Abstract
Microwave-assisted synthetic techniques were used to quickly and reproducibly produce silica nanoparticle sols using an acid catalyst with nanoparticle diameters ranging from 30-250 nm by varying the reaction conditions. Through the selection of a microwave compatible solvent, silicic acid precursor, catalyst, and microwave irradiation time, these microwave-assisted methods were capable of overcoming the previously reported shortcomings associated with synthesis of silica nanoparticles using microwave reactors. The siloxane precursor was hydrolyzed using the acid catalyst, HCl. Acetone, a low-tan δ solvent, mediates the condensation reactions and has minimal interaction with the electromagnetic field. Condensation reactions begin when the silicic acid precursor couples with the microwave radiation, leading to silica nanoparticle sol formation. The silica nanoparticles were characterized by dynamic light scattering data and scanning electron microscopy, which show the materials' morphology and size to be dependent on the reaction conditions. Microwave-assisted reactions produce silica nanoparticles with roughened textured surfaces that are atypical for silica sols produced by Stöber's methods, which have smooth surfaces.
Keywords: Chemistry, Issue 82, Chemistry, chemical manufacturing, chemistry (general), materials (general), nanocomposites, catalysts (chemical), chemistry of compounds, Chemistry and Materials (General), Composite Materials, Inorganic, Organic and Physical Chemistry, Engineering (General), Microwave, nanoparticle, silica, silicic acid, NP, SiO2, synthesis
Introduction
Silica nanoparticles (SiO2 NPs) were first synthesized by Stöber1 and through modifications2-7 have become the preferred method for SiO2 NPs synthesis. Typically, Stöber reactions are catalyzed by alkaline conditions where silica sols are formed. Acid-catalyzed reactions are used less frequently than alkaline-catalyzed reactions due to the greater degree of difficulty of hydrolysis of the siloxane precursor. Unlike alkaline-catalyzed reactions, acid-catalyzed reactions preferentially form silica gels.8
Microwave-assisted chemical reactions are an emerging technique within the scientific community and in literature due to the associated benefits to the techniques9-18. Specifically, microwave-assisted techniques have been shown to be advantageous in the synthesis of nanomaterials where the promotion of spontaneous nucleation events is desired. Microwave conditions are advantageous because microwave reactors deliver controlled power quickly to the reaction10. Until recently19, the synthesis of SiO2 NPs using microwave reactors have been used with limited success mainly as a result of issues with reproducibility20-22.
The details and procedural methods often reported in the literature on the synthesis of nanomaterials often tend to be obscure and sometimes seen as an "art-form." Combining microwave-assisted synthetic techniques and nanomaterial synthesis can compound the subject even further. The purpose of this manuscript is to guide researchers in the synthesis of nanomaterials by microwave-assisted techniques, eliminate obscurity associated with these techniques, and point out common mistakes associated with these techniques.
In a microwave chemical reaction, any molecular species containing a permanent dipole is capable of interacting and perturbing the electromagnetic (EM) field. These species are not limited solely to the reagents and solvents used within the reaction, but can be any substance placed in the EM field i.e. glass vials, salts, ionic liquids.
The ability for a specific substance to effectively convert EM energy into heat is defined as the loss factor of a material or tan δ. Solvents are commonly classified by their loss factor where values for tan δ > 0.5 are considered high, 0.1 > tan δ > 0.5 are considered medium, and tan δ < 0.1 are considered low. These loss factors values relate the ability for a particular solvent to couple or absorb microwave energy and convert that energy into heat. Thermal energy is generated through molecular friction of species attempting to align with the oscillating EM field. If solvents with high tan δ values are used within a reaction, the solvent will dominate the microwave absorption events, masking the precursor or reagents, leading to bulk heating as a result of the solvent strongly coupling with the EM field.
Typically, polar solvents are used in SiO2 NP synthesis to ensure reagent solubility and for donation of labile protons23. Common solvents used in SiO2 NP syntheses are alcohol solvents such as ethanol, methanol or 2-propanol. These solvents all have high tan δ values (0.941, 0.799, and 0.659 for ethanol, 2-propanol and methanol, respectively) making them poor solvent choices for microwave-assisted chemical reactions of SiO2 NPs as they efficiently couple with the EM field. It is our belief that microwave-assisted reactions are most effective when low tan δ solvents are used in combination with polar molecular precursors in synthetic reactions. These circumstances allow for the molecular precursors to couple with the EM field, providing molecular heating, while the solvent interacts minimally. For microwave-assisted reactions in this manuscript, acetone is used as an alternative to the commonly used alcohol solvents associated with SiO2 NPs synthesis. Acetone is considered a low loss factor solvent (tan δ = 0.054), which limits the solvent interactions within the EM field allowing selective microwave absorption with the reactants meditating silica condensation reactions.
In this manuscript, we outline the procedures associated with the microwave-assisted synthesis of SiO2 NPs which are accurate, precise and quick. SiO2 NP growth is achieved by effective coupling of the precursor with the EM field while the solvent has a minimal role in heating. Hydrolysis of the siloxane precursor is achieved by using hydrochloric acid, which leads to slower rates of hydrolysis and limits further condensation reactions. Alkaline-catalyzed reactions have much faster reaction rates and can complicate growth processes when used with microwave-assisted techniques. The resultant SiO2 NPs synthesized by these techniques range in diameters from 30 nm up to diameters greater than 250 nm. SiO2 NP size is controlled by varying the precursor concentration and exposure time to the microwave radiation.
Protocol
1. Preparation and Calculations
Prepare 1 mM HCl solution using concentrated hydrochloric acid, 37%, and water. Note: Caution should always be used when handling concentrated acids. Concentrated acid should always be added to water, never add water to concentrated acid.
Determine the desired concentration of TMOS, siloxane precursor, for the microwave reaction. A TMOS concentration of 25 mM will be used for procedural demonstrations.
2. Hydrolysis of TMOS and Preparation of Reaction Solution
For a 25 mM reaction solution of TMOS in acetone, obtain a 50 ml plastic conical tube, TMOS, and the prepared 1 mM HCl solution.
Using a micropipetter, add 850 µl of the 1 mM HCl solution to the plastic conical tube.
Using a micropipetter, add 150 µl of TMOS to the plastic conical tube containing the 850 µl of 1 mM HCl.
Observe that the mixture of TMOS and acid are originally immiscible, resulting in two clear colorless layers.
Vortex the solution. A quick burst will cause solution to turn slightly cloudy and pale white. Vortexing the solution again will yield a clear colorless solution. Total vortex time is ~10 sec. At this point, the TMOS is hydrolyzed and in the form of silicic acid.
Add acetone to the plastic conical tube containing the silicic acid solution and dilute to a total volume of 40 ml. Ensure proper mixing by vortexing the solution after diluting the solution with acetone.
Note: Prior to microwave heating, the silicic acid / acetone solution will be stable for several hours. Silica condensation was not observed in similar prepared solutions which were stable for several weeks without any evidence of condensation. Although for best results, reaction solutions should be used upon day of preparation to ensure reproducible results.
3. Create Microwave Method on Microwave Reactor
Create a microwave method within the provided software for SiO2 NP formation. Typical reaction parameters include: power = 300 W, temperature = 125 °C and time = 60 sec. Using an external laptop the temperature, pressure, and power profiles can be stored, plotted, and compared with future experiments as in Figure 1.
4. Microwave-assisted Synthesis of SiO2 Nanoparticles
Obtain a 10 ml microwave reaction vial, stir bar, and snap cap.
Add the stir bar to the vial and using a micropipetter add 5 ml of the silicic acid/acetone reaction solution to the microwave vial. A reaction volume of 5 ml should be used to provide adequate headspace to accommodate the generation of pressure from superheated acetone.
Place the snap cap on 10 ml vial. Ensure cap seats properly on the vial to reduce loss of solvent when superheated.
Place vial in microwave reactor.
Press the start button on the laptop to start the microwave heating procedure. Observe proper placement of pressure head unit. Observe that the proper microwave power settings are correct once the reaction begins: correct power setting, increase in reaction temperature, and generation of reaction pressure. When the reaction time has expired, compressed air will quickly cool the reaction.
Remove the reaction vial from microwave reactor after cooling.
Note: Typically for a 5 ml reaction, a measured conversion of ~40-50% has been reported19 in the preparation of SiO2 NPs by these microwave-assisted methods.
5. DLS Size Analysis of Reaction Solutions
Obtain a polystyrene disposable cuvette.
Add 900 µl of distilled water to the cuvette.
Add 100 µl of reaction solution containing SiO2 NP to the cuvette containing the water and mix.
Setup procedures for particle measurements by following DLS analyzer software and analyze the solution for particle size.
6. SiO2 NP Cleanup Procedure
Add 1 ml of SiO2 NP reaction solution to an Eppendorf tube.
Centrifuge at 15.7k x g for 30-60 min depending on particle diameter.
Carefully decant excess solution from SiO2 NP pellet.
Add 1 ml fresh acetone and resuspend SiO2 NP by placing in an ultrasonic bath for 5-10 min.
Repeat (centrifuging, decanting, resuspending) two more times for adequate removal of residual silicic acid.
7. SEM Sample Preparation
Clean the highly polished single crystal silicon wafers in an ultrasonic bath for approximately 30 min prior to use.
Place the silicon wafers in piranha solution (3:1 conc. H2SO4:30% H2O2) at a temperature of 80 °C for 1 hr after removing from the ultrasonic bath. Wash the silicon wafers with distilled water and dry. Note: Caution should be used when handling concentrated acids, especially piranha solution.
Drop-cast clean or not clean solutions of SiO2 NPs onto the clean prepared silicon wafer.
Place prepared samples on silicon wafer in the cylindrical tube of the sputter system.
Vacuum the cylindrical tube of the sputter system to 40 mTorr.
Purge the cylindrical tube of the sputter system with Argon gas until a pressure of approximately 200 mTorr is reached.
Decrease the pressure to 80 mTorr and apply 15 mA of voltage and keep constant. Sputter platinum on prepared samples for 60 sec.
Representative Results
Temperature, pressure, and microwave power traces for a representative SiO2 NP microwave-assisted reaction are presented in Figure 1. The microwave reaction plot is divided into three sections - ramping, reaction, and cooling. A reaction temperature of 125 °C and reaction time of 60 sec are used in this representative SiO2 NP reaction. During the ramping portion, the power is maximized at 300 W (or near max power) so that the reaction temperature can be reached quickly without over shooting the targeted reaction temperature. The reaction begins to pressurize once the temperature surpasses the boiling point of the solvent. Upon reaching the reaction temperature, microwave power is decreased and the power oscillates to maintain the selected targeted temperature. After the allotted reaction time has expired, the power is reduced to 0 W and compressed air is blown across the reaction vial quickly reducing the temperature and quenching particle growth. Temperature measurements are recorded using an IR thermometer.
Reaction vials containing prepared SiO2 NPs ranging in size from 40-270 nm in diameter are presented in Figure 2. The prepared reaction vials consist of SiO2 NPs in a solution of acetone. The SiO2 NP particle diameters are measured by dynamic light scattering (DLS) and are zeta averages based on intensity measurements. Reaction solutions are clear and colorless for SiO2 NP with particle diameters less than 125 nm while solutions are pale white in color for particle diameters greater than 125 nm. As the size and concentration of NPs increases, the reaction solution will become more colored and turbid. In reaction solutions with smaller sized NPs, the particles will stay suspended in acetone without precipitating out of solution. At these smaller diameters, particles remain suspended in the solvent since particle - particle interactions are stabilized by ionic charge of the particle surface limiting particle agglomeration and precipitation. When NP concentrations increase, SiO2 NPs will begin to settle out of solution over time. Minimal Ostwald ripening of the NPs is observed even when the synthesized materials stay suspended in acetone for extended time19.
A 3-D bar graph is presented in Figure 3 demonstrating the range of SiO2 NPs that can be synthesized using these microwave-assisted techniques by controlling the initial TMOS concentration and reaction time at 125 °C. The TMOS concentration varies from 10 - 50 mM and the reaction time varies from 0-60 sec resulting in SiO2 NPs diameters ranging in size from 30 nm to over 275 nm. The SiO2 NP diameters used in Figure 3 were measured using intensity DLS measurements.
Scanning electron micrographs of SiO2 NPs prepared by microwave-assisted synthetic techniques are shown in Figure 4. Micrographs in Figures 4a and 4b are prepared from solutions of SiO2 NPs that were not cleaned by the cleaning procedures described earlier. In Figure 4a, a glass-like silica film can be observed covering the entire prepared silicon wafer. At this magnification the SiO2 NPs cannot be observed although large cracks can be visualized as a result of the evaporation of the acetone from preparation. In Figure 4b, a more detailed view of the silica film can be observed as well as the trapped SiO2 NPs. The silica film results from the incomplete conversion of silicic acid to SiO2 NPs during the microwave reaction as previously noted19. As the solvent evaporates, the unreacted silicic acid begins to condense trapping the NPs in the film. SiO2 NPs in Figure 4c and d are prepared from solutions where the reaction solution have been cleaned using the provided cleanup procedure. By cleaning the reaction solution using the cleanup procedure, the majority of the unreacted silicic acid is removed and individual SiO2 NPs can be visualized. SiO2 NPs in Figure 4c have an average size of 70±7 nm while NPs in Figure 4d have average diameters of 200±20 nm.
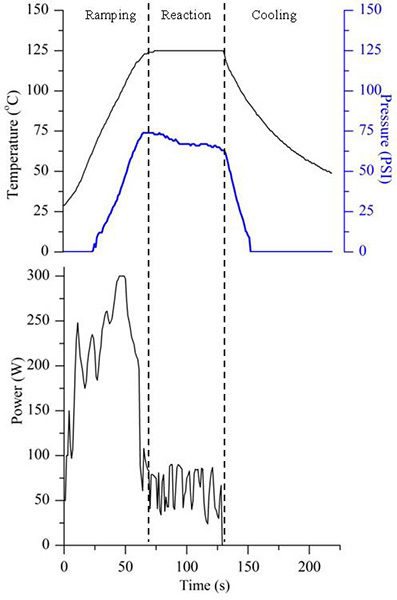 Figure 1. Representative temperature, pressure, and power curves for a SiO2 NP reaction are presented. The plot is representative for a reaction volume of 5 ml, reaction temperature of 125 °C, and reaction time of 60 sec. Click here to view larger image.
Figure 1. Representative temperature, pressure, and power curves for a SiO2 NP reaction are presented. The plot is representative for a reaction volume of 5 ml, reaction temperature of 125 °C, and reaction time of 60 sec. Click here to view larger image.
 Figure 2. Reaction vials containing solutions of different sized SiO2 NPs produced by microwave-assisted techniques are presented. Differences in solution color are based upon NP size and concentration within the reaction solution. The color of reaction solution ranges from colorless to pale white for NPs with sizes between 40-270 nm, respectively. Teflon stir bars are still present in the reaction vials. Click here to view larger image.
Figure 2. Reaction vials containing solutions of different sized SiO2 NPs produced by microwave-assisted techniques are presented. Differences in solution color are based upon NP size and concentration within the reaction solution. The color of reaction solution ranges from colorless to pale white for NPs with sizes between 40-270 nm, respectively. Teflon stir bars are still present in the reaction vials. Click here to view larger image.
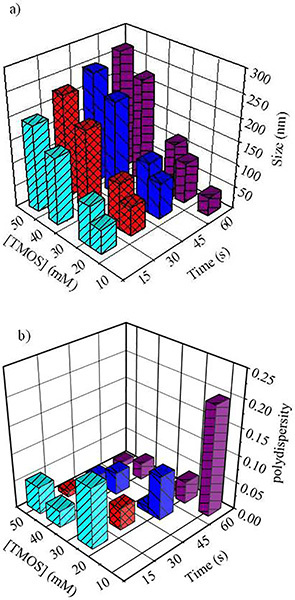 Figure 3. Plots illustrating the range of sizes (3a) and polydispersity (3b) for SiO2 NPs that can be prepared by controlling the initial concentration of TMOS and total time of microwave heating are presented. SiO2 NP sizes and polydispersity are measured by dynamic light scattering. Click here to view larger image.
Figure 3. Plots illustrating the range of sizes (3a) and polydispersity (3b) for SiO2 NPs that can be prepared by controlling the initial concentration of TMOS and total time of microwave heating are presented. SiO2 NP sizes and polydispersity are measured by dynamic light scattering. Click here to view larger image.
 Figure 4. Scanning electron micrographs of SiO2 NPs prepared by microwave-assisted techniques are presented. Images a) and b) are prepared from a solution that did not follow the cleanup procedure while c) and d) are prepared from a solution following the cleanup procedure. Microscope magnification equals 500X, 60,000X, 150,000X, and 80,000X, respectively. Click here to view larger image.
Figure 4. Scanning electron micrographs of SiO2 NPs prepared by microwave-assisted techniques are presented. Images a) and b) are prepared from a solution that did not follow the cleanup procedure while c) and d) are prepared from a solution following the cleanup procedure. Microscope magnification equals 500X, 60,000X, 150,000X, and 80,000X, respectively. Click here to view larger image.
Discussion
The microwave-assisted methods described in this manuscript are advantageous over conventional heating methods because SiO2 NPs can be synthesized accurately, precisely, and quickly. The following criteria should be followed to eliminate any potential issues associated with SiO2 NPs formation by these microwave-associated techniques: 1) use of a catalyst such as 1 mM HCl, 2) hydrolysis of the TMOS should be completed before addition of acetone, 3) use of an aprotic solvent such as acetone, 4) use of stir bars to ensure solvent homogeneity, and 5) use of a reaction system that can be pressurized. Discussions describing the selection and advantages of solvent, catalyst, and reaction conditions are briefly described below, but in-depth discussions can be elsewhere19.
The formation of silica sols has been documented by a variety of different methods including Stöber's method and reverse micelle1,24-25. The specific mechanism governing the formation of SiO2 is limited to the rate of condensation of the silicon species, which is directly controlled by the local environment of the silicon species. Specifically, the catalyst and solvent have direct effects on the local silicon environment.
Typically, alkaline catalysts, such as ammonium hydroxide, have been used for SiO2 sol formation due to the increased rates of reaction in silica formation compared to the observed rates by acid-catalysis. When alkaline catalysts are used in conjunction with microwave-assisted techniques, controlling the growth of the NPs becomes difficult because there are two mechanisms governing condensation reactions and particle formation. SiO2 reactions catalyzed by acid catalysis have much slower condensation reactions rates allowing particle formation from the exposure to microwave radiation and not by the acid catalyst.
TMOS is the preferred siloxane precursor for these microwave-assisted reactions because TMOS is easily hydrolyzed at low pH compared to the commonly used ethyl version, tetraethyl orthosilicate. The hydrolysis of TMOS prior to the addition of acetone is critical in the formation of SiO2 NPs. If the solvent is added prior to hydrolysis minimal to no SiO2 NP growth will be observed. Additionally, it should be noted the rate of polymerization or siloxane bond formation is reduced when silica reactions are performed at low pH. This reduction is also important and a necessary step for the production of SiO2 NPs by microwave-assisted techniques.
Proper solvent selection is another critical component for the synthesis of SiO2 NPs by microwave-assisted techniques which separates microwave-assisted heating from conventional heating. If the solvent can effectively couple with the EM field, heating will mimic conventional heating as the solvent is the most concentrated species in the reaction solution. In microwave heating and with a properly chosen solvent, heat is generated by the molecular precursor(s) coupling with the EM field. Solvents such as methanol or ethanol (tan δ values 0.659 and 0.941, respectively) have been reported in microwave-assisted reactions and the preparation of SiO2 NPs have inconsistent results20,22. Inconsistent results could result from the use of a high tan δ solvent in these microwave reactions resulting in inadequate coupling of the silioxane precursors with the EM field. These alcohol solvents can aid in siloxane condensation, which can further complicate NP formation. Acetone is an ideal solvent for microwave-assisted reactions because it is a low tan δ solvent (0.054) allowing for molecular heating from the silicon precursors19,26. Another benefit of acetone is the condensation of silicic acid is impeded under acid catalysis due to acetone being aprotic23. This observation was consistent since prepared reaction solutions did not lead to SiO2 condensation unless exposed to microwave radiation.
To effectively ensure NP homogeneity, reaction solutions should be stirred while irradiated by microwaves. Heating occurs at the molecular level as a result of the molecular precursors coupling with the EM field when low loss solvents are used. As a result, hot spots can occur within the reaction solution which can be minimized with stirring the reaction solution ensuring homogeneity.
Lastly, the choice of microwave reactor is important for SiO2 formation. The reactor should allow for vials to be sealed, solvents superheated, and reaction vessels pressurized. Performing NP reactions in a closed system allows reaction vessel pressurization which is important in minimizing particle-particle aggregation from solvent evaporation or loss. The generation of pressure during SiO2 NP reactions has been reported to aid siloxane condensation27. The synthesis of SiO2 NP by conventional heating techniques using standard laboratory glassware would be limited to the boiling point of the solvent (acetone, 56 °C). Attempts to perform these reactions would be unsafe due of the pressure generated at elevated temperatures. Pressurization systems, such as Parr bombs, would be needed where control over particle size becomes challenging because longer reaction times are needed to account for the diffusion of heat through the pressurization vessel.
Silica is a dielectric material of great interest for industrial applications because of its properties- highly transparent, highly functional surface, and low toxicity28-33. The microwave-assisted techniques presented in this manuscript have significance over existing methods because they provide a quick and reproducible synthesis for SiO2 NPs with varying diameters under microwave heating and acid catalysis.
Future applications for these microwave-assisted methods include the development of a continuous flow system for large batch production of SiO2 NPs for industrial needs. Through modifications of the synthetic methods, these microwave-assisted methods have utility for other encapsulating other substrates, such as quantum dots or other semiconductor nanomaterials. Substrates encapsulated with silica shells provide a protective layer against oxidation creating a water soluble material with a surface that can be functionalized. Specifically, CdSe quantum dots and carbon nanotubes are being investigated as substrates to encapsulate with a silica shell with the microwave-assisted methods. CdSe quantum dots and carbon nanotubes are being targeted as substrates for silica encapsulation because of their applications associated with sensors and bio-related applications.
Disclosures
We have nothing to disclose.
Acknowledgments
Funding was provided by the Defense Threat Reduction Agency, Physical Science and Technology Division, Protection and Hazard Mitigation technical area. This research was supported in part by an appointment to the Postgraduate Research Participation Program at the Air Force Research Laboratory administered by the Oak Ridge Institute for Science and Education (ORISE) through an interagency agreement between the U.S. Department of Energy and the Air Force Research Laboratory, Materials and Manufacturing Directorate, Airbase Technologies Division (AFRL/RXQ).
References
- Stober W, Fink A, Bohn E. CONTROLLED GROWTH OF MONODISPERSE SILICA SPHERES IN MICRON SIZE RANGE. J. Colloid Interface Sci. 1968;26:62. [Google Scholar]
- Chiang YD, et al. Controlling Particle Size and Structural Properties of Mesoporous Silica Nanoparticles Using the Taguchi Method. J. Phys. Chem. C. 2011;115:13158–13165. [Google Scholar]
- Hartlen KD, Athanasopoulos APT, Kitaev V. Facile preparation of highly monodisperse small silica spheres (15 to > 200 nm) suitable for colloidal templating and formation of ordered arrays. Langmuir. 2008;24:1714–1720. doi: 10.1021/la7025285. [DOI] [PubMed] [Google Scholar]
- Finnie KS, Bartlett JR, Barbe CJA, Kong LG. Formation of silica nanoparticles in microemulsions. Langmuir. 2007;23:3017–3024. doi: 10.1021/la0624283. [DOI] [PubMed] [Google Scholar]
- El Hawi N, et al. Silica Nanoparticles Grown and Stabilized in Organic Nonalcoholic Media. Langmuir. 2009;25:7540–7546. doi: 10.1021/la9011789. [DOI] [PubMed] [Google Scholar]
- Qiao ZA, Zhang L, Guo MY, Liu YL, Huo QS. Synthesis of Mesoporous Silica Nanoparticles via Controlled Hydrolysis and Condensation of Silicon Alkoxide. Chem. Mater. 2009;21:3823–3829. [Google Scholar]
- Yu QY, et al. Hydrothermal Synthesis of Hollow Silica Spheres under Acidic Conditions. Langmuir. 2011;27:7185–7191. doi: 10.1021/la200719g. [DOI] [PubMed] [Google Scholar]
- Chen SL, Dong P, Yang GH, Yang JJ. Kinetics of formation of monodisperse colloidal silica particles through the hydrolysis and condensation of tetraethylorthosilicate. Ind. Eng. Chem. Res. 1996;35:4487–4493. [Google Scholar]
- Caddick S, Fitzmaurice R. Microwave enhanced synthesis. Tetrahedron. 2009;65:3325–3355. [Google Scholar]
- Baghbanzadeh M, Carbone L, Cozzoli PD, Kappe CO. Microwave-Assisted Synthesis of Colloidal Inorganic Nanocrystals. Angew. Chem. Int. Edit. 2011;50:11312–11359. doi: 10.1002/anie.201101274. [DOI] [PubMed] [Google Scholar]
- Tompsett GA, Conner WC, Yngvesson KS. Microwave synthesis of nanoporous materials. ChemPhysChem. 2006;7:296–319. doi: 10.1002/cphc.200500449. [DOI] [PubMed] [Google Scholar]
- Lovingood DD, Strouse GF. Microwave Induced In-Situ Active Ion Etching of Growing. InP Nanocrystals. Nano Lett. 2008;8:3394–3397. doi: 10.1021/nl802075j. [DOI] [PubMed] [Google Scholar]
- Washington AL, Strouse GF. Microwave Synthetic Route for Highly Emissive TOP/TOP-S Passivated CdS Quantum Dots. Chem. Mater. 2009;21:3586–3592. [Google Scholar]
- Washington AL, Strouse GF. Microwave synthesis of CdSe and CdTe nanocrystals in nonabsorbing alkanes. J. Am. Chem. Soc. 2008;130:8916–8922. doi: 10.1021/ja711115r. [DOI] [PubMed] [Google Scholar]
- Washington AL, Strouse GF. Selective Microwave Absorption by Trioctyl Phosphine Selenide: Does It Play a Role in Producing Multiple Sized Quantum Dots in a Single Reaction? Chem. Mater. 2009;21:2770–2776. [Google Scholar]
- Gerbec JA, Magana D, Washington A, Strouse GF. Microwave-enhanced reaction rates for nanoparticle synthesis. J. Am. Chem. Soc. 2005;127:15791–15800. doi: 10.1021/ja052463g. [DOI] [PubMed] [Google Scholar]
- Kappe CO. Controlled microwave heating in modern organic synthesis. Angew. Chem. Int. Edit. 2004;43:6250–6284. doi: 10.1002/anie.200400655. [DOI] [PubMed] [Google Scholar]
- Nuchter M, Ondruschka B, Bonrath W, Gum A. Microwave assisted synthesis - a critical technology overview. Green Chem. 2004;6:128–141. [Google Scholar]
- Lovingood DD, Owens JR, Seeber M, Kornev KG, Luzinov I. Controlled Microwave-Assisted Growth of Silica Nanoparticles under Acid Catalysis. ACS Appl. Mater. Interfaces. 2012;4:6875–6883. doi: 10.1021/am3020247. [DOI] [PubMed] [Google Scholar]
- Davies GL, Barry A, Gun'ko YK. Preparation and size optimisation of silica nanoparticles using statistical analyses. Chem. Phys. Lett. 2009;468:239–244. [Google Scholar]
- Park SE, Kim DS, Chang JS, Kim WY. Synthesis of MCM-41 using microwave heating with ethylene glycol. Catal. Today. 1998;44:301–308. [Google Scholar]
- Mily E, Gonzalez A, Iruin JJ, Irusta L, Fernandez-Berridi MJ. Silica nanoparticles obtained by microwave assisted sol-gel process: multivariate analysis of the size and conversion dependence. J. Sol-Gel Sci. Technol. 2010;53:667–672. [Google Scholar]
- Brinker CJ. HYDROLYSIS AND CONDENSATION OF SILICATES - EFFECTS ON STRUCTURE. J. Non-Cryst. Solids. 1988;100:31–50. [Google Scholar]
- Arriagada FJ, Osseo-Asare K. Synthesis of nanosize silica in a nonionic water-in-oil microemulsion: Effects of the water/surfactant molar ratio and ammonia concentration. J. Colloid Interface Sci. 1999;211:210–220. doi: 10.1006/jcis.1998.5985. [DOI] [PubMed] [Google Scholar]
- Burda C, Chen XB, Narayanan R, El-Sayed MA. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 2005;105:1025–1102. doi: 10.1021/cr030063a. [DOI] [PubMed] [Google Scholar]
- Iler RK. The Chemistry of Silica - Solubility, Polymerization, Colloid and Surface Properties and Biochemistry. Plenum Press; 1979. [Google Scholar]
- Artaki I, Sinha S, Irwin AD, Jonas J. 29Si NMR study of the initial stage of the sol-gel process under high pressure. J. Non-Cryst. Solids. 1985;72:391–402. [Google Scholar]
- Sorensen L, Strouse GF, Stiegman AE. Fabrication of stable low-density silica aerogels, containing luminescent ZnS capped CdSe quantum dots. Adv. Mater. 2006;18:1965. [Google Scholar]
- Lita A, Washington AL, Lvan de Burgt , Strouse GF, Stiegman AE. Stable Efficient Solid-State White-Light-Emitting Phosphor with a High Scotopic/Photopic Ratio Fabricated from Fused CdSe-Silica Nanocomposites. Adv. Mater. 2010;22:3987–3991. doi: 10.1002/adma.201000493. [DOI] [PubMed] [Google Scholar]
- Halas NJ. Nanoscience under glass: The versatile chemistry of silica nanostructures. ACS Nano. 2008;2:179–183. doi: 10.1021/nn800052e. [DOI] [PubMed] [Google Scholar]
- Tang FQ, Li LL, Chen D. Mesoporous Silica Nanoparticles: Synthesis, Biocompatibility and Drug Delivery. Adv. Mater. 2012;24:1504–1534. doi: 10.1002/adma.201104763. [DOI] [PubMed] [Google Scholar]
- Wang YJ, Price AD, Caruso F. Nanoporous colloids: building blocks for a new generation of structured materials. J. Mater. Chem. 2009;19:6451–6464. [Google Scholar]
- Guerrero-Martinez A, Perez-Juste J, Liz-Marzan LM. Recent Progress on Silica Coating of Nanoparticles and Related Nanomaterials. Adv. Mater. 2010;22:1182–1195. doi: 10.1002/adma.200901263. [DOI] [PubMed] [Google Scholar]


