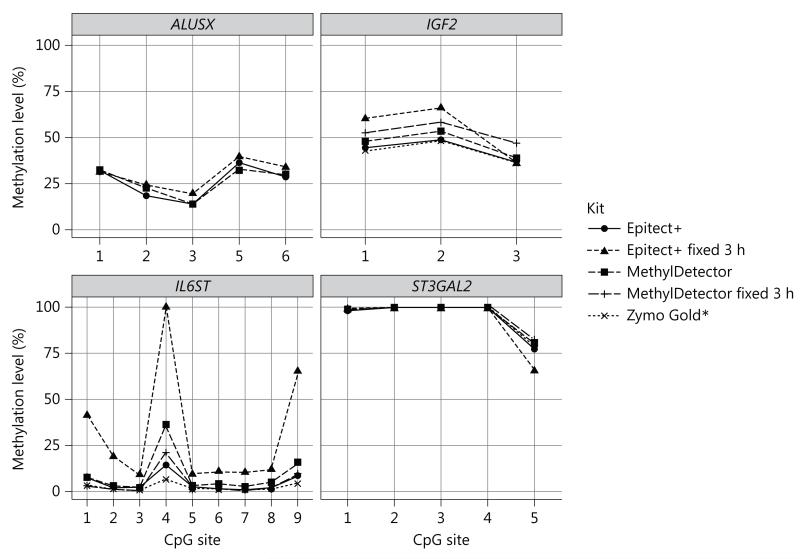
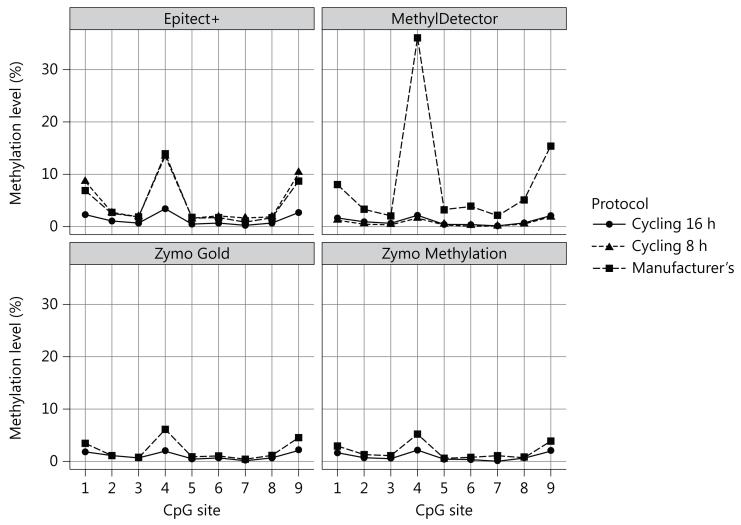
Four of the kits tested (Epitect+, MethylDetector, Zymo Gold and Zymo Methylation) differ with respect to their conversion/incubation protocol. To assess the potential impact of the incubation protocol, we tested two different incubation sequences of temperatures for each kit. The first protocol, designated as ‘cycling 16 h’ is recommended by the Zymo Methylation kit manufacturer instructions to be used when performing Illumina Infinium 450K array analysis. It consists of 16 cycles of 95°C for 30 s, 50°C for 1 h, and subsequently holding the temperature constant at 4°C. The second protocol, ‘cycling 8 h’, was custom designed to evaluate the impact of a shorter cycling protocol on the cytosine conversion. The custom protocol consists of eight cycles of 95°C for 6 min, 60°C for 30 min and subsequently holding the temperature at4°C. The ‘cycling 16 h’ incubation generated more efficient conversion for all four kits tested at the IL6ST locus. When using the MethylDetector kit from Active Motif (fig. 2), IL6ST CpG4 methylation was ten times lower after the application of the ‘cycling 16 h’ incubation protocol (fig. 3), Smaller differences, following a similar trend, were observed for the Zymo and Epitect+ kits (fig. 3), No differences were observed for the other genes studied (data not shown). When applying the ‘cycling 8 h’ protocol, better conversion was achieved for the MethylDetector, the Zymo Gold and Zymo Methylation kits (with comparable values to the ‘cycling 16 h’ protocol). The ‘cycling 8 h’ protocol did not increase the cytosine conversion using the Epitect+ kit (fig. 3).
Fig. 2.
Effect of the fixed/short incubation protocols on conversion efficiency (individual A), ALUSX, IGF2, IL6ST and ST3GAL2 methylation (%) estimates assessed on human whole-blood DNA from individual A using the Epitect+, MethylDetector and Zymo Gold kits according to the manufacturer’s protocols, respectively, or by applying the ‘fixed 3 h’ incubation protocol (see table 1), Data are reported as mean of two different bisulfite treatments after which two technical replicates have been performed (SD across bisulfite and technical replicates was <5% for each of the CpG sites analyzed).
Fig. 3.
Effect of the cycling incubation protocols on conversion efficiency for IL6ST methylation (individual A), Comparison of different DNA incubation protocols (‘cycling 16 h’, ‘cycling 8 h’ and ‘manufacturer’s’, see table 1) in determining IL6ST methylation (%) estimates assessed on human whole-blood DNA from individual A using the Epitect+, MethylDetector, Zymo Gold and Zymo Methylation kits. Data are reported as mean of two different bisulfite treatments after which two technical replicates have been performed (SD across bisulfite and technical replicates was <5% for each of the CpG sites analyzed).
To confirm that long incubation with cycling (‘cycling 16 h’ protocol) indeed generates the best conversion efficiency of unmethylated cytosines, we also used a third protocol (‘fixed T’) for both the Active Motif and Epitect+ kits. This manufacturer’s protocol for the Zymo Methylation Gold kit consists of incubation at 98°C for 10 min, 64°C for 2.5 h, and a final holding temperature at 4°C. The Zymo Gold performance according to the manufacturer’s protocol is shown in figure 1. The ‘fixed 3 h’ protocol did not effectively convert cytosines when using either MethylDetector or Epitect+ kits, with higher variability in the methylation values detected across the four regions included in this study (fig. 2).
Discussion
Most technologies used for DNA methylation analyses rely on bisulfite treatment of DNA, which introduces methylation-dependent sequence changes through selective chemical conversion of nonmethylated cytosine to uracil. Currently, a number of commercial kits are available for bisulfite conversion.
We evaluated the conversion performance of four of the most commonly used bisulfite treatment kits (MethylDetector, Epitect+, Zymo Methylation, and Zymo Gold) and the recently released fast bisulfite kit from Zymo (Zymo Lightning) by pyrosequencing four candidate regions with different methylation values. These regions included a repetitive element (ALUSX), a low methylated gene (IL6ST) , an imprinted gene with expected methylation values around 50% (IGF2) and a fully methylated gene (ST3GAL2). Generally, all kits have been designed to ensure minimal DNA degradation during conversion and minimal DNA loss during purification to avoid low DNA yield, highly fragmented DNA, and irreproducible conversion rates. Sulfonation and cytosine deamination for all kits used in this study occur during DNA incubation with the respective conversion reagents, while desulfonation occurs in on-column steps that include DNA purification as well. However, some differences in reagents and sample preparation protocol exist and might result in different conversion rates. There are several differences in the material prescribed in the protocols between kits. These include specific buffers, such as the ‘DNA Protect Buffer’ in the Epitect+ kit that contains a pH indicator dye as a mixing control in the setup of the reaction, allowing confirmation of the correct pH necessary for cytosine conversion. Unlike Zymo Gold and Epitect+, the MethylDetector and Zymo Methylation protocols separate the alkaline/denaturation step from the conversion step itself. Additionally, Epitect+ and MethylDetector utilize flat filters for the on-column steps, while Zymo kits utilize a conical filter. Active Motif provides a positive PCR control in its MethylDetector kit, which amplifies a specific region of the pl6 locus on chromosome 9 to check for conversion of the DNA samples.
Grunau et al. [12] observed that at a given bisulfite concentration, the reaction kinetics of the conversion of cytosine to uracil are nonlinear and depend on the reaction temperature and the reaction time. Accordingly, we found that the variability of the conversion reaction is associated with incubation duration (short vs. longtime) and type (fixed vs. cycling program). Despite their differences, all Zymo kits were highly consistent in their performance, even when different protocols of incubation were applied (fig. 1, 2), We observed similar conversion efficiency for IL6ST, for which we expected to see higher variability, when applying a long-cycling incubation to the MethylDetector and Epitect+ kits (fig. 3), When using each of the manufacturer-recommended protocols, we did not observe the same conversion rate (fig. 1), A short incubation program (‘fixed 3 h’ protocol) at a fixed temperature confirmed the latter finding, as it generated highly variable methylation results across all regions studied (fig. 2).
Performing a reproducible bisulfite conversion reaction is necessary for both genome-wide and gene-specific DNA methylation studies. Particularly, genome-wide data need to be comparable among laboratories to integrate studies or validate DNA methylation findings [6], At the moment, several tools and techniques are available to assess genome-wide DNA methylation levels; two in particular, reduced representation of bisulfite sequencing [20, 21] and the Infinium Human Methylation 450 Bead Chip Array [22, 23], have become increasingly popular due to their ability to deliver site-specific detail with a relatively high sensitivity. However, different bisulfite treatment kits are often used for these two methods (Epitect+ and Zymo Methylation, respectively) [21,23,24] with different incubation duration and type during conversion. Epigenome-wide association studies using reduced representation of bisulfite sequencing or the 450K Bead Chip have reported small but significant methylation differences between the groups that are comparable in magnitude to the variability observed among the kits used in our study for the low methylated IL6ST region [6], Increased technical variation among hypomethylated regions decreases our ability to identify significant changes in these distinct genomic contexts that may have biologically relevant regulation. Accurate and precise methylation estimates are also required for candidate gene studies. Genomic imprinting-dependent DNA methylation [17, 25] and stochastic epigenetic variability observed in cancer [26-28] are quantitative traits and require reproducible assays given their possible application as diagnostic and/or prognostic markers. Small differences in methylation have been reported to impact gene expression [29].
In conclusion, we have compared the performance of five commercially available bisulfite treatment kits by pyrosequencing regions representative of different methylation levels and genomic context. Our results suggest that duration (short vs. long) and type (fixed temperature vs. cycling) of DNA incubation can impact the conversion reaction. While variability of conversion was observed among kits from different manufacturers, all three Zymo kits tested produced consistent bisulfite conversion across all genes analyzed.
Using a long-cycling protocol during conversion may provide a means of standardizing bisulfite treatment of genomic DNA across different laboratories and bisulfite kits, and their subsequent application in gene-specific analysis, genome-wide arrays and bisulfite-based sequencing methodologies.
Acknowledgements
We thank Amy L. Non, Rebecca C. Rancourt, Timothy M. Barrow, and Jessica Larocca from the Michels’ lab for the constructive discussions.
Alexandra Binder was supported by Training Grant T32HD060454 in Reproductive, Perinatal and Pediatric Epidemiology from the National Institute of Child Health and Human Development, National Institutes of Health. Dr. Karin Michels was supported in part by Public Health Service Grant 1R01CA158313 from the National Cancer Institute/National Institute of Environmental Health Sciences, National Institutes of Health.
References
- 1.Laird PW. Principles and challenges of genome wide DNA methylation analysis. Nat Rev Genet. 2010;ll:191–203. doi: 10.1038/nrg2732. [DOI] [PubMed] [Google Scholar]
- 2.Harrison A, Parle-Mcdermott A. DNA methylation: a timeline of methods and applications. Front Genet. 2011;2:74. doi: 10.3389/fgene.2011.00074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xu J, Pope SD, Jazirehi AR, Attema JL, Papathanasiou P, Watts JA, Zaret KS, Weissman IL, Smale ST. Pioneer factor interactions and unmethylated CpG dinucleotides mark silent tissue-specific enhancers in embryonic stem cells. Proc Natl Acad Sci USA. 2007;104:12377–12382. doi: 10.1073/pnas.0704579104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nativio R, Sparago A, Ito Y, Weksberg R, Riccio A, Murrell A. Disruption of genomic neighbourhood at the imprinted IGF2-H19 locus in Beckwith-Wiedemann syndrome and Silver-Russell syndrome. Hum Mol Genet. 2011;20:1363–1374. doi: 10.1093/hmg/ddr018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gibney ER, Nolan CM. Epigenetics and gene expression. Heredity (Edinb) 2010;105:4–13. doi: 10.1038/hdy.2010.54. [DOI] [PubMed] [Google Scholar]
- 6.Michels KB, Binder AM, Dedeurwaerder S, Epstein CB, Greally JM, Gut I, Houseman EA, Izzi B, Kelsey KT, Meissner A, Milosavljevic A, Siegmund KD, Bock C, Irizarry RA. Recommendations for the design and analysis of epigenome-wide association studies. Nat Methods. 2013;10:949–955. doi: 10.1038/nmeth.2632. [DOI] [PubMed] [Google Scholar]
- 7.Hayatsu H, Wataya Y, Kai K, Iida S. Reaction of sodium bisulfite with uracil, cytosine, and their derivatives. Biochemistry. 1970;9:2858–2865. doi: 10.1021/bi00816a016. [DOI] [PubMed] [Google Scholar]
- 8.Hayatsu H, Wataya Y, Kazushige K. The addition of sodium bisulfite to uracil and to cytosine. J Am Chem Soc. 1970;92:724–726. doi: 10.1021/ja00706a062. [DOI] [PubMed] [Google Scholar]
- 9.Frommer M, Mcdonald LE, Millar DS, Collis CM, Watt F, Grigg GW, Molloy PL, Paul CL. A genomic sequencing protocol that yields a positive display of 5-methylcytosine residues in individual DNA strands. Proc Natl Acad Sci USA. 1992;89:1827–1831. doi: 10.1073/pnas.89.5.1827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Clark SJ, Harrison J, Paul CL, Frommer M. High sensitivity mapping of methylated cytosines. Nucleic Acids Res. 1994;22:2990–2997. doi: 10.1093/nar/22.15.2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Genereux DP, Johnson WC, Burden AF, Stoger R, Laird CD. Errors in the bisulfite conversion of DNA: modulating inappropriate- and failed-conversion frequencies. Nucleic Acids Res. 2008;36:el50. doi: 10.1093/nar/gkn691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Grunau C, Clark SJ, Rosenthal A. Bisulfite genomic sequencing: systematic investigation of critical experimental parameters. Nucleic Acids Res. 2001;29:E65–65. doi: 10.1093/nar/29.13.e65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Laird CD, Pleasant ND, Clark AD, Sneeden JL, Hassan KM, Manley NC, Vary JC, Jr, Morgan T, Hansen RS, Stoger R. Hairpin-bisulfite PCR: assessing epigenetic methylation patterns on complementary strands of individual DNA molecules. Proc Natl Acad Sci USA. 2004;101:204–209. doi: 10.1073/pnas.2536758100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stoger R, Kajimura TM, Brown WT, Laird CD. Epigenetic variation illustrated by DNA methylation patterns of the fragile-X gene FMR1. Hum Mol Genet. 1997;6:1791–1801. doi: 10.1093/hmg/6.11.1791. [DOI] [PubMed] [Google Scholar]
- 15.Shiraishi M, Hayatsu H. High-speed conversion of cytosine to uracil in bisulfite genomic sequencing analysis of DNA methylation. DNA Res. 2004;11:409–415. doi: 10.1093/dnares/11.6.409. [DOI] [PubMed] [Google Scholar]
- 16.Byun HM, Nordio F, Coull BA, Tarantini L, Hou L, Bonzini M, Apostoli P, Bertazzi PA, Baccarelli A. Temporal stability of epigenetic markers: sequence characteristics and predictors of short-term DNA methylation variations. PLoS One. 2012;7:e39220. doi: 10.1371/journal.pone.0039220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rancourt RC, Harris HR, Barault L, Michels KB. The prevalence of loss of imprinting of H19 and IGF2 at birth. FASEB J. 2013;27:3335–3343. doi: 10.1096/fj.12-225284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rancourt RC, Harris HR, Michels KB. Methylation levels at imprinting control regions are not altered with ovulation induction or in vitro fertilization in a birth cohort. Hum Reprod. 2012;27:2208–2216. doi: 10.1093/humrep/des151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Barault L, Ellsworth RE, Harris HR, Valente AL, Shriver CD, Michels KB. Leukocyte DNA as surrogate for the evaluation of imprinted Loci methylation in mammarytissue DNA. PLoS One. 2013;8:e55896. doi: 10.1371/journal.pone.0055896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Meissner A, Gnirke A, Bell GW, Ramsahoye B, Lander ES, Jaenisch R. Reduced representation bisulfite sequencing for comparative high-resolution DNA methylation analysis. Nucleic Acids Res. 2005;33:5868–5877. doi: 10.1093/nar/gki901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Boyle P, Clement K, Gu H, Smith ZD, Ziller M, Fostel JL, Holmes L, Meldrim J, Kelley F, Gnirke A, Meissner A. Gel-free multiplexed reduced representation bisulfite sequencing for large-scale DNA methylation profiling. Genome Biol. 2012;13:R92. doi: 10.1186/gb-2012-13-10-r92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bibikova M, Barnes B, Tsan C, Ho V, Klotzle B, Le JM, Delano D, Zhang L, Schroth GP, Gunderson KL, Fan JB, Shen R. High density DNA methylation array with single CpG site resolution. Genomics. 2011;98:288–295. doi: 10.1016/j.ygeno.2011.07.007. [DOI] [PubMed] [Google Scholar]
- 23.Bibikova M, Le J, Barnes B, Saedinia-Melnyk S, Zhou L, Shen R, Gunderson KL. Genome-wide DNA methylation profiling using Infinium(R) assay. Epigenomics. 2009;1:177–200. doi: 10.2217/epi.09.14. [DOI] [PubMed] [Google Scholar]
- 24.Gu H, Smith ZD, Bock C, Boyle P, Gnirke A, Meissner A. Preparation of reduced representation bisulfite sequencing libraries for genome-scale DNA methylation profiling. Nat Protoc. 2011;6:468–481. doi: 10.1038/nprot.2010.190. [DOI] [PubMed] [Google Scholar]
- 25.Turan N, Katari S, Gerson LF, Chalian R, Foster MW, Gaughan JP, Coutifaris C, Sapienza C. Inter- and intra-individual variation in allele-specific DNA methylation and gene expression in children conceived using assisted reproductive technology. PLoS Genet. 2010;6:el001033. doi: 10.1371/journal.pgen.1001033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hansen KD, Timp W, Bravo HC, Sabunciyan S, Langmead B, Mcdonald OG, Wen B, Wu H, Liu Y, Diep D, Briem E, Zhang K, Irizarry RA, Feinberg AP. Increased methylation variation in epigenetic domains across cancer types. Nat Genet. 2011;43:768–775. doi: 10.1038/ng.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Teschendorff AE, Jones A, Fiegl H, Sargent A, Zhuang JJ, Kitchener HC, Widschwendter M. Epigenetic variability in cells of normal cytology is associated with the risk of future morphological transformation. Genome Med. 2012;4:24. doi: 10.1186/gm323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Teschendorff AE, Menon U, Gentry-Maharaj A, Ramus SJ, Gayther SA, Apostolidou S, Jones A, Lechner M, Beck S, Jacobs IJ, Widschwendter M. An epigenetic signature in peripheral blood predicts active ovarian cancer. PLoS One. 2009;4:e8274. doi: 10.1371/journal.pone.0008274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Izzi B, Francois I, Labarque V, Thys C, Wittevrongel C, Devriendt K, Legius E, Van Den Bruel A, D’hooghe M, Lambrechts D, De Zegher F, Van Geet C, Freson K. Methylation defect in imprinted genes detected in patients with an Albright’s hereditary osteodystrophy like phenotype and platelet Gs hypofunction. PLoS One. 2012;7:e38579. doi: 10.1371/journal.pone.0038579. [DOI] [PMC free article] [PubMed] [Google Scholar]